Galera Therapeutics, Inc. - Annual Report: 2022 (Form 10-K)
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
☒ |
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2022
or
☐ |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 For the transition period from to |
Commission File Number 001-39114
Galera Therapeutics, Inc.
(Exact name of Registrant as specified in its Charter)
Delaware |
46-1454898 |
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
P.O. Box 134 Malvern, Pennsylvania |
19355 |
(Address of principal executive offices) |
(Zip Code) |
(610) 725-1500
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Act:
Title of each class |
Trading Symbol(s) |
Name of each exchange on which registered |
Common Stock, $0.001 par value per share |
GRTX |
The Nasdaq Stock Market LLC (Nasdaq Global Market) |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. YES ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act. YES ☐ No ☒
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ NO ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ NO ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer |
|
☐ |
|
Accelerated filer |
|
☐ |
Non-accelerated filer |
|
☒ |
|
Smaller reporting company |
|
☒ |
|
|
|
|
Emerging growth company |
|
☒ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☒
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15-U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. ☐
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to § 240.10D-1(b). ☐
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). YES ☐ NO ☒
At June 30, 2022, the last business day of the registrant’s most recently completed second fiscal quarter, the aggregate market value of the voting and non-voting common equity held by non-affiliates of the registrant was approximately $32.2 million. Solely for purposes of this disclosure, shares of common stock held by executive officers, directors and certain stockholders of the registrant as of such date have been excluded because such holders may be deemed to be affiliates.
The number of shares of registrant’s Common Stock outstanding as of March 4, 2023 was 42,830,066.
DOCUMENTS INCORPORATED BY REFERENCE
Portions of the registrant’s definitive Proxy Statement, relating to its 2023 Annual Meeting of Stockholders to be filed with the Securities and Exchange Commission are incorporated by reference into Part III of this Annual Report on Form 10-K.
CAUTIONARY NOTE REGARDING FORWARD LOOKING STATEMENTS
This Annual Report on Form 10-K contains forward-looking statements. All statements other than statements of historical facts contained in this Annual Report on Form 10-K are forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “will,” “should,” “expect,” “plan,” “anticipate,” “could,” “intend,” “target,” “project,” “contemplate,” “believe,” “estimate,” “predict,” “potential” or “continue” or the negative of these terms or other similar expressions, although not all forward-looking statements contain these words. All statements other than statements of historical fact contained in this Annual Report on Form 10-K, including without limitation statements regarding our plans to develop and commercialize our product candidates, the timing of our ongoing or planned clinical trials and data readouts, the timing of and our ability to obtain and maintain regulatory approvals, the clinical utility and potential benefits of our product candidates, our commercialization, marketing and manufacturing capabilities and strategy, our expectations about the willingness of healthcare professionals to use our product candidates, expected coverage and reimbursement for avasopasem and our other product candidates; the sufficiency of our cash, cash equivalents and short-term investments and our ability to raise additional capital to fund our operations, our plans to mitigate the risk that we are unable to continue as a going concern, the anticipated impact of the COVID-19 pandemic on our business, and the plans and objectives of management for future operations and capital expenditures are forward-looking statements.
The forward-looking statements in this Annual Report on Form 10-K are only predictions and are based largely on our current expectations and projections about future events and financial trends that we believe may affect our business, financial condition and results of operations. These forward-looking statements speak only as of the date of this Annual Report on Form 10-K and are subject to a number of known and unknown risks, uncertainties and assumptions, including those described under the sections in this Annual Report on Form 10-K entitled “Summary Risk Factors,” “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” and elsewhere in this Annual Report on Form 10-K.
Because forward-looking statements are inherently subject to risks and uncertainties, some of which cannot be predicted or quantified and some of which are beyond our control, you should not rely on these forward-looking statements as predictions of future events. The events and circumstances reflected in our forward-looking statements may not be achieved or occur and actual results could differ materially from those projected in the forward-looking statements. Moreover, we operate in an evolving environment. New risk factors and uncertainties may emerge from time to time, and it is not possible for management to predict all risk factors and uncertainties. Except as required by applicable law, we do not plan to publicly update or revise any forward-looking statements contained herein, whether as a result of any new information, future events, changed circumstances or otherwise. We intend the forward-looking statements contained in this Annual Report on Form 10-K to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act of 1933, as amended, or the Securities Act, and Section 21E of the Securities Exchange Act of 1934, as amended, or the Exchange Act.
ii
SUMMARY RISK FACTORS
Our business is subject to numerous risks and uncertainties, including those described in Part I, Item 1A. “Risk Factors” in this Annual Report on Form 10-K. You should carefully consider these risks and uncertainties when investing in our common stock. The principal risks and uncertainties affecting our business include the following:
iii
Table of Contents
|
|
Page |
PART I |
|
|
Item 1. |
1 |
|
Item 1A. |
41 |
|
Item 1B. |
99 |
|
Item 2. |
99 |
|
Item 3. |
99 |
|
Item 4. |
99 |
|
|
|
|
PART II |
|
|
Item 5. |
103 |
|
Item 6. |
103 |
|
Item 7. |
Management’s Discussion and Analysis of Financial Condition and Results of Operations |
104 |
Item 7A. |
118 |
|
Item 8. |
119 |
|
Item 9. |
Changes in and Disagreements with Accountants on Accounting and Financial Disclosure |
141 |
Item 9A. |
141 |
|
Item 9B. |
141 |
|
Item 9C. |
Disclosure Regarding Foreign Jurisdictions that Prevent Inspections |
141 |
|
|
|
PART III |
|
|
Item 10. |
142 |
|
Item 11. |
142 |
|
Item 12. |
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
142 |
Item 13. |
Certain Relationships and Related Transactions, and Director Independence |
143 |
Item 14. |
143 |
|
|
|
|
PART IV |
|
|
Item 15. |
144 |
|
Item 16. |
147 |
iv
PART I
Item 1. Business.
Overview
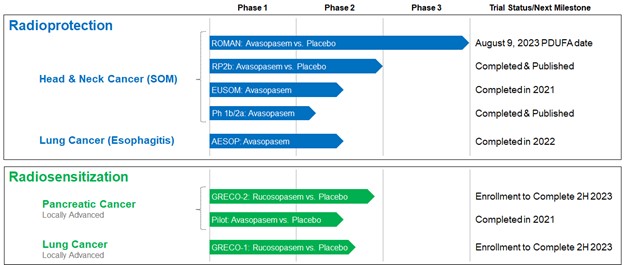
We are a clinical stage biopharmaceutical company focused on developing and commercializing a pipeline of novel, proprietary therapeutics that have the potential to transform radiotherapy in cancer. We leverage our expertise in superoxide dismutase mimetics to design drugs to reduce normal tissue toxicity from radiotherapy and to increase the anti-cancer efficacy of radiotherapy. Avasopasem manganese (avasopasem, or GC4419) is a highly selective small molecule dismutase mimetic in development for the reduction of severe oral mucositis, or SOM, in patients with head and neck cancer, or HNC, and for the reduction of esophagitis in patients with lung cancer. We are also exploring the potential for avasopasem to reduce cisplatin-induced kidney damage. SOM is a common, debilitating complication of radiotherapy in patients with HNC. In February 2018, the U.S. Food and Drug Administration, or FDA, granted Breakthrough Therapy Designation to avasopasem for the reduction of SOM induced by radiotherapy. In February 2023, the FDA accepted and granted Priority Review designation to our New Drug Application, or NDA, for avasopasem for this indication. Our second dismutase mimetic product candidate, rucosopasem manganese (rucosopasem, or GC4711), is in clinical-stage development to augment the anti-cancer efficacy of stereotactic body radiation therapy, or SBRT, in patients with non-small cell lung cancer, or NSCLC, and locally advanced pancreatic cancer, or LAPC.
Radiotherapy-induced SOM can lead to devastating complications for patients. A majority will suffer severe pain which is often managed with the use of opioids. Patients with SOM are at risk of dehydration and malnutrition as a result of the inability to eat or drink, and often require nutrition through an intravenous line or surgical placement of a feeding tube. Each year in the United States, approximately 67,000 patients are diagnosed with HNC, according to the American Cancer Society. In the five largest European markets, approximately 68,000 patients are diagnosed annually with HNC, and an additional 23,000 in Japan. We estimate that approximately 65% of patients diagnosed with HNC will be treated with radiotherapy. All patients with HNC treated with radiotherapy are at risk for developing SOM, which suggests a target patient population of approximately 43,500 patients in the United States alone for our lead indication. We believe that SOM in patients with HNC represents a total market opportunity of more than $1.5 billion in the United States based on branded supportive care price analogs. There are currently no FDA-approved drugs for SOM in these patients and we believe avasopasem, which to date is not approved for any indication, has the potential to become the standard of care for the reduction of SOM in patients with HNC receiving radiotherapy.
In December 2021, we announced topline efficacy results from a Phase 3 trial of avasopasem for the reduction of radiotherapy-induced SOM in patients with locally advanced HNC, which we refer to as the Reduction in Oral Mucositis with Avasopasem Manganese, or ROMAN, trial. The results demonstrated efficacy across multiple SOM endpoints with a statistically significant reduction on the primary endpoint of reduction in the incidence of SOM and a statistically significant reduction on the secondary endpoint of number of days of SOM, with a median of 18 days in the placebo arm versus 8 days in the avasopasem arm. We had previously announced topline results from the ROMAN trial in October 2021 that incorrectly stated the reduction on the primary endpoint was not statistically significant. Upon further analysis, an error by the contract research organization was identified in the statistical program. Correction of this error yielded the correct, statistically significant p-values for the primary and a key secondary endpoint. Exploratory analyses, such as time to SOM onset and SOM incidence at various landmarks of radiotherapy delivered, further demonstrated clinical efficacy of avasopasem in reducing the burden of SOM. Avasopasem appeared to be generally well tolerated compared to placebo.
In October 2022, we announced the presentation of the one-year tumor and renal function outcomes data from the ROMAN trial as well as topline results from a recently completed meta-analysis of the ROMAN and GT-201 (Phase 2b) SOM trial results at the 2022 American Society for Radiation Oncology, or ASTRO, Annual Meeting. After one-year follow-up, patients with HNC treated with avasopasem in combination with the standard-of-care regimen (intensity-modulated radiation therapy, or IMRT, plus cisplatin) demonstrated comparable tumor outcomes and overall survival to patients in the placebo arm, showing that avasopasem protected HNC patients from SOM without affecting the treatment benefit of standard-of-care chemoradiotherapy. In addition, after one year of follow-up, patients treated with avasopasem in combination with IMRT plus cisplatin had a 10% incidence of
1
chronic kidney disease, or CKD, compared to 20% of patients in the placebo arm, which was a pre-defined exploratory endpoint evaluating renal function. CKD (eGFR <60) is a known toxicity risk with cisplatin, which can have significant long-term consequences. The prospective exploration of this potential benefit of avasopasem was driven by published preclinical data and a post hoc assessment of patients from the GT-201 trial presented at the 2020 American Society of Clinical Oncology, or ASCO, Annual Meeting. We believe these CKD data suggest another potential benefit of avasopasem for these patients beyond reducing SOM. In addition to the ROMAN long-term endpoints, a meta-analysis of the Company’s two randomized placebo-controlled trials (ROMAN and GT-201; n=551) was included in the ASTRO presentation; these results reinforced that across both trials avasopasem therapy resulted in clinically meaningful improvements in radiotherapy-induced SOM, including reductions in the incidence, number of days, severity, and delay in the onset of SOM compared to placebo.
In December 2022, we submitted an NDA to the FDA for avasopasem for the reduction of radiotherapy-induced SOM in patients with HNC undergoing standard-of-care treatment. The NDA is supported by the data from the two randomized, double-blinded, placebo-controlled trials (ROMAN and GT-201), as well as data from other clinical trials of avasopasem in the proposed indication. In February 2023, the FDA accepted the NDA for filing and granted priority review with a Prescription Drug User Fee Act, or PDUFA, target date of August 9, 2023. The FDA indicated in its acceptance of filing letter that it is not planning to hold an advisory committee meeting on the application.
In December 2021, we also announced topline results from a Phase 2a multi-center trial in Europe assessing the safety and efficacy of avasopasem in patients with HNC undergoing standard-of-care radiotherapy, which we refer to as the EUSOM trial. Avasopasem appeared to be generally well tolerated, and the incidence of SOM and median number of days of SOM observed in the EUSOM trial were in line with the ROMAN trial results. We plan to meet with the European Medicines Agency, or EMA, in 2023 to discuss the potential registration pathway in Europe for avasopasem for radiotherapy-induced SOM.
In May 2022, we announced topline results from an open-label, single-arm Phase 2a trial evaluating avasopasem for its ability to reduce the incidence of radiotherapy-induced esophagitis in patients with lung cancer, which we refer to as the AESOP trial. This multi-center trial enrolled 39 patients (62 screened) of which 35 completed treatment with 60 gray of radiotherapy plus chemotherapy over six weeks. Of these 35 patients, 29 received at least five weeks of 90 mg of avasopasem on the days they underwent radiotherapy. These 29 patients were evaluated as the pre-specified per protocol population. The results demonstrated that avasopasem substantially reduced the incidence of severe esophagitis in patients with lung cancer receiving chemoradiotherapy compared to expectations based on review of historical data in the literature. Avasopasem was generally well tolerated. The adverse events experienced are comparable to those expected with chemoradiotherapy.
There are currently no FDA-approved drugs and no established guidelines for the treatment of radiotherapy-induced esophagitis. We intend to pursue a strategy for avasopasem, if approved for reduction in the incidence of SOM, that involves presenting the AESOP clinical data to entities like the National Comprehensive Cancer Network, or NCCN, to support the use of avasopasem to reduce esophagitis as a medically accepted indication in published drug compendia, notwithstanding that this indication may not be approved by the FDA.
In addition to developing avasopasem for the reduction of normal tissue toxicity from radiotherapy, we are developing our second dismutase mimetic product candidate, rucosopasem, to increase the anti-cancer efficacy of higher daily doses of radiotherapy, or SBRT. SBRT typically involves a patient receiving one to five large doses of radiotherapy, in contrast to the 30 to 35 small daily doses typical of intensity modulated radiation therapy, or IMRT. Clinically, SBRT is increasingly used in patients with certain tumors, such as LAPC and NSCLC, that are less responsive to the small daily doses typical of IMRT. Even with the use of SBRT, there is need for improvement in treatment outcomes for certain tumors. In September 2021, in support of rucosopasem, we announced final results from our pilot Phase 1/2 safety and anti-cancer efficacy trial of avasopasem in combination with SBRT in patients with unresectable or borderline resectable LAPC. In this proof-of-concept trial, improvements were observed with avasopasem plus SBRT in overall survival, progression-free survival, local tumor control and time to distant metastases relative to patients treated with placebo plus SBRT.
We used our observations from the pilot LAPC trial to inform the design of our rucosopasem clinical trials in combination with SBRT. We have successfully completed Phase 1 trials of intravenous rucosopasem in healthy volunteers and initiated a Phase 1/2 trial in patients with NSCLC in October 2020, which we refer to as the
2
GRECO-1 trial, and in May 2021, initiated a Phase 2b trial in patients with LAPC, which we refer to as the GRECO-2 trial.
The GRECO-1 trial is supported in part by a Small Business Innovation Research grant from the National Cancer Institute, or NCI, of the National Institutes of Health, or NIH, for the investigation of our dismutase mimetics in combination with SBRT for the treatment of lung cancer. We intend for this trial to assess the anti-cancer efficacy and safety of rucosopasem in combination with SBRT. In June 2022, we reported interim results from the open-label Phase 1 stage of the trial with six months follow-up on all seven patients. Rucosopasem in combination with SBRT appeared to be well tolerated through the cutoff date of June 14, 2022. The most frequent adverse events were fatigue, cough, and nausea, which are common in patients with lung cancer receiving radiotherapy. Through six months, in-field partial responses were observed in three patients and stable disease was observed in three others based on RECIST criteria. These results included target tumor reductions in five patients of 61%, 58%, 33%, 29% and 27% and one patient with an 8% increase. Preservation of pulmonary lung function was also observed compared to our expectations based on review of historical literature evaluating pulmonary function in a similar patient population with SBRT alone. We expect to complete enrollment in the randomized, placebo-controlled Phase 2 stage of this trial in the second half of 2023.
The GRECO-2 trial is designed to assess rucosopasem in combination with SBRT in patients with LAPC, based on our observations from the pilot LAPC trial with avasopasem. The primary endpoint of this trial is overall survival. We expect to complete enrollment in the GRECO-2 trial in the second half of 2023.
Our management team has extensive drug development and commercialization experience ranging from discovery through registrational trials and commercial launch.
Our Pipeline
The following table summarizes our product candidates:
Our Strategy
Our mission is to transform cancer therapy by reducing normal tissue toxicity induced by radiotherapy and increasing the anti-cancer efficacy of radiotherapy with the use of our dismutase mimetics. By doing this we seek to improve the lives of patients with cancer. Key elements of our strategy are as follows:
3
Background on Superoxide and Superoxide Dismutase
Superoxide is similar to the molecular oxygen, O2, that is essential to breathing and life, except it carries one more electron. This extra electron, shown in the chemical formula O2•-, makes superoxide a reactive oxygen species that can react with a variety of biological molecules. Superoxide is produced constantly in every living cell by normal activities such as mitochondrial respiration, and if not removed rapidly, it causes damage to lipids, proteins, DNA and other critical biological molecules. As a result, it can harm or kill cells and has been implicated in a variety of biological disorders, including cancer. As protection, human cells produce superoxide
4
dismutase enzymes, or SODs, to eliminate superoxide by rapidly and selectively converting it to hydrogen peroxide at rates of 107 molecules per second or higher. Hydrogen peroxide is much less toxic than superoxide to normal cells and is subsequently broken down by various enzymes, such as catalase (the natural disposal enzyme for hydrogen peroxide), to molecular oxygen and water. The SOD pathway is depicted below.
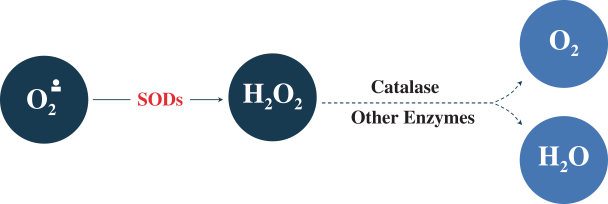
Radiotherapy induces bursts of superoxide in the irradiated tissues well in excess of normal amounts, which can overwhelm native SOD activity. It generates superoxide directly, by splitting water molecules immediately, and indirectly, by activating enzymes that produce large amounts of superoxide following radiation. In addition, once tissue damage has begun, inflammatory cells attracted to the irradiated region also produce superoxide prodigiously. The resulting high levels of superoxide can induce significant damage in normal cells, and, depending on which organs fall within the irradiated field, can drive a variety of normal tissue toxicities. A condition referred to as mucositis occurs when the cells lining the gastro-intestinal tract, known as the mucosa, are damaged or killed.
Scientific literature suggests that metabolic differences make cancer cells much less sensitive than normal cells to elevated superoxide; elevated superoxide levels may even be typical of some cancers. As a result, the removal of the excess superoxide generated by radiotherapy does not decrease the anti-cancer efficacy of radiotherapy. Meanwhile, scientific literature also suggests that cancer cells are much more sensitive than normal cells to elevated hydrogen peroxide, so the conversion of excess superoxide to hydrogen peroxide by SODs may contribute to the anti-cancer efficacy of radiotherapy.
Artificially increasing SOD levels, by gene overexpression or administering recombinant SOD enzyme, has been shown in third-party preclinical and clinical studies to reduce radiotherapy-induced normal tissue toxicities, including mucositis. The preclinical studies have also suggested that increasing SOD levels can increase the anti-cancer efficacy of radiotherapy. Current therapeutic applications of the SODs themselves, however, have been limited by their following characteristics:
Our Superoxide Dismutase Mimetics
We believe low molecular weight drugs that can mimic native SODs can overcome the limitations of using the native enzymes therapeutically. The challenge has been finding small molecule dismutase mimetics with similarly fast catalytic rates and high selectivity for superoxide that are also stable, safe and suitable for manufacturing. We are developing our dismutase mimetics to address this challenge.
5
Our class of dismutase mimetics are based on a common core structure, where a macrocyclic ring positions five nitrogen atoms to tightly hold a manganese atom in the ring’s center. These pentaaza macrocycles are manufactured with the manganese in the +2 oxidation state, or Mn+2. In solution, this Mn+2 reacts rapidly with the protonated form of superoxide, which has the chemical formula HO2• and is constantly in equilibrium with regular superoxide. In this reaction, Mn+2 gives up an electron and is oxidized to Mn+3, making hydrogen peroxide. Then, as quickly as superoxide can reach the Mn+3, it takes superoxide’s extra electron, reducing back to Mn+2, making molecular oxygen and bringing the dismutase mimetic full circle back to where it started.
Our Dismutase Mimetics Core Structure:
Pentaaza Macrocycles

We have designed, and are developing, our dismutase mimetics to have each of the following essential features:
In radiotherapy, we believe our dismutase mimetics have the potential to reduce normal tissue toxicity by removing excessive superoxide. We have demonstrated this in preclinical models not only of mucositis, but also radiotherapy damage to the lungs, liver and other organs. Importantly, our dismutase mimetics do not interfere with the anti-cancer efficacy of radiotherapy, as demonstrated in preclinical tumor models and in our placebo-controlled SOM trials in patients with HNC.
There is also the potential to increase the anti-cancer efficacy of SBRT, where our dismutase mimetics generate high daily doses of hydrogen peroxide. Preclinically, we have shown this effect in a variety of cancer types, including head and neck, pancreatic, lung and breast cancer, and when SBRT is combined with immune checkpoint
6
inhibitor therapy. Given the combination of reduced normal tissue toxicity and increased anti-cancer efficacy of radiotherapy, we believe that our dismutase mimetics can transform radiotherapy.
In chemotherapy, we further believe our dismutase mimetics have the potential to reduce normal tissue toxicity by removing excessive superoxide produced by some commonly used chemotherapeutic agents. We have demonstrated this in preclinical models of cisplatin damage to the kidneys. Importantly, our dismutase mimetics do not interfere with the anti-cancer efficacy of cisplatin and other commonly used chemotherapeutic agents, as demonstrated in preclinical tumor models.
We currently have two dismutase mimetic candidates in clinical development, avasopasem and rucosopasem. We also believe the technology is amenable to development of additional candidates for intravenous or other routes of administration.
Disease Overview and Our Product Pipeline
Reducing Radiotherapy-Induced Toxicities in Patients with Cancer (Radioprotection)
The American Cancer Society estimates approximately two million new cancer cases will be diagnosed in the United States in 2023 and over 50% of patients with cancer will be treated with radiotherapy at some time in their treatment cycle. While radiotherapy has variable success depending on the cancer being treated, the toxicity or side effects associated with its use can limit its effectiveness. Radiotherapy causes acute and late toxicities that affect various organs and functions.
One of the most common radiotherapy-induced toxicities results in a condition referred to as mucositis which occurs when cells lining the gastro-intestinal tract, known as the mucosa, are damaged or killed. The oral mucosa is a common location for mucositis to occur, particularly for patients with HNC receiving radiotherapy. Another common location for mucositis to occur in patients receiving radiotherapy is the esophagus, referred to as esophagitis.
Oral Mucositis
OM occurs when radiotherapy induces the production of superoxide that attacks and breaks down the epithelial cells lining the mouth. The severity of OM is commonly measured using the WHO scale, which is also used by the FDA as a basis for product approvals. The scale consists of five Grades: Grade 0 through Grade 4. SOM is commonly defined as Grade 3 or Grade 4 OM.
Grade |
|
WHO Scale Description |
0 |
|
No OM |
1 |
|
Erythema (redness) and soreness |
2 |
|
Erythema and ulcers but patients can swallow solid food |
3 |
|
Ulcers with extensive erythema and patients cannot swallow solid food |
4 |
|
Oral alimentation (solid or liquid) is not possible |
SOM can lead to devastating complications, including:
7
Each year in the United States, approximately 67,000 patients are diagnosed with HNC, according to the American Cancer Society and we estimate that approximately 65% will be treated with standard-of-care radiotherapy. All patients with HNC treated with radiotherapy are at risk for developing SOM, which suggests a target patient population of approximately 43,500 patients in the United States alone for our lead indication. Based on observations from multiple studies, we estimate that approximately 70% will develop SOM and between 20% to 30% will develop Grade 4 OM.
In the five largest European markets, approximately 68,000 patients are diagnosed annually with HNC, and an additional 23,000 in Japan.
In a survey we conducted in 2018 of 150 U.S. radiation oncologists, OM was identified as the most burdensome side effect caused by radiotherapy in patients being treated for HNC. In a survey we conducted in 2022 of 100 U.S. radiation oncologists and 100 U.S. medical oncologists, SOM was considered to cause a major impact to patients and their care, with the condition leading to increased treatment delays, treatment discontinuations, and hospitalizations.
Current Treatment Landscape and Limitations
There are currently no FDA-approved drugs for the treatment of OM in patients with HNC. In 2020, the Multinational Association of Supportive Care in Cancer and International Society of Oral Oncology, or MASCC / ISOO, published an update to the leading clinical practice guidelines for the management of OM. These guidelines, which are summarized below, underscore how limited the existing approaches are for the management of OM in patients with HNC, and that these approaches have been largely palliative to date.
8
These MASCC / ISOO guidelines demonstrate that there is a high unmet need for the treatment or prevention of OM in patients with HNC, driven by the lack of clear efficacy of the existing treatment options. This unmet need is further demonstrated by the findings from our 2018 survey of 150 U.S. radiation oncologists, where only 19% and 21% of physicians, respectively, stated that topical agents are effective in preventing or reducing the incidence of SOM and in treating or reducing the duration of SOM in patients with HNC. Our 2022 survey reinforced that the majority of radiation and medical oncologists are not satisfied with current options available to treat SOM and that there is a high unmet need for the treatment or prevention of OM. The FDA has also acknowledged this unmet need and the lack of effective therapies for the reduction of the duration, incidence and severity of SOM induced by radiotherapy by granting avasopasem Fast Track and Breakthrough Therapy Designation, and priority review for the NDA. We believe that SOM in patients with HNC represents a total market opportunity of more than $1.5 billion in the United States.
Our Solution: Avasopasem for Radiotherapy-Induced Severe Oral Mucositis
Avasopasem is a highly selective small molecule dismutase mimetic we are developing for the reduction of SOM in patients with HNC. We believe avasopasem, which to date is not approved for any indication, has the potential to address shortcomings associated with current approaches and become the standard of care treatment for SOM in patients with locally advanced HNC.
Potential Benefits of Avasopasem for Severe Oral Mucositis
We believe that avasopasem has the potential to be the first FDA-approved drug and the standard of care for the reduction of SOM in patients with HNC receiving radiotherapy, with the following benefits:
Our market research surveys conducted with radiation and medical oncologists suggest avasopasem has a clinically meaningful product profile based on the safety and efficacy data from our two randomized, double-blinded placebo-controlled trials. Respondents in the various rounds of market research conducted between 2018 and 2022 projected the use of avasopasem in a range of 48% to 69% of their eligible patients, with a majority of physicians suggesting they would adopt avasopasem within the first 12 months of it becoming available.
9
Clinical Development of Avasopasem for Severe Oral Mucositis
New Drug Application (NDA)
Avasopasem has been granted Fast Track and Breakthrough Therapy Designation by the FDA for the reduction of SOM induced by radiotherapy. As a benefit of such designations, we had multiple interactions with the FDA in preparation for the submission of the NDA. Based on these interactions, it was determined that our two randomized, double-blinded, placebo-controlled trials are sufficient for review of the NDA, and that the FDA would review an Integrated Summary of Efficacy based on a meta-analysis of the integrated ROMAN and GT-201 trial results.
In December 2022, we submitted the NDA to the FDA. The NDA is supported by the data from the two randomized, double-blinded, placebo-controlled trials (ROMAN and GT-201), as well as data from other clinical trials of avasopasem in the proposed indication.
In February 2023, the FDA accepted the NDA for filing and granted Priority Review designation with a PDUFA target date of August 9, 2023. The FDA indicated in its acceptance of filing letter that it is not planning to hold an advisory committee meeting on the application.
ROMAN Trial (Phase 3)
In December 2021, we announced positive topline efficacy results from the ROMAN trial. We had previously announced topline results from the ROMAN trial in October 2021. Upon further analysis following the October topline data announcement, an error by the contract research organization was identified in the statistical program. Correction of this error yielded the correct, statistically significant p-values for the primary and a key secondary endpoint. The trial was a randomized, double-blinded, multicenter, placebo-controlled trial assessing the effects of avasopasem on the incidence, duration and severity of SOM. 455 patients were enrolled in the trial and randomized 3:2 in favor of the avasopasem 90 mg treatment arm. Like our Phase 1b/2a and GT-201 trials, the eligible population was patients with locally advanced, squamous cell HNC who were eligible for seven weeks of standard-of-care radiotherapy.
ROMAN Trial Design (n=455 patients)
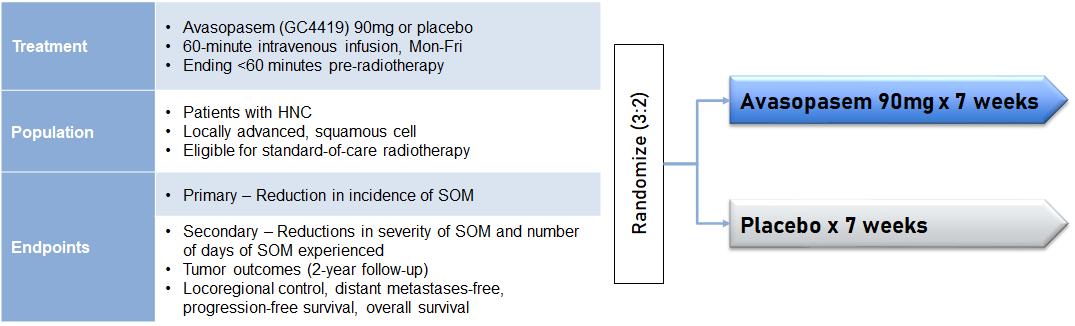
The primary endpoint of the ROMAN trial was the reduction in the incidence of SOM through the radiotherapy period for patients being treated with 90 mg of avasopasem as compared to placebo received as a 60-minute intravenous infusion less than 60 minutes before radiation, Monday to Friday, for seven weeks. All patients were assessed twice weekly for OM by trained evaluators during the course of their radiotherapy treatment.
Additional endpoints included, among others, reduction in the number of days of SOM experienced by all patients and reduction in the severity of SOM, as well as the effect of treatment on tumor outcomes measured by overall survival, or OS, progression-free survival, or PFS, locoregional control, or LRC, and distant metastasis-free,
10
or DMF, rates. Adverse events were monitored during the trial period. One-year tumor outcomes and two-year survival rates will be collected.
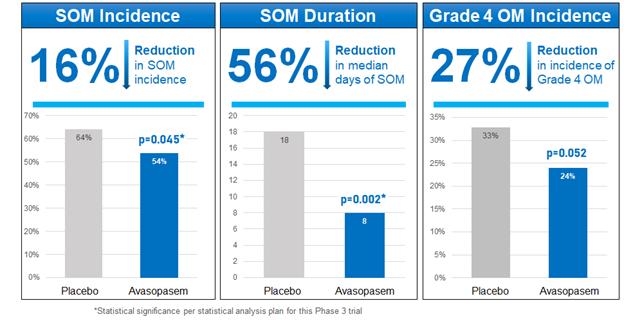
In this trial, avasopasem demonstrated efficacy across SOM endpoints with a statistically significant 16% relative reduction on the primary endpoint of reduction in the incidence of SOM (p=0.045) and a statistically significant 56% relative reduction in the number of days of SOM (p=0.002), with a median of 18 days in the placebo arm versus 8 days in the avasopasem arm. The severity of SOM (incidence of Grade 4 OM) was reduced by 27% in the avasopasem arm compared to placebo (p=0.052).
Relative Reduction Across SOM Endpoints
Exploratory analyses, such as time to SOM onset and SOM incidence at various landmarks of radiotherapy delivered, also demonstrated clinical efficacy of avasopasem in reducing the burden of SOM. The median time to onset of SOM for all patients was delayed by 11 days, from 38 days in the placebo arm to 49 days in the avasopasem arm. The incidence of SOM at all radiotherapy landmarks for patients on avasopasem was reduced compared to placebo, with the relative reductions greater than the primary endpoint both earlier during the course of therapy and during the two-week observation period after radiotherapy, as summarized in the following chart. The gray, or Gy, is the International System of Units unit of absorbed radiation dose.
11
Incidence of SOM Reduced at All Landmarks of Radiation Therapy
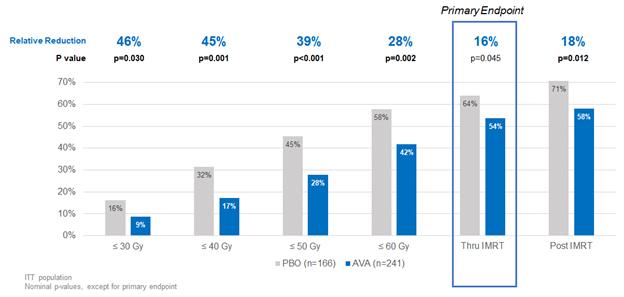
We believe the data in the above chart further demonstrate the potential clinical activity of avasopasem and the potential benefit to patients over the course of their radiotherapy.
In addition, after one year of post treatment follow-up, patients treated with avasopasem in combination with IMRT plus cisplatin had a 10% incidence of chronic kidney disease, or CKD, which was a pre-defined exploratory endpoint evaluating renal function, compared to 20% of patients in the placebo arm (p=0.0043). CKD (eGFR <60) is a known toxicity risk with cisplatin, which can have significant long-term consequences. The prospective exploration of this potential benefit of avasopasem was driven by published preclinical data and a post hoc assessment of patients from the GT-201 trial presented at the 2020 ASCO Annual Meeting. We believe this CKD data suggest another potential benefit of avasopasem for these patients beyond reducing SOM.
We also followed patients from this trial for tumor outcomes out to one year following radiotherapy and continue to follow patients out to two years for overall survival. In the one-year assessment of tumor outcomes, we observed similar outcomes among patients in the avasopasem and placebo arms in OS, PFS, LRC and DMF rates, demonstrating that avasopasem protected HNC patients from SOM without affecting the treatment benefit of standard-of-care chemoradiotherapy.
12
Tumor Outcomes Maintained through One Year
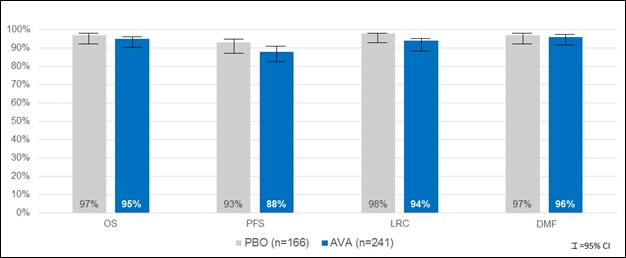
Avasopasem appeared to be generally well tolerated compared to placebo. No difference was observed in the severity of adverse events and the most frequent adverse events were similar between the treatment and placebo arms. The percentages of patients with the most common adverse events in the ROMAN trial are shown in the table below.
Most Frequent Adverse Events Similar
Across Active and Placebo Arms

GT-201 Trial (Phase 2b) in Patients with HNC
In December 2017, we announced positive topline data from the GT-201 trial in 223 patients with locally advanced HNC being treated with IMRT and concurrent cisplatin at multiple sites in the United States and Canada. The trial was a randomized, double-blinded, placebo-controlled trial assessing the effects of avasopasem on the median duration, incidence and severity of SOM. Patients received 30 mg of avasopasem, 90 mg of avasopasem or placebo as a 60-minute infusion less than 60 minutes before radiation, Monday to Friday, for seven weeks. All patients were assessed twice weekly for OM by trained evaluators during the course of their radiotherapy treatment. If SOM was present in a patient at the end of the course of his or her radiotherapy treatment, that patient continued to be evaluated weekly for up to eight additional weeks.
13
GT-201 Trial Design (n=223)
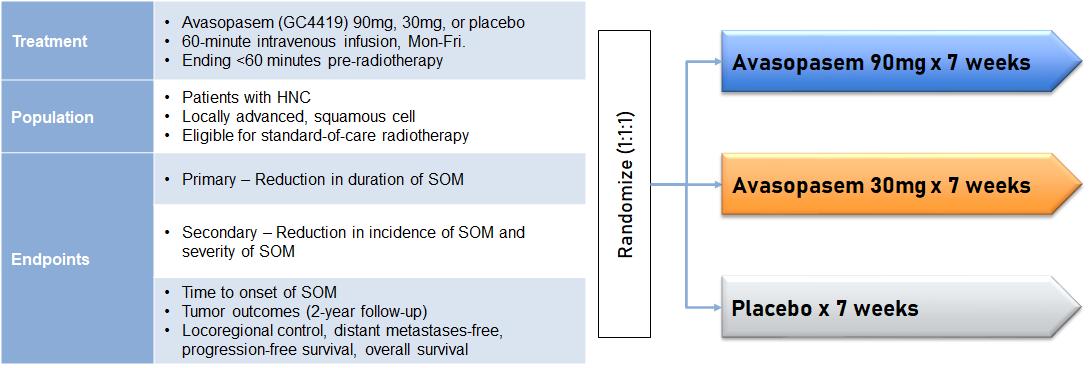
The primary endpoints of the trial were reduction in the duration of SOM in the 90 mg and 30 mg treatment arms. Duration was defined as the number of days from when a patient was first assessed with SOM until the first day that patient was assessed with Grade 2 or less OM, with no subsequent occurrences of SOM.
In this trial, the 90 mg treatment arm of avasopasem demonstrated a statistically significant reduction compared to placebo on the primary endpoint (p=0.024). The median duration of SOM in this arm was 1.5 days, a 92% reduction compared to placebo.
Secondary endpoints included reduction in the incidence and severity of SOM in each of the 90 mg and 30 mg treatment arms. For these purposes, we define the severity of SOM as the incidence of Grade 4 OM. The incidence of SOM in the 90 mg treatment arm was reduced by 36% through 60 Gy and 34% through the full course of radiotherapy treatment compared to placebo and the severity of SOM in the 90 mg treatment arm was reduced by 47% through the full course of radiotherapy treatment compared to placebo.
In the 30 mg treatment arm, intermediate reductions compared to placebo were observed in median duration of SOM (58%), incidence of SOM through 60 Gy (31%) and through the full course of radiotherapy treatment (8%), and in severity of SOM (30%) through the full course of radiotherapy treatment.
14
Relative Reduction Across SOM Endpoints
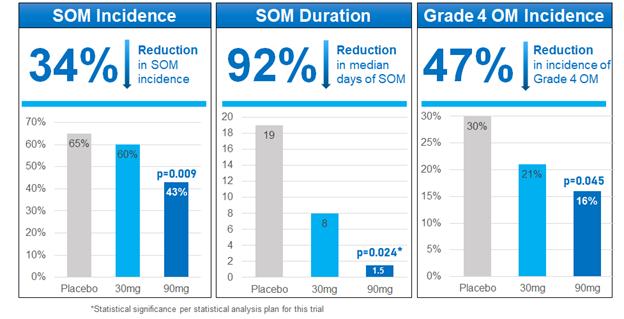
In the trial, we also observed an apparent delay in the onset of SOM in the 90 mg treatment arm compared to placebo, reduced usage of opioids in both the 30 mg and 90 mg treatment arms compared to placebo, and reduced placement and use of gastrostomy tubes in the 90 mg treatment arm compared to placebo.
The following chart depicts the course of SOM in each patient in the 90 mg treatment arm or the placebo arm who experienced at least one episode of SOM during the course of his or her treatment and follow-up. Each bar represents a single patient and illustrates the length of time between that patient’s first evaluated instance of SOM and his or her last evaluated instance of SOM, along with the severity of his or her SOM during that interval.
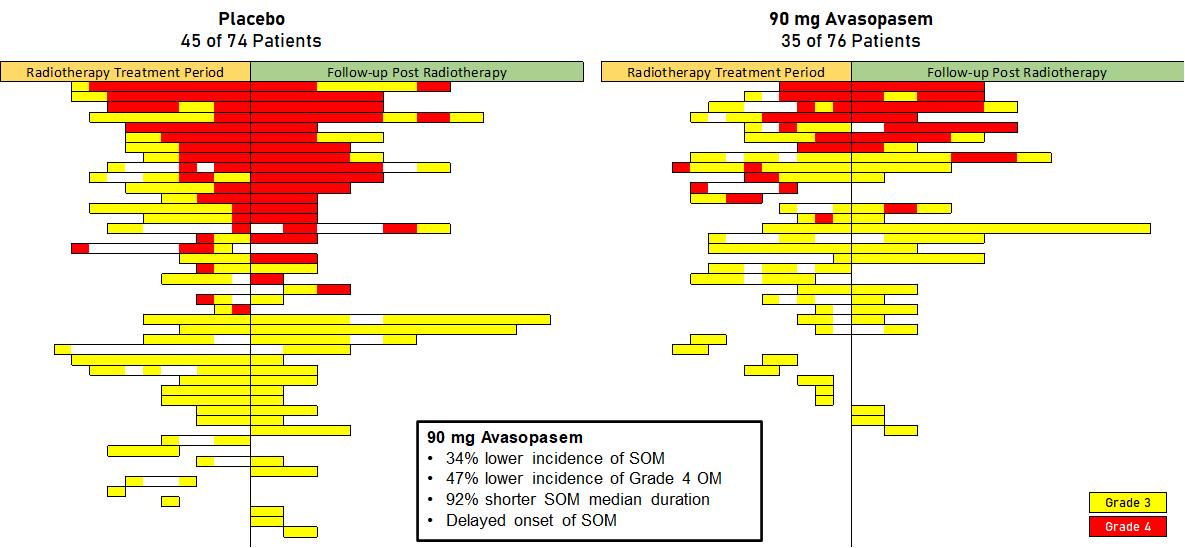
This chart demonstrates that (1) fewer patients in the 90 mg treatment arm developed SOM than in the placebo arm, (2) fewer patients in the 90 mg treatment arm developed Grade 4 OM than in the placebo arm, and (3)
15
on average, SOM did not last as long for patients in the 90 mg treatment arm. This is consistent with the observed reductions in the individual numerical endpoints of median duration, incidence and severity.
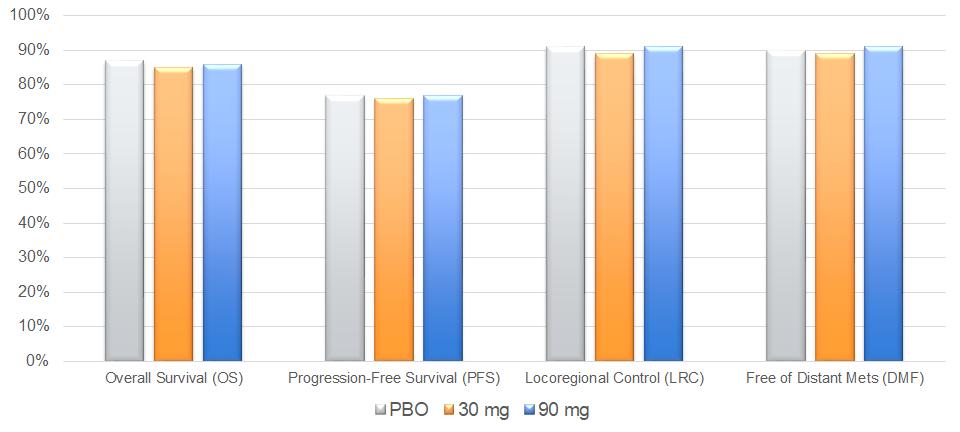
We followed patients from this trial for tumor outcomes out to two years following radiotherapy. In the two-year assessment of tumor outcomes, we observed similar outcomes among the three arms in OS, PFS, LRC and DMF rates.
Tumor Outcomes Maintained through Two Years
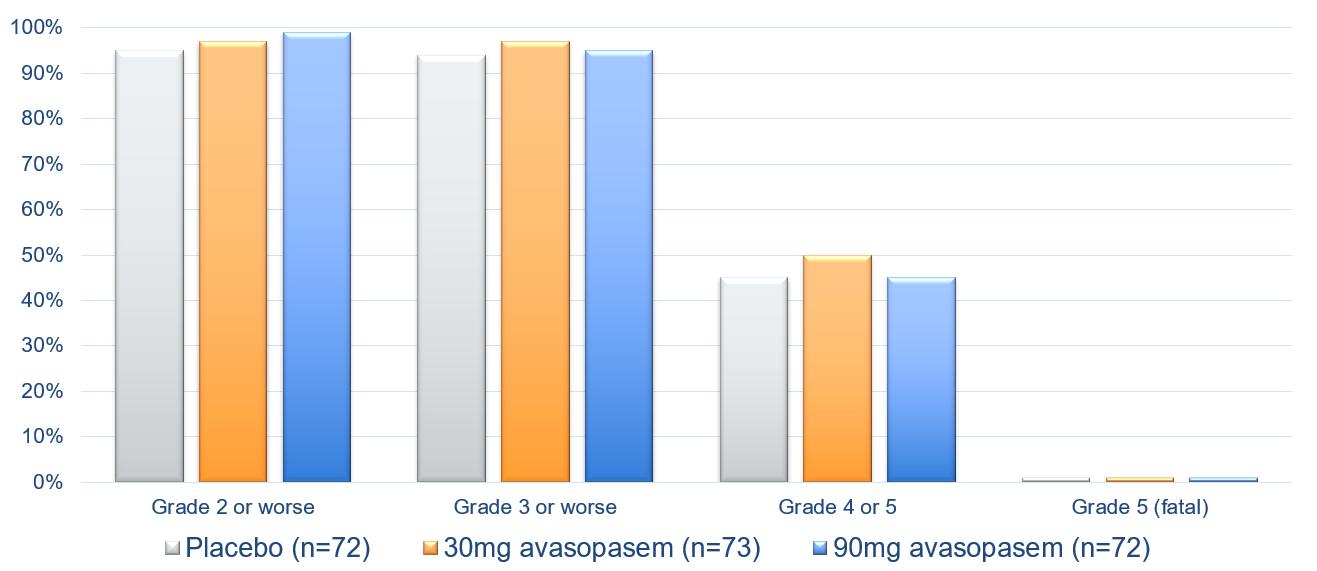
No difference was observed in the severity of adverse events among the three arms in the trial and the most frequent adverse events were similar among the three arms.
Safety Profile of Both Avasopasem Doses was Comparable to
Standard-of-Care Chemoradiotherapy (Placebo Arm)
16
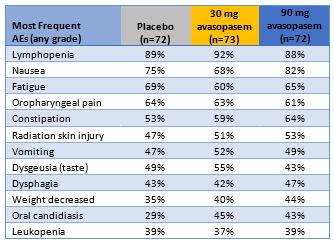
The percentages of patients with the most common adverse events in the GT-201 trial are shown in the table below.
Most Frequent Adverse Events Similar
Across Active and Placebo Arms
Meta-Analysis of Integrated ROMAN and GT-201 Trials
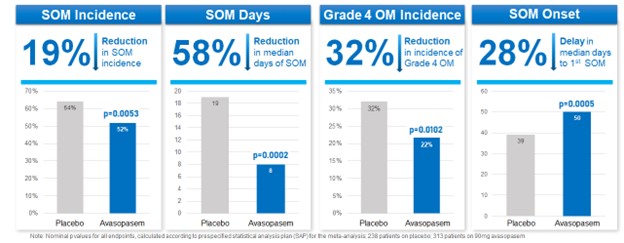
In October 2022, we announced the presentation of a meta-analysis of our two randomized placebo-controlled trials (ROMAN and GT-201; n=551) at the 2022 ASTRO Annual Meeting. These results reinforced that across both trials avasopasem therapy resulted in clinically meaningful improvements in radiotherapy-induced SOM, including reductions in the incidence, number of days, severity, and delay in the onset of SOM compared to placebo.
Reduction Across SOM Endpoints in Meta-Analysis
Phase 2a Trial in Patients with HNC in Europe (EUSOM)
In December 2021, we announced topline results from EUSOM, a Phase 2a multi-center trial of avasopasem in Europe evaluating avasopasem in combination with IMRT and concurrent cisplatin in patients with locally advanced HNC. This trial was conducted in twelve centers across six countries in Europe and enrolled 38 patients, of which 33 completed full treatment.
17
The primary objective of this trial was to assess the safety of avasopasem in combination with IMRT and concurrent cisplatin. Secondary objectives included, among others, the reduction in the incidence of SOM through the radiotherapy period.
Avasopasem appeared to be generally well tolerated. The incidence of SOM was 54.5% and the median number of days of SOM was 9 days for patients who completed treatment in the EUSOM trial, in line with the ROMAN trial, in which the incidence of SOM in the avasopasem arm was 54% and the median number of days of SOM was 8 days.
Phase 1b/2a Trial in Patients with HNC
In August 2016, we completed a Phase 1b/2a, open-label, multi-center, dose escalation trial of the safety, tolerability, pharmacodynamic and pharmacokinetic properties of avasopasem in combination with radiotherapy and concurrent cisplatin in 46 patients with locally advanced HNC. Doses ranged from 15 mg to 112 mg. The objectives of this trial were to evaluate the safety and tolerability of avasopasem in combination with IMRT and cisplatin, to determine a maximum tolerated dose and to assess the potential of avasopasem to reduce the duration, incidence and severity of SOM.
In this trial, patients were assigned to treatment duration groups based upon the dose and duration of dosing of avasopasem received and we observed that the incidence, duration, and severity of SOM through six weeks of radiotherapy (with patients receiving a cumulative radiotherapy dose of 60 Gy) decreased for patients who received six to seven weeks of avasopasem. In the group receiving six to seven weeks of avasopasem, 29% of patients experienced SOM, with a median duration of 2.5 days, and no patients experienced Grade 4 OM. Avasopasem was well tolerated and a maximum tolerated dose was not reached.
Patients in the trial were followed for tumor outcomes at one-year post-radiotherapy. The observed LRC, DMF, PFS, and OS rates in 44 patients evaluable for tumor outcome at one year were 93%, 93%, 84% and 93%, respectively. We believe these outcomes are similar to the outcomes observed in historical control studies, suggesting that avasopasem does not decrease the anti-cancer efficacy of radiotherapy.
Radiotherapy-Induced Esophagitis
Radiotherapy-induced esophagitis is a common and debilitating adverse effect that develops in patients receiving radiotherapy, most commonly for lung, esophageal, breast or head and neck cancers or for lymphoma. Radiotherapy-induced esophagitis is inflammation, edema, erythema, and erosion of the mucosal surface of the esophagus caused by radiotherapy. Esophagitis can be life-threatening, and symptoms include an inability to swallow, severe pain, ulceration, infection, bleeding and weight loss and may require hospitalization. The severity of esophagitis is graded using the National Cancer Institute, or NCI, Common Terminology Criteria for Adverse Events, which is a five-point grading scale:
Grade |
|
Description |
1 |
|
Patients are asymptomatic with only clinical observations |
2 |
|
Patients are symptomatic with altered eating or swallowing, with oral supplements indicated |
3 |
|
Patients exhibit severely altered eating or swallowing requiring tube feeding, total parenteral nutrition or hospitalization |
4 |
|
Patient requires urgent operative intervention; condition is life-threatening |
5 |
|
Results in death |
Radiotherapy-induced esophagitis potentially represents a larger market opportunity than OM. In lung cancer (our first target market for esophagitis), there are approximately 238,000 new patients annually in the United States, of which approximately 50,000 are treated with radiotherapy. The overall frequency of Grade 2 or higher esophagitis in patients receiving radiotherapy for the treatment of lung cancer is approximately 50% and approximately 20-30% will experience Grade 3 or higher. The results of our 2018 survey of 150 U.S. radiation oncologists suggested that they view OM data as being representative of potential efficacy in esophagitis, which we believe supports the feasibility of exploring the use of avasopasem for the reduction of esophagitis.
18
Current Treatment Landscape and its Limitations
There are currently no FDA-approved drugs and no established guidelines for the treatment of radiotherapy-induced esophagitis. Treatment options are not only ineffective but also largely symptomatic in nature, with medications being administered in conjunction with a focus on adequate hydration and nutrition. These approaches, which include various analgesics such as topical lidocaine and opioids, and tube or intravenous feeding, do not treat the underlying cause of radiotherapy-induced esophagitis.
Our Solution: Avasopasem for Radiotherapy-Induced Esophagitis
Unlike existing treatment options that are largely palliative in nature, we believe avasopasem has the potential to address and mitigate the root cause of radiotherapy-induced esophagitis. By removing superoxide, avasopasem is designed to reduce the damage radiotherapy ordinarily causes to the patient’s esophageal mucosa, and thereby reduce the incidence of radiotherapy-induced esophagitis. We believe avasopasem has the potential to become the standard of care for the reduction in the incidence of radiotherapy-induced esophagitis in patients with lung cancer.
Clinical Development of Avasopasem for Esophagitis
Phase 2a Trial in Patients with Lung Cancer (AESOP Trial)
In May 2022, we announced topline results from an open-label, single-arm Phase 2a trial evaluating avasopasem for its potential to reduce the incidence of radiotherapy-induced esophagitis in patients with lung cancer, which we refer to as the AESOP trial. This multi-center trial enrolled 39 patients (62 screened) of which 35 completed treatment with 60 Gy of radiotherapy plus chemotherapy over six weeks. Of these 35 patients, 29 received at least five weeks of 90 mg of avasopasem on the days they underwent radiotherapy. These 29 patients were evaluated as the pre-specified per protocol population. The results demonstrated that avasopasem substantially reduced the incidence of severe esophagitis in patients with lung cancer receiving chemoradiotherapy compared to our expectations based on review of historical data in the literature. Avasopasem was generally well tolerated. The adverse events experienced are comparable to those expected with chemoradiotherapy.
Based on these data, we intend to pursue a strategy for avasopasem, if approved for the reduction of SOM, that involves presenting the AESOP clinical data to entities like the National Comprehensive Cancer Network, or NCCN, to support the use of avasopasem to reduce esophagitis as a medically accepted indication in published drug compendia, notwithstanding that this indication may not be approved by the FDA.
Increasing Anti-Cancer Efficacy of Radiotherapy (Radiosensitization)
As cancer cells are much more sensitive than normal cells to elevated hydrogen peroxide, we believe the conversion of excess superoxide to hydrogen peroxide by our dismutase mimetics has the potential to increase the anti-cancer efficacy of radiotherapy. We are developing rucosopasem with the goal to increase the anti-cancer efficacy of high daily doses of radiotherapy, which we have demonstrated in our preclinical studies. A preclinical research article was published in Science Translational Medicine in May 2021 describing the synergy of our selective dismutase mimetics in combination with SBRT in killing tumors. This increased efficacy could be particularly important in settings where the current anti-cancer efficacy of radiotherapy alone is insufficient to achieve the desired outcome.
Locally Advanced Pancreatic Cancer Overview
Pancreatic cancer is a disease in which solid tumors form in the tissues of the pancreas. It is a particularly aggressive form of cancer and represents the third-leading cause of cancer deaths in the United States with approximately 64,000 new diagnoses and 50,500 deaths estimated in 2023. Globally, pancreatic cancer accounted for almost as many deaths (446,000) as new diagnoses (496,000) in 2020. Over 30% of newly diagnosed patients have non-metastatic disease that is unresectable due to the location of the primary tumor or its relationship to the surrounding vasculature. The first line of treatment for patients with unresectable tumors is chemotherapy. For
19
those patients whose tumors remain unresectable following chemotherapy, SBRT is an emerging treatment option. Even with SBRT as an option, patients with pancreatic cancer often have a poor prognosis, with a five-year survival rate of only approximately 10%. As a result, there remains a large unmet need to increase the effectiveness of disease management and ultimately improve outcomes for patients.
Non-Small Cell Lung Cancer Overview
According to the NCI, lung cancer is the leading cause of cancer-related mortality in the United States. The NCI estimates that in 2023 there will be approximately 238,000 new cases of lung cancer (both NSCLC and small cell lung cancer) in the United States and approximately 127,000 deaths. Approximately 195,000 patients will be diagnosed with NSCLC in the United States in 2023 and are typically treated with some combination of surgery, radiotherapy, chemotherapy and immunotherapy, depending on the severity of their disease, and SBRT is an established radiotherapy treatment for some forms of NSCLC. Even with all these current treatment options, NSCLC remains the leading cause of cancer deaths in the United States. As such, improving the effectiveness of lung cancer treatment and improving patient outcomes represents a significant unmet need.
Our Solution: Rucosopasem (GC4711) for Increasing Anti-Cancer Efficacy in Patients Receiving SBRT
Rucosopasem is our second dismutase mimetic product candidate. We are specifically developing rucosopasem, an analog of avasopasem, with the goal of increasing the anti-cancer efficacy of SBRT. Based on our extensive preclinical data and positive data from our proof-of-concept pilot LAPC trial, we believe rucosopasem has the potential to increase the anti-cancer efficacy of radiotherapy. By adding rucosopasem to an SBRT regimen, we believe that our dismutase mimetics’ conversion of superoxide to hydrogen peroxide may increase the anti-cancer efficacy of radiotherapy at current doses.
Phase 1 Trials
In December 2017, we completed a Phase 1 single-dose trial of intravenously administered rucosopasem in Australia. In March 2020, we completed a second Phase 1 single-ascending dose and multiple-dose trial of rucosopasem administered by 15-minute intravenous infusions to healthy volunteers in Australia.
In these trials, rucosopasem was observed to be well tolerated. There were no Grade 3, 4, or 5 adverse events, and no adverse events led to withdrawal from these trials. We used the results of these trials to identify the dose and schedule of rucosopasem to be studied in future trials and to support an Investigational New Drug Application, or IND, submission to initiate clinical trials evaluating intravenous rucosopasem delivered via 15-minute infusion.
Preclinical Results
We have observed in multiple xenograft and syngeneic tumor mouse models a strong correlation between the daily dose of radiation and the increase in anti-cancer efficacy with our selective dismutase mimetics. Notably, we observed that many of the mice at the highest daily dose of radiotherapy with a dismutase mimetic became tumor-free. The results of one such study, in which mice bearing NSCLC xenograft tumors received 24 mg/kg of avasopasem daily for five days concurrent with one of four different radiotherapy dosage regimens, are depicted below. For example, 5 Gy x 5 RT indicates that the mice received five daily doses of five Gy each. These radiotherapy regimens were selected because, without the addition of our dismutase mimetic, each should produce an equivalent reduction in tumor growth. The data reflects that expected result, but the increase in anti-cancer efficacy with addition of the dismutase mimetic increases significantly at the higher daily doses of radiotherapy.
20
H1299 Human NSCLC Tumors in Mice

In another preclinical study, mice bearing pancreatic cancer xenograft tumors treated with a single 12 Gy dose demonstrated a meaningful decrease in tumor volume when avasopasem was added, as depicted below. We believe that this result shows that our dismutase mimetics have the potential to synergize with SBRT to rapidly convert superoxide to hydrogen peroxide and exploit cancer cells’ increased sensitivity to hydrogen peroxide to promote cancer cell death.
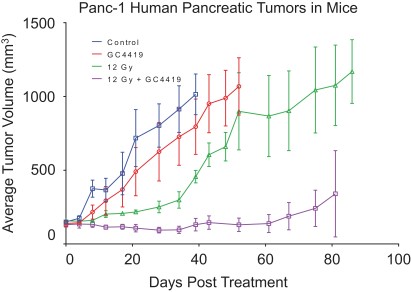
Additional preclinical studies have provided further evidence supporting our dismutase mimetics’ biological mechanism in combination with radiotherapy in solid tumors. To test the hypothesis that our dismutase mimetics’ conversion of superoxide to hydrogen peroxide increases the anti-cancer efficacy of radiotherapy, we genetically engineered NSCLC tumors to overexpress catalase enzyme when triggered. This overexpression of catalase, a native enzyme that rapidly removes hydrogen peroxide, blocked the dismutase mimetic’s synergy with radiotherapy in an experiment similar to the ones described above.
21

We believe the results of our studies represent significant potential in the treatment of cancer, particularly as recent advances in radiotherapy, such as SBRT, are capable of administering targeted, high daily doses of radiotherapy to solid tumors. SBRT utilizes several beams of various intensities aimed at different angles to precisely target the tumor, with the goal of delivering the highest possible dose of radiotherapy to kill cancer cells while minimizing exposure to normal cells. For example, SBRT is an established radiotherapy treatment for NSCLC, used increasingly for small, peripheral lung tumors. Data to date suggest that SBRT could also increase the anti-cancer efficacy and safety of radiotherapy for many other patients with NSCLC, LAPC and other cancers. SBRT application for large or centrally located NSCLC tumors, however, faces unique challenges, as lung and other toxicities limit the amount of radiotherapy patients can tolerate. As such, the most suitable patients for this procedure currently are those with smaller, well-defined tumors who are ineligible for or cannot tolerate surgery.
The increase in anti-cancer efficacy of SBRT with our dismutase mimetics has been shown in a variety of models of lung, pancreatic, head and neck, breast and other cancers. In addition, because low oxygen levels typically found deep in larger tumors can interfere with the anti-cancer efficacy of radiotherapy, it is important that our dismutase mimetics appear to also increase anti-cancer efficacy in hypoxic tumor models. Further, they may also reduce the normal tissue toxicities that restrict the patients now eligible for SBRT. Because of this we believe that the combination of rucosopasem and SBRT has the potential to further increase the anti-cancer efficacy of and to broaden the group of patients who can benefit from SBRT.
The clinical research community is also exploring the possibility of increasing the anti-cancer efficacy of SBRT by combining it with checkpoint inhibitor immunotherapy, merging the targeted efficacy of radiotherapy with the demonstrated durability of checkpoint therapy. In preclinical models combining our dismutase mimetics with SBRT and anti-PD-1, anti-PD-L1 or anti-CTLA4 checkpoint therapy, this triple combination was more effective than combinations of SBRT combined with checkpoint therapy or SBRT combined with dismutase mimetic. The triple combination increased control of the irradiated primary tumors and also appeared to reduce the metastatic spread of the cancer and even controlled pre-existing tumors outside the radiation field. Based upon these data, we believe there is an opportunity to assess the combination of SBRT, checkpoint therapy and rucosopasem as a novel approach to treating various cancers.
Clinical Development for Increasing Anti-Cancer Efficacy
Phase 1/2 Pilot Trial of Avasopasem in Patients with LAPC
In September 2021, we announced final results from a pilot Phase 1/2 safety and anti-cancer efficacy trial of avasopasem in combination with SBRT in patients with unresectable or borderline resectable LAPC. The primary objective of this trial was to determine the maximum tolerated daily dose of SBRT in conjunction with our dismutase mimetic, with secondary measures assessing, among others, OS, PFS, resectability and overall response rate compared to placebo. The trial was designed to evaluate three dose levels of SBRT, with each patient receiving five doses of SBRT. SBRT daily dose levels ranged from 10 Gy/dose to 12 Gy/dose.
22
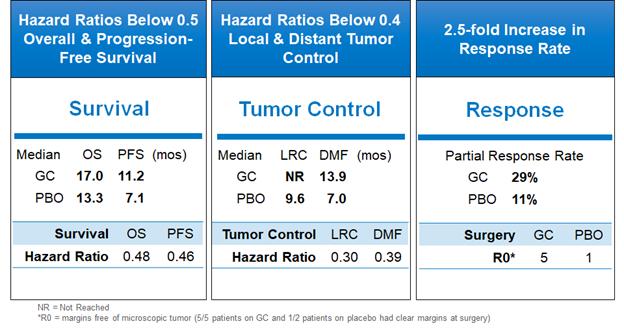
The results included a minimum follow up of one year on all 42 patients enrolled in the trial. In this proof-of-concept trial, relative improvements were observed in OS, PFS, local tumor control and time to distant metastases. 46% of patients in the active arm were alive at last follow-up (11 out of 24) compared to 33% in the placebo arm (6 out of 18). 29% of patients in the active arm achieved a 30% or greater decrease in primary tumor size (partial response) compared to 11% of patients in the placebo arm. Avasopasem was well tolerated, with similar rates of early and late adverse events in the active and placebo arms. The data from this trial enabled us to select the SBRT regimen for our subsequent trial in this indication, the GRECO-2 trial, of five daily doses at 10 Gy/dose.
Improvements Across All Efficacy Parameters
Ongoing Phase 1/2 Trial of rucosopasem in Patients with NSCLC (GRECO-1 Trial)
In October 2020, we initiated a Phase 1/2 trial of rucosopasem in combination with SBRT in patients with NSCLC, which we refer to as the GRECO-1 trial. The GRECO-1 trial is supported in part by a Small Business Innovation Research grant from the NCI for the investigation of our dismutase mimetics in combination with SBRT for the treatment of lung cancer. We intend for this trial to assess the anti-cancer efficacy and safety of rucosopasem in combination with SBRT. Subsequently, in a separate trial, we intend to assess the anti-cancer efficacy and safety of rucosopasem in combination with SBRT and a checkpoint inhibitor.
The trial was designed to enroll approximately five patients with locally advanced NSCLC as part of the Phase 1 open-label safety run-in portion of the trial. Patients received 100 mg of rucosopasem with SBRT over five consecutive weekdays. Following the safety run-in cohort, up to 66 NSCLC patients with locally advanced disease will receive 100 mg of rucosopasem with SBRT or placebo with SBRT over five consecutive weekdays in the randomized, blinded, placebo-controlled Phase 2 portion of trial.
The primary objective of the trial is to assess safety with secondary measures assessing, among others, objective response rate, PFS and OS.
In June 2022, we reported interim results from the Phase 1 open-label stage of the trial with six months follow-up on all seven patients enrolled. Rucosopasem in combination with SBRT appeared to be well tolerated through the cutoff date of June 14, 2022. The most frequent adverse events were fatigue, cough, and nausea, which are common in patients with lung cancer receiving radiotherapy. Through six months, in-field partial responses were observed in three patients and stable disease was observed in three others based on RECIST criteria. These results
23
include target tumor reductions in five patients of 61%, 58%, 33%, 29% and 27% and one patient with an 8% increase as of the cutoff date. Preservation of pulmonary lung function was also observed compared to expectations based on review of historical literature evaluating pulmonary function in a similar patient population with SBRT alone.
We expect to complete enrollment in the randomized, placebo-controlled Phase 2 stage of the GRECO-1 trial in the second half of 2023.
Ongoing Phase 2b Trial of rucosopasem in Patients with LAPC (GRECO-2 Trial)
In May 2021, we initiated a randomized, double-blinded, multicenter, placebo-controlled Phase 2b trial of rucosopasem in combination with SBRT in patients with LAPC, which we refer to as the GRECO-2 trial. We expect to enroll approximately 160 patients in this trial.
The primary objective of this trial is to determine the impact on OS of adding 100 mg of rucosopasem to SBRT following chemotherapy in patients with unresectable or borderline resectable nonmetastatic pancreatic cancer. Key secondary objectives, among others, will include PFS, locoregional tumor control, time to distant metastases, surgical resection outcomes, and objective response rate. We expect to complete enrollment in the GRECO-2 trial in the second half of 2023.
Oral and Other Formulations
Preclinical studies conducted by us suggest that rucosopasem and other novel dismutase mimetics in our portfolio can also be delivered by other routes of administration beyond intravenous, and one or more of these may be considered for future development.
Manufacturing
We do not own or operate, and currently have no plans to establish, any manufacturing facilities. We currently rely, and expect to continue to rely, on third party contract manufacturing organizations, or CMOs, for the supply of current good manufacturing practice-, or cGMP-, grade clinical trial materials and commercial quantities of our product candidates and products, if approved. We require all of our CMOs to conduct manufacturing activities in compliance with cGMP requirements, and we maintain our product candidates in refrigerated conditions prior to intravenous infusion. We have assembled a team of experienced employees and consultants to provide the necessary technical, quality and regulatory oversight of our CMOs.
We anticipate that these CMOs will have the capacity to support both clinical supply and commercial-scale production. We have a formal agreement with Patheon Manufacturing Services LLC, or Patheon, for commercial production of avasopasem, if approved. See “Management’s Discussion and Analysis of Financial Condition and Results of Operations-Liquidity and Capital Resources-Patheon Manufacturing Agreements” in Part II, Item 7 of this Annual Report on Form 10-K.
We also may elect to pursue additional CMOs for manufacturing supplies of drug substance and finished drug product in the future. We believe that our standardized manufacturing process can be transferred to a number of other CMOs for the production of clinical and commercial supplies of our product candidates in the ordinary course of business.
Commercialization
Our aim is to become a fully integrated biopharmaceutical company. At our current stage, we have a small commercial organization but have not yet established sales or distribution capabilities. We intend to commercialize avasopasem, if approved, by expanding our commercial organization by building a specialized sales and marketing organization in the United States focused on radiation and medical oncologists, who are the primary decision makers in the prescribing and/or infusion of therapies in patients with HNC undergoing standard-of-care treatment.
24
We believe a scientifically oriented, customer-focused team of approximately 40 sales representatives would allow us to effectively reach the approximately 5,000 radiation oncologists in the United States, who treat patients using an even smaller number of radiation machines. There are approximately 2,500 radiotherapy treatment sites in the United States. Based on a third-party claims database, we estimate that over 80% of radiotherapy treatments for HNC patients are performed at approximately 700 sites. Most of these treatment sites are hospitals and the majority have the ability to administer intravenous infusion therapies either in the radiation or medical oncology suite and/or in the hospital outpatient infusion department. In the hospital setting, infusions are often administered on the same floor or in the same building where the radiation therapy is delivered.
We expect avasopasem to be reimbursed as an outpatient therapy through a “buy-and-bill” model of reimbursement under Medicare Part B or a medical benefit under commercial insurance. Under the “buy-and-bill” model, institutions or providers will buy the drug and then submit a claim for reimbursement from the payer once the drug has been administered to the patient. Hospital care data suggests the payer mix for patients with HNC to be approximately 50% Medicare, 30% commercial, 12% Medicaid, 3% self-pay and 5% other (which may include payers such as the U.S. Department of Veteran Affairs, TRICARE and workers compensation). Data also suggests that a majority of the hospital treatment sites are eligible for the 340B Drug Pricing Program.
Because of the limited number of physicians concentrated around a smaller number of radiation machines, we believe we can effectively commercialize avasopasem, if approved, in the United States with a small, focused commercial organization. We also expect to leverage this sales organization to commercialize rucosopasem, if approved, and any of our future product candidates in the United States. We may seek to establish collaborations for the commercialization of avasopasem, rucosopasem, and our other product candidates in the United States and in other regions such as Europe and Asia.
Competition
The biotechnology and pharmaceutical industries put significant emphasis and resources into the development of novel and proprietary therapies for cancer treatment. While we believe that our knowledge, experience and scientific resources provide us with competitive advantages, we face potential competition from many different sources, including large and specialty pharmaceutical and biotechnology companies, academic research institutions and governmental agencies and public and private research institutions. Any product candidates that we successfully develop and commercialize will compete with existing treatment options and new therapies that may become available in the future.
Many of our potential competitors may have significantly greater financial resources, a more established presence in the market, and more expertise in research and development, manufacturing, preclinical and clinical testing, obtaining regulatory approvals and reimbursement, and marketing approved products than we do. Mergers and acquisitions in the pharmaceutical, biotechnology and diagnostic industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller or early-stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These potential competitors may also compete with us in recruiting and retaining top qualified scientific, sales, marketing and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
The key competitive factors affecting the success of avasopasem, rucosopasem and any of our other product candidates, if approved, are likely to be their efficacy, safety, convenience, price, the level of generic competition and the availability of reimbursement from government and other third-party payors. There are currently no FDA-approved drugs for the treatment of OM in patients with HNC and no FDA-approved drugs or established guidelines for the treatment of radiotherapy-induced esophagitis.
A number of large pharmaceutical and biotechnology companies that currently market and sell drugs or biologics are pursuing the development of therapies in the fields in which we are interested. Our commercial opportunity for any of our product candidates could be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, less expensive, more convenient or easier to administer, or have fewer or less severe side effects, than any products that we may develop. Because our product candidates are designed to reduce the side effects, or to increase the anti-cancer efficacy, of radiotherapy, our commercial
25
opportunities could also be reduced or eliminated if radiotherapy methods are improved in a way that reduces the incidence of such side effects or increases anti-cancer efficacy, or if new therapies are developed which effectively treat cancer without causing such side effects. Our competitors also may obtain FDA, EMA or other regulatory approval for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we are able to enter the market.
Intellectual Property
Our commercial success depends in part on our ability to obtain and maintain proprietary protection for avasopasem, rucosopasem and any of our other product candidates, manufacturing methods and processes, novel discoveries, and other know-how; to operate without infringing on or otherwise violating the proprietary rights of others; and to prevent others from infringing or otherwise violating our proprietary rights. Our policy is to seek to protect our proprietary position by, among other methods, filing or in-licensing U.S. and foreign patents and patent applications related to our product candidates and other proprietary technologies, inventions and improvements, including claims related to composition of matter and methods of use, that are important to the development and implementation of our business. We also rely on trademarks, trade secrets, know-how, continuing technological innovation and potential in-licensing opportunities to develop and maintain our proprietary position. For more information, please see “Risk Factors—Risks Related to Intellectual Property.”
Patents and Patent Applications
As of December 31, 2022, our owned and currently pending and/or in-force patent portfolio consisted of approximately 18 issued U.S. patents, 12 pending U.S. patent applications, 112 issued foreign patents including 4 issued European patents that have been validated in many European countries, and 107 pending foreign applications.
The term of individual patents depends upon the legal term for patents in the countries in which they are obtained. In most countries in which we file, including the United States, the patent term is 20 years from the earliest filing date of a non-provisional patent application. In the United States, a patent’s term may be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the USPTO, in examining and granting a patent, or may be shortened if a patent is terminally disclaimed over an earlier expiring patent. In some instances, such a patent term adjustment may result in the term of a United States patent extending beyond 20 years from the earliest filing date of a non-provisional patent application. In the United States, the term of a patent that covers a drug product may also be eligible for patent term extension when regulatory approval is granted, provided the legal requirements are met. This permits patent term restoration as compensation for the patent term lost during the FDA regulatory review process. The Hatch-Waxman Act permits a patent term extension of up to a maximum of five years beyond the expiration of the patent if the patent is eligible for such an extension under the Hatch-Waxman Act. The length of the patent term extension is related to the length of time the drug is under regulatory review; however, it cannot extend the remaining term of a patent beyond a total of 14 years from the date of product approval. For patents that might expire during the application phase, the patent owner may request an interim patent extension. An interim patent extension increases the patent term by one year and may be renewed up to four times. For each interim patent extension granted, the post-approval patent extension is reduced by one year. The director of the USPTO must determine that approval of the drug covered by the patent for which a patent extension is being sought is likely. Interim patent extensions are not available for a drug for which an NDA has not been submitted. Only one patent applicable to an approved drug may be extended. Similar provisions are available in Europe and certain other jurisdictions to extend the term of a patent that covers an approved drug. In the future, if and when our drug candidates receive approval by the FDA or foreign regulatory authorities, we expect to apply for patent term extensions on issued patents covering those drugs, depending upon the length of the clinical trials for each drug and other factors.
The two most advanced product candidates in our portfolio, avasopasem and rucosopasem, are protected by issued patents with claims directed to composition of matter and method of use. Avasopasem is covered by a composition of matter patent in the United States that had a natural expiration date in March 2022. The U.S. patent family covering the method of treating OM has a natural expiration date in late 2027, and if we are successful in obtaining a patent term extension of approximately two and a half years which we believe should be available, the extension would result in an expiration date in 2031. The U.S. patent family covering treating tissue damage resulting from radiation therapy, chemotherapy or a combination thereof by administering high doses of
26
avasopasem, including that tested in the ROMAN Phase 3 trial, has a natural expiration date in 2032, and if we are successful in obtaining a patent term extension which we believe should be available, the extension would result in an expiration date in late 2034 to early 2035. In any event, we can only extend one applicable patent for each approved drug. Rucosopasem is covered by a composition of matter patent in the United States, which also covers oral bioavailability of the product candidate, and has a natural expiration date in 2036. However, we believe the rucosopasem composition of matter patent may be eligible for a patent term extension of at least about two years which, if granted, would result in an expiration date in 2038. When including only current issued patents and related potential patent term extensions, our product candidate patent portfolio is projected to expire between 2031 and 2038 in the United States. Additional pending or future patent applications may supplement or extend this patent portfolio.
However, there can be no assurance that any of our pending patent applications will issue or that we will benefit from any patent term extension or favorable adjustment to the term of any of our patents. The applicable authorities, including the FDA in the United States, may not agree with our assessment of whether such patent term extensions should be granted, and if granted, they may grant more limited extensions than we request.
We also have pending patent families in the United States that cover certain combinations of our product candidates with several oncology products and therapies that may provide protection for the use of our product candidates in connection with those oncology products and therapies, which, if granted, are projected to expire between 2037 and 2041.
Trademarks and Trade Secrets
As of December 31, 2022, our owned and currently pending and/or in-force trademark portfolio consisted of 3 registered U.S. trademarks, 9 pending U.S. trademark applications, 26 registered foreign trademarks, and 9 pending foreign trademark applications.
Furthermore, we rely upon trade secrets, know-how, continuing technological innovation and potential in-licensing opportunities to develop and maintain our competitive position. We seek to protect our proprietary information, in part, using confidentiality and invention assignment agreements with our commercial partners, collaborators, employees, and consultants. These agreements are designed to protect our proprietary information and, in the case of the invention assignment agreements, to grant us ownership of technologies that are developed through a relationship with an employee or a third party. These agreements may be breached, and we may not have adequate remedies for any such breach. In addition, our trade secrets may otherwise become known or be independently discovered by competitors. To the extent that our commercial partners, collaborators, employees, and consultants use intellectual property owned by others in their work for us, disputes may arise as to the rights in related or resulting know-how and inventions.
Royalty Agreement with Blackstone Life Sciences (Formerly Known as Clarus Ventures)
In November 2018, we entered into the Royalty Agreement with Blackstone Life Sciences. Pursuant to the Royalty Agreement, Blackstone agreed to pay us, in the aggregate, up to $80.0 million, or the Royalty Purchase Price, in four tranches of $20.0 million each upon the achievement of specified clinical milestones in our ROMAN trial. We agreed to apply the proceeds from such payments primarily to support clinical development and regulatory activities for avasopasem, rucosopasem and any pharmaceutical product comprising or containing avasopasem or rucosopasem, or, collectively, the Products, as well as to satisfy working capital obligations and for general corporate expenses. We received the first tranche of the Royalty Purchase Price in November 2018, the second tranche of the Royalty Purchase Price in April 2019, and the third tranche of the Royalty Purchase Price in February 2020, in each case in connection with the achievement of the first three milestones, respectively, under the Royalty Agreement.
In May 2020, we entered into Amendment No. 1 to the Royalty Agreement, or the Amendment, with Clarus IV Galera Royalty AIV, L.P., or the Blackstone Purchaser. The Blackstone Purchaser is affiliated with Blackstone Life Sciences, successor in interest to Clarus Ventures. The Amendment increased the Royalty Purchase Price by $37.5 million to $117.5 million by increasing the fourth tranche from $20.0 million to $37.5 million and adding a new $20.0 million tranche upon the achievement of an additional clinical enrollment milestone. We
27
received the new $20.0 million tranche of the Amendment in June 2021, in connection with the enrollment of the first patient in the GRECO-2 trial. Also in June 2021, we completed enrollment in the ROMAN trial, thereby achieving the milestone associated with the fourth tranche, and received the associated $37.5 million in July 2021.
Pursuant to the amended Royalty Agreement, in connection with the payment of each tranche of the Royalty Purchase Price, we have agreed to sell, convey, transfer and assign to Blackstone all of our right, title and interest in a high single-digit percentage of (i) worldwide net sales of the Products and (ii) all amounts received by us or our affiliates, licensees and sublicensees with respect to Product-related damages (collectively, the Product Payments) during the Royalty Period. The Royalty Period means, on a Product-by-Product and country-by-country basis, the period of time commencing on the commercial launch of such Product in such country and ending on the latest to occur of (i) the 12th anniversary of such commercial launch, (ii) the expiration of all valid claims of our patents covering such Product in such country, and (iii) the expiration of regulatory data protection or market exclusivity or similar regulatory protection afforded by the health authorities in such country, to the extent such protection or exclusivity effectively prevents generic versions of such Product from entering the market in such country.
The amended Royalty Agreement will remain in effect until the date on which the aggregate amount of the Product Payments paid to Blackstone exceeds a fixed single-digit multiple of the actual amount of the Royalty Purchase Price received by us, unless earlier terminated pursuant to the mutual written agreement of us and Blackstone. If no Products are commercialized, we would not have an obligation to make Product Payments to Blackstone, which is the sole mechanism for repaying the liability.
In May 2020, as partial consideration for the Amendment, we issued two warrants to the Blackstone Purchaser to purchase an aggregate of 550,661 shares of our common stock at an exercise price equal to $13.62 per share, each of which became exercisable upon the receipt by Galera of the applicable specified milestone payment. The issued warrants expire six years after the initial exercise date of each respective warrant.
Government Regulation
The FDA and comparable regulatory authorities in state and local jurisdictions and in other countries impose substantial and burdensome requirements upon companies involved in the clinical development, manufacture, marketing and distribution of drugs, such as those we are developing. These agencies and other federal, state and local entities regulate, among other things, the research and development, testing, manufacture, quality control, safety, effectiveness, labeling, storage, record keeping, approval, advertising and promotion, distribution, post-approval monitoring and reporting, sampling and export and import of our product candidates.
U.S. Government Regulation
In the United States, the FDA regulates drugs under the Federal Food, Drug, and Cosmetic Act, or FDCA, and its implementing regulations. The process of obtaining regulatory approvals and the subsequent compliance with appropriate federal, state, local and foreign statutes and regulations requires the expenditure of substantial time and financial resources. Failure to comply with the applicable U.S. requirements at any time during the product development process, approval process or after approval, may subject an applicant to a variety of administrative or judicial sanctions, such as the FDA’s refusal to approve pending NDAs, withdrawal of an approval, imposition of a clinical hold, issuance of warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement or civil or criminal penalties.
The process required by the FDA before a drug may be marketed in the United States generally involves the following:
28
Preclinical Studies
Preclinical studies include laboratory evaluation of product chemistry, toxicity and formulation, as well as animal studies to assess potential safety and efficacy. An IND sponsor must submit the results of the preclinical studies, together with manufacturing information, analytical data and any available clinical data or literature, among other things, to the FDA as part of an IND. Some preclinical studies may continue even after the IND is submitted. An IND automatically becomes effective 30 days after receipt by the FDA, unless before that time the FDA raises concerns or questions related to one or more proposed clinical trials and places the clinical trial on a clinical hold. In such a case, the IND sponsor and the FDA must resolve any outstanding concerns before the clinical trial can begin. As a result, submission of an IND may not result in the FDA allowing clinical trials to commence.
Clinical Trials
Clinical trials involve the administration of the investigational new drug to human subjects under the supervision of qualified investigators in accordance with GCP requirements, which include the requirement that all research subjects provide their informed consent in writing for their participation in any clinical trial. Clinical trials are conducted under protocols detailing, among other things, the objectives of the trial, the parameters to be used in monitoring safety, and the effectiveness criteria to be evaluated. A protocol for each clinical trial and any subsequent protocol amendments must be submitted to the FDA as part of the IND. In addition, an IRB at each institution participating in the clinical trial must review and approve the plan for any clinical trial before it commences at that institution. Information about certain clinical trials must be submitted within specific timeframes to the NIH for public dissemination on their www.clinicaltrials.gov website.
Human clinical trials are typically conducted in three sequential phases, which may overlap or be combined:
29
Progress reports detailing the results of the clinical trials must be submitted at least annually to the FDA and more frequently if serious adverse events occur. Phase 1, Phase 2 and Phase 3 clinical trials may not be completed successfully within any specified period, or at all. Furthermore, the FDA or the sponsor may suspend or terminate a clinical trial at any time on various grounds, including a finding that the research subjects are being exposed to an unacceptable health risk. Similarly, an IRB can suspend or terminate approval of a clinical trial at its institution if the clinical trial is not being conducted in accordance with the IRB’s requirements or if the drug has been associated with unexpected serious harm to patients.
Marketing Approval
Assuming successful completion of the required clinical testing, the results of the preclinical studies and clinical trials, together with detailed information relating to the product’s chemistry, manufacture, controls and proposed labeling, among other things, are submitted to the FDA as part of an NDA requesting approval to market the product for one or more indications. In most cases, the submission of an NDA is subject to a substantial application user fee.
In addition, under the Pediatric Research Equity Act of 2003, as amended and reauthorized, certain NDAs or supplements to an NDA must contain data that are adequate to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations, and to support dosing and administration for each pediatric subpopulation for which the product is safe and effective. The FDA may, on its own initiative or at the request of the applicant, grant deferrals for submission of some or all pediatric data until after approval of the product for use in adults, or full or partial waivers from the pediatric data requirements.
The FDA conducts a preliminary review of all NDAs within the first 60 days after submission, before accepting them for filing, to determine whether they are sufficiently complete to permit substantive review. The FDA may request additional information rather than accept an NDA for filing. In this event, the application must be resubmitted with the additional information. The resubmitted application is also subject to review before the FDA accepts it for filing. Once the submission is accepted for filing, the FDA begins an in-depth substantive review. The FDA reviews an NDA to determine, among other things, whether the drug is safe and effective and whether the facility in which it is manufactured, processed, packaged or held meets standards designed to assure the product’s continued safety, quality and purity. Under the PDUFA guidelines that are currently in effect, the FDA has a goal of ten months from the date of “filing” of a standard NDA for a new molecular entity to review and act on the submission. This review typically takes twelve months from the date the NDA is submitted to FDA because the FDA has approximately two months to make a “filing” decision. The actual review time may be significantly longer, depending on the complexity of the review, FDA requests for additional information and the sponsor’s submission of additional information.
The FDA may refer an application for a novel drug to an advisory committee. An advisory committee is a panel of independent experts, including clinicians and other scientific experts, that reviews, evaluates and provides a recommendation as to whether the application should be approved and under what conditions. The FDA is not bound by the recommendations of an advisory committee, but it considers such recommendations carefully when making decisions.
30
Before approving an NDA, the FDA typically will inspect the facility or facilities where the product is manufactured. The FDA will not approve an application unless it determines that the manufacturing processes and facilities are in compliance with cGMP requirements and adequate to assure consistent production of the product within required specifications. Additionally, before approving an NDA, the FDA may inspect one or more clinical trial sites to assure compliance with GCP requirements.
After evaluating the NDA and all related information, including the advisory committee recommendation, if any, and inspection reports regarding the manufacturing facilities and clinical trial sites, the FDA may issue an approval letter, or, in some cases, a complete response letter. A complete response letter generally contains a statement of specific conditions that must be met in order to secure final approval of the NDA and may require additional clinical trials or preclinical studies in order for FDA to reconsider the application. Even with submission of this additional information, the FDA ultimately may decide that the application does not satisfy the regulatory criteria for approval. If and when those conditions have been met to the FDA’s satisfaction, the FDA will typically issue an approval letter. An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications.
Even if the FDA approves a product, it may limit the approved indications for use of the product, require that contraindications, warnings or precautions be included in the product labeling, require that post-approval studies, including Phase 4 clinical trials, be conducted to further assess a drug’s safety after approval, require testing and surveillance programs to monitor the product after commercialization, or impose other conditions, including distribution and use restrictions or other risk management mechanisms under a REMS, which can materially affect the potential market and profitability of the product. The FDA may prevent or limit further marketing of a product based on the results of post-marketing studies or surveillance programs. After approval, some types of changes to the approved product, such as adding new indications, manufacturing changes, and additional labeling claims, are subject to further testing requirements and FDA review and approval.
FDA Expedited Programs
The FDA offers a number of expedited development and review programs for qualifying product candidates. To be eligible for a fast track designation, the FDA must determine, based on the request of a sponsor, that a product candidate is intended to treat a serious or life-threatening disease or condition and demonstrates the potential to address an unmet medical need for such disease or condition. The sponsor of a fast track-designated product candidate has opportunities for more frequent interactions with the applicable FDA review team during product development. In addition, the FDA may review sections of the NDA for a fast track-designated product candidate on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the NDA, the FDA agrees to accept sections of the NDA and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the NDA.
The FDA may give a priority review designation to NDAs for drugs that offer major advances in treatment or provide a treatment where no adequate therapy exists. A priority review means that the goal for the FDA to review an application is six months, rather than the standard review of ten months under current PDUFA guidelines. Under the current PDUFA agreement, these six and ten month review periods are measured from the “filing” date rather than the receipt date for NDAs for new molecular entities, which typically adds approximately two months to the timeline for review and decision from the date of submission.
In addition, products studied for their safety and effectiveness in treating serious or life-threatening illnesses and that provide meaningful advantages over existing treatments may be eligible for accelerated approval and may be approved upon a determination that the product candidate has an effect on a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity or prevalence of the condition and the availability or lack of alternative treatments. As a condition of approval, the FDA may require a sponsor of a drug receiving accelerated approval to perform post-marketing studies to verify and describe the predicted effect on irreversible morbidity or mortality or other clinical endpoint, and the drug may be subject to accelerated withdrawal procedures if the sponsor fails to conduct the required post-marketing studies in a timely manner or if such studies fail to verify the predicted clinical benefit. In addition, the FDA currently requires as a condition for accelerated approval pre-approval of promotional materials, which could adversely impact the timing of the commercial launch of the product.
31
The FDA may also designate a product candidate as a breakthrough therapy, which is defined as a drug that is intended, alone or in combination with one or more other drugs or biologics, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The designation includes all of the fast track program features, as well as more intensive FDA interaction and guidance beginning as early as Phase 1 and an organizational commitment to expedite the development and review of the product candidate, including involvement of senior managers.
Fast track designation, breakthrough therapy designation, priority review, and accelerated approval do not change the standards for approval and approval is not guaranteed. Such designation may, however, expedite the development or approval process. Even if a product qualifies for one or more of these programs, the FDA may later decide that the product no longer meets the conditions for qualification or decide that the time period for FDA review or approval will not be shortened. We may explore some of these opportunities for our product candidates as appropriate.
Post-Approval Requirements
Drugs manufactured or distributed pursuant to FDA approvals are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims are subject to prior FDA review and approval. There also are continuing annual user fee requirements for any marketed products and the establishments at which such products are manufactured, as well as new application fees for supplemental applications with clinical data.
The FDA may impose a number of post-approval requirements as a condition of approval of an NDA. For example, the FDA may require post-marketing testing, including Phase 4 clinical trials, and surveillance to further assess and monitor the product’s safety and effectiveness after commercialization.
In addition, drug manufacturers and other entities involved in the manufacture and distribution of approved drugs are required to register their establishments with the FDA and state agencies and are subject to periodic unannounced inspections by the FDA and these state agencies for compliance with cGMP requirements. Changes to the manufacturing process are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from cGMP requirements and impose reporting and documentation requirements upon the sponsor and any third-party manufacturers that the sponsor may decide to use. Accordingly, manufacturers must continue to expend time, money, and effort in the area of production and quality control to maintain cGMP compliance.
Once an approval is granted, the FDA may withdraw the approval if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in mandatory revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. Other potential consequences include, among other things:
32
The FDA strictly regulates marketing, labeling, advertising and promotion of products that are placed on the market. Drugs may be promoted only for the approved indications and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant liability. Physicians may prescribe legally available products for uses that are not described in the product’s labeling and that differ from those tested by us and approved by the FDA. Such off-label uses are common across medical specialties. Physicians may believe that such off-label uses are the best treatment for many patients in varied circumstances. The FDA does not regulate the behavior of physicians in their choice of treatments. The FDA does, however, restrict manufacturer’s communications on the subject of off-label use of their products.
In addition, the distribution of prescription pharmaceutical products is subject to the Prescription Drug Marketing Act, or PDMA, which regulates the distribution of drugs and drug samples at the federal level and sets minimum standards for the registration and regulation of drug distributors by the states. Both the PDMA and state laws limit the distribution of prescription pharmaceutical product samples and impose requirements to ensure accountability in distribution.
The Hatch-Waxman Act and Marketing Applications for Follow-On Drugs
In 1984, with passage of the Hatch-Waxman Amendments to the FDCA, Congress authorized the FDA to approve generic drugs that are the same as drugs previously approved by the FDA under the NDA provisions of the statute and also enacted Section 505(b)(2) of the FDCA. To obtain approval of a generic drug, an applicant must submit an abbreviated new drug application, or ANDA, to the agency. In support of such applications, a generic manufacturer may rely on the preclinical and clinical testing conducted for a drug product previously approved under an NDA, known as the reference listed drug, or RLD. Specifically, in order for an ANDA to be approved, the FDA must find that the generic version is identical to the RLD with respect to the active ingredients, the route of administration, the dosage form, and the strength of the drug. At the same time, the FDA must also determine that the generic drug is “bioequivalent” to the innovator drug.
Orange Book Listing
In seeking approval for a drug through an NDA, applicants are required to list with the FDA each patent that has claims that cover the applicant’s product or method of therapeutic use. Upon approval of a drug, each of the patents listed in the application for the drug is then published in the FDA’s Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the Orange Book. If an applicable patent issues between filing and approval, the applicant is required to amend the application to include that patent. Companies also may list applicable patents in the Orange Book after receiving product approval so long as the patent is submitted to FDA within 30 days of patent issuance. Drugs listed in the Orange Book can, in turn, be cited by potential generic competitors in support of approval of an ANDA. The ANDA requests permission to market a drug product that has the same active ingredients in the same strengths and dosage form as the RLD and has been shown through bioequivalence testing to be therapeutically equivalent to the RLD. Other than the requirement for bioequivalence testing, ANDA applicants are not required to conduct, or submit results of, nonclinical or clinical tests to prove the safety or effectiveness of their drug product. Drugs approved in this way are commonly referred to as “generic equivalents” to the innovator drug and can often be substituted by pharmacists under prescriptions written for the original listed drug referenced by the ANDA applicant if the FDA’s listing for the generic drug in the Orange Book indicates that it is “therapeutically equivalent” to the RLD.
In contrast, Section 505(b)(2) permits the filing of an NDA where at least some of the information required for approval comes from studies not conducted by or for the applicant and for which the applicant has not obtained a right of reference. A Section 505(b)(2) applicant may eliminate the need to conduct certain preclinical or clinical studies if it can establish that reliance on studies conducted for a previously approved product is scientifically appropriate. Unlike the ANDA pathway used by developers of bioequivalent versions of innovator drugs, which does not allow applicants to submit new clinical data other than bioavailability or bioequivalence data, the 505(b)(2) regulatory pathway does not preclude the possibility that a follow-on applicant would need to conduct additional clinical trials or nonclinical studies; for example, it may be seeking approval to market a previously
33
approved drug for new indications or for a new patient population that would require new clinical data to demonstrate safety or effectiveness.
When an ANDA applicant submits its application to the FDA, it is required to certify to the FDA concerning any patents listed for the approved product in the FDA’s Orange Book. Specifically, the applicant must certify that: (i) the required patent information has not been filed; (ii) the listed patent has expired; (iii) the listed patent has not expired but will expire on a particular date and approval is sought after patent expiration; or (iv) the listed patent is invalid or will not be infringed by the new product. The ANDA applicant may also elect to submit a section viii statement, certifying that its proposed ANDA label does not contain or carves out any language regarding the patented method-of-use, rather than certify to a listed method-of-use patent. Moreover, to the extent that the Section 505(b)(2) NDA applicant is relying on studies conducted for an already approved product, the applicant also is required to certify to the FDA concerning any patents listed for the approved product in the Orange Book to the same extent that an ANDA applicant would.
If the applicant does not challenge the innovator’s listed patents, FDA will not approve the ANDA or 505(b)(2) application until all the listed patents claiming the referenced product have expired. A certification that the new product will not infringe the already approved product’s listed patents, or that such patents are invalid, is called a Paragraph IV certification. If the ANDA applicant has provided a Paragraph IV certification to the FDA, the applicant must also send notice of the Paragraph IV certification to the NDA and patent holders once the ANDA has been accepted for filing by the FDA. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of the Paragraph IV certification. The filing of a patent infringement lawsuit within 45 days of the receipt of a Paragraph IV certification automatically prevents the FDA from approving the ANDA until the earlier of 30 months, expiration of the patent, settlement of the lawsuit, or a decision in the infringement case that is favorable to the ANDA applicant.
An ANDA or 505(b)(2) application also will not be approved until any applicable non-patent exclusivity listed in the Orange Book for the referenced product has expired.
Non-Patent Exclusivity
Upon NDA approval of a new chemical entity or NCE, which is a drug that contains no active moiety that has been approved by the FDA in any other NDA, that drug receives five years of non-patent data exclusivity during which time the FDA cannot receive any ANDA seeking approval of a generic version of that drug. During the exclusivity period, the FDA cannot accept for review any ANDA or 505(b)(2) NDA submitted by another company for another version of such drug where the applicant does not own or have a legal right of reference to all the data required for approval. However, an application may be submitted one year before NCE exclusivity expires if a Paragraph IV certification is filed on an NCE patent and any time after approval if the application is filed based on a new indication or a new formulation.
The Hatch-Waxman Act also provides three years of data exclusivity for a NDA, 505(b)(2) NDA or supplement to an existing NDA if new clinical investigations, other than bioavailability studies, that were conducted or sponsored by the applicant are deemed by the FDA to be essential to the approval of the application, for example, new indications, dosages or strengths of an existing drug. This three-year exclusivity covers only the conditions of use associated with the new clinical investigations and does not prohibit the FDA from approving follow-on applications for drugs containing the original active agent. If there is no listed patent in the Orange Book, there may not be a Paragraph IV certification, and, thus, no ANDA or 505(b)(2) NDA may be filed before the expiration of the exclusivity period. Five-year and three-year exclusivity also will not delay the submission or approval of a traditional NDA filed under Section 505(b)(1) of the FDCA. However, an applicant submitting a traditional NDA would be required to either conduct or obtain a right of reference to all of the preclinical studies and adequate and well-controlled clinical trials necessary to demonstrate safety and effectiveness.
Other Healthcare Laws
Pharmaceutical companies are subject to additional healthcare regulation and enforcement by the federal government and by authorities in the states and foreign jurisdictions in which they conduct their business. Such laws include, without limitation, U.S. federal and state anti-kickback, fraud and abuse, false claims, consumer fraud, pricing reporting, and transparency laws and regulations with respect to drug pricing and payments and other
34
transfers of value made to physicians and other healthcare professionals, as well as similar foreign laws in the jurisdictions outside the U.S. Violations of such laws, or any other governmental regulations that apply, may result in significant penalties, including, without limitation, administrative, civil and criminal penalties, damages, fines, additional reporting and oversight obligations, the curtailment or restructuring of operations, exclusion from participation in governmental healthcare programs and individual imprisonment.
Coverage and Reimbursement
Sales of any pharmaceutical product depend, in part, on the extent to which such product will be covered by third-party payors, such as federal, state and foreign government healthcare programs, commercial insurance and managed healthcare organizations, and the level of reimbursement for such product by third-party payors. Significant uncertainty exists as to the coverage and reimbursement status of any newly approved product. Decisions regarding the extent of coverage and amount of reimbursement to be provided are made on a plan-by-plan basis. With respect to off-label uses, third-party payors may provide coverage and reimbursement under certain limited circumstances. By way of example, Medicare covers off-label uses of FDA-approved drugs if the use is supported as a medically accepted indication by certain compendia and is not otherwise listed as unsupported, not indicated, not recommended, or equivalent terms, in any such compendia. For products administered under the supervision of a physician, obtaining coverage and adequate reimbursement may be particularly difficult because of the higher prices often associated with such drugs. Additionally, separate reimbursement for the product itself or the treatment or procedure in which the product is used may not be available, which may impact physician utilization.
In addition, third-party payors are increasingly reducing reimbursements for pharmaceutical products and services. Moreover, for drugs and biologics administered under the supervision of a physician, obtaining coverage and adequate reimbursement may be particularly difficult because of the higher prices often associated with such products. The U.S. government and state legislatures have continued implementing cost-containment programs, including price controls, restrictions on coverage and reimbursement and requirements for substitution of generic products. Adoption of price controls and cost-containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit sales of any product. Decreases in third-party reimbursement for any product or a decision by a third-party payor not to cover a product could reduce physician usage and patient demand for the product.
In international markets, reimbursement and healthcare payment systems vary significantly by country, and many countries have instituted price ceilings on specific products and therapies. For example, the European Union provides options for its member states to restrict the range of medicinal products for which their national health insurance systems provide reimbursement and to control the prices of medicinal products for human use. A member state may approve a specific price for the medicinal product, or it may instead adopt a system of direct or indirect controls on the profitability of the company placing the medicinal product on the market. Products may face competition from lower-priced products in foreign countries that have placed price controls on pharmaceutical products and may also compete with imported foreign products.
Furthermore, there is no assurance that a product will be considered medically reasonable and necessary for a specific indication, be considered cost-effective by third-party payors, that an adequate level of reimbursement will be established even if coverage is available or that the third-party payors’ reimbursement policies will not adversely affect the ability for manufacturers to sell products profitably.
Healthcare Reform
In the United States and certain foreign jurisdictions, there have been, and we expect there will continue to be, a number of legislative and regulatory changes to the healthcare system that could affect the pharmaceutical industry. In March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, or collectively the ACA, was signed into law, which substantially changed the way healthcare is financed by both governmental and private insurers in the United States. The ACA contained a number of provisions, including those governing enrollment in federal healthcare programs and reimbursement adjustments. Additionally, the ACA increased the minimum level of Medicaid rebates payable by manufacturers of brand name drugs from 15.1% to 23.1% of the average manufacturer price; required collection of rebates for drugs paid by Medicaid managed care organizations; required manufacturers to participate in a coverage gap discount program,
35
under which they must agree to offer 70 percent point-of-sale discounts off negotiated prices of applicable brand drugs to eligible beneficiaries during their coverage gap period, as a condition for the manufacturer’s outpatient drugs to be covered under Medicare Part D; imposed a non-deductible annual fee on pharmaceutical manufacturers or importers who sell “branded prescription drugs” to specified federal government programs, implemented a new methodology by which rebates owed by manufacturers under the Medicaid Drug Rebate Program are calculated for drugs that are inhaled, infused, instilled, implanted, or injected; expanded the eligibility criteria for Medicaid programs, created a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research; and established a Center for Medicare and Medicaid Innovation at the Centers for Medicare & Medicaid Services, or CMS, to test innovative payment and service delivery models to lower Medicare and Medicaid spending, potentially including prescription drug spending.
Since its enactment, there have been judicial, executive and Congressional challenges to certain aspects of the ACA. On June 17, 2021, the U.S. Supreme Court dismissed the most recent judicial challenge to the ACA brought by several states without specifically ruling on the constitutionality of the ACA. In addition, other legislative changes have been proposed and adopted since the ACA was enacted, including aggregate reductions of Medicare payments to providers until 2032, which was temporarily suspended from May 1, 2020 through March 31, 2022, and further reduced payments to several types of Medicare providers. Moreover, there has recently been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several presidential executive orders. Congressional inquiries and proposed and enacted legislation designed, among other things, to bring more transparency to product pricing, review the relationship between pricing and manufacturer patient programs and reform government program reimbursement methodologies for drug products. On March 11, 2021, the American Rescue Plan Act of 2021 was signed into law, which eliminates the statutory Medicaid drug rebate cap, currently set at 100% of a drug’s average manufacturer price, or AMP, beginning January 1, 2024. More recently, on August 16, 2022, the Inflation Reduction Act of 2022, or IRA, was signed into law. Among other things, the IRA requires manufacturers of certain drugs to engage in price negotiations with Medicare (beginning in 2026), with prices that can be negotiated subject to a cap; imposes rebates under Medicare Part B and Medicare Part D to penalize price increases that outpace inflation (first due in 2023); and replaces the Part D coverage gap discount program with a new discounting program (beginning in 2025). The IRA permits the Secretary of the Department of Health and Human Services (HHS) to implement many of these provisions through guidance, as opposed to regulation, for the initial years. For that and other reasons, it is currently unclear how the IRA will be effectuated. Individual states in the United States have also become increasingly active in implementing regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures and, in some cases, mechanisms to encourage importation from other countries and bulk purchasing. Further, it is possible that additional government action is taken in response to the COVID-19 pandemic.
Foreign Regulation
In addition to regulations in the United States, we will be subject to a variety of regulations in other jurisdictions governing, among other things, clinical trials and commercial sales and distribution of our product candidates. Whether or not we obtain FDA approval for a product, we must obtain the requisite approvals from the comparable regulatory authorities in foreign countries prior to the commencement of clinical trials or marketing of the product candidates in those countries. The requirements and process governing the conduct of clinical trials, product licensing, pricing and reimbursement vary from country to country. Failure to comply with applicable foreign regulatory requirements, may be subject to, among other things, fines, suspension or withdrawal of regulatory approvals, product recalls, seizure of products, operating restrictions and criminal prosecution.
Non-Clinical Studies and Clinical Trials
Similar to the United States, the various phases of non-clinical and clinical research in the European Union, or EU, are subject to significant regulatory controls.
Non-clinical studies are performed to demonstrate the health or environmental safety of new biological substances. Non-clinical studies must be conducted in compliance with the principles of good laboratory practice, or GLP, as set forth in EU Directive 2004/10/EC. In particular, non-clinical studies, both in vitro and in vivo, must be
36
planned, performed, monitored, recorded, reported and archived in accordance with the GLP principles, which define a set of rules and criteria for a quality system for the organizational process and the conditions for non-clinical studies. These GLP standards reflect the Organization for Economic Co-operation and Development requirements.
Clinical trials of medicinal products in the EU must be conducted in accordance with EU and national regulations and the International Conference on Harmonization, or ICH, guidelines on Good Clinical Practices, or GCP, as well as the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki. If the sponsor of the clinical trial is not established within the EU, it must appoint an EU entity to act as its legal representative. The sponsor must take out a clinical trial insurance policy, and in most EU member states, the sponsor is liable to provide ‘no fault’ compensation to any study subject injured in the clinical trial.
The regulatory landscape related to clinical trials in the EU has been subject to recent changes. The EU Clinical Trials Regulation, or CTR, which was adopted in April 2014 and repeals the EU Clinical Trials Directive, became applicable on January 31, 2022. Unlike directives, the CTR is directly applicable in all EU member states without the need for member states to further implement it into national law. The CTR notably harmonizes the assessment and supervision processes for clinical trials throughout the EU via a Clinical Trials Information System, which contains a centralized EU portal and database.
While the Clinical Trials Directive required a separate clinical trial application, or CTA, to be submitted in each member state, to both the competent national health authority and an independent ethics committee, much like the FDA and IRB respectively, the CTR introduces a centralized process and only requires the submission of a single application to all member states concerned. The CTR allows sponsors to make a single submission to both the competent authority and an ethics committee in each member state, leading to a single decision per member state. The CTA must include, among other things, a copy of the trial protocol and an investigational medicinal product dossier containing information about the manufacture and quality of the medicinal product under investigation. The assessment procedure of the CTA has been harmonized as well, including a joint assessment by all member states concerned, and a separate assessment by each member state with respect to specific requirements related to its own territory, including ethics rules. Each member state’s decision is communicated to the sponsor via the centralized EU portal. Once the CTA is approved, clinical study development may proceed.
The CTR foresees a three-year transition period. The extent to which ongoing and new clinical trials will be governed by the CTR varies. Clinical trials for which an application was submitted (i) prior to January 31, 2022 under the Clinical Trials Directive, or (ii) between January 31, 2022 and January 31, 2023 and for which the sponsor has opted for the application of the Clinical Trials Directive remain governed by said Directive until January 31, 2025. After this date, all clinical trials (including those which are ongoing) will become subject to the provisions of the CTR.
Marketing Authorization
In the EU, medicinal products can only be placed on the market after obtaining a marketing authorization, or MA. To obtain regulatory approval of an investigational medicinal product under EU regulatory systems, we must submit an MA application, or MAA. The process for doing this depends, among other things, on the nature of the medicinal product.
Essentially, there are two types of MAs:
37
Under the centralized procedure, the maximum timeframe for the evaluation of a MAA by the EMA is 210 days. In exceptional cases, the CHMP might perform an accelerated review of a MAA in no more than 150 days (not including clock stops). Innovative products that target an unmet medical need and are expected to be of major public health interest may be eligible for a number of expedited development and review programs, such as the PRIME scheme, which provides incentives similar to the breakthrough therapy designation in the U.S. PRIME is a voluntary scheme aimed at enhancing the EMA’s support for the development of medicines that target unmet medical needs. It is based on increased interaction and early dialogue with companies developing promising medicines, to optimize their product development plans and speed up their evaluation to help them reach patients earlier. Product developers that benefit from PRIME designation can expect to be eligible for accelerated assessment but this is not guaranteed. The benefits of a PRIME designation include the appointment of a CHMP rapporteur before submission of a MAA, early dialogue and scientific advice at key development milestones, and the potential to qualify products for accelerated review earlier in the application process.
Moreover, in the EU, a “conditional” MA may be granted in cases where all the required safety and efficacy data are not yet available. The conditional MA is subject to conditions to be fulfilled for generating the missing data or ensuring increased safety measures. It is valid for one year and has to be renewed annually until fulfillment of all the conditions. Once the pending studies are provided, it can become a “normal” MA. However, if the conditions are not fulfilled within the timeframe set by the EMA, the MA ceases to be renewed. Furthermore, MA may also be granted “under exceptional circumstances” when the applicant can show that it is unable to provide comprehensive data on the efficacy and safety under normal conditions of use even after the product has been authorized and subject to specific procedures being introduced. This may arise in particular when the intended indications are very rare and, in the present state of scientific knowledge, it is not possible to provide comprehensive information, or when generating data may be contrary to generally accepted ethical principles. This MA is close to the conditional MA as it is reserved to medicinal products to be approved for severe diseases or unmet medical needs and the applicant does not hold the complete data set legally required for the grant of a MA. However, unlike the conditional MA, the applicant does not have to provide the missing data and will never have to. Although the MA “under exceptional circumstances” is granted definitively, the risk-benefit balance of the medicinal product is reviewed annually and the MA is withdrawn in case the risk-benefit ratio is no longer favorable.
Under the above described procedures, in order to grant the MA, the EMA or the competent authorities of the EU member states make an assessment of the risk benefit balance of the product on the basis of scientific criteria concerning its quality, safety and efficacy.
MAs have an initial duration of five years. After these five years, the authorization may be renewed for an unlimited period on the basis of a reevaluation of the risk-benefit balance.
Data and Marketing Exclusivity
The EU also provides opportunities for market exclusivity. Upon receiving MA, new chemical entities generally receive eight years of data exclusivity and an additional two years of market exclusivity. If granted, data exclusivity prevents regulatory authorities in the EU from referencing the innovator’s data to assess a generic or biosimilar application. During the additional two‑year period of market exclusivity, a generic/biosimilar MA can be submitted, and the innovator’s data may be referenced, but no generic/biosimilar product can be marketed until the
38
expiration of the market exclusivity. The overall ten-year market exclusivity period may be extended to a maximum of eleven years if, during the first eight years, a new therapeutic indication with significant clinical benefit over existing therapies is approved. However, there is no guarantee that a product will be considered by the EU or member state regulatory authorities to be a new chemical entity, and products may not qualify for data exclusivity.
Post-Approval Requirements
Similar to the United States, both MA holders and manufacturers of medicinal products are subject to comprehensive regulatory oversight by the EMA, the European Commission and/or the competent regulatory authorities of the member states. The holder of a MA must establish and maintain a pharmacovigilance system and appoint an individual qualified person for pharmacovigilance who is responsible for oversight of that system. Key obligations include expedited reporting of suspected serious adverse reactions and submission of periodic safety update reports, or PSURs.
All new MAAs must include a risk management plan, or RMP, describing the risk management system that the company will put in place and documenting measures to prevent or minimize the risks associated with the product. The regulatory authorities may also impose specific obligations as a condition of the MA. Such risk-minimization measures or post-authorization obligations may include additional safety monitoring, more frequent submission of PSURs, or the conduct of additional clinical trials or post-authorization safety studies.
The advertising and promotion of medicinal products is also subject to laws concerning promotion of medicinal products, interactions with physicians, misleading and comparative advertising and unfair commercial practices. All advertising and promotional activities for the product must be consistent with the approved summary of product characteristics, and therefore all off-label promotion is prohibited. Direct-to-consumer advertising of prescription medicines is also prohibited in the EU. Although general requirements for advertising and promotion of medicinal products are established under EU directives, the details are governed by regulations in each member state and can differ from one country to another.
Failure to comply with EU and member state laws that apply to the conduct of clinical trials, manufacturing approval, MA of medicinal products and marketing of such products, both before and after grant of the MA, manufacturing of pharmaceutical products, statutory health insurance, bribery and anti-corruption or with other applicable regulatory requirements may result in administrative, civil or criminal penalties. These penalties could include delays or refusal to authorize the conduct of clinical trials, or to grant MA, product withdrawals and recalls, product seizures, suspension, withdrawal or variation of the MA, total or partial suspension of production, distribution, manufacturing or clinical trials, operating restrictions, injunctions, suspension of licenses, fines and criminal penalties.
The aforementioned EU rules are generally applicable in the European Economic Area, or EEA, which consists of the 27 EU member states plus Norway, Liechtenstein and Iceland.
For other countries outside of the European Union, such as countries in Latin America or Asia, the requirements governing the conduct of clinical studies, product licensing, pricing and reimbursement vary from country to country. In all cases, again, the clinical studies are conducted in accordance with GCP and the applicable regulatory requirements and the ethical principles that have their origin in the Declaration of Helsinki.
Brexit and the Regulatory Framework in the United Kingdom
Since the end of the Brexit transition period on January 1, 2021, Great Britain (England, Scotland and Wales) has not been subject to EU laws, however under the terms of the Ireland/Northern Ireland Protocol, Northern Ireland continues to follow EU law. The EU laws that have been transposed into United Kingdom, or UK, law through secondary legislation remain applicable in Great Britain. However, under the Retained EU Law (Revocation and Reform) Bill 2022, which is currently before the UK parliament, any retained EU law not expressly preserved and “assimilated” into domestic law or extended by ministerial regulations (to no later than 23 June 2026) will automatically expire and be revoked by December 31, 2023. In addition, new legislation such as the EU CTR will not be applicable. The UK government has passed a new Medicines and Medical Devices Act 2021, which introduces delegated powers in favor of the Secretary of State or an ‘appropriate authority’ to amend or supplement
39
existing regulations in the area of medicinal products and medical devices. This allows new rules to be introduced in the future by way of secondary legislation, which aims to allow flexibility in addressing regulatory gaps and future changes in the fields of human medicines, clinical trials and medical devices.
As of January 1, 2021, the Medicines and Healthcare products Regulatory Agency, or MHRA, is the UK’s standalone medicines and medical devices regulator. The MHRA has introduced changes to national licensing procedures, including procedures to prioritize access to new medicines that will benefit patients, including a 150-day assessment and a rolling review procedure. All existing EU MAs for centrally authorized products were automatically converted or grandfathered into UK MAs, effective in Great Britain (only), free of charge on January 1, 2021, unless the MA holder chose to opt-out. After Brexit, companies established in the UK cannot use the centralized procedure and instead must follow one of the UK national authorization procedures or one of the remaining post-Brexit international cooperation procedures to obtain an MA to commercialize products in the UK. For a period of three years from 1 January 2021, the MHRA may rely on a decision taken by the EU Commission on the approval of a new (centralized procedure) MA when determining an application for a Great Britain authorization; or use the MHRA’s decentralized or mutual recognition procedures which enable MAs approved in EU member states (or Iceland, Liechtenstein, Norway) to be granted in Great Britain.
Data Privacy and Security Laws
Numerous state, federal and foreign laws govern the collection, dissemination, use, access to, confidentiality and security of personal information, including health-related information. In the United States, numerous federal and state laws and regulations, including data breach notification laws, health information privacy and security laws, and consumer protection laws and regulations govern the collection, use, disclosure, and protection of health-related and other personal information and could apply to our operations or the operations of our partners. In addition, certain foreign laws, such as the UK General Data Protection Regulation and Data Protection Act 2018 (collectively, the “UK GDPR”), govern the privacy and security of personal data, including health-related data. Failure to comply with these laws, where applicable, can result in the imposition of significant civil and/or criminal penalties and private litigation. Privacy and security laws, regulations, and other obligations are constantly evolving, may conflict with each other to complicate compliance efforts, and can result in investigations, proceedings, or actions that lead to significant civil and/or criminal penalties and restrictions on data processing.
Employees
As of March 1, 2023, we had 31 employees. None of our employees is subject to a collective bargaining agreement or represented by a trade or labor union. We consider our relationship with our employees to be good.
Corporate Information
We were incorporated in Delaware in November 2012. Our address is P.O. Box 134, Malvern, Pennsylvania 19355. Our common stock is listed on the Nasdaq Global Market under the symbol “GRTX.”
Available Information
Our internet website address is www.galeratx.com. In addition to the information about us and our subsidiaries contained in this Annual Report on Form 10-K, information about us can be found on our website. Our website and information included in or linked to our website are not part of this Annual Report on Form 10-K.
Our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K and amendments to those reports filed or furnished pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934, as amended, are available free of charge through our website as soon as reasonably practicable after they are electronically filed with or furnished to the Securities and Exchange Commission, or SEC. Additionally the SEC maintains an internet site that contains reports, proxy and information statements and other information. The address of the SEC's website is www.sec.gov.
40
Item 1A. Risk Factors.
Investing in our common stock involves a high degree of risk. You should carefully consider the risks described below, as well as the other information in this Annual Report on Form 10-K, including our consolidated financial statements and the related notes and “Management’s Discussion and Analysis of Financial Condition and Results of Operations,” before deciding whether to invest in our common stock. The occurrence of any of the events or developments described below could adversely affect our business, financial condition, results of operations and growth prospects. In such an event, the market price of our common stock could decline, and you may lose all or part of your investment. Additional risks and uncertainties not presently known to us or that we currently deem immaterial also may impair our business operations.
Risks Related to Our Financial Position and Capital Needs
We are a clinical stage biopharmaceutical company with a limited operating history and have not generated any revenue from product sales. We have incurred significant operating losses since our inception and anticipate that we will incur continued losses for the foreseeable future.
We have incurred losses in each year since our inception in 2012 and anticipate incurring losses for the foreseeable future. To date, we have invested substantially all of our efforts and financial resources in identifying, acquiring, in-licensing and developing our product candidates, including commencing and conducting clinical trials and providing general and administrative support for these operations. Our future success is dependent on our ability to develop, obtain regulatory approval for and successfully commercialize one or more of our product candidates. We have not yet demonstrated our ability to obtain regulatory approvals, manufacture a drug at commercial scale, or conduct sales and marketing activities. We currently generate no revenue from sales of any products, and we may never be able to develop or commercialize a marketable product. Biopharmaceutical product development is a highly speculative undertaking and involves a substantial degree of risk. Typically, it takes many years to develop one new drug from the time it is discovered to when it is available for treating patients, and development may cease for a number of reasons.
We have incurred significant losses related to expenses for research and development and our ongoing operations. Our net losses for the years ended December 31, 2022 and 2021 were $62.2 million and $80.5 million, respectively. As of December 31, 2022, we had an accumulated deficit of $378.3 million. We expect to continue to incur losses for the foreseeable future, and we anticipate these losses will increase substantially as we:
41
To become and remain profitable, we must succeed in developing and eventually commercializing product candidates that generate significant revenue. This will require us to be successful in a range of challenging activities, including completing preclinical studies and clinical trials of our product candidates, obtaining regulatory approval, and manufacturing, marketing and selling any product candidates for which we may obtain regulatory approval, as well as discovering and developing additional product candidates. We are only in the preliminary stages of most of these activities. We may never succeed in these activities and, even if we do, may never generate revenue that is significant enough to achieve profitability.
In cases where we are successful in obtaining regulatory approval to market one or more of our product candidates, our revenue will be dependent, in part, upon the size of the markets in the territories for which we gain regulatory approval, the accepted price for the product, the ability to obtain coverage and reimbursement, and whether we own the commercial rights for that territory. If the number of our addressable patients is not as significant as we estimate, the indication approved by regulatory authorities is narrower than we expect, or the treatment population is narrowed by competition, physician choice or treatment guidelines, we may not generate significant revenue from sales of such products, even if approved.
Because of the numerous risks and uncertainties associated with product development, we are unable to accurately predict the timing or amount of expenses or when, or if, we will be able to achieve profitability. If we are required by regulatory authorities to perform studies in addition to those expected, or if there are any delays in the initiation and completion of our clinical trials or the development of any of our product candidates, our expenses could increase.
Further, the net losses we incur may fluctuate significantly from quarter-to-quarter and year-to-year, such that a period-to-period comparison of our results of operations may not be a good indication of our future performance. We expect to incur additional costs associated with operating as a public company. Even if we achieve profitability in the future, we may not be able to sustain profitability in subsequent periods. Our prior losses, combined with expected future losses, have had and will continue to have an adverse effect on our stockholders’ equity and working capital.
Our recurring losses from operations raise substantial doubt regarding our ability to continue as a going concern.
We have incurred significant losses since our inception and have never generated revenue or profit, and it is possible we will never generate revenue or profit. As of December 31, 2022, we had $31.6 million in cash, cash equivalents and short-term investments and an accumulated deficit of $378.3 million. Based on our current operating plan and assumptions, we believe that our existing cash, cash equivalents and short-term investments as of December 31, 2022, together with the net proceeds from our February 2023 registered direct offering, will be sufficient to enable us to fund our operating expenses and capital expenditure requirements into the fourth quarter of 2023, but not for more than one year after the date of the filing of this Annual Report on Form 10-K. These factors raise substantial doubt about our ability to continue as a going concern. We will need to raise additional capital to fund our future operations and remain as a going concern. There can be no assurance that we will be able to obtain additional funding on acceptable terms, if at all. To the extent that we raise additional capital through future equity offerings, the ownership interest of common stockholders will be diluted, which dilution may be significant.
42
However, we cannot guarantee that we will be able to obtain any or sufficient additional funding or that such funding, if available, will be obtainable on terms satisfactory to us. In the event that we are unable to obtain any or sufficient additional funding, there can be no assurance that we will be able to continue as a going concern, and we will be forced to delay, reduce or discontinue our product development programs or commercialization efforts.
Substantial doubt about our ability to continue as a going concern may materially and adversely affect the price per share of our common stock, and it may be more difficult for us to obtain financing. If potential collaborators decline to do business with us or potential investors decline to participate in any future financings due to such concerns, our ability to increase our cash position may be limited. The perception that we may not be able to continue as a going concern may cause others to choose not to deal with us due to concerns about our ability to meet our contractual obligations.
We have prepared our consolidated financial statements on a going concern basis, which contemplates the realization of assets and the satisfaction of liabilities and commitments in the normal course of business. Our consolidated financial statements included in this Annual Report on Form 10-K do not include any adjustments to reflect the possible inability to continue as a going concern within one year after the date of the filing of this Annual Report on Form 10-K. If we are unable to continue as a going concern, you could lose all or part of your investment.
We will need substantial funding to meet our financial obligations and to pursue our business objective. If we are unable to raise capital when needed, we could be forced to curtail our planned operations and the pursuit of our growth strategy.
Identifying potential product candidates and conducting preclinical studies and clinical trials is a time-consuming, expensive and uncertain process that takes years to complete, and we may never generate the necessary data or results required to obtain regulatory approval and achieve product sales. We expect our expenses to increase in connection with our ongoing development activities related to avasopasem for the reduction in the incidence of severe oral mucositis, or SOM, in patients with locally advanced HNC, seek marketing approval for avasopasem, pursue clinical trials and marketing approval of avasopasem in other indications, pursue clinical trials and marketing approval of rucosopasem and advance any of our other product candidates we may develop or otherwise acquire. In addition, if we obtain marketing approval for any of our product candidates, we expect to incur significant commercialization expenses related to manufacturing, product sales, marketing and distribution. We may also need to raise additional funds sooner if we choose to pursue additional indications for our product candidates or otherwise expand more rapidly than we presently anticipate. Furthermore, we expect to continue to incur significant costs associated with operating as a public company. Accordingly, we will need to obtain substantial additional funding in connection with our continuing operations. If we are unable to raise capital when needed on attractive terms, if at all, we will be forced to delay, reduce or eliminate certain of our clinical development plans, research and development programs or future commercialization efforts.
The development process for our product candidates is highly uncertain, and we cannot estimate with certainty the actual amounts necessary to successfully complete the development, regulatory approval process and commercialization of our product candidates. Based on our current operating plan and assumptions, we believe that our existing cash, cash equivalents and short-term investments as of December 31, 2022, together with the net proceeds from our February 2023 registered direct offering, will be sufficient to enable us to fund our operating expenses and capital expenditure requirements into the fourth quarter of 2023. Our operating plans may change as a result of many factors currently unknown to us, and we may need to seek additional funds sooner than expected, through public or private equity, debt financings or other sources. Our future capital requirements will depend on and could increase significantly as a result of many factors, including:
43
Any additional fundraising efforts may divert our management from their day-to-day activities, which may adversely affect our ability to develop and commercialize our product candidates. Dislocations in the financial markets may make equity and debt financing more difficult to obtain and may have a material adverse effect on our ability to meet our fundraising needs when they arise. Additional funds may not be available when we need them, on terms that are acceptable to us, or at all. If we are unable to obtain funding on a timely basis, we may be required to significantly curtail, delay or discontinue one or more of our preclinical studies, clinical trials or other research or development programs, the commercialization of any product candidate. We may also be unable to expand our operations or otherwise capitalize on our business opportunities or may be required to relinquish rights to our product candidates or products. Any of these occurrences could materially affect our business, financial condition and results of operations.
Raising additional capital may cause dilution to our stockholders, restrict our operations or require us to relinquish rights to our technologies or product candidates.
Until such time as we can generate substantial product revenues, if ever, we expect to finance our cash needs through securities offerings or debt financings, or possibly, license and collaboration agreements or research grants. The terms of any financing may adversely affect the holdings or the rights of our stockholders and our issuance of additional securities, whether equity or debt, or the possibility of such issuance, may cause the market price of our common stock to decline. The sale of additional equity or convertible securities would dilute all of our
44
stockholders, including your ownership interest. The incurrence of indebtedness would result in increased fixed or variable payment obligations, and we may be required to agree to certain restrictive covenants, such as limitations on our ability to incur additional debt, limitations on our ability to acquire, sell or license intellectual property rights and other operating restrictions that could adversely impact our ability to conduct our business. We could also be required to seek funds through arrangements with collaborators or otherwise at an earlier stage than otherwise would be desirable and we may be required to relinquish rights to some of our technologies, product candidates or future revenue streams, or otherwise agree to terms unfavorable to us, any of which may have a material adverse effect on our business, operating results and prospects. If we raise funds through research grants, we may be subject to certain requirements, which may limit our ability to use the funds or require us to share information from our research and development. Raising additional capital through any of these or other means could adversely affect our business and the holdings or rights of our stockholders and may cause the market price of our shares to decline.
Risks Related to the Discovery and Development of Our Product Candidates
We are heavily dependent on the success of our lead product candidate, avasopasem, and if avasopasem does not receive regulatory approval, our business may be harmed.
We currently have no products that are approved for commercial sale. We expect that a substantial portion of our efforts and expenditures over the next few years will be devoted to the advancement of avasopasem, through the regulatory approval process, as well as the commercialization of avasopasem following regulatory approval, if received.
We cannot be certain that avasopasem will receive regulatory approval, or be successfully commercialized even if we receive regulatory approval. We have not completed the development of any product candidates and we may never be able to develop marketable products. The research, testing, manufacturing, labeling, approval, sale, marketing and distribution of products are, and will remain, subject to extensive regulation by the FDA and other regulatory authorities in the United States and other countries that each have differing regulations. We are not permitted to market avasopasem in the United States until we receive approval of a New Drug Application, or NDA, or in any foreign country until we receive the requisite approvals from the appropriate authorities in such countries for marketing authorization.
We have not yet demonstrated our ability to obtain regulatory approval for any of our product candidates, and there can be no assurance that the results from our Phase 3 ROMAN trial together with the randomized Phase 2b GT-201 trial of avasopasem will be sufficient for the FDA to approve the NDA for the reduction of SOM in patients with HNC that we submitted to the FDA in December 2022.
While we are currently continuing our ongoing clinical trials, the COVID-19 pandemic and related precautions have directly or indirectly impacted the timeline for certain of our clinical trials of avasopasem. We delayed the initiation of the Phase 2a multi-center trial in Europe assessing the safety of avasopasem in patients with HNC undergoing standard-of-care radiotherapy due to concerns with clinical trial enrollment in Europe during the COVID-19 pandemic. The first patient was dosed in this trial in June 2020, and target enrollment was decreased to approximately 35 patients due to this delay. This trial was expected to contribute to the safety database for avasopasem in patients with HNC receiving radiotherapy. As a result of the delay in initiating the trial in Europe, the target enrollment for the ROMAN trial was increased to approximately 450 patients in order to ensure we were positioned to maintain the overall planned size of the safety database in a timely manner.
While our Phase 3 ROMAN trial did demonstrate a statistically significant difference for the active 90 mg dose compared to placebo for the primary endpoint and a key secondary endpoint, we do not know whether the FDA will find these results together with the results from the randomized Phase 2b GT-201 trial of avasopasem in patients with HNC sufficient to approve the NDA for avasopasem for the reduction of SOM in patients with HNC.
In December 2022, we submitted to the FDA the NDA for avasopasem for the reduction of SOM in patients with HNC. Obtaining approval of an NDA or similar regulatory approval is an extensive, lengthy, expensive and inherently uncertain process, and the FDA or other foreign regulatory authorities may delay, limit or deny approval of any of our current or future product candidates for many reasons, including:
45
Clinical drug development involves a lengthy and expensive process with uncertain timelines and outcomes, and results of earlier studies and trials may not be predictive of future trial results. If development of our product candidates is unsuccessful or delayed, we may be unable to obtain required regulatory approvals and be unable to commercialize our product candidates on a timely basis, if at all.
Clinical testing is expensive and can take many years to complete, and its outcome is inherently uncertain. Failure or delay can occur at any time during the clinical trial process. Success in preclinical studies and early clinical trials does not ensure that later clinical trials will be successful. A number of companies in the
46
pharmaceutical industry, including biotechnology companies, have suffered significant setbacks in clinical trials, even after promising results in earlier preclinical studies or clinical trials. These setbacks have been caused by, among other things, preclinical findings made while clinical trials were underway and safety or efficacy observations made in clinical trials, including previously unreported adverse events. The results of preclinical studies and clinical trials of our product candidates may not be predictive of the results of later-stage clinical trials. Product candidates in later stages of clinical trials may fail to show the desired safety and efficacy traits despite having progressed through preclinical studies and initial clinical trials. Notwithstanding any potential promising results in earlier studies, we cannot be certain that we will not face similar setbacks. Even if our clinical trials are completed, the results may not be sufficient to obtain regulatory approval for our product candidates.
Furthermore, we rely on CROs and clinical trial sites to ensure the proper and timely conduct of our clinical trials. While we have agreements with our CROs governing their committed activities, and the ability to audit their performance, we have limited influence over their actual performance. We rely on third-party vendors, such as CROs, scientists and collaborators to provide us with significant data and other information related to our preclinical studies or clinical trials and our business. If such third parties provide inaccurate, misleading or incomplete data, our business, prospects and results of operations could be materially adversely affected. For example, in October 2021, we announced topline data from the Phase 3 ROMAN trial of avasopasem in SOM and reported that the trial did not achieve statistical significance on the primary endpoint. Upon further analysis of the ROMAN data, an error by the CRO was identified in the statistical program. Correction of this error yielded the correct, statistically significant p-values for the primary and a key secondary endpoint. We announced the correct topline results in December 2021.
We may experience delays in initiating our clinical trials and we cannot be certain that the trials or any other future clinical trials for our product candidates will begin on time, need to be redesigned, enroll an adequate number of patients on time or be completed on schedule, if at all. Clinical trials can be delayed or terminated for a variety of reasons, including delay or failure related to:
47
We could also encounter delays if a clinical trial is suspended or terminated by us, by the IRBs of the institutions in which such trials are being conducted, by the Data Safety Monitoring Board, or DSMB, for such trial or by the FDA or other regulatory authorities, such as the competent authorities of the member states of the EU. Such authorities may suspend or terminate a clinical trial due to a number of factors, including failure to conduct the clinical trial in accordance with regulatory requirements or our clinical protocols, inspection of the clinical trial operations or trial site by the FDA or other regulatory authorities, such as the competent authorities of the member states of the EU, resulting in the imposition of a clinical hold, unforeseen safety issues or adverse side effects, failure to demonstrate a benefit from using a drug, changes in governmental regulations or administrative actions or lack of adequate funding to continue the clinical trial.
Further, conducting clinical trials in foreign countries, as we plan to do for our product candidates, presents additional risks that may delay completion of our clinical trials. These risks include the failure of enrolled patients in foreign countries to adhere to clinical protocol as a result of differences in healthcare services or cultural customs, managing additional administrative burdens associated with foreign regulatory schemes, as well as political and economic risks relevant to such foreign countries.
If we experience delays in the completion, or termination, of any clinical trial of our product candidates, the commercial prospects of our product candidates may be harmed, and our ability to generate product revenues from any of these product candidates will be delayed or not realized at all. In addition, any delays in completing our clinical trials will increase our costs, slow down our product candidate development and approval process and jeopardize our ability to commence product sales and generate revenues. Any of these occurrences may significantly harm our business, financial condition and prospects. In addition, many of the factors that cause, or lead to, a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of regulatory approval of our product candidates.
In addition, the FDA’s and other regulatory authorities’ policies with respect to clinical trials may change and additional government regulations may be enacted. For instance, the regulatory landscape related to clinical trials in the EU recently evolved. The EU Clinical Trials Regulation, or CTR, which was adopted in April 2014 and repeals the EU Clinical Trials Directive, became applicable on January 31, 2022. While the Clinical Trials Directive required a separate clinical trial application, or CTA, to be submitted in each member state, to both the competent national health authority and an independent ethics committee, the CTR introduces a centralized process and only requires the submission of a single application to all member states concerned. The CTR allows sponsors to make a single submission to both the competent authority and an ethics committee in each member state, leading to a single decision per member state. The assessment procedure of the CTA has been harmonized as well, including a joint assessment by all member states concerned, and a separate assessment by each member state with respect to specific requirements related to its own territory, including ethics rules. Each member state’s decision is communicated to the sponsor via the centralized EU portal. Once the CTA is approved, clinical study development may proceed. The CTR foresees a three-year transition period. The extent to which ongoing and new clinical trials will be governed by the CTR varies. Clinical trials for which an application was submitted (i) prior to January 31, 2022 under the Clinical Trials Directive, or (ii) between January 31, 2022 and January 31, 2023 and for which the sponsor has opted for the application of the Clinical Trials Directive remain governed by said Directive until January 31, 2025. After this date, all clinical trials (including those which are ongoing) will become subject to the provisions of the CTR. Compliance with the CTR requirements by us and our third-party service providers, such as CROs, may impact our developments plans.
The United Kingdom, or UK, is no longer part of the EU, and since the end of the Brexit transition period on January 1, 2021, the EU regulatory regime no longer applies in Great Britain (England, Wales and Scotland). Under the terms of the Ireland/Northern Ireland Protocol, the provisions of the CTR generally apply to clinical trials taking place in Northern Ireland.
It is currently unclear to what extent the UK Government will seek to align the regulations in Great Britain with the EU CTR. The Great Britain regulatory framework in relation to clinical trials is still derived from the EU Clinical Trials Directive (as implemented into Great Britain law, through secondary legislation). On January 17, 2022, the UK Medicines and Healthcare products Regulatory Agency, or MHRA, carried out an eight-week consultation on a set of proposals aimed at improving and strengthening the clinical trials regime across the UK. The consultation closed on March 14, 2022 and aims to streamline clinical trials approvals, enable innovation, enhance
48
clinical trials transparency, enable greater risk proportionality, and promote patient and public involvement in clinical trials. The outcome of the consultation is closely watched and will determine whether the UK chooses to align with the EU CTR or diverge from it to maintain regulatory flexibility. A decision by the UK Government not to closely align its regulations with the new EU approach may have an effect on the cost of conducting clinical trials in the UK as opposed to other countries and/or make it harder to seek a MA in the EU for our product candidates on the basis of clinical trials conducted in the UK.
If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies governing clinical trials, our development plans may be impacted.
If we encounter difficulties or delays enrolling patients in our clinical trials, our clinical development activities could be delayed or otherwise adversely affected.
The timely completion of clinical trials in accordance with their protocols depends, among other things, on our ability to enroll a sufficient number of patients who remain in the study until its conclusion. We may experience difficulties in patient enrollment in our clinical trials for a variety of reasons. The enrollment of patients depends on many factors, including:
In addition, our clinical trials will compete with other clinical trials for product candidates that are in the same therapeutic areas as our product candidates, and this competition will reduce the number and types of patients available to us, because some patients who might have opted to enroll in our trials may instead opt to enroll in a trial being conducted by one of our competitors. Since the number of qualified clinical investigators is limited, we expect to conduct some of our clinical trials at the same clinical trial sites that some of our competitors use, which will reduce the number of patients who are available for our clinical trials in such clinical trial site.
Delays in patient enrollment may result in increased costs or may affect the timing or outcome of the planned clinical trials, which could prevent completion of these trials and adversely affect our ability to advance the development of our product candidates.
Success in preclinical studies or earlier clinical trials may not be indicative of results in future clinical trials.
Success in preclinical studies and early clinical trials does not ensure that later clinical trials will generate the same results or otherwise provide adequate data to demonstrate the efficacy and safety of a product candidate. Preclinical studies and early-stage clinical trials are primarily designed to test safety, to study pharmacokinetics and pharmacodynamics and to understand the side effects of product candidates at various doses and schedules. Success in preclinical studies and early clinical trials does not ensure that later, large-scale efficacy trials will be successful, nor does it predict final results. Our product candidates may fail to show the desired safety
49
and efficacy in clinical development despite positive results in preclinical studies or having successfully advanced through initial clinical trials.
In addition, the design of a clinical trial can determine whether its results will support approval of a product, and flaws in the design of a clinical trial may not become apparent until the clinical trial is well advanced. As an organization, we have limited experience designing clinical trials and may be unable to design and execute a clinical trial sufficient to support regulatory approval. Many companies in the pharmaceutical and biotechnology industries have suffered significant setbacks in late-stage clinical trials even after achieving promising results in preclinical studies and earlier-stage clinical trials. Data obtained from preclinical and clinical activities are subject to varying interpretations, which may delay, limit or prevent regulatory approval. In addition, we may experience regulatory delays or rejections as a result of many factors, including changes in regulatory policy during the period of our product candidate development. Any such delays could negatively impact our business, financial condition, results of operations and prospects.
We plan to conduct clinical trials for our product candidates outside the United States and the FDA may not accept data from such trials.
We have conducted certain of our clinical trials outside the United States, and we plan to conduct additional clinical trials outside the United States. For example, we conducted a Phase 1 dose and schedule escalation study of rucosopasem in healthy volunteers in Australia. Although the FDA may accept data from clinical trials conducted outside the United States, acceptance of such study data by the FDA is subject to certain conditions. For example, for clinical trials not otherwise subject to an IND, such clinical trials must be conducted in accordance with good clinical practices, or GCP, requirements and the FDA must be able to validate the data from the clinical trial through an onsite inspection if it deems such inspection necessary.
Where data from foreign clinical trials are intended to serve as the sole basis for marketing approval in the United States, the FDA will not approve the application on the basis of foreign data alone unless those data are applicable to the U.S. population and U.S. medical practice, the clinical trials were performed by clinical investigators of recognized competence, and the data are considered valid without the need for an on-site inspection by the FDA or, if the FDA considers such an inspection to be necessary, the FDA is able to validate the data through an on-site inspection or other appropriate means. In addition, such clinical trials would be subject to the applicable local laws of the foreign jurisdictions where the clinical trials are conducted.
There can be no assurance the FDA will accept data from clinical trials conducted outside of the United States. There can also be no assurance that the comparable foreign regulatory authority in any jurisdiction in which we seek marketing approval for our product candidates will accept data from clinical trials conducted outside such jurisdiction. If the FDA or any such foreign regulatory authority does not accept any such data, it would likely result in the need for additional clinical trials, which would be costly and time-consuming and delay aspects of our development plan. In addition, the conduct of clinical trials outside the United States could have a significant impact on us. Risks inherent in conducting international clinical trials include:
50
Our product candidates may cause undesirable side effects or have other properties that could delay or prevent their regulatory approval, cause us to suspend or discontinue clinical trials, limit the commercial profile of an approved label, or result in significant negative consequences following marketing approval, if any.
Undesirable side effects caused by our product candidates could cause us or regulatory authorities to interrupt, delay or halt clinical trials and could result in a more restrictive label or the delay or denial of regulatory approval by the FDA or comparable foreign regulatory authorities, such as the EMA or the competent authorities of the member states of the EU. Results of our clinical trials could reveal a high and unacceptable severity and prevalence of side effects or unexpected characteristics. To date, patients treated with our product candidates have experienced drug-related side effects including lymphopenia, nausea, fatigue, oropharyngeal pain, constipation, radiation skin injury and vomiting.
If unacceptable side effects arise in the development of our product candidates, we, the FDA, the IRBs at the institutions in which our studies are conducted, or the DSMB could suspend or terminate our clinical trials or the FDA or comparable foreign regulatory authorities could order us to cease clinical trials or deny approval of our product candidates for any or all targeted indications. Treatment-related side effects could also affect patient recruitment or the ability of enrolled patients to complete the trial or result in potential product liability claims. In addition, these side effects may not be appropriately recognized or managed by the treating medical staff. We expect to have to train medical personnel using our product candidates to understand the side effect profiles for our clinical trials and upon any commercialization of any of our product candidates. Inadequate training in recognizing or managing the potential side effects of our product candidates could result in patient injury or death. Any of these occurrences may harm our business, financial condition and prospects significantly.
Our clinical trials include cancer patients who are very sick and whose health may deteriorate, and we expect that additional clinical trials of our other product candidates will include similar patients with potentially deteriorating health. It is possible that some may die during our clinical trials for various reasons, including because the patient’s underlying disease continues to advance despite treatment, or because the patient experiences medical problems that may not be related to our product candidate. For example, during the treatment phase of our Phase 2b trial of avasopasem, there was one non-treatment-related death in each of the placebo, 30 mg treatment and 90 mg treatment arms. Even if the deaths are not related to our product candidate, the deaths could affect perceptions regarding the safety of our product candidates.
In addition, if any of our product candidates receives marketing approval, and we or others later identify undesirable side effects caused by such products, a number of potentially significant negative consequences could result, including:
51
Any of the foregoing events could prevent us from achieving or maintaining market acceptance of the particular product candidate, if approved, and result in the loss of significant revenues to us, which would materially and adversely affect our results of operations and business.
Interim, topline or preliminary data from our clinical trials that we announce or publish from time to time may change as more patient data become available and are subject to audit and verification procedures that could result in material changes in the final data.
From time to time, we may publicly disclose interim, topline, or preliminary data from our clinical trials, which is based on a preliminary analysis of then-available data, and the results and related findings and conclusions are subject to change following a full analyses of all data related to the particular trial. We also make assumptions, estimations, calculations and conclusions as part of our analyses of data, and we may not have received or had the opportunity to fully and carefully evaluate all data. As a result, the interim, topline, or preliminary results that we report may differ from future results of the same trials, or different conclusions or considerations may qualify such results, once additional data have been received and fully evaluated. Topline and preliminary data also remain subject to audit and verification procedures that may result in the final data being materially different from the preliminary data we previously published. As a result, topline and preliminary data should be viewed with caution until the final data are available.
We may also disclose interim data from our clinical trials. Interim data from clinical trials that we may complete are subject to the risk that one or more of the clinical outcomes may materially change as patient enrollment continues and more patient data become available. Adverse differences between interim, top-line, or preliminary data and final data could significantly harm our business prospects.
Further, others, including regulatory agencies, may not accept or agree with our assumptions, estimates, calculations, conclusions or analyses or may interpret or weigh the importance of data differently, which could impact the value of the particular program, the approvability or commercialization of the particular product candidate or product and our business in general. In addition, the information we choose to publicly disclose regarding a particular study or clinical trial is based on what is typically extensive information, and you or others may not agree with what we determine is the material or otherwise appropriate information to include in our disclosure, and any information we determine not to disclose may ultimately be deemed significant with respect to future decisions, conclusions, views, activities or otherwise regarding a particular drug, product candidate or our business. If the interim, topline, or preliminary data that we report differ from actual results, or if others, including regulatory authorities, disagree with the conclusions reached, our ability to obtain approval for and commercialize our product candidates, our business, operating results, prospects or financial condition may be harmed.
The regulatory approval process is lengthy, expensive and uncertain, and we may be unable to obtain regulatory approval for our product candidates under applicable regulatory requirements. The denial or delay of any such approval would delay commercialization of our product candidates and adversely impact our ability to generate revenue, our business and our results of operations.
The development, research, testing, manufacturing, labeling, approval, selling, import, export, marketing, promotion and distribution of drug products are subject to extensive and evolving regulation by federal, state and local governmental authorities in the United States, principally the FDA, and by foreign authorities, such as the EU institutions or the competent authorities of the member states of the EU, which regulations differ from country to country. Neither we nor any future collaborator is permitted to market any of our product candidates in the United States or foreign jurisdictions until we receive regulatory approval of an NDA from the FDA or similar approval from foreign regulatory authorities.
Obtaining regulatory approval of an NDA or a similar foreign application can be a lengthy, expensive and uncertain process. Prior to obtaining approval to commercialize a product candidate in the United States or
52
abroad, we or our collaborators must demonstrate with substantial evidence from well-controlled clinical trials, and to the satisfaction of the FDA or other foreign regulatory agencies, that such product candidates are safe and effective for their intended uses. The number of preclinical studies and clinical trials that will be required for FDA or a foreign regulatory authority's approval varies depending on the product candidate, the disease or condition that the product candidate is designed to address, and the regulations applicable to any particular product candidate.
Results from preclinical studies and clinical trials can be interpreted in different ways. Even if we believe the preclinical or clinical data for our product candidates are promising, such data may not be sufficient to support approval by the FDA and other regulatory authorities. Administering product candidates to humans may produce undesirable side effects, which could interrupt, delay or halt clinical trials and result in the FDA or other regulatory authorities denying approval of a drug candidate for any or all indications. The FDA or foreign regulatory authorities may also require us to conduct additional studies or trials for our product candidates either prior to or post-approval, such as additional drug-drug interaction studies or safety or efficacy studies or trials, or it may object to elements of our clinical development program such as the number of subjects in our current clinical trials from the United States or abroad. We may experience difficulty in identifying and enrolling patients in such a trial, if one were to be required, which could interrupt, delay or halt the process of obtaining regulatory approval of our product candidates.
The FDA or any foreign regulatory bodies can delay, limit or deny approval of our product candidates or require us to conduct additional preclinical studies or clinical testing or abandon a program for many reasons, including:
Of the large number of drugs in development, only a small percentage successfully complete the FDA or other regulatory approval processes and are commercialized. The lengthy approval process as well as the unpredictability of future clinical trial results may result in our failing to obtain regulatory approval to market our
53
product candidates, which would significantly harm our business, financial condition, results of operations and prospects.
Even if we receive FDA approval of an NDA or foreign marketing application for avasopasem or our other product candidates, the FDA or the applicable foreign regulatory agency may grant approval contingent on the performance of costly additional clinical trials, including Phase 4 clinical trials, and/or in the case of the FDA, the implementation of a REMS, which may be required to ensure safe use of the drug after approval. The FDA or the applicable foreign regulatory agency also may approve a product candidate for a more limited indication or a narrower patient population than we originally requested, and the FDA or applicable foreign regulatory agency may not approve the labeling that we believe is necessary or desirable for the successful commercialization of a product candidate. Any delay in obtaining, or inability to obtain, applicable regulatory approval would delay or prevent commercialization of that product candidate and would materially adversely impact our business and prospects.
Changes in methods of product candidate manufacturing or formulation may result in additional costs or delay.
As product candidates proceed through preclinical studies to late-stage clinical trials towards potential approval and commercialization, it is common that various aspects of the development program, such as manufacturing methods and formulation, are altered along the way in an effort to optimize processes and results. Such changes carry the risk that they will not achieve these intended objectives. Any of these changes could cause our product candidates to perform differently and affect the results of planned clinical trials or other future clinical trials conducted with the altered materials. Such changes may also require additional testing, notification to the FDA or foreign regulatory authorities or approval by the FDA or foreign regulatory authorities. This could delay completion of clinical trials, require the conduct of bridging clinical trials or the repetition of one or more clinical trials, increase clinical trial costs, delay approval of our product candidates and jeopardize our ability to commence sales and generate revenue.
For example, in an effort to optimize scale-up efficiencies for avasopasem, we implemented certain changes to the manufacturing process related to the order of addition of ingredients. However, subsequent to this manufacturing change trace amounts of visible fine particles were observed in the drug product. Following notification to the FDA in April 2019 that we had voluntarily suspended dosing of avasopasem in all active clinical trials until we were able to resolve the issue, our INDs for avasopasem were temporarily placed on clinical hold. While we have now modified the manufacturing process and the FDA lifted the clinical holds in August 2019, and subsequently we added a filtration step to the preparation procedure for both avasopasem and placebo before administration to trial subjects to remove any particles that might form in the future, there can be no assurance that a similar or different manufacturing issue will not occur and one or more of our programs will not be placed on clinical hold in the future.
We may expend our limited resources to pursue a particular product candidate or indication and fail to capitalize on product candidates or indications that may be more profitable or for which there is a greater likelihood of success.
Because we have limited financial and management resources, we focus on development programs and product candidates that we identify for specific indications. As such, we are currently primarily focused on the development of avasopasem and rucosopasem. As a result, we may forego or delay pursuit of opportunities with other product candidates or for other indications for avasopasem or rucosopasem that later prove to have greater commercial potential. Our resource allocation decisions may cause us to fail to capitalize on viable commercial products or profitable market opportunities. Our spending on current and future development programs and product candidates for specific indications may not yield any commercially viable products. If we do not accurately evaluate the commercial potential or target market for a particular product candidate, we may relinquish valuable rights to that product candidate through collaboration, licensing or other royalty arrangements in cases in which it would have been more advantageous for us to retain sole development and commercialization rights to such product candidate.
54
While we have received Breakthrough Therapy Designation for avasopasem, we may not receive such designation for our other product candidates, and such designation for avasopasem or any other product candidate may not lead to a faster development or regulatory review or approval process and will not increase the likelihood that our product candidates will receive marketing approval.
We have received Breakthrough Therapy Designation from the FDA for avasopasem for the reduction of SOM induced by radiotherapy. We may also seek Breakthrough Therapy Designation for any other product candidates that we may develop. A Breakthrough Therapy is defined as a product candidate that is intended, alone or in combination with one or more other drugs or biologics, to treat a serious or life-threatening disease or condition, and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints. For product candidates that have been designated as Breakthrough Therapies, interaction and communication between the FDA and the sponsor of the trial can help to identify the most efficient path for clinical development while minimizing the number of patients placed in ineffective control regimens. Drugs designated as Breakthrough Therapies also receive the same benefits associated with Fast Track designation, including eligibility for rolling review of a submitted NDA, if the relevant criteria are met.
Designation as a Breakthrough Therapy is within the discretion of the FDA. Accordingly, even if we believe that a product candidate meets the criteria for designation as a breakthrough therapy, the FDA may disagree and instead determine not to make such designation. In any event, the receipt of Breakthrough Therapy Designation for a product candidate may not result in a faster development process, review or approval compared to products considered for approval under conventional FDA procedures and does not assure ultimate approval by the FDA. In addition, even if one or more product candidates qualify as breakthrough therapies, the FDA may later decide that the products no longer meet the conditions for qualification and rescind the designation, or otherwise decide that the time period required for FDA review or approval will not be reduced. Similarly, our products may not qualify for similar programs in other jurisdictions, such as the PRIME scheme in the EU.
We have received Fast Track Designation for avasopasem, and we may seek such designation for some or all of our other product candidates. We may not receive such designation, and even for those product candidates for which we do, it may not lead to a faster development or regulatory review or approval process and will not increase the likelihood that product candidates will receive marketing approval.
We have received Fast Track Designation from the FDA for avasopasem for the reduction of the severity and incidence of radiation and chemotherapy-induced OM, and we may seek Fast Track Designation for some or all of our other product candidates. If a drug is intended for the treatment of a serious or life-threatening condition or disease, and preclinical or clinical data demonstrate the potential to address an unmet medical need, the product may qualify for Fast Track Designation, for which sponsors must apply. The sponsor of a Fast Track product candidate has opportunities for more frequent interactions with the applicable FDA review team during product development and, once an NDA is submitted, the product candidate may be eligible for priority review. A Fast Track product candidate may also be eligible for rolling review, where the FDA may consider for review sections of the NDA on a rolling basis before the complete application is submitted, if the sponsor provides a schedule for the submission of the sections of the NDA, the FDA agrees to accept sections of the NDA and determines that the schedule is acceptable, and the sponsor pays any required user fees upon submission of the first section of the NDA. The FDA has broad discretion whether or not to grant this designation. Thus, even if we believe a particular product candidate is eligible for this designation, the FDA may decide not to grant it. Moreover, even if we do receive Fast Track Designation, we or our collaborators may not experience a faster development process, review or approval compared to conventional FDA procedures. In addition, the FDA may withdraw Fast Track Designation if it believes that the designation is no longer supported by data from our clinical development program. Many drugs that have received Fast Track Designation have failed to obtain approval.
Obtaining and maintaining regulatory approval of our product candidates in one jurisdiction does not mean that we will be successful in obtaining regulatory approval of our product candidates in other jurisdictions.
Obtaining and maintaining regulatory approval of our product candidates in one jurisdiction does not guarantee that we will be able to obtain or maintain regulatory approval in any other jurisdiction, while a failure or delay in obtaining regulatory approval in one jurisdiction may have a negative effect on the regulatory approval
55
process in others. For example, even if the FDA grants marketing approval of a product candidate, comparable regulatory authorities in foreign jurisdictions, such as the European Commission, or the competent authorities of the EU member states, must also approve the manufacturing and marketing of the product candidate in those countries. Approval procedures vary among jurisdictions and can involve requirements and administrative review periods different from, and greater than, those in the United States, including additional preclinical studies or clinical trials, as studies conducted in one jurisdiction may not be accepted by regulatory authorities in other jurisdictions. In many jurisdictions outside the United States, a product candidate must be approved for reimbursement before it can be approved for sale in that jurisdiction. In some cases, the price that we intend to charge for our products is also subject to approval.
Obtaining foreign regulatory approvals and compliance with foreign regulatory requirements could result in significant delays, difficulties and costs for us and could delay or prevent the introduction of our products in certain countries. If we fail to comply with the regulatory requirements in international markets and/or receive applicable marketing approvals, our target market size will be reduced and our ability to realize the full market potential of our product candidates will be harmed.
Since the end of the Brexit transition period on January 1, 2021, however, Great Britain (England, Scotland and Wales) has operated under a separate regulatory regime to the EU. EU laws regarding medicinal products only apply in respect of the United Kingdom to Northern Ireland (as set out in the Protocol on Ireland/Northern Ireland). The EU laws that have been transposed into United Kingdom law through secondary legislation remain applicable in Great Britain. While the United Kingdom has indicated a general intention that new laws regarding the development, manufacture and commercialization of medicinal products in the United Kingdom will align closely with EU law, there are limited detailed proposals for future regulation of medicinal products. The trade and cooperation agreement includes specific provisions concerning medicinal products, which include the mutual recognition of Good Manufacturing Practice, or GMP, inspections of manufacturing facilities for medicinal products and GMP documents issued (such mutual recognition can be rejected by either party in certain circumstances) but does not foresee wholesale mutual recognition of United Kingdom and EU pharmaceutical regulations. For example, it is not clear to what extent the United Kingdom will adopt legislation aligned with, or similar to, the EU CTR which became applicable on January 31, 2022 and which significantly reforms the assessment and supervision processes for clinical trials throughout the EU. Therefore, there remains political and economic uncertainty regarding to what extent the regulation of medicinal products will differ between the United Kingdom and the EU in the future. Any divergences will increase the cost and complexity of running our business, including with respect to the conduct of clinical trials.
Brexit also materially impacted the regulatory regime with respect to the approval of our product candidates. Great Britain is no longer covered by the EU’s procedures for the grant of marketing authorizations (Northern Ireland is covered by the centralized authorization procedure and can be covered under the decentralized or mutual recognition procedures). As of January 1, 2021, all existing centralized marketing authorizations were automatically converted into United Kingdom marketing authorizations effective in Great Britain and issued with a United Kingdom marketing authorization number on January 1, 2021 (unless marketing authorization holders opted out of this scheme). A separate marketing authorization is now required to market drugs in Great Britain. It is currently unclear whether the regulator in the United Kingdom, the MHRA is sufficiently prepared to handle the increased volume of marketing authorization applications that it is likely to receive. Further, the United Kingdom’s withdrawal from the EU has resulted in the relocation of the EMA from the United Kingdom to the Netherlands. This relocation has caused, and may continue to cause, disruption in the administrative and medical scientific links between the EMA and the MHRA, including delays in granting clinical trial authorization or marketing authorization, disruption of importation and export of active substance and other components of new drug formulations, and disruption of the supply chain for clinical trial product and final authorized formulations. The cumulative effects of the disruption to the regulatory framework may add considerably to the development lead time to marketing authorization and commercialization of products in the EU and/or the United Kingdom. Any delay in obtaining, or an inability to obtain, any marketing approvals, as a result of Brexit or otherwise, would prevent us from commercializing our product candidates in the United Kingdom and/or the EU and restrict our ability to generate revenue and achieve and sustain profitability. If any of these outcomes occur, we may be forced to restrict or delay efforts to seek regulatory approval in the United Kingdom and/or EU for our product candidates, which could significantly and materially harm our business.
56
Even if we receive regulatory approval of our product candidates, we will be subject to ongoing regulatory obligations and continued regulatory review, which may result in significant additional expense, and we may be subject to penalties if we fail to comply with regulatory requirements or experience unanticipated problems with our product candidates.
Any regulatory approvals that we receive for our product candidates may be subject to limitations on the approved indicated uses for which the product may be marketed or the conditions of approval, or contain requirements for potentially costly post-market testing and surveillance to monitor the safety and efficacy of the product candidate. The FDA may also require a REMS as a condition of approval of our product candidates, which could include requirements for a medication guide, physician communication plans or additional elements to ensure safe use, such as restricted distribution methods, patient registries and other risk minimization tools. Similar requirements may be requested by foreign regulatory authorities. In addition, if the FDA or a comparable foreign regulatory authority approves our product candidates, the manufacturing processes, labeling, packaging, distribution, adverse event reporting, storage, advertising, promotion, import, export and recordkeeping for our product candidates will be subject to extensive and ongoing regulatory requirements. These requirements include submissions of safety and other post-marketing information and reports, registration, as well as continued compliance with current good manufacturing practice-grade, or cGMP, or similar foreign requirements and GCP requirements for any clinical trials that we conduct post-approval. Later discovery of previously unknown problems with our product candidates, including adverse events of unanticipated severity or frequency, or with our third-party manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
Any government investigation of alleged violations of law could require us to expend significant time and resources in response and could generate negative publicity. Any failure to comply with ongoing regulatory requirements may significantly and adversely affect our ability to commercialize and generate revenue from our products. If regulatory sanctions are applied or if regulatory approval is withdrawn, the value of our company and our operating results will be adversely affected.
The FDA’s and other regulatory authorities’ policies may change and additional government regulations may be enacted that could prevent, limit or delay regulatory approval of our product candidates.
We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action, either in the United States or abroad. If we are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we are not able to maintain regulatory compliance, we may be subject to enforcement action and we may not achieve or sustain profitability.
Disruptions at the FDA and other government agencies caused by funding shortages or global health concerns could hinder their ability to hire, retain or deploy key leadership and other personnel, or otherwise prevent new or modified products from being developed, approved or commercialized in a timely manner or at all, which could negatively impact our business.
The ability of the FDA and foreign regulatory authorities to review and approve new products can be affected by a variety of factors, including government budget and funding levels, statutory, regulatory, and policy
57
changes, the FDA’s and foreign regulatory authorities’ ability to hire and retain key personnel and accept the payment of user fees, and other events that may otherwise affect the FDA’s and foreign regulatory authorities’ ability to perform routine functions. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, over the last several years, the U.S. government has shut down several times and certain regulatory agencies, such as the FDA, have had to furlough critical FDA employees and stop critical activities.
Separately, in response to the COVID-19 pandemic, the FDA postponed most inspections of domestic and foreign manufacturing facilities at various points. Even though the FDA has since resumed standard inspection operations of domestic facilities where feasible, the FDA has continued to monitor and implement changes to its inspection activities to ensure the safety of its employees and those of the firms it regulates as it adapts to the evolving COVID-19 pandemic, and any resurgence of the virus or emergence of new variants may lead to further inspectional delays. Regulatory authorities outside the United States have adopted similar restrictions or other policy measures in response to the COVID-19 pandemic. If a prolonged government shutdown occurs, or if global health concerns continue to prevent the FDA or other regulatory authorities from conducting their regular inspections, reviews, or other regulatory activities, it could significantly impact the ability of the FDA or other regulatory authorities to timely review and process our regulatory submissions, which could have a material adverse effect on our business.
The FDA and other regulatory agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses. If we are found or alleged to have improperly promoted off-label uses, we may become subject to significant liability.
The FDA and other regulatory agencies, including the competent authorities of the EU member states, strictly regulate the promotional claims that may be made about prescription products, as our product candidates would be, if approved. In particular, a product may not be promoted for uses that are not approved by the FDA or such other regulatory agencies as reflected in the product’s approved labeling. Physicians may nevertheless prescribe such drugs to their patients in a manner that is inconsistent with the approved label. For example, if we obtain approval for avasopasem for the reduction in the incidence of SOM in patients with locally advanced HNC receiving radiotherapy, we may pursue a strategy for avasopasem for the reduction of radiotherapy-induced esophagitis by presenting clinical data to entities like the National Comprehensive Cancer Network, or NCCN, to support use of avasopasem under these circumstances as a medically accepted indication in published drug compendia, notwithstanding the fact that we may not seek approval for avasopasem for radiotherapy-induced esophagitis by the FDA. Even if we are successful in obtaining Category 1 or Category 2A status from NCCN for avasopasem for the reduction of esophagitis, we will nevertheless be restricted from marketing and promoting the product for the reduction of esophagitis unless and until it is approved by the FDA for such indication.
If we are found to have promoted off-label uses, or if the government takes the position that our presenting clinical data related to off-label uses of avasopasem to NCCN or other drug compendia publishers to establish compendia-listed indications constitutes off-label promotion, we may become subject to significant liability. The federal government has levied large civil and criminal fines against companies for alleged improper promotion and has enjoined several companies from engaging in off-label promotion. The FDA has also requested that companies enter into consent decrees or permanent injunctions under which specified promotional conduct is changed or curtailed. If we cannot successfully manage the promotion of our product candidates, if approved, we could become subject to significant liability, which would materially adversely affect our business and financial condition. The same applies in foreign jurisdictions, including the EU.
58
Risks Related to Our Dependence on Third Parties
We rely, and will continue to rely, on third parties to conduct our clinical trials for our product candidates, and those third parties may not perform satisfactorily, including failing to meet deadlines for the completion of such trials.
We have relied, and expect to continue to rely, on CROs for the conduct of preclinical studies and clinical trials of avasopasem, rucosopasem and/or any other product candidates that we may progress to clinical development. We expect to continue to rely on third parties, such as clinical data management organizations, medical institutions and clinical investigators, to conduct those clinical trials. If any of our relationships with these third parties terminate, we may not be able to timely enter into arrangements with alternative third parties or to do so on commercially reasonable terms, if at all. In addition, any third parties conducting our clinical trials will not be our employees, and except for remedies available to us under our agreements with such third parties, we cannot control whether or not they devote sufficient time and resources to our clinical programs. We have no control over the ability of our CROs to maintain adequate quality control, quality assurance and qualified personnel. For example, in October 2021, we announced topline data from the Phase 3 ROMAN trial of avasopasem in SOM and reported that the trial did not achieve statistical significance on the primary endpoint. Upon further analysis of the ROMAN data, an error by the CRO was identified in the statistical program. Correction of this error yielded the correct, statistically significant p-values for the primary and a key secondary endpoint. We announced the correct topline results in December 2021. If our CROs and other third parties do not successfully carry out their contractual duties or obligations or meet expected deadlines, if they need to be replaced or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols, regulatory requirements, their standard operating procedures and policies, or for other reasons, our clinical trials may be extended, delayed or terminated and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates. Consequently, our results of operations and the commercial prospects for our product candidates would be harmed, our costs could increase substantially and our ability to generate revenue could be delayed significantly.
Switching or adding CROs involves substantial cost and requires management time and focus. In addition, there is a natural transition period when a new CRO commences work. As a result, delays occur, which can materially impact our ability to meet our desired clinical development timelines. Though we intend to carefully manage our relationships with our CROs, there can be no assurance that we will not encounter challenges or delays in the future or that these delays or challenges will not have a material adverse impact on our business, financial condition and prospects.
We rely on these parties for execution of our preclinical studies and clinical trials, and generally do not control their activities. Our reliance on these third parties for research and development activities will reduce our control over these activities but will not relieve us of our responsibilities. For example, we will remain responsible for ensuring that each of our clinical trials is conducted in accordance with the general investigational plan and protocols for the trial. Moreover, the FDA and foreign regulatory authorities require us to comply with GCPs for conducting, recording and reporting the results of clinical trials to assure that data and reported results are credible and accurate and that the rights, integrity and confidentiality of trial participants are protected. We are also required to register ongoing clinical trials and post the results of completed clinical trials on a government-sponsored database, ClinicalTrials.gov, within specified timeframes. Failure to do so can result in fines, adverse publicity and civil and criminal sanctions. If we or any of our CROs or other third parties, including trial sites, fails to comply with applicable GCPs, the clinical data generated in our clinical trials may be deemed unreliable and the FDA, EMA or comparable foreign regulatory authorities may require us to perform additional clinical trials before approving our marketing applications. We cannot assure you that upon inspection by a given regulatory authority, such regulatory authority will determine that any of our clinical trials complies with GCP regulations. In addition, our clinical trials must be conducted with product produced under cGMP or similar foreign conditions. Our failure to comply with these regulations may require us to repeat clinical trials, which would delay the regulatory approval process.
In addition, principal investigators for our clinical trials may serve as scientific advisors or consultants to us from time to time and receive compensation in connection with such services. Under certain circumstances, we may be required to report some of these relationships to the FDA or foreign regulatory authorities. The FDA or foreign regulatory authorities may conclude that a financial relationship between us and a principal investigator has created a conflict of interest or otherwise affected interpretation of the trial. The FDA or foreign regulatory
59
authorities may therefore question the integrity of the data generated at the applicable clinical trial site and the utility of the clinical trial itself may be jeopardized. This could result in a delay in approval, or rejection, of our marketing applications by the FDA or foreign regulatory authorities and may ultimately lead to the denial of marketing approval of avasopasem, rucosopasem and any other product candidates.
We also expect to rely on other third parties to store and distribute product supplies for our clinical trials. Any performance failure on the part of our distributors could delay clinical development or marketing approval of our product candidates or commercialization of our products, producing additional losses and depriving us of potential revenue.
We contract with third parties for the manufacture and supply of our product candidates for preclinical and clinical testing and expect to continue to do so for commercialization. This reliance on third parties increases the risk that we will not have sufficient quantities of our product candidates or such quantities at an acceptable cost, which could delay, prevent or impair our development or commercialization efforts.
We do not have any manufacturing facilities or personnel. We do not have any long-term contractual arrangements with manufacturers and instead rely on third parties to manufacture our product candidates on a purchase-order or work-order basis. We currently have limited manufacturing arrangements, and we cannot be certain that we will be able to establish redundancy in manufacturers for our product candidates, which could lead to reliance on a limited number of manufacturers for one or more of our product candidates. This reliance increases the risk that we will not have sufficient quantities of our drug candidates or products, if approved, or such quantities at an acceptable cost or quality, which could delay, prevent or impair our development or commercialization efforts.
We also expect to rely on third-party manufacturers or third-party collaborators for the manufacture of commercial supply of avasopasem, if approved, and any other product candidates for which we obtain marketing approval. The facilities used by our contract manufacturing organizations, or CMOs, to manufacture our product candidates must be approved by the FDA or other regulatory authorities for the manufacture of our product candidates pursuant to inspections that will be conducted after we submit our NDA or comparable marketing application to the FDA or other regulatory authority. We do not have control over a supplier’s or manufacturer’s compliance with laws, regulations and applicable cGMP standards and other laws and regulations, such as those related to environmental health and safety matters. If our CMOs cannot successfully manufacture material that conforms to our specifications and the strict regulatory requirements of the FDA or others, they will not be able to secure and maintain regulatory approval for their manufacturing facilities. In addition, we have no control over the ability of our CMOs to maintain adequate quality control, quality assurance and qualified personnel. If the FDA or a comparable foreign regulatory authority does not approve these facilities for the manufacture of our product candidates or if it withdraws any such approval in the future, we may need to find alternative manufacturing facilities, which would significantly impact our ability to develop, obtain regulatory approval for or market our product candidates, if approved. If our current or future suppliers are unable to supply us with sufficient raw materials for our preclinical studies and clinical trials, we may experience delays in our development efforts as we locate and qualify new raw material manufacturers.
We may be unable to establish any agreements with future third-party manufacturers or to do so on acceptable terms. Even if we are able to establish agreements with third-party manufacturers, qualifying and validating such manufacturers may take a significant period of time and reliance on third-party manufacturers entails additional risks, including:
60
Third-party manufacturers may not be able to comply with cGMP regulations or other regulatory requirements outside the United States. Our failure, or the failure of our third-party manufacturers, to comply with applicable regulations could result in sanctions being imposed on us, including clinical holds, fines, injunctions, civil penalties, delays, suspension or withdrawal of approvals, license revocation, seizures or recalls of product candidates or drugs, operating restrictions and criminal prosecutions, any of which could significantly and adversely affect supplies of our products.
Our product candidates and any drugs that we may develop may compete with other product candidates and drugs for access to manufacturing facilities. There are no assurances we would be able to enter into similar commercial arrangements with other manufacturers that operate under cGMP regulations or other regulatory requirements outside the United States and that might be capable of manufacturing for us. Any performance failure on the part of our existing or future manufacturers could delay clinical development or marketing approval.
We may seek collaborations with third parties for the development or commercialization of our product candidates. If those collaborations are not successful, we may not be able to capitalize on the market potential of these product candidates.
We may seek third-party collaborators for the development and commercialization of our product candidates, including for the commercialization of any of our product candidates that are approved for marketing outside the United States. Our likely collaborators for any collaboration arrangements include large and mid-size pharmaceutical companies, regional and national pharmaceutical companies and biotechnology companies. If we do enter into any such arrangements with any third parties, we will likely have limited control over the amount and timing of resources that our collaborators dedicate to the development or commercialization of our product candidates. Our ability to generate revenue from these arrangements will depend on our collaborators’ abilities to successfully perform the functions assigned to them in these arrangements.
Collaborations involving our product candidates would pose the following risks to us:
61
Collaboration agreements may not lead to development or commercialization of product candidates in the most efficient manner or at all. If a present or future collaborator of ours were to be involved in a business combination, the continued pursuit and emphasis on our product development or commercialization program could be delayed, diminished or terminated.
If we seek, but are not able to establish, collaborations, we may have to alter our development and commercialization plans.
Our product development programs and the potential commercialization of our product candidates will require substantial additional capital. For some of our product candidates, we may decide to collaborate with pharmaceutical and biotechnology companies for the development and potential commercialization of those product candidates.
We face significant competition in seeking appropriate collaborators. Whether we reach a definitive agreement for a collaboration will depend, among other things, upon our assessment of the collaborator’s resources and expertise, the terms and conditions of the proposed collaboration and the proposed collaborator’s evaluation of a number of factors. Those factors may include the design or results of clinical trials, the likelihood of approval by the FDA or comparable regulatory authorities outside the United States, the potential market for the subject product candidate, the costs and complexities of manufacturing and delivering such product candidate to patients, the potential of competing products, the existence of uncertainty with respect to our ownership of technology, which can exist if there is a challenge to such ownership without regard to the merits of the challenge and industry and market conditions generally. The collaborator may also consider alternative product candidates or technologies for similar indications that may be available to collaborate on and whether such a collaboration could be more attractive than the one with us for our product candidate. Collaborations are complex and time-consuming to negotiate and document. In addition, there have been a significant number of recent business combinations among large pharmaceutical companies that have resulted in a reduced number of potential future collaborators.
We may not be able to negotiate collaborations on a timely basis, on acceptable terms, or at all. If we are unable to do so, we may have to curtail the development of such product candidate, reduce or delay its development program or one or more of our other development programs, delay its potential commercialization or reduce the scope of any sales or marketing activities, or increase our expenditures and undertake development or commercialization activities at our own expense. If we elect to increase our expenditures to fund development or commercialization activities on our own, we may need to obtain additional capital, which may not be available to us
62
on acceptable terms or at all. If we do not have sufficient funds, we may not be able to further develop our product candidates or bring them to market and generate revenue.
Risks Related to Commercialization
Even if any of our product candidates receives marketing approval, it may fail to achieve the degree of market acceptance by physicians, patients, third-party payors and others in the medical community necessary for commercial success.
If any of our product candidates, including avasopasem, receives marketing approval, it may nonetheless fail to gain sufficient market acceptance by physicians, patients, third-party payors and others in the medical community. If our product candidates do not achieve an adequate level of acceptance, we may not generate significant revenue and we may not become profitable. The degree of market acceptance of our product candidates, if approved for commercial sale, will depend on a number of factors, including:
63
If we are unable to establish our own sales, marketing and distribution capabilities, or enter into agreements with third parties to sell and market avasopasem or any other product candidates, we may not be successful in commercializing our product candidates if and when they are approved, and we may not be able to generate any revenue.
We do not currently have a sales, marketing or distribution infrastructure. We have never sold, marketed or distributed any therapeutic products. To achieve commercial success for any approved product candidate, we will need to establish a sales and marketing organization. Under the amended Royalty Agreement with Blackstone, we are required to establish a trained sales force sufficiently in advance of any anticipated commercial launch in a country where we seek to commercialize avasopasem or related product candidates. We expect to build a specialized sales and marketing organization of approximately 40 sales representatives to market our product candidates to the approximately 5,000 radiation oncologists in the United States. There are risks involved with establishing our own sales and marketing capabilities. For example, recruiting and training a sales force is expensive and time consuming and could delay any drug launch. If the commercial launch of a product candidate for which we recruit a sales force and establish marketing capabilities is delayed or does not occur for any reason, we would have prematurely or unnecessarily incurred these commercialization expenses. This may be costly, and our investment would be lost if we cannot retain or reposition our sales and marketing personnel.
Factors that may inhibit our efforts to commercialize our product candidates on our own include:
If we are unable to establish our own sales, marketing and distribution capabilities and are forced to enter into arrangements with, and rely on, third parties to perform these services, our revenue and our profitability, if any, are likely to be lower than if we had developed such capabilities ourselves. In addition, we may not be successful in entering into arrangements with third parties to sell, market and distribute our product candidates or may be unable to do so on terms that are favorable to us. We likely will have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market our products effectively. If we do not establish sales, marketing and distribution capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our product candidates.
The incidence and prevalence for target patient populations of our product candidates have not been established with precision. If the market opportunities for our product candidates are smaller than we estimate, or if any approval that we obtain is based on a narrower definition of the patient population, our revenue and ability to achieve profitability may be materially adversely affected.
The precise incidence and prevalence for all the conditions we aim to address with our product candidates are unknown and cannot be precisely determined. Our projections of both the number of people who have these diseases, as well as the subset of people with these diseases who have the potential to benefit from
64
treatment with our product candidates, are based on beliefs and estimates. These estimates have been derived from a variety of sources, including the scientific literature, surveys of clinics, patient foundations or market research, and may prove to be incorrect. Further, new trials may change the estimated incidence or prevalence of these diseases.
The total addressable market across all of our product candidates will ultimately depend upon, among other things, the diagnosis criteria included in the final label for each of our product candidates approved for sale for these indications, acceptance by the medical community and patient access, drug pricing and reimbursement. The number of patients in the United States and other major markets and elsewhere may turn out to be lower than expected, patients may not be otherwise amenable to treatment with our products or new patients may become increasingly difficult to identify or gain access to, all of which would adversely affect our results of operations and our business. Further, even if we obtain significant market share for our product candidates, because the potential target populations are very small, we may never achieve profitability despite obtaining such significant market share.
The successful commercialization of avasopasem or any other product candidates will depend in part on the extent to which governmental authorities and health insurers establish coverage, adequate reimbursement levels and pricing policies. Failure to obtain or maintain coverage and adequate reimbursement for our product candidates, if approved, could limit our ability to market those products and decrease our ability to generate revenue.
The availability of coverage and adequacy of reimbursement by governmental healthcare programs such as Medicare and Medicaid, private health insurers and other third-party payors are essential for most patients to be able to afford medical services and pharmaceutical products such as our product candidates, assuming FDA approval. Our ability to achieve acceptable levels of coverage and reimbursement for our products or procedures using our products by governmental authorities, private health insurers and other organizations will have an effect on our ability to successfully commercialize our product candidates. Obtaining coverage and adequate reimbursement for our products may be particularly difficult because of the higher prices often associated with drugs administered under the supervision of a physician. Separate reimbursement for the product itself or the treatment or procedure in which our product is used may not be available. A decision by a third-party payor not to cover or separately reimburse for our products or procedures using our products, could reduce physician utilization of our products once approved. Assuming there is coverage for our product candidates or procedures using our product candidates by a third-party payor, the resulting reimbursement payment rates may not be adequate or may require co-payments that patients find unacceptably high. We cannot be sure that coverage and reimbursement in the United States, the EU or elsewhere will be available for our product candidates or any product that we may develop, and any reimbursement that may become available may not be adequate or may be decreased or eliminated in the future.
Third-party payors increasingly are challenging prices charged for pharmaceutical products and services, and many third-party payors may refuse to provide coverage and reimbursement for particular drugs when an equivalent generic drug, biosimilar or a less expensive therapy is available. It is possible that a third-party payor may consider our product candidates as substitutable and only offer to reimburse patients for the less expensive product. Even if we show improved efficacy or improved convenience of administration with our product candidates, pricing of existing third-party therapeutics may limit the amount we will be able to charge for our product candidates. These payors may deny or revoke the reimbursement status of a given product or establish prices for new or existing marketed products at levels that are too low to enable us to realize an appropriate return on our investment in our product candidates. If reimbursement is not available or is available only at limited levels, we may not be able to successfully commercialize our product candidates and may not be able to obtain a satisfactory financial return on our product candidates.
There is significant uncertainty related to the insurance coverage and reimbursement of newly approved products. In the United States, third-party payors, including private and governmental payors, such as the Medicare and Medicaid programs, play an important role in determining the extent to which new drugs will be covered. The Medicare and Medicaid programs increasingly are used as models in the United States for how private payors and other governmental payors develop their coverage and reimbursement policies for drugs. Some third-party payors may require pre-approval of coverage for new or innovative devices or drug therapies before they will reimburse healthcare providers who use such therapies. We cannot predict at this time what third-party payors will decide with respect to the coverage and reimbursement for our product candidates.
65
No uniform policy for coverage and reimbursement for products exists among third-party payors in the United States. Therefore, coverage and reimbursement for products can differ significantly from payor to payor. As a result, the coverage determination process is often a time-consuming and costly process that will require us to provide scientific and clinical support for the use of our product candidates to each payor separately, with no assurance that coverage and adequate reimbursement will be applied consistently or obtained in the first instance. Furthermore, rules and regulations regarding reimbursement change frequently, in some cases on short notice, and we believe that changes in these rules and regulations are likely.
Outside the United States, international operations are generally subject to extensive governmental price controls and other market regulations, and we believe the increasing emphasis on cost-containment initiatives in Europe and other countries have and will continue to put pressure on the pricing and usage of our product candidates. In many countries, the prices of medical products are subject to varying price control mechanisms as part of national health systems. Other countries allow companies to set their own prices for medical products but monitor and control company profits. Additional foreign price controls or other changes in pricing regulation could restrict the amount that we are able to charge for our product candidates. Accordingly, in markets outside the United States, the reimbursement for our product candidates may be reduced compared with the United States and may be insufficient to generate commercially reasonable revenue and profits.
Moreover, increasing efforts by governmental and third-party payors in the United States and abroad to cap or reduce healthcare costs may cause such organizations to limit both coverage and the level of reimbursement for newly approved products and, as a result, they may not cover or provide adequate payment for our product candidates. We expect to experience pricing pressures in connection with the sale of our product candidates due to the trend toward managed health care, the increasing influence of health maintenance organizations and additional legislative changes. The downward pressure on healthcare costs in general, particularly prescription drugs and surgical procedures and other treatments, has become intense. As a result, increasingly high barriers are being erected to the entry of new products.
Enacted and future healthcare legislation may increase the difficulty and cost for us to obtain marketing approval of and commercialize our product candidates and may affect the prices we may set.
In the United States, the EU and other jurisdictions, there have been, and we expect there will continue to be, a number of legislative and regulatory changes and proposed changes to the healthcare system that could affect our future results of operations. In particular, there have been and continue to be a number of initiatives at the U.S. federal and state levels that seek to reduce healthcare costs and improve the quality of healthcare. For example, in March 2010, the Patient Protection and Affordable Care Act, as amended by the Health Care and Education Reconciliation Act, or collectively the ACA, was enacted, which substantially changed the way healthcare is financed by both governmental and private insurers. Among the provisions of the ACA, those of greatest importance to the pharmaceutical and biotechnology industries include the following:
66
Since its enactment, there have been judicial, executive and Congressional challenges to certain aspects of the ACA. On June 17, 2021, the U.S. Supreme Court dismissed the most recent judicial challenge to the ACA brought by several states without specifically ruling on the constitutionality of the ACA. Prior to the Supreme Court’s decision, President Biden issued an Executive Order to initiate a special enrollment period for purposes of obtaining health insurance coverage through the ACA marketplace. The Executive Order also instructed certain governmental agencies to review and reconsider their existing policies and rules that limit access to healthcare, including among others, reexamining Medicaid demonstration projects and waiver programs that include work requirements, and policies that create unnecessary barriers to obtaining access to health insurance coverage through Medicaid or the ACA.
In addition, other legislative changes have been proposed and adopted in the United States since the ACA was enacted. For example, the Budget Control Act of 2011, among other things, led to aggregate reductions of Medicare payments to providers, which went into effect in April 2013 and, due to subsequent legislative amendments to the statute will remain in effect through 2032, with the exception of a temporary suspension from May 1, 2020 through March 31, 2022, unless additional action is taken by Congress. On March 11, 2021, President Biden signed the American Rescue Plan Act of 2021 into law, which eliminates the statutory Medicaid drug rebate cap, currently set at 100% of a drug's average manufacturer price, for single source and innovator multiple source drugs, beginning January 1, 2024. In addition, the American Taxpayer Relief Act of 2012, which, among other things, further reduced Medicare payments to several types of providers, including hospitals, imaging centers and cancer treatment centers, and increased the statute of limitations period for the government to recover overpayments to providers from three to five years. These new laws or any other similar laws introduced in the future may result in additional reductions in Medicare and other health care funding, which could negatively affect our customers and accordingly, our financial operations.
Moreover, payment methodologies may be subject to changes in healthcare legislation and regulatory initiatives. For example, CMS may develop new payment and delivery models, such as bundled payment models. In addition, recently there has been heightened governmental scrutiny over the manner in which manufacturers set prices for their marketed products, which has resulted in several U.S. Congressional inquiries, and Congress has proposed and enacted federal legislation designed to, among other things, bring more transparency to drug pricing, reduce the cost of prescription drugs under Medicare, and review the relationship between pricing and manufacturer patient programs. On August 16, 2022, the Inflation Reduction Act of 2022, or IRA, was signed into law. Among other things, the IRA requires manufacturers of certain drugs to engage in price negotiations with Medicare (beginning in 2026), with prices that can be negotiated subject to a cap; imposes rebates under Medicare Part B and Medicare Part D to penalize price increases that outpace inflation (first due in 2023); and replaces the Part D coverage gap discount program with a new discounting program (beginning in 2025). The IRA permits the Secretary of the Department of Health and Human Services (HHS) to implement many of these provisions through guidance, as opposed to regulation, for the initial years. For that and other reasons, it is currently unclear how the IRA will be effectuated. Individual states in the United States have also increasingly passed legislation and implemented regulations designed to control pharmaceutical product pricing, including price or patient reimbursement constraints, discounts, restrictions on certain product access and marketing cost disclosure and transparency measures, and, in some cases, designed to encourage importation from other countries and bulk purchasing. In addition, regional healthcare authorities and individual hospitals are increasingly using bidding procedures to determine what pharmaceutical products and which suppliers will be included in their prescription drug and other healthcare programs. This could reduce the ultimate demand for our product candidates or put pressure on our product pricing. We expect that additional U.S. healthcare reform measures will be adopted in the future, any of which could limit the amounts paid for healthcare products and services, which could result in reduced demand for our product candidates or additional pricing pressures.
67
In the EU, similar political, economic and regulatory developments may affect our ability to profitably commercialize our product candidates, if approved. In addition to continuing pressure on prices and cost containment measures, legislative developments at the EU or member state level may result in significant additional requirements or obstacles that may increase our operating costs. The delivery of healthcare in the EU, including the establishment and operation of health services and the pricing and reimbursement of medicines, is almost exclusively a matter for national, rather than EU, law and policy. National governments and health service providers have different priorities and approaches to the delivery of health care and the pricing and reimbursement of products in that context. In general, however, the healthcare budgetary constraints in most EU member states have resulted in restrictions on the pricing and reimbursement of medicines by relevant health service providers. Coupled with ever-increasing EU and national regulatory burdens on those wishing to develop and market products, this could prevent or delay marketing approval of our product candidates, restrict or regulate post-approval activities and affect our ability to commercialize our product candidates, if approved.
In markets outside of the United States and the EU, reimbursement and healthcare payment systems vary significantly by country, and many countries have instituted price ceilings on specific products and therapies.
We cannot predict the likelihood, nature or extent of government regulation that may arise from future legislation or administrative action in the United States, the EU or any other jurisdiction. If we or any third parties we may engage are slow or unable to adapt to changes in existing requirements or the adoption of new requirements or policies, or if we or such third parties are not able to maintain regulatory compliance, our product candidates may lose any regulatory approval that may have been obtained and we may not achieve or sustain profitability.
Our future growth may depend, in part, on our ability to penetrate foreign markets, where we would be subject to additional regulatory burdens and other risks and uncertainties.
Our future profitability may depend, in part, on our ability to commercialize our product candidates in foreign markets for which we may rely on collaboration with third parties. We are evaluating the opportunities for the development and commercialization of our product candidates in foreign markets. We are not permitted to market or promote any of our product candidates before we receive regulatory approval from the applicable regulatory authority in that foreign market, and we may never receive such regulatory approval for any of our product candidates. To obtain separate regulatory approval in many other countries we must comply with numerous and varying regulatory requirements of such countries regarding safety and efficacy and governing, among other things, clinical trials and commercial sales, pricing and distribution of our product candidates, and we cannot predict success in these jurisdictions. If we obtain approval of our product candidates and ultimately commercialize our product candidates in foreign markets, we would be subject to additional risks and uncertainties, including:
68
Foreign sales of our product candidates could also be adversely affected by the imposition of governmental controls, political and economic instability, trade restrictions and changes in tariffs.
In some countries, particularly the countries in Europe, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a drug. To obtain reimbursement or pricing approval in some countries, we may be required to conduct a clinical trial that compares the cost-effectiveness of our product candidate to other available therapies. If reimbursement of our products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business could be harmed, possibly materially.
Product liability lawsuits against us could cause us to incur substantial liabilities and could limit commercialization of any product candidates that we may develop.
We will face an inherent risk of product liability exposure related to the testing of our product candidates in human clinical trials and will face an even greater risk if we commercially sell any product candidates that we may develop. If we cannot successfully defend ourselves against claims that our product candidates caused injuries, we could incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
Although we maintain product liability insurance coverage, it may not be adequate to cover all liabilities that we may incur and is subject to deductibles and coverage limitations. We anticipate that we will need to increase our insurance coverage when and if we successfully commercialize any product candidate. Insurance coverage is increasingly expensive. We may not be able to maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise. If we are unable to obtain insurance at acceptable cost or otherwise protect against potential product liability claims, we will be exposed to significant liabilities, which may materially and adversely affect our business and financial position. These liabilities could prevent or interfere with our commercialization efforts.
69
Risks Related to Competition, Retaining Key Employees and Managing Growth
We face substantial competition, which may result in others discovering, developing or commercializing drugs before or more successfully than we do.
The development and commercialization of new drugs and biologics is highly competitive. We face competition with respect to our current product candidates and will face competition with respect to any product candidates that we may seek to develop or commercialize in the future, from major pharmaceutical companies, specialty pharmaceutical companies and biotechnology companies worldwide. There are a number of large pharmaceutical and biotechnology companies that currently market and sell drugs or biologics that are pursuing the development of therapies in the fields in which we are interested. Some of these competitive products and therapies are based on entirely different scientific approaches to our approach. Potential competitors also include academic institutions, government agencies and other public and private research organizations that conduct research, seek patent protection and establish collaborative arrangements for research, development, manufacturing and commercialization.
Many of the companies against which we are competing or against which we may compete in the future have significantly greater financial resources, a more established presence in the market, and more expertise in research and development, manufacturing, preclinical studies and clinical trials, obtaining regulatory approvals and reimbursement and marketing approved products than we do. Mergers and acquisitions in the pharmaceutical, biotechnology and diagnostic industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller or early stage companies may also prove to be significant competitors, particularly through collaborative arrangements with large and established companies. These competitors also compete with us in recruiting and retaining highly qualified scientific, sales, marketing and management personnel and establishing clinical trial sites and patient registration for clinical trials, as well as in acquiring technologies complementary to, or necessary for, our programs.
Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize products that are safer, more effective, have fewer or less severe side effects, are more convenient or are less expensive than any drugs that we or our collaborators may develop. Because our product candidates are designed to reduce normal tissue toxicity from radiotherapy, or to increase the anti-cancer efficacy, our commercial opportunities could also be reduced or eliminated if radiotherapy methods are improved in a way that reduces normal tissue toxicity or increases anti-cancer efficacy, or if new therapies are developed which effectively treat cancer with less or without normal tissue toxicity. Our competitors also may obtain FDA or other regulatory approval for their products more rapidly than we may obtain approval for ours, which could result in our competitors establishing a strong market position before we or our collaborators are able to enter the market. The key competitive factors affecting the success of all of our product candidates, if approved, are likely to be their efficacy, safety, convenience, price, the effectiveness of companion diagnostics in guiding the use of related products, market acceptance by physicians and patients, the level of generic competition and the availability of reimbursement from government and other third-party payors.
Our future success depends on our ability to retain key executives and to attract, retain and motivate qualified personnel.
We have a limited operating history and are highly dependent on the research and development, clinical, commercial and business development expertise of the principal members of our management, scientific and clinical team. Although we have entered into employment agreements with our executive officers, each of them may terminate their employment with us at any time. We do not maintain “key person” insurance for any of our executives or other employees. In addition, we rely on consultants and advisors, including scientific and clinical advisors, to assist us in formulating our research and development and commercialization strategy. Our consultants and advisors may be employed by employers other than us and may have commitments under consulting or advisory contracts with other entities that may limit their availability to us. If we are unable to continue to attract and retain high quality personnel, our ability to pursue our growth strategy will be limited.
Recruiting and retaining qualified scientific, clinical, manufacturing and sales and marketing personnel will also be critical to our success. The failure to recruit, or the loss of the services of our executive officers or other
70
key employees could impede the achievement of our research, development and commercialization objectives and seriously harm our ability to successfully implement our business strategy. Furthermore, replacing executive officers and key employees may be difficult and may take an extended period of time because of the limited number of individuals in our industry with the breadth of skills and experience required to successfully develop, gain regulatory approval of and commercialize products. Competition to hire from this limited pool is intense, and we may be unable to hire, train, retain or motivate these key personnel on acceptable terms given the competition among numerous pharmaceutical and biotechnology companies for similar personnel. We also experience competition for the hiring of scientific and clinical personnel from universities and research institutions. Failure to succeed in clinical trials may make it more challenging to recruit and retain qualified scientific personnel. If we are not able to continue to attract and retain, on acceptable terms, the qualified personnel necessary for the continued development of our business, we may not be able to sustain our operations or growth.
We will need to develop and expand our company, and we may encounter difficulties in managing this development and expansion, which could disrupt our operations.
We expect to increase our number of employees and the scope of our operations. To manage our anticipated development and expansion, we must continue to implement and improve our managerial, operational and financial systems, expand our facilities and continue to recruit and train additional qualified personnel. Also, our management may need to divert a disproportionate amount of its attention away from its day-to-day activities and devote a substantial amount of time to managing these development activities. Due to our limited resources, certain employees may need to perform activities that are beyond their regular scope of work, and we may not be able to effectively manage the expansion of our operations or recruit and train additional qualified personnel. This may result in weaknesses in our infrastructure, give rise to operational mistakes, loss of business opportunities, loss of employees and reduced productivity among remaining employees. The physical expansion of our operations may lead to significant costs and may divert financial resources from other projects, such as the development of our product candidates. If our management is unable to effectively manage our expected development and expansion, our expenses may increase more than expected, our ability to generate revenue could be reduced and we may not be able to implement our business strategy. Our future financial performance and our ability to commercialize our product candidates, if approved, and compete effectively will depend, in part, on our ability to effectively manage the future development and expansion of our company.
We may not be successful in executing our growth strategy to identify, discover, develop, in-license or acquire additional product candidates or our growth strategy may not deliver the anticipated results.
We plan to source new product candidates that are complementary to our existing product candidates through our internal discovery program, or in-licensing or acquiring them from other companies or academic institutions. If we are unable to identify, discover, develop, in-license or acquire and integrate product candidates in accordance with this strategy, our ability to pursue this part of our growth strategy would be limited.
Research programs and business development efforts to identify new product candidates require substantial technical, financial and human resources. We may focus our efforts and resources on potential programs or product candidates that ultimately prove to be unsuccessful. In-licensing and acquisitions of technology often require significant payments, expenses and will consume additional resources. We will need to devote a substantial amount of time and personnel to research, develop and commercialize any acquired technology, in addition to our existing portfolio of programs. Our research programs, business development efforts or licensing attempts may fail to yield additional complementary or successful product candidates for clinical development and commercialization for a number of reasons, including, but not limited to, the following:
71
If any of these events occurs, we may not be successful in executing our growth strategy or our growth strategy may not deliver the anticipated results.
Risks Related to Intellectual Property
If we are unable to adequately protect our proprietary technology and product candidates, if the scope of the patent protection obtained is not sufficiently broad, or if the terms of our patents are insufficient to protect our product candidates for an adequate amount of time, our competitors could develop and commercialize technology and products similar or identical to ours, and our ability to successfully commercialize our product candidates may be materially impaired.
We rely primarily upon a combination of patents, trademarks, trade secret protection, and other intellectual property rights as well as nondisclosure, confidentiality and other contractual agreements to protect the intellectual property related to our brands, product candidates, including avasopasem and rucosopasem, and other proprietary technologies. Our success depends on our ability to develop, manufacture, market and sell our product candidates, if approved, and use our proprietary technologies without alleged or actual infringement, misappropriation or other violation of the patents and other intellectual property rights of third parties. There have been many lawsuits and other proceedings asserting patents and other intellectual property rights in the pharmaceutical and biotechnology industries. We cannot assure you that our product candidates, including avasopasem and rucosopasem, will not infringe existing or future third-party patents. Because patent applications can take many years to issue and may be confidential for 18 months or more after filing, there may be applications now pending of which we are unaware and which may later result in issued patents that we may infringe by commercializing our product candidates, including avasopasem and rucosopasem. There may also be issued patents or pending patent applications that we are aware of, but that we think are irrelevant to our product candidates, including avasopasem and rucosopasem, which may ultimately be found to be infringed by the manufacture, sale, or use of our product candidates, including avasopasem and rucosopasem. Moreover, we may face claims from non-practicing entities that have no relevant product revenue and against whom our own patent portfolio may thus have no deterrent effect. In addition, many of our product candidates, including avasopasem and rucosopasem, have a
72
complex structure that makes it difficult to conduct a thorough search and review of all potentially relevant third-party patents. Because we have not yet conducted a formal freedom to operate analysis for patents related to our product candidates, we may not be aware of issued patents that a third party might assert are infringed by one of our current or future product candidates, which could materially impair our ability to commercialize our product candidates. Even if we diligently search third-party patents for potential infringement by our products or product candidates, including avasopasem or rucosopasem, we may not successfully find patents that our products or product candidates, including avasopasem or rucosopasem, may infringe. If we are unable to secure and maintain freedom to operate, others could preclude us from commercializing our product candidates.
The process of obtaining patent protection is expensive and time-consuming, and we may not be able to prosecute all necessary or desirable patent applications at a reasonable cost or in a timely manner. We may choose not to seek patent protection for certain innovations or products and may choose not to pursue patent protection in certain jurisdictions, and under the laws of certain jurisdictions, patents or other intellectual property rights may be unavailable or limited in scope and, in any event, any patent protection we obtain may be limited. As a result, in some jurisdictions some of our products currently or in the future may not be, protected by patents. We generally apply for patents in those countries where we intend to make, have made, use, offer for sale, or sell products and where we assess the risk of infringement to justify the cost of seeking patent protection. However, we may not accurately predict all the countries where patent protection would ultimately be desirable. If we fail to timely file a patent application in any such country or major market, we may be precluded from doing so at a later date. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and, further, may export otherwise infringing products to territories in which we have patent protection that may not be sufficient to terminate infringing activities. In addition, the actual protection afforded by a patent varies on a product-by-product basis, from country to country, and depends upon many factors, including the type of patent, the scope of its coverage, the availability of regulatory-related extensions, the availability of legal remedies in a particular country and the validity and enforceability of the patent.
Furthermore, we cannot guarantee that any patents will be issued from any pending or future owned or licensed patent applications, or that any current or future patents will provide us with any meaningful protection or competitive advantage. Even if issued, existing or future patents may be challenged, including with respect to ownership, narrowed, invalidated, held unenforceable or circumvented, any of which could limit our ability to prevent competitors and other third parties from developing and marketing similar products or limit the length of terms of patent protection we may have for our product candidates, including avasopasem and rucosopasem, and technologies. Moreover, should we be unable to obtain meaningful patent coverage for clinically relevant infusion rates for avasopasem and rucosopasem in jurisdictions with commercially significant markets, our ability to extend and reinforce patent protection for these product candidates in those jurisdictions may be adversely impacted, which could limit our ability to prevent competitors and other third parties from developing and marketing similar products or limit the length of terms of patent protection we may have for those product candidates. Other companies may also design around technologies we have patented, licensed or developed. In addition, the issuance of a patent does not give us the right to practice the patented invention. Third parties may have blocking patents that could prevent us from marketing our products or practicing our own patented technology.
The patent positions of biotechnology and pharmaceutical companies can be highly uncertain and involve complex legal, scientific and factual questions for which important legal principles remain unresolved. As a result, the issuance, scope, validity, enforceability and commercial value of our patent rights may be uncertain. The standards that the United States Patent and Trademark Office, or the USPTO, and its foreign counterparts use to grant patents are not always applied predictably or uniformly. Changes in either the patent laws, implementing regulations or the interpretation of patent laws may diminish the value of our rights. The legal systems of certain countries do not protect intellectual property rights to the same extent as the laws of the United States, and many companies have encountered significant problems in protecting and defending such rights in foreign jurisdictions. For example, patent laws in various jurisdictions, including significant commercial markets such as Europe, restrict the patentability of methods of treatment of the human body more than United States law does. In addition, many countries, including certain countries in Europe, have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties (for example, the patent owner has failed to “work” the invention in that country, or the third party has patented improvements). In addition, many countries limit the enforceability of patents against government agencies or government contractors. In these countries, the patent owner may have limited remedies, which could materially diminish the value of the patent. Moreover, the legal systems of certain
73
countries, particularly certain developing countries, do not favor the aggressive enforcement of patent and other intellectual property protection, which makes it difficult to stop infringement.
Because patent applications in the United States, Europe and many other jurisdictions are typically not published until 18 months after filing, or in some cases not at all, and because publications of discoveries in scientific literature lag behind actual discoveries, we cannot be certain that we were the first to conceive or reduce to practice the inventions claimed in our issued patents or pending patent applications, or that we were the first to file for protection of the inventions set forth in our patents or pending patent applications. We can give no assurance that all of the potentially relevant art relating to our patents and patent applications has been found; overlooked prior art could be used by a third party to challenge the validity, enforceability and scope of our patents or prevent a patent from issuing from a pending patent application. As a result, we may not be able to obtain or maintain protection for certain inventions. Therefore, the validity, enforceability and scope of our patents in the United States, Europe and in other countries cannot be predicted with certainty and, as a result, any patents that we own or license may not provide sufficient protection against our competitors.
Third parties may challenge any existing patent or future patent we own or license through adversarial proceedings in the issuing offices or in court proceedings, including as a response to any assertion of our patents against them. In any of these proceedings, a court or agency with jurisdiction may find our patents invalid and/or unenforceable, or even if valid and enforceable, insufficient to provide protection against competing products and services sufficient to achieve our business objectives. We may be subject to a third-party pre-issuance submission of prior art to the USPTO, or reexamination by the USPTO if a third party asserts a substantial question of patentability against any claim of a U.S. patent we own or license. The adoption of the Leahy-Smith America Invents Act, or the Leahy-Smith Act, in September 2011 established additional opportunities for third parties to invalidate U.S. patent claims, including inter partes review and post-grant review proceedings. Outside of the United States, patents we own or license may become subject to patent opposition or similar proceedings, which may result in loss of scope of some claims or the entire patent. In addition, such proceedings are very complex and expensive, and may divert our management’s attention from our core business. If any of our patents are challenged, invalidated, circumvented by third parties or otherwise limited or expire prior to the commercialization of our products, and if we do not own or have exclusive rights to other enforceable patents protecting our products or other technologies, competitors and other third parties could market products and use processes that are substantially similar to, or superior to, ours and our business would suffer.
The degree of future protection for our proprietary rights is uncertain because legal means afford only limited protection and may not adequately protect our rights or permit us to gain or keep a competitive advantage. For example:
We are generally also subject to all of the same risks with respect to protection of intellectual property that we license as we are for intellectual property that we own. We currently in-license certain intellectual property from third parties to be able to use such intellectual property in our products and product candidates and to aid in our research activities. In the future, we may in-license intellectual property from additional licensors. We may rely on certain of these licensors to file and prosecute patent applications and maintain, or assist us in the maintenance of, patents and otherwise protect the intellectual property we license from them. We may have limited control over
74
these activities or any other intellectual property that may be related to our in-licensed intellectual property. For example, we cannot be certain that such activities by these licensors have been or will be conducted diligently or in compliance with applicable laws and regulations or will result in valid and enforceable patents and other intellectual property rights. We may have limited control over the manner in which our licensors initiate, or support our efforts to initiate, an infringement proceeding against a third-party infringer of the intellectual property rights, or defend certain of the intellectual property that is licensed to us. If we or our licensors fail to adequately protect this intellectual property, our ability to commercialize products could suffer.
We may become involved in lawsuits to protect or enforce our patents or other intellectual property, which could be expensive, time-consuming and unsuccessful.
Competitors may infringe, misappropriate or otherwise violate our patents, trademarks, copyrights, trade secrets or other intellectual property, or those of our licensors. To counter infringement, misappropriation, unauthorized use or other violations, we may be required to file legal claims, which can be expensive and time consuming and divert the time and attention of our management and scientific personnel. In some cases, it may be difficult or impossible to detect third-party infringement or misappropriation of our intellectual property rights, even in relation to issued patent claims, and proving any such infringement may be even more difficult.
We may not be able to prevent, alone or with our licensees or any future licensors, infringement, misappropriation or other violations of our intellectual property rights, particularly in countries where the laws may not protect those rights as fully as in the United States. Any claims we assert against perceived infringers could provoke these parties to assert counterclaims against us alleging that we infringe their patents. In patent litigation in the United States, defendant counterclaims alleging invalidity or unenforceability are commonplace. The outcome following legal assertions of invalidity and unenforceability is unpredictable. We cannot be certain that there is no invalidating prior art, of which we and the patent examiner were unaware during prosecution. If a third party or a defendant were to prevail on a legal assertion of invalidity or unenforceability, we would lose at least part, and perhaps all, of any future patent protection on our current or future product candidates, including avasopasem and rucosopasem. Such a loss of patent protection could harm our business. In addition, in a patent infringement proceeding, there is a risk that a court will decide that a patent of ours is invalid or unenforceable, in whole or in part, and that we do not have the right to stop the other party from exploiting the claimed subject matter at issue. There is also a risk that, even if the validity of such patents is upheld, the court will construe the patent’s claims narrowly or decide that we do not have the right to stop the other party from exploiting its technology on the grounds that our patents do not cover such technology. An adverse outcome in a litigation or proceeding involving our patents could limit our ability to assert our patents against those parties or other competitors and may curtail or preclude our ability to exclude third parties from making, using, importing and selling similar or competitive products. Any of these occurrences could adversely affect our competitive business position, business prospects and financial condition. Similarly, if we assert trademark infringement claims, a court may determine that the marks we have asserted are invalid or unenforceable, or that the party against whom we have asserted trademark infringement has superior rights to the marks in question. In this case, we could ultimately be forced to cease use of such trademarks.
In any infringement, misappropriation or other intellectual property litigation, any award of monetary damages we receive may not be commercially valuable. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during litigation. Moreover, there can be no assurance that we will have sufficient financial or other resources to file and pursue such infringement claims, which typically last for years before they are concluded. Even if we ultimately prevail in such claims, the monetary cost of such litigation and the diversion of the attention of our management and scientific personnel could outweigh any benefit we receive as a result of the proceedings. We may not be able to detect or prevent misappropriation of our intellectual property rights, particularly in countries where the laws may not protect those rights as fully as in the United States. Our business could be harmed if in litigation the prevailing party does not offer us a license on commercially reasonable terms. Any litigation or other proceedings to enforce our intellectual property rights may fail, and even if successful, may result in substantial costs and distract our management and other employees.
75
Our commercial success depends significantly on our ability to operate without infringing upon the intellectual property rights of third parties.
The biotechnology and pharmaceutical industries are subject to rapid technological change and substantial litigation regarding patent and other intellectual property rights. Our competitors in both the United States and abroad, many of which have substantially greater resources and have made substantial investments in patent portfolios and competing technologies, may have applied for or obtained or may in the future apply for or obtain, patents that will prevent, limit or otherwise interfere with our ability to make, use and sell our product candidates, including avasopasem and rucosopasem, and services. Numerous third-party patents exist in the fields relating to our products and services, and it is difficult for industry participants, including us, to identify all third-party patent rights relevant to our product candidates, including avasopasem and rucosopasem, services and technologies. As the biotechnology and pharmaceutical industries expand and more patents are issued, the risk increases that our product candidates may give rise to claims of infringement of the patent rights of others. Moreover, because some patent applications are maintained as confidential for a certain period of time, we cannot be certain that third parties have not filed patent applications that cover our product candidates, including avasopasem and rucosopasem, services and technologies. Therefore, it is uncertain whether the issuance of any third-party patent would require us to alter our development or commercial strategies for our product candidates, including avasopasem and rucosopasem, or processes, or to obtain licenses or cease certain activities.
Patents could be issued to third parties that we may ultimately be found to infringe. Third parties may have or obtain valid and enforceable patents or proprietary rights that could block us from developing products using our technology. If any third-party patents were held by a court of competent jurisdiction to cover the manufacturing process of our product candidates, constructs or molecules used in or formed during the manufacturing process, or any final product itself, the holders of any such patents may be able to block our ability to commercialize the product candidate unless we obtain a license under the applicable patents, or until such patents expire or they are determined to be held invalid or unenforceable. Our failure to obtain or maintain a license to any technology that we require to develop or commercialize our current and future product candidates, including avasopasem and rucosopasem, may materially harm our business, financial condition and results of operations. Furthermore, we would be exposed to a threat of litigation.
From time to time, we may be party to, or threatened with, litigation or other proceedings with third parties, including non-practicing entities, who allege that our product candidates, including avasopasem and rucosopasem, components of our product candidates, including avasopasem and rucosopasem, services, and/or proprietary technologies infringe, misappropriate or otherwise violate their intellectual property rights. The types of situations in which we may become a party to such litigation or proceedings include:
76
These lawsuits and proceedings, regardless of merit, are time-consuming and expensive to initiate, maintain, defend or settle, and could divert the time and attention of managerial and technical personnel, which could materially adversely affect our business. Any such claim could also force use to do one or more of the following:
Some of our competitors may be able to sustain the costs of complex intellectual property litigation more effectively than we can because they have substantially greater resources. In addition, intellectual property litigation, regardless of its outcome, may cause negative publicity, adversely impact prospective customers, cause product shipment delays, or prohibit us from manufacturing, marketing or otherwise commercializing our products,
77
services and technology. Any uncertainties resulting from the initiation and continuation of any litigation could have a material adverse effect on our ability to raise additional funds or otherwise have a material adverse effect on our business, results of operation, financial condition or cash flows.
In addition, we may indemnify our customers and distributors against claims relating to the infringement of intellectual property rights of third parties related to our product candidates, including avasopasem and rucosopasem. Third parties may assert infringement claims against our customers or distributors. These claims may require us to initiate or defend protracted and costly litigation on behalf of our customers or distributors, regardless of the merits of these claims. If any of these claims succeed, we may be forced to pay damages on behalf of our customers, suppliers or distributors, or may be required to obtain licenses for the product candidates, including avasopasem and rucosopasem, or services they use. If we cannot obtain all necessary licenses on commercially reasonable terms, our customers may be forced to stop using our products or services.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. There could also be public announcements of the results of hearings, motions or other interim proceedings or developments, which could have a material adverse effect on the price of our common stock. If securities analysts or investors perceive these results to be negative, it could have a material adverse effect on the price of our common stock. The occurrence of any of these events may have a material adverse effect on our business, results of operation, financial condition or cash flows.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position may be harmed.
In addition to patent and trademark protection, we also rely on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain our competitive position. Because we expect to rely on third parties to manufacture our product candidates, including avasopasem and rucosopasem, and we expect to continue to collaborate with third parties on the development of our product candidates, including avasopasem and rucosopasem, we must, at times, share trade secrets with them. We seek to protect our trade secrets, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them prior to disclosing our proprietary information, such as our consultants and vendors, or our former or current employees. These agreements typically limit the rights of third parties to use or disclose our confidential information, including our trade secrets. We also enter into confidentiality and invention assignment agreements with our employees and consultants. Despite these efforts, however, any of these parties may breach the agreements and disclose our trade secrets and other unpatented or unregistered proprietary information, and once disclosed, we are likely to lose trade secret protection. Monitoring unauthorized uses and disclosures of our intellectual property is difficult, and we do not know whether the steps we have taken to protect our intellectual property will be effective. In addition, we may not be able to obtain adequate remedies for any such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States are less willing or unwilling to enforce trade secret protection. A competitor’s discovery of our trade secrets would impair our competitive position and have an adverse impact on our business, operating results and financial condition. Additionally, we cannot be certain that competitors will not gain access to our trade secrets and other proprietary confidential information or independently develop substantially equivalent information and techniques.
Changes in patent law could diminish the value of patents in general, thereby impairing our ability to protect our existing and future product candidates, including avasopasem and rucosopasem, and processes.
As is the case with other biotechnology and pharmaceutical companies, our success is heavily dependent on intellectual property, particularly patents. Obtaining and enforcing patents in the biotechnology and pharmaceutical industries involves both technological and legal complexity, and is therefore costly, time consuming, and inherently uncertain. In addition, the United States has recently enacted and is currently implementing wide-ranging patent reform legislation. Recent patent reform legislation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents. On September 16, 2011, the Leahy-Smith Act was signed into law. The Leahy-Smith Act includes a number of significant changes to U.S. patent law. These include provisions that affect the way patent applications are
78
prosecuted, redefine prior art, may affect patent litigation, and switched the United States patent system from a “first-to-invent” system to a “first-to-file” system. Under a “first-to-file” system, assuming the other requirements for patentability are met, the first inventor to file a patent application generally will be entitled to the patent on an invention regardless of whether another inventor had conceived or reduced to practice the invention earlier. The USPTO recently developed new regulations and procedures to govern administration of the Leahy-Smith Act, and many of the substantive changes to patent law associated with the Leahy-Smith Act, in particular, the first-to-file provisions, only became effective on March 16, 2013. Accordingly, it is not clear what, if any, impact the Leahy-Smith Act will have on the operation of our business. The Leahy-Smith Act and its implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business and financial condition.
In addition, patent reform legislation may pass in the future that could lead to additional uncertainties and increased costs surrounding the prosecution, enforcement and defense of our patents and pending patent applications. Recent U.S. Supreme Court rulings have narrowed the scope of patent protection available in certain circumstances and weakened the rights of patent owners in certain situations. Furthermore, the U.S. Supreme Court and the U.S. Court of Appeals for the Federal Circuit have made, and will likely continue to make, changes in how the patent laws of the United States are interpreted. Similarly, foreign courts have made, and will likely continue to make, changes in how the patent laws in their respective jurisdictions are interpreted. We cannot predict future changes in the interpretation of patent laws or changes to patent laws that might be enacted into law by United States and foreign legislative bodies. Those changes may materially affect our patents or patent applications and our ability to obtain additional patent protection in the future.
The United States federal government retains certain rights in inventions produced with its financial assistance under the Patent and Trademark Law Amendments Act, or the Bayh-Dole Act. The federal government retains a “nonexclusive, nontransferable, irrevocable, paid-up license” for its own benefit. The Bayh-Dole Act also provides federal agencies with “march-in rights.” March-in rights allow the government, in specified circumstances, to require the contractor or successors in title to the patent to grant a “nonexclusive, partially exclusive, or exclusive license” to a “responsible applicant or applicants.” If the patent owner refuses to do so, the government may grant the license itself. We partner with a number of universities, including the University of Iowa, Northwestern University, and the University of Texas Southwestern Medical Center, with respect to certain of our research, development and manufacturing. While it is our policy to avoid engaging our university partners in projects in which there is a risk that federal funds may be commingled, we cannot be sure that any co-developed intellectual property will be free from government rights pursuant to the Bayh-Dole Act. If, in the future, we co-own or license in technology which is critical to our business that is developed in whole or in part with federal funds subject to the Bayh-Dole Act, our ability to enforce or otherwise exploit patents covering such technology may be adversely affected.
If we do not obtain patent term extensions in the United States under the Hatch-Waxman Act and in foreign countries under similar legislation with respect to our product candidates, including avasopasem and rucosopasem, thereby potentially extending the term of marketing exclusivity for such product candidates, including avasopasem and rucosopasem, our business may be harmed.
In the United States, a patent that covers an FDA-approved drug or biologic may be eligible for a term extension designed to restore the period of the patent term that is lost during the premarket regulatory review process conducted by the FDA. Depending upon the timing, duration and conditions of FDA marketing approval of our product candidates, including avasopasem and rucosopasem, one or more of our U.S. patents may be eligible for limited patent term extension under the Drug Price Competition and Patent Term Restoration Act of 1984, or the Hatch-Waxman Act, which permits a patent term extension of up to a maximum of five years beyond the normal expiration of the patent if the patent is eligible for such an extension under the Hatch-Waxman Act as compensation for patent term lost during development and the FDA regulatory review process, which is limited to the approved indication (and potentially additional indications approved during the period of extension) covered by the patent. This extension is limited to only one patent that covers the approved product, the approved use of the product, or a method of manufacturing the product. However, the applicable authorities, including the FDA and the USPTO in the United States, and any equivalent regulatory authority in other countries, may not agree with our assessment of whether such extensions are available, and may refuse to grant extensions to our patents, or may grant more limited extensions than we request.
79
We may not receive an extension if we fail to apply within applicable deadlines, fail to apply prior to expiration of relevant patents or otherwise fail to satisfy applicable requirements. Even if we are granted such extension, the duration of such extension may be less than our request and the patent term may still expire before or shortly after we receive FDA marketing approval. If we are unable to extend the expiration date of our existing patents or obtain new patents with longer expiry dates, our competitors may be able to take advantage of our investment in development and clinical trials by referencing our clinical and preclinical data to obtain approval of competing products following our patent expiration and launch their product earlier than might otherwise be the case.
Obtaining and maintaining patent protection depends on compliance with various procedural, document submission, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment, and other similar provisions during the patent application process. In addition, periodic maintenance fees on issued patents often must be paid to the USPTO and foreign patent agencies over the lifetime of the patent. While an unintentional lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If we fail to maintain the patents and patent applications covering our product candidates, including avasopasem and rucosopasem, or procedures, we may not be able to stop a competitor from marketing products that are the same as or similar to our own, which would have a material adverse effect on our business.
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
During trademark registration proceedings, our trademark application(s) may be rejected. Although we are given an opportunity to respond to those rejections, we may be unable to overcome such rejections. In addition, in the USPTO and in comparable agencies in many foreign jurisdictions, third parties can oppose pending trademark applications and seek to cancel registered trademarks. Opposition or cancellation proceedings may be filed against our trademarks, and our trademarks may not survive such proceedings. Moreover, any name we propose to use with our product candidate(s), including avasopasem and rucosopasem, in the United States must be approved by the FDA, regardless of whether we have registered it, or applied to register it, as a trademark. The FDA typically conducts a review of proposed product names, including an evaluation of potential for confusion with other product names. If the FDA objects to any of our proposed proprietary product names, we may be required to expend significant additional resources in an effort to identify a suitable substitute name that would qualify under applicable trademark laws, not infringe the existing rights of third parties and be acceptable to the FDA.
Our registered or unregistered trademarks or trade names may be challenged, infringed, circumvented, declared generic or determined to be infringing on other marks. We may not be able to protect our rights in these trademarks and trade names, which we need in order to build name recognition with potential partners or customers in our markets of interest. In addition, third parties have used trademarks similar and identical to our trademarks in foreign jurisdictions and have filed or may in the future file for registration of such trademarks. If they succeed in registering or developing common law rights in such trademarks, and if we are not successful in challenging such third-party rights, we may not be able to use these trademarks to market our products in those countries. In any case, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be adversely affected.
We may not be able to adequately protect our intellectual property rights throughout the world.
Certain of our key patent families have been filed in the United States, as well as in numerous jurisdictions outside the United States. However, our intellectual property rights in certain jurisdictions outside the United States may be less robust. The laws of some foreign countries do not protect intellectual property rights to
80
the same extent as the laws of the United States. For example, the requirements for patentability may differ in certain countries, particularly developing countries, and we may be unable to obtain issued patents that contain claims that adequately cover or protect our current or future product candidates, including avasopasem and rucosopasem. Many companies have encountered significant problems in protecting and defending intellectual property rights in certain foreign jurisdictions. The legal systems of some countries, particularly developing countries, do not favor the enforcement of patents and other intellectual property protection, especially those relating to life sciences. This could make it difficult for us to stop the infringement of our patents or the misappropriation of our other intellectual property rights. For example, many foreign countries have compulsory licensing laws under which a patent owner must grant licenses to third parties. In addition, many countries limit the enforceability of patents against third parties, including government agencies or government contractors. In these countries, patents may provide limited or no benefit.
Proceedings to enforce our patent rights in foreign jurisdictions, whether or not successful, could result in substantial costs and divert our efforts and attention from other aspects of our business. Furthermore, while we intend to protect our intellectual property rights in our expected significant markets, we cannot ensure that we will be able to initiate or maintain similar efforts in all jurisdictions in which we may wish to market current or future product candidates, including avasopasem and rucosopasem. Consequently, we may not be able to prevent third parties from practicing our technology in all countries outside the United States, or from selling or importing products made using our technology in and into those other jurisdictions where we do not have intellectual property rights. Competitors may use our technologies in jurisdictions where we have not obtained patent protection to develop their own products and may also export infringing products to territories where we have patent protection, but where enforcement is not as strong as that in the United States. These products may compete with our product candidates, including avasopasem and rucosopasem, and our patents or other intellectual property rights may not be effective or sufficient to prevent them from competing. Accordingly, our efforts to protect our intellectual property rights in such countries may be inadequate. In addition, changes in the law and legal decisions by courts in the United States and foreign countries may affect our ability to obtain and enforce adequate intellectual property protection for our technology.
We may not identify relevant third-party patents or may incorrectly interpret the relevance, scope or expiration of a third-party patent which might adversely affect our ability to develop and market our product candidates, including avasopasem and rucosopasem.
We cannot guarantee that any of our or our licensors’ patent searches or analyses, including the identification of relevant patents, the scope of patent claims or the expiration of relevant patents, are complete or thorough, nor can we be certain that we have identified each and every third-party patent and pending application in the United States and abroad that is relevant to or necessary for the commercialization of our product candidates, including avasopasem and rucosopasem, in any jurisdiction. For example, U.S. patent applications filed before November 29, 2000 and certain U.S. patent applications filed after that date that will not be filed outside the United States remain confidential until patents issue. Patent applications in the United States and elsewhere are published approximately 18 months after the earliest filing for which priority is claimed, with such earliest filing date being commonly referred to as the priority date. Therefore, patent applications covering our product candidates, including avasopasem and rucosopasem could have been filed by others without our knowledge. Additionally, pending patent applications that have been published can, subject to certain limitations, be later amended in a manner that could cover our product candidates, including avasopasem and rucosopasem, or the use of our products. The scope of a patent claim is determined by an interpretation of the law, the written disclosure in a patent and the patent’s prosecution history. Our interpretation of the relevance or the scope of a patent or a pending application may be incorrect, which may negatively impact our ability to market our product candidates, including avasopasem and rucosopasem. We may incorrectly determine that our product candidates, including avasopasem and rucosopasem, are not covered by a third-party patent or may incorrectly predict whether a third party’s pending patent application will issue with claims of relevant scope. Our determination of the expiration date of any patent in the United States or abroad that we consider relevant may be incorrect, which may negatively impact our ability to develop and market our product candidates, including avasopasem and rucosopasem, and services. Our failure to identify and correctly interpret relevant patents may negatively impact our ability to develop and market our product candidates, including avasopasem and rucosopasem, and services.
81
If we fail to identify and correctly interpret relevant patents, we may be subject to infringement claims. We cannot guarantee that we will be able to successfully settle or otherwise resolve such infringement claims. If we fail in any such dispute, in addition to being forced to pay damages, we may be temporarily or permanently prohibited from commercializing any of our product candidates, including avasopasem and rucosopasem, that are held to be infringing. We might, if possible, also be forced to redesign products, product candidates, including avasopasem and rucosopasem, or services so that we no longer infringe the third-party intellectual property rights. Any of these events, even if we were ultimately to prevail, could require us to divert substantial financial and management resources that we would otherwise be able to devote to our business.
Patent terms may be inadequate to protect our competitive position on our product candidates, including avasopasem and rucosopasem, for an adequate amount of time.
Patents have a limited lifespan, and the protection patents afford is limited. In the United States, if all maintenance fees are timely paid, the natural expiration of a patent is generally 20 years from its earliest U.S. non-provisional filing date. Even if patents covering our product candidates, including avasopasem and rucosopasem, are obtained, once the patent life has expired for patents covering a product or product candidate, we may be open to competition from competitive products and services. As a result, our patent portfolio may not provide us with sufficient rights to exclude others from commercializing products similar or identical to ours.
Intellectual property rights do not necessarily address all potential threats to our business.
While we seek broad coverage under our existing patent applications, there is always a risk that an alteration to products or processes may provide sufficient basis for a competitor to avoid infringing our patent claims. In addition, patents, if granted, expire and we cannot provide any assurance that any potentially issued patents will adequately protect our product candidates, including avasopasem and rucosopasem. Once granted, patents may remain open to invalidity challenges including opposition, interference, re-examination, post-grant review, inter partes review, nullification or derivation action in court or before patent offices or similar proceedings for a given period after allowance or grant, during which time third parties can raise objections against such grant. In the course of such proceedings, which may continue for a protracted period of time, the patent owner may be compelled to limit the scope of the allowed or granted claims thus attacked or may lose the allowed or granted claims altogether.
In addition, the degree of future protection afforded by our intellectual property rights is uncertain because even granted intellectual property rights have limitations, and may not adequately protect our business, provide a barrier to entry against our competitors or potential competitors or permit us to maintain our competitive advantage. Moreover, if a third party has intellectual property rights that cover the practice of our technology, we may not be able to fully exercise or extract value from our intellectual property rights. The following examples are illustrative:
82
Should any of these events occur, they could have a material adverse effect on our business, financial condition, results of operations and prospects.
We may be subject to claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of their former employers or other third parties.
We do and may employ individuals who were previously employed at universities or other biotechnology or pharmaceutical companies, including our licensors, competitors or potential competitors. Although we try to ensure that our employees, consultants and independent contractors do not use the proprietary information or know-how of others in their work for us, and we are not currently subject to any claims that our employees, consultants or independent contractors have wrongfully used or disclosed confidential information of third parties, we may in the future be subject to such claims.
Litigation may be necessary to defend against these claims. If we fail in defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Such intellectual property rights could be awarded to a third party, and we could be required to obtain a license from such third party to commercialize our technology or product candidates, including avasopasem and rucosopasem. Such a license may not be available on commercially reasonable terms or at all. Even if we are successful in defending against such claims, litigation could result in substantial costs and be a distraction to management and other employees and could result in customers seeking other sources for the technology or in ceasing from doing business with us.
Our intellectual property agreements with third parties may be subject to disagreements over contract interpretation, which could narrow the scope of our rights to the relevant intellectual property or technology.
Certain provisions in our intellectual property agreements may be susceptible to multiple interpretations. The resolution of any contract interpretation disagreement that may arise could affect the scope of our rights to the relevant intellectual property or technology or affect financial or other obligations under the relevant agreement, either of which could have a material adverse effect on our business, financial condition, results of operations and prospects.
In addition, while we typically require our employees, consultants and contractors who may be involved in the conception or development of intellectual property to execute agreements assigning such intellectual property
83
to us, we may be unsuccessful in executing such an agreement with each party who in fact conceives or develops intellectual property that we regard as our own. To the extent that we fail to obtain such assignments, such assignments do not contain a self-executing assignment of intellectual property rights or such assignment agreements are breached, we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property and this may interfere with our ability to capture the commercial value of such intellectual property. If we fail in prosecuting or defending any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel. Such intellectual property rights could be awarded to a third party, and we could be required to obtain a license from such third party to commercialize our technology or products. Such a license may not be available on commercially reasonable terms or at all. Even if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and be a distraction to our management and scientific personnel. Disputes regarding ownership or inventorship of intellectual property can also arise in other contexts, such as collaborations and sponsored research. We may be subject to claims that former collaborators or other third parties have an ownership interest in our patents or other intellectual property. If we are subject to a dispute challenging our rights in or to patents or other intellectual property, such a dispute could be expensive and time-consuming. If we are unsuccessful, we could lose valuable rights in intellectual property that we regard as our own.
We may not be successful in obtaining necessary intellectual property rights to future products through acquisitions and in-licenses.
Although we intend to develop products and technology through our own internal research, we may also seek to acquire or in-license technologies to grow our product offerings and technology portfolio. However, we may be unable to acquire or in-license intellectual property rights relating to, or necessary for, any such products or technology from third parties on commercially reasonable terms or at all. In that event, we may be unable to develop or commercialize such products or technology. We may also be unable to identify products or technology that we believe are an appropriate strategic fit for our Company and protect intellectual property relating to, or necessary for, such products and technology.
The in-licensing and acquisition of third-party intellectual property rights for product candidates, including avasopasem and rucosopasem, is a competitive area, and a number of more established companies are also pursuing strategies to in-license or acquire third-party intellectual property rights for products that we may consider attractive or necessary. These established companies may have a competitive advantage over us due to their size, cash resources and greater clinical development and commercialization capabilities. Furthermore, companies that perceive us to be a competitor may be unwilling to assign or license rights to us. If we are unable to successfully obtain rights to additional technologies or products, our business, financial condition, results of operations and prospects for growth could suffer.
In addition, we expect that competition for the in-licensing or acquisition of third-party intellectual property rights for products and technologies that are attractive to us may increase in the future, which may mean fewer suitable opportunities for us as well as higher acquisition or licensing costs. We may be unable to in-license or acquire the third-party intellectual property rights for products or technology on terms that would allow us to make an appropriate return on our investment.
Other Risks Related to Our Business
The COVID-19 pandemic has adversely impacted and could continue to adversely impact, our business, including our clinical trials, results of operations and financial condition.
The COVID-19 pandemic and government measures taken in response have had a significant impact, both direct and indirect, on businesses and commerce. While we are currently continuing our ongoing clinical trials, the COVID-19 pandemic and related precautions have directly or indirectly impacted the timeline for certain of our clinical trials. In April 2020, we delayed the initiation of the Phase 2a multi-center trial in Europe assessing the safety of avasopasem manganese in patients with HNC undergoing standard-of-care radiotherapy. The first patient was dosed in the trial in June 2020, and target enrollment was decreased to approximately 35 patients due to the delay. This trial was expected to contribute to the safety database for avasopasem in patients with HNC receiving radiotherapy. As a result of the delay in initiating the trial in Europe, the target enrollment for the ROMAN trial was
84
increased to approximately 450 patients in order to ensure we are positioned to maintain the planned size of the safety database in a timely manner. We have since completed the enrollment in the Phase 2a trial in Europe and the ROMAN trial. We are continuing to monitor the impact of the COVID-19 pandemic on our operations and ongoing clinical development activity, generally. As a result of the COVID-19 pandemic, we may experience further disruptions that could severely impact our business, preclinical studies and clinical trials, including:
The extent to which the COVID-19 pandemic may further impact our business, including our preclinical studies and clinical trials, results of operations and financial condition, will depend on future developments which are highly uncertain and cannot be predicted with confidence. Such factors include but are not limited to the duration of the pandemic, travel restrictions, quarantines, business closures or business disruptions, the effectiveness of vaccines and vaccine distribution efforts, the availability and effectiveness of COVID-19 testing, the ultimate impact of COVID-19 on financial markets and the global economy, and the effectiveness of other actions taken in the United States and other countries to contain and treat the disease.
85
Our business operations and current and future relationships with investigators, healthcare professionals, consultants, third-party payors, patient organizations and customers will be subject to applicable healthcare regulatory laws, which could expose us to penalties.
Our business operations and current and future arrangements with investigators, healthcare professionals, consultants, third-party payors, patient organizations and customers, may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations. These laws may constrain the business or financial arrangements and relationships through which we conduct our operations, including how we research, market, sell and distribute our product candidates, if approved. Such laws include:
86
Ensuring that our internal operations and future business arrangements with third parties comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business practices do not comply with current or future statutes, regulations, agency guidance or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of the laws described above or any other governmental laws and regulations that may apply to us, we may be subject to significant penalties, including civil, criminal and administrative penalties, damages, fines, exclusion from government-funded healthcare programs, such as Medicare and Medicaid or similar programs in other countries or jurisdictions, integrity oversight and reporting obligations to resolve allegations of non-compliance, disgorgement, individual imprisonment, contractual damages, reputational harm, diminished profits and the curtailment or restructuring of our operations. If any of the physicians or other providers or entities with whom we expect to do business are found to not be in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs and imprisonment, which could affect our ability to operate our business. Further, defending against any such actions can be costly, time-consuming and may require significant personnel resources. Therefore, even if we are successful in defending against any such actions that may be brought against us, our business may be impaired.
Unfavorable global economic conditions could adversely affect our business, financial condition or results of operations.
Our results of operations could be adversely affected by general conditions in the global economy and in the global financial markets. The global economy, including credit and financial markets, has recently experienced extreme volatility and disruptions, including severely diminished liquidity and credit availability, rising interest and inflation rates, declines in consumer confidence, declines in economic growth, increases in unemployment rates and uncertainty about economic stability. A severe or prolonged economic downturn, such as the global financial crisis, could result in a variety of risks to our business, including, weakened demand for our product candidates and our ability to raise additional capital when needed on acceptable terms, if at all. A weak or declining economy could also strain our suppliers, possibly resulting in supply disruption, or cause our customers to delay making payments for our services. Doing business internationally involves a number of risks, including but not limited to:
87
Any of the foregoing could harm our business and we cannot anticipate all of the ways in which the current economic climate and financial market conditions could adversely impact our business.
Our business and operations may suffer in the event of information technology system failures, cyberattacks or deficiencies in our cybersecurity.
Despite the implementation of security measures, our information technology systems and those of our third-party CMOs, CROs, contractors and consultants are vulnerable to attack, interruption and damage from computer viruses and malware (e.g. ransomware), malicious code, natural disasters, terrorism, war, telecommunication and electrical failures, hacking, cyberattacks, phishing attacks and other social engineering schemes, employee theft or misuse, human error, fraud, denial or degradation of service attacks, sophisticated nation-state and nation-state-supported actors or unauthorized access or use by persons inside our organization, or persons with access to systems inside our organization. Attacks upon information technology systems are increasing in their frequency, levels of persistence, sophistication and intensity, and are being conducted by sophisticated and organized groups and individuals with a wide range of motives and expertise. As a result of the COVID-19 pandemic, we may also face increased cybersecurity risks due to our reliance on internet technology and the number of our employees who are working remotely, which may create additional opportunities for cybercriminals to exploit vulnerabilities. Furthermore, because the techniques used to obtain unauthorized access to, or to sabotage, systems change frequently and often are not recognized until launched against a target, we may be unable to anticipate these techniques or implement adequate preventative measures. We may also experience security breaches that may remain undetected for an extended period. Even if identified, we may be unable to adequately investigate or remediate incidents or breaches due to attackers increasingly using tools and techniques that are designed to circumvent controls, to avoid detection, and to remove or obfuscate forensic evidence.
While we do not believe that we have experienced any significant system failure or accident, from time to time, we have been the target of cybersecurity breach attempts and we expect them to continue as cybersecurity threats have been rapidly evolving in sophistication and becoming more prevalent. We do not believe that these cybersecurity breaches have had a material impact on our operations, but future breaches may have such impact. If such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our programs. For example, the loss of clinical trial data for our product candidates could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach results in a loss of or damage to our data or applications or other data or applications relating to our technology or product candidates, or inappropriate disclosure or theft of confidential or proprietary information, we could incur liabilities and the further development of our product candidates could be delayed. Federal, state and international laws and regulations could expose us to enforcement actions and investigations by regulatory authorities, and potentially result in regulatory penalties, fines and significant legal liability, if our information technology security efforts fail. We maintain cyber liability insurance; however, this insurance may not be sufficient to cover the financial, legal, business or reputational losses that may result from an interruption or breach of our systems.
88
Actual or perceived failures to comply with applicable data protection, privacy and security laws, regulations, standards and other requirements could adversely affect our business, results of operations, and financial condition.
The global data protection landscape is rapidly evolving, and we are or may become subject to numerous state, federal and foreign laws, requirements and regulations governing the collection, use, disclosure, retention, and security of personal data, such as information that we may collect in connection with clinical trials in the U.S. and abroad. Implementation standards and enforcement practices are likely to remain uncertain for the foreseeable future, and we cannot yet determine the impact future laws, regulations, standards, or perception of their requirements may have on our business. This evolution may create uncertainty in our business, affect our ability to operate in certain jurisdictions or to collect, store, transfer use and share personal information, necessitate the acceptance of more onerous obligations in our contracts, result in liability or impose additional costs on us. The cost of compliance with these laws, regulations and standards is high and is likely to increase in the future. Any failure or perceived failure by us to comply with federal, state or foreign laws or regulation, our internal policies and procedures or our contracts governing our processing of personal information could result in negative publicity, government investigations and enforcement actions, claims by third parties and damage to our reputation, any of which could have a material adverse effect on our operations, financial performance and business.
Most healthcare providers, including research institutions from which we obtain patient health information, are subject to privacy and security regulations promulgated under HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act, or collectively, HIPAA. HIPAA imposes, among other things, certain standards relating to the privacy, security, transmission and breach reporting of individually identifiable health information. While we do not believe we are currently acting or regulated as a covered entity or business associate under HIPAA and thus are not directly regulated under HIPAA, any person may be prosecuted under HIPAA’s criminal provisions either directly or under aiding-and-abetting or conspiracy principles. Consequently, depending on the facts and circumstances, we could face substantial criminal penalties if we knowingly receive individually identifiable health information.
Certain states have also adopted comparable privacy and security laws and regulations, which govern the privacy, processing and protection of health-related and other personal information. For example, the CCPA went into effect on January 1, 2020. The CCPA creates individual privacy rights for California consumers and increases the privacy and security obligations of entities handling certain personal information. The CCPA provides for civil penalties for violations, as well as a private right of action for data breaches has increased the likelihood of, and risks associated with data breach litigation. Further, the CPRA generally went into effect on January 1, 2023 and significantly amends the CCPA. The CPRA imposes additional data protection obligations on covered businesses, including additional consumer rights processes, limitations on data uses, new audit requirements for higher risk data, and opt outs for certain uses of sensitive data. It also creates a new California data protection agency authorized to issue substantive regulations and could result in increased privacy and information security enforcement. Additional compliance and business process changes may be required. Similar laws have passed in Virginia, Connecticut, Utah and Colorado and have been proposed in other states and at the federal level, reflecting a trend toward more stringent privacy legislation in the United States. The enactment of such laws could have potentially conflicting requirements that would make compliance challenging. In the event that we are subject to or affected by HIPAA, the CCPA, the CPRA or other domestic privacy and data protection laws, any liability from failure to comply with the requirements of these laws could adversely affect our financial condition.
Our operations abroad, including our clinical trial programs outside the United States may also be subject to increased scrutiny or attention from data protection authorities. Our activities outside the United States impose additional compliance requirements and generate additional risks of enforcement for noncompliance. In Europe, the GDPR went into effect in May 2018 and imposes strict requirements for processing the personal data of individuals within the European Economic Area, or EEA. Companies that must comply with the GDPR face increased compliance obligations and risk, including more robust regulatory enforcement of data protection requirements and potential fines for noncompliance of up to €20 million or 4% of the annual global revenues of the noncompliant company, whichever is greater. In addition to fines, a breach of the GDPR may result in regulatory investigations, reputational damage, orders to cease/ change our data processing activities, enforcement notices, assessment notices (for a compulsory audit) and/ or civil claims (including class actions). Among other requirements, the GDPR regulates transfers of personal data subject to the GDPR to third countries that have not
89
been found to provide adequate protection to such personal data, including the United States; in July 2020, the Court of Justice of the EU, or CJEU, limited how organizations could lawfully transfer personal data from the EU/EEA to the United States by invalidating the Privacy Shield for purposes of international transfers and imposing further restrictions on the use of standard contractual clauses, or SCCs. In March 2022, the US and EU announced a new regulatory regime intended to replace the invalidated regulations; however, this new EU-US Data Privacy Framework has not been implemented beyond an executive order signed by President Biden on October 7, 2022 on Enhancing Safeguards for United States Signals Intelligence Activities. European court and regulatory decisions subsequent to the CJEU decision of July 16, 2020 have taken a restrictive approach to international data transfers. As supervisory authorities issue further guidance on personal data export mechanisms, including circumstances where the SCCs cannot be used, and/or start taking enforcement action, we could suffer additional costs, complaints and/or regulatory investigations or fines, and/or if we are otherwise unable to transfer personal data between and among countries and regions in which we operate, it could affect the manner in which we conduct our business, the geographical location or segregation of our relevant systems and operations, and could adversely affect our financial results.
Further, from January 1, 2021, companies have had to comply with the GDPR and also the United Kingdom GDPR, or UK GDPR, which, together with the amended UK Data Protection Act 2018, retains the GDPR in UK national law. The UK GDPR mirrors the fines under the GDPR, i.e., fines up to the greater of €20 million (£17.5 million) or 4% of global turnover. As we continue to expand into other foreign countries and jurisdictions, we may be subject to additional laws and regulations that may affect how we conduct business.
Although we work to comply with applicable laws, regulations and standards, our contractual obligations and other legal obligations, these requirements are evolving and may be modified, interpreted and applied in an inconsistent manner from one jurisdiction to another, and may conflict with one another or other legal obligations with which we must comply. Any failure or perceived failure by us, our third-party CMOs, CROs, contractors, or consultants to comply with applicable federal, state or local regulatory requirements, we could be subject to a range of regulatory actions that could affect our or our contractors’ ability to develop and commercialize our product candidates and could harm or prevent sales of any affected products that we are able to commercialize, or could substantially increase the costs and expenses of developing, commercializing and marketing our products. Claims that we have violated individuals’ privacy rights or breached our contractual obligations, even if we are not found liable, could be expensive and time-consuming to defend and could result in adverse publicity that could harm our business. Any threatened or actual government enforcement action could also generate adverse publicity and require that we devote substantial resources that could otherwise be used in other aspects of our business. Increasing use of social media could give rise to liability, breaches of data security or reputational damage.
Violations of or liabilities under environmental, health and safety laws and regulations could subject us to fines, penalties or other costs that could have a material adverse effect on the success of our business.
We are subject to numerous environmental, health and safety laws and regulations, including those governing laboratory procedures, the handling, use, storage, treatment and disposal of hazardous materials and wastes and the cleanup of contaminated sites. Our operations involve the use of potentially hazardous and flammable materials, including chemicals and biological materials. Our operations also produce hazardous waste products. We could incur substantial costs as a result of violations of or liabilities under environmental requirements in connection with our operations or property, including fines, penalties and other sanctions, investigation and cleanup costs and third-party claims. Although we generally contract with third parties for the disposal of hazardous materials and wastes from our operations, we cannot eliminate the risk of contamination or injury from these materials. In the event of contamination or injury resulting from our use of hazardous materials, we could be held liable for any resulting damages, and any liability could exceed our resources.
Furthermore, environmental laws and regulations are complex, change frequently and have tended to become more stringent. We cannot predict the impact of changes to applicable laws and regulations and cannot be certain of our future compliance. In addition, we may incur substantial costs in order to comply with current or future environmental, health and safety laws and regulations. These current or future laws and regulations may impair our research, development or production efforts.
90
Although we maintain workers’ compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. We do not maintain insurance for environmental liability or toxic tort claims that may be asserted against us in connection with our storage or disposal of biological, hazardous or radioactive materials.
The increasing focus on environmental sustainability and social initiatives could increase our costs, harm our reputation and adversely impact our financial results.
There has been increasing public focus by investors, environmental activists, the media and governmental and nongovernmental organizations on a variety of environmental, social and other sustainability matters. We may experience pressure to make commitments relating to sustainability matters that affect us, including the design and implementation of specific risk mitigation strategic initiatives relating to sustainability. If we are not effective in addressing environmental, social and other sustainability matters affecting our business, or setting and meeting relevant sustainability goals, our reputation and financial results may suffer. In addition, we may experience increased costs in order to execute upon our sustainability goals and measure achievement of those goals, which could have an adverse impact on our business and financial condition.
In addition, this emphasis on environmental, social and other sustainability matters has resulted and may result in the adoption of new laws and regulations, including new reporting requirements. If we fail to comply with new laws, regulations or reporting requirements, our reputation and business could be adversely impacted.
Insurance policies are expensive and protect us only from some business risks, which leaves us exposed to uninsured liabilities.
Some of the insurance policies we currently maintain include general liability, employment practices liability, property, workers’ compensation, umbrella, and directors’ and officers’ insurance. These policies may not adequately cover all categories of risk that our business may encounter.
Any additional product liability insurance coverage we acquire in the future may not be sufficient to reimburse us for any expenses or losses we may suffer. Moreover, insurance coverage is becoming increasingly expensive and in the future we may not be able to maintain insurance coverage at a reasonable cost or in sufficient amounts to protect us against losses due to liability. If we obtain marketing approval for avasopasem, we intend to acquire insurance coverage to include the sale of commercial products; however, we may be unable to obtain product liability insurance on commercially reasonable terms or in adequate amounts. A successful product liability claim or series of claims brought against us could cause our share price to decline and, if judgments exceed our insurance coverage, could adversely affect our results of operations and business, including preventing or limiting the development and commercialization of any product candidates we develop. We do not carry specific biological or hazardous waste insurance coverage, and our property, casualty and general liability insurance policies specifically exclude coverage for damages and fines arising from biological or hazardous waste exposure or contamination. Accordingly, in the event of contamination or injury, we could be held liable for damages or be penalized with fines in an amount exceeding our resources, and our clinical trials or regulatory approvals could be suspended.
We also expect that operating as a public company will make it more difficult and more expensive for us to obtain director and officer liability insurance, and we may be required to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. As a result, it may be more difficult for us to attract and retain qualified people to serve on our board of directors, our board committees or as executive officers. We do not know, however, if we will be able to maintain existing insurance with adequate levels of coverage. Any significant uninsured liability may require us to pay substantial amounts, which would adversely affect our cash position and results of operations.
91
We and our employees are increasingly utilizing social media tools as a means of communication both internally and externally.
Despite our efforts to monitor evolving social media communication guidelines and comply with applicable rules, there is risk that the use of social media by us or our employees to communicate about our product candidates or business may cause us to be found in violation of applicable requirements. In addition, our employees may knowingly or inadvertently make use of social media in ways that may not comply with our social media policy or other legal or contractual requirements, which may give rise to liability, lead to the loss of trade secrets or other intellectual property or result in public exposure of personal information of our employees, clinical trial patients, customers and others. Furthermore, negative posts or comments about us or our product candidates in social media could seriously damage our reputation, brand image and goodwill. Any of these events could have a material adverse effect on our business, prospects, operating results and financial condition and could adversely affect the price of our common stock.
Our employees and independent contractors, including consultants, vendors, and any third parties we may engage in connection with development and commercialization may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could harm our business.
Misconduct by our employees and independent contractors, including consultants, vendors, and any third parties we may engage in connection with development and commercialization, could include intentional, reckless or negligent conduct or unauthorized activities that violate: (i) the laws and regulations of the FDA and other comparable regulatory authorities, including those laws that require the reporting of true, complete and accurate information to such authorities; (ii) manufacturing standards; (iii) data privacy, security, fraud and abuse and other healthcare laws and regulations; or (iv) laws that require the reporting of true, complete and accurate financial information and data. Specifically, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, misconduct, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Activities subject to these laws could also involve the improper use or misrepresentation of information obtained in the course of clinical trials, creation of fraudulent data in preclinical studies or clinical trials or illegal misappropriation of drug product, which could result in regulatory sanctions and cause serious harm to our reputation. It is not always possible to identify and deter misconduct by employees and other third parties, and the precautions we take to detect and prevent this activity may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to comply with such laws or regulations. Additionally, we are subject to the risk that a person or government could allege such fraud or other misconduct, even if none occurred. If any such actions are instituted against us, and we are not successful in defending ourselves or asserting our rights, those actions could have a significant impact on our business and results of operations, including the imposition of significant civil, criminal and administrative penalties, damages, monetary fines, disgorgements, possible exclusion from participation in Medicare, Medicaid, other U.S. federal healthcare programs or healthcare programs in other jurisdictions, integrity oversight and reporting obligations to resolve allegations of non-compliance, individual imprisonment, other sanctions, contractual damages, reputational harm, diminished profits and future earnings, and curtailment of our operations.
We or the third parties upon whom we depend may be adversely affected by natural disasters and our business continuity and disaster recovery plans may not adequately protect us from a serious disaster.
Natural disasters could severely disrupt our operations and have a material adverse effect on our business, results of operations, financial condition and prospects. If a natural disaster, power outage, public health emergency, such as the novel coronavirus, or other event occurred that prevented us from using all or a significant portion of our headquarters, that damaged critical infrastructure, such as the manufacturing facilities on which we rely, or that otherwise disrupted operations, it may be difficult or, in certain cases, impossible for us to continue our business for a substantial period of time. The disaster recovery and business continuity plans we have in place may prove inadequate in the event of a serious disaster or similar event. We may incur substantial expenses as a result of the limited nature of our disaster recovery and business continuity plans, which could have a material adverse effect on our business.
92
Our ability to use our net operating losses to offset future taxable income may be subject to certain limitations.
In general, under Section 382 of the Code, a corporation that undergoes an “ownership change,” generally defined as a greater than 50% change by value in its equity ownership over a three-year period, is subject to limitations on its ability to utilize its pre change net operating losses, or NOLs, to offset future taxable income. Our existing NOLs may be subject to limitations arising from ownership changes that we might have undergone in the past. Future changes in our stock ownership, some of which might be beyond our control, could result in an ownership change under Section 382 of the Code, further limiting our ability to utilize a material portion of the NOLs even if we attain profitability.
We are a multinational company that faces complex taxation regimes in various jurisdictions. Audits, investigations, and tax proceedings could have a material adverse effect on our business, results of operations, and financial condition.
We are subject to income and non-income taxes in multiple jurisdictions. Income tax accounting often involves complex issues, and judgment is required in determining our worldwide provision for income taxes and other tax liabilities. In particular, the jurisdictions in which we operate have detailed transfer pricing rules, which require that all transactions with non-resident related parties be priced using arm’s length pricing principles within the meaning of such rules. We could be subject to tax audits involving transfer pricing issues. We believe that our tax positions are reasonable and our tax reserves are adequate to cover any potential liability. However, tax authorities in certain jurisdictions may disagree with our position, including the propriety of our related party arm’s length transfer pricing policies and the tax treatment of corresponding expenses and income. If any of these tax authorities were successful in challenging our positions, we may be liable for additional income tax and penalties and interest related thereto in excess of any reserves established therefor, which may have a significant impact on our results and operations and future cash flow.
Risks Related to Our Common Stock
Our common stock may be delisted from The Nasdaq Global Market if we cannot maintain compliance with Nasdaq’s continued listing requirements, which could harm our business, the trading price of our common stock, our ability to raise additional capital and the liquidity of the market for our common stock.
Our common stock is currently listed on The Nasdaq Global Market. To maintain the listing of our common stock on The Nasdaq Global Market, we are required to meet certain listing requirements, including related to the price of our common stock. On June 8, 2022, we received written notice, or the Initial Notice, from The Nasdaq Stock Market LLC, or Nasdaq, indicating that we were no longer in compliance with the minimum Market Value of Listed Securities, or MVLS, of $50,000,000 required for continued listing on The Nasdaq Global Market, as set forth in Nasdaq Listing Rule 5450(b)(2)(A), or the MVLS Requirement. The Initial Notice had no immediate effect on the listing of our common stock, which continued to trade on The Nasdaq Global Market under the symbol “GRTX.”
In accordance with Nasdaq Listing Rule 5810(c)(3)(C), we had a period of 180 calendar days, or until December 5, 2022, or the Compliance Date, to regain compliance with the MVLS Requirement. To regain compliance, our MVLS had to close at $50,000,000 or more for a minimum of 10 consecutive business days prior to the Compliance Date. On December 6, 2022, we received a letter from Nasdaq indicating that we did not regain compliance with the MVLS Requirement by the Compliance Date and that, unless we request a hearing before a Nasdaq hearings panel, or Panel, to appeal Nasdaq’s delisting determination by December 13, 2022, trading of our common stock would be suspended on December 15, 2022, and our common stock would be delisted from The Nasdaq Global Market. On December 13, 2022, we requested a hearing before a Panel. On January 24, 2023, prior to the scheduled hearing date, we received a letter from Nasdaq notifying us that we had regained compliance with the MVLS Requirement, as our MVLS had closed at over $50,000,000 for 10 consecutive business days, and that the hearing had been cancelled.
There can be no assurance that we will be able to maintain compliance with Nasdaq Listing Rule 5450(b)(2)(A) or any other listing requirements, or satisfy the requirements necessary to transfer the listing of our common stock to The Nasdaq Capital Market. Delisting from the Nasdaq Global Market or any Nasdaq market could make trading our common stock more difficult for investors, potentially leading to declines in our share price
93
and liquidity. In addition, without a Nasdaq market listing, stockholders may have a difficult time getting a quote for the sale or purchase of our common stock, the sale or purchase of our common stock would likely be made more difficult and the trading volume and liquidity of our common stock could decline. Delisting from Nasdaq could also result in negative publicity and could also make it more difficult for us to raise additional capital. The absence of such a listing may adversely affect the acceptance of our common stock as currency or the value accorded by other parties. If our common stock is delisted by Nasdaq, our common stock may be eligible to trade on an over-the-counter quotation system, such as the OTCQB market, where an investor may find it more difficult to sell our common stock or obtain accurate quotations as to the market value of our common stock. We cannot assure you that our common stock, if delisted from Nasdaq, will be listed on another national securities exchange or quoted on an over-the counter quotation system.
Our directors, officers and principal stockholders own a significant percentage of our stock and, if they choose to act together, are able to exercise influence over matters submitted to stockholders for approval.
Our officers, directors and principal stockholders each holding more than 5% of our common stock, collectively, control approximately 38% of our outstanding common stock as of December 31, 2022. Accordingly, these stockholders, if they act together, will be able to exert a significant degree of influence over our management and affairs of our company and most matters requiring stockholder approval, including the election of directors and approval of significant corporate transactions. The interests of these stockholders may not be the same as or may even conflict with your interests. For example, these stockholders could attempt to delay or prevent a change in control of us, even if such change in control would benefit our other stockholders, which could deprive our stockholders of an opportunity to receive a premium for their common stock as part of a sale of us or our assets, and might affect the prevailing market price of our common stock due to investors’ perceptions that conflicts of interest may exist or arise. As a result, this concentration of ownership may not be in the best interests of our other stockholders.
We are an “emerging growth company,” and the reduced disclosure requirements applicable to emerging growth companies may make our common stock less attractive to investors.
We are an “emerging growth company,” as defined in the JOBS Act. We will remain an emerging growth company until the earlier of (a) the last day of the fiscal year in which we have total annual gross revenues of $1.235 billion or more, (b) the last day of the fiscal year following the fifth anniversary of the date of the completion of our initial public offering, or IPO (December 31, 2024), (c) the date on which we have issued more than $1 billion in nonconvertible debt during the previous three years, or (d) the date on which we are deemed to be a large accelerated filer under the rules of the SEC, which means the market value of our common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter. For so long as we remain an emerging growth company, we are permitted and intend to rely on exemptions from certain disclosure requirements that are applicable to other public companies that are not emerging growth companies. These exemptions include:
94
We may choose to take advantage of some, but not all, of the available exemptions. In particular, we have provided only two years of audited financial statements and have not included all of the executive compensation information that would be required if we were not an emerging growth company. We cannot predict whether investors will find our common stock less attractive if we rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our shares price may be more volatile.
We are a “smaller reporting company” and the reduced disclosure requirements applicable to smaller reporting companies may make our common stock less attractive to investors.
We are considered a “smaller reporting company.” We are therefore entitled to rely on certain reduced disclosure requirements, such as an exemption from providing selected financial data and executive compensation information. These exemptions and reduced disclosures in our SEC filings due to our status as a smaller reporting company may make it harder for investors to analyze our results of operations and financial prospects. We cannot predict if investors will find our common stock less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock prices may be more volatile.
We have incurred and expect to continue to incur increased costs as a result of operating as a public company, and our management will be required to devote substantial time to new compliance initiatives.
As a public company, we have incurred, and particularly after we are no longer an “emerging growth company,” expect to continue to incur significant legal, accounting and other expenses that we did not incur as a private company. In addition, the Sarbanes-Oxley Act of 2002 and rules subsequently implemented by the SEC and Nasdaq have imposed various requirements on public companies, including establishment and maintenance of effective disclosure and financial controls and corporate governance practices. Our management and other personnel need to devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations have increased our legal and financial compliance costs and have made some activities more time-consuming and costly. For example, we expect that these rules and regulations may make it more difficult and more expensive for us to obtain director and officer liability insurance.
Pursuant to Section 404, we are required to furnish a report by our management on our internal control over financial reporting. However, while we remain an emerging growth company, we will not be required to include an attestation report on internal control over financial reporting issued by our independent registered public accounting firm. To achieve compliance with Section 404 within the prescribed period, we have engaged in a process to document and evaluate our internal control over financial reporting, which has been both costly and challenging. In this regard, we will need to continue to dedicate internal resources, potentially engage outside consultants and adopt a detailed work plan to assess and document the adequacy of internal control over financial reporting, continue steps to improve control processes as appropriate, validate through testing that controls are functioning as documented and implement a continuous reporting and improvement process for internal control over financial reporting. Despite our efforts, there is a risk that neither we nor our independent registered public accounting firm, as applicable, will be able to conclude within the prescribed timeframe that our internal control over financial reporting is effective as required by Section 404. If we identify one or more material weaknesses, it could cause us to need to restate our previously issued financial statements and could result in an adverse reaction in the financial markets due to a loss of confidence in the reliability of our financial statements.
Provisions in our amended and restated certificate of incorporation and amended and restated bylaws and under Delaware law could make an acquisition of our company, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our amended and restated certificate of incorporation and our amended and restated bylaws may discourage, delay or prevent a merger, acquisition or other change in control of our company that stockholders may consider favorable, including transactions in which you might otherwise receive a premium for your shares. These provisions could also limit the price that investors might be willing to pay in the future for shares of our common stock, thereby depressing the market price of our common stock. In addition, because our board of directors is responsible for appointing the members of our management team, these provisions may frustrate or
95
prevent any attempts by our stockholders to replace or remove our current management by making it more difficult for stockholders to replace members of our board of directors. Among other things, these provisions include those establishing:
Moreover, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the General Corporation Law of the State of Delaware, which prohibits a person who owns in excess of 15% of our outstanding voting stock from merging or combining with us for a period of three years after the date of the transaction in which the person acquired in excess of 15% of our outstanding voting stock, unless the merger or combination is approved in a prescribed manner.
Our amended and restated certificate of incorporation provides that the Court of Chancery of the State of Delaware will be the exclusive forum for substantially all disputes between us and our stockholders, which could limit our stockholders’ ability to obtain a favorable judicial forum for disputes with us or our directors, officers or employees.
Our amended and restated certificate of incorporation provides that, unless we consent in writing to the selection of an alternative forum to the fullest extent permitted by law, the Court of Chancery of the State of Delaware will be the sole and exclusive forum for (1) any derivative action or proceeding brought on our behalf, (2) any action asserting a claim for breach of a fiduciary duty owed by any of our directors, officers, other employees or our stockholders to us or our stockholders, (3) any action asserting a claim arising pursuant to any provision of the General Corporation Law of the State of Delaware, our amended and restated certificate of incorporation or our amended and restated bylaws, or (4) any action asserting a claim governed by the internal affairs doctrine. Under our
96
amended and restated certificate of incorporation, this exclusive forum provision will not apply to claims which are vested in the exclusive jurisdiction of a court or forum other than the Court of Chancery of the State of Delaware, or for which the Court of Chancery of the State of Delaware does not have subject matter jurisdiction. For instance, the provision would not apply to actions arising under federal securities laws, including suits brought to enforce any liability or duty created by the Securities Act, the Exchange Act, or the rules and regulations thereunder. In addition, our bylaws provide that the federal district courts of the United States are the exclusive forum for any complaint raising a cause of action arising under the Securities Act. Any person or entity purchasing or otherwise acquiring any interest in shares of our capital stock shall be deemed to have notice of and to have consented to the provisions of our restated certificate of incorporation and bylaws described above.
These exclusive forum provisions may have the effect of discouraging lawsuits against us and our directors, officers and other employees. The enforceability of similar choice of forum provisions in other companies’ certificates of incorporation has been challenged in legal proceedings, and it is possible that, in connection with any applicable action brought against us, a court could find the choice of forum provisions contained in our amended and restated certificate of incorporation or bylaws to be inapplicable or unenforceable in such action. If a court were to find the choice of forum provisions contained in our amended and restated certificate of incorporation or bylaws to be inapplicable or unenforceable in an action, we may incur additional costs associated with resolving such action in other jurisdictions, which could adversely affect our business, financial condition or results of operations.
Because we do not anticipate paying any cash dividends on our capital stock in the foreseeable future, capital appreciation, if any, will be your sole source of gain.
We have never declared or paid cash dividends on our capital stock. We currently intend to retain all of our future earnings, if any, to finance the growth and development of our business. Additionally, the proposal to pay future dividends to stockholders will effectively be at the sole discretion of our board of directors after taking into account various factors our board of directors deems relevant, including our business prospects, capital requirements, financial performance and new product development. As a result, capital appreciation, if any, of our common stock will be your sole source of gain for the foreseeable future.
General Risk Factors
We may acquire businesses, or products or product candidates, or form strategic alliances, in the future, and we may not realize the benefits of such acquisitions.
We have acquired and in-licensed, and may acquire or in-license, additional businesses or products, from other companies or create joint ventures with third parties that we believe will complement or augment our existing business. If we acquire businesses with promising markets or technologies, we may not be able to realize the benefit of acquiring such businesses if we are unable to successfully integrate them with our existing operations and company culture. We may encounter numerous difficulties in developing, manufacturing and marketing any new products resulting from a strategic alliance or acquisition that delay or prevent us from realizing their expected benefits or enhancing our business. We cannot assure you that, following any such acquisition or license, we will achieve the expected synergies to justify the transaction.
New tax legislation may impact our results of operations and financial condition.
On December 22, 2017, the United States enacted the Tax Cuts and Jobs Act (the “Tax Act”), which significantly reformed the U.S. Internal Revenue Code of 1986, as amended, or the Code. Among a number of significant changes to the current U.S. federal income tax rules, the Tax Act reduced the marginal U.S. corporate income tax rate from 35% to 21%, limited the deduction for net interest expense, shifted the United States toward a more territorial tax system, and imposed new taxes to combat erosion of the U.S. federal income tax base. The financial statements contained herein reflect the effects of the Tax Act based on current guidance. However, there remain uncertainties and ambiguities in the application of certain provisions of the Tax Act, and, as a result, we made certain judgments and assumptions in the interpretation thereof. More recently, on August 16, 2022, the United States enacted the Inflation Reduction Act introducing, among other changes, a 15% corporate minimum tax on certain United States corporations and a 1% excise tax on certain stock redemptions by United States corporations. As we further analyze the impact of the Tax Act, the Inflation Reduction Act and any new tax
97
legislation and collect relevant information to complete our computations of the related accounting impact, we may make adjustments to the provisional amounts that could materially affect our results of operations and financial condition.
An active trading market for our common stock may not be sustained.
An active public trading market for our common stock may not be sustained. The lack of an active market may impair your ability to sell your shares at the time you wish to sell them or at a price that you consider reasonable. The lack of an active market may also reduce the fair value of your shares. An inactive market may also impair our ability to raise capital to continue to fund operations by selling shares and may impair our ability to acquire other companies or technologies by using our shares as consideration.
The price of our common stock is likely to be volatile and fluctuate substantially, which could result in substantial losses for purchasers of our common stock.
Our share price is likely to be volatile. The stock market in general and the market for biopharmaceutical companies in particular have experienced extreme volatility that has often been unrelated to the operating performance of particular companies. As a result of this volatility, you may not be able to sell your common stock at a price that you consider reasonable. The market price for our common stock may be influenced by many factors, including:
98
If securities or industry analysts do not publish research or reports, or publish unfavorable research or reports, about us, our business or our market, our shares price and trading volume could decline.
The trading market for our common stock will be influenced by the research and reports that equity research analysts publish about us and our business. We do not have any control over the analysts or the content and opinions included in their reports. The price of our shares could decline if one or more equity research analysts downgrades our shares or issues other unfavorable commentary or research. If one or more equity research analysts ceases coverage of our company or fails to publish reports on us regularly, demand for our common stock could decrease, which in turn could cause the price of our common stock or its trading volume to decline.
Future sales and issuances of our common stock or rights to purchase common stock, including pursuant to our equity incentive plans, could result in dilution of the percentage ownership of our stockholders and could cause our common stock price to fall.
We will need additional capital in the future to continue our planned operations. To the extent we raise additional capital by issuing additional common stock or other equity securities, our stockholders may experience substantial dilution. We may sell common stock, convertible securities or other equity securities in one or more transactions at prices and in a manner we determine from time to time. If we sell common stock, convertible securities or other equity securities in more than one transaction, investors may be materially diluted by subsequent sales. These sales may also result in material dilution to our existing stockholders, and new investors could gain rights superior to our existing stockholders.
Item 1B. Unresolved Staff Comments.
None.
Item 2. Properties.
Our principal office was located at 2 W. Liberty Blvd, Suite 100, Malvern, Pennsylvania 19355, where we leased approximately 12,200 square feet of office space under a lease that terminated on February 28, 2023. In October 2022, we entered into a new lease agreement for approximately 6,900 square feet of office space at 45 Liberty Blvd, Suite 230, Malvern, Pennsylvania 19355, which will serve as our new principal office location. The lease commencement date is expected to occur in May 2023 and will terminate 7.4 years after the lease commencement date. We may add new facilities or space as we add employees, and we believe that suitable additional or substitute space will be available as needed to accommodate any such expansion of our operations.
Item 3. Legal Proceedings.
We are not subject to any material legal proceedings.
Item 4. Mine Safety Disclosures.
Not applicable.
99
INFORMATION ABOUT OUR EXECUTIVE OFFICERS AND DIRECTORS
The following table sets forth information regarding our executive officers and directors as of the date of this Annual Report on Form 10-K.
Name |
Age |
Position |
Executive Officers |
|
|
J. Mel Sorensen, M.D. |
66 |
President, Chief Executive Officer and Director |
Christopher Degnan |
43 |
Chief Financial Officer |
Mark Bachleda |
48 |
Chief Commercial Officer |
Robert A. Beardsley, Ph.D. |
62 |
Chief Operating Officer |
Eugene Kennedy, M.D., FACS |
54 |
Chief Medical Officer |
Jennifer Evans Stacey |
58 |
Chief Legal & Compliance Officer and Secretary |
Non-Employee Directors |
|
|
Michael Powell, Ph.D.(3) |
68 |
Chairman of the Board |
Lawrence Alleva(1)(2) |
73 |
Director |
Emmett Cunningham, M.D., Ph.D., MPH |
62 |
Director |
Kevin Lokay(1)(2)(3) |
66 |
Director |
Linda West (1)(2)(3) |
63 |
Director |
(1) Member of the Audit Committee.
(2) Member of the Compensation Committee.
(3) Member of the Nominating and Corporate Governance Committee.
Executive Officers
J. Mel Sorensen, M.D. has served as Director, Chief Executive Officer and President of Galera since 2012. Dr. Sorensen serves on the boards of directors of several private companies including Esanik Therapeutics, Medsyn Biopharma and PlanetVerify Ltd. He is an advisor to the Biomarkers Consortium of the National Institutes of Health and to the Irish Cancer Society. Dr. Sorensen holds an M.B., B.Ch. and B.A.O. from University College, Dublin. Dr. Sorensen’s postgraduate education and work has been in the United States, including an internal medicine residency in St. Louis and medical oncology fellowship at the Mayo Clinic, seven years at the National Cancer Institute as Senior Investigator in the Cancer Therapy Evaluation Program and four years each with Bayer and GlaxoSmithKline. Dr. Sorensen served as Director, Chief Executive Officer and President of Ascenta Therapeutics from 2004 until he joined Galera. We believe Dr. Sorensen’s experience in the industry, his role as our Chief Executive Officer and President and his knowledge of the Company enable him to make valuable contributions to our board of directors.
Christopher Degnan has served as our Chief Financial Officer since October 2019 and served as our Secretary from October 2019 to October 2021. Mr. Degnan was most recently the Chief Financial Officer at Verrica Pharmaceuticals Inc., a public, late-stage biotechnology company focused on medical dermatology, from March 2018 to October 2019. Prior to Verrica, Mr. Degnan held roles of increasing responsibility at Endo International plc, a generics and specialty branded pharmaceutical company, beginning in November 2014, where he most recently served as the Vice President of Finance, Corporate FP&A and International Pharmaceuticals Segment Chief Financial Officer from December 2016 to March 2018. Prior to that, he served as the Vice President of Finance, Chief Financial Officer for Endo’s U.S. Branded Pharmaceuticals segment from March 2016 to December 2016, and as the Senior Finance Director, U.S. Branded Pharmaceuticals from November 2014 to March 2016. Prior to joining Endo, Mr. Degnan held roles of increasing responsibility at AstraZeneca plc, a global biopharmaceutical company, beginning in 2004, most recently as Senior Finance Director, U.S. Commercial Finance from July 2013 to November 2014. He is a Certified Public Accountant in the State of Pennsylvania (voluntary inactive status). Mr. Degnan holds a B.B.A. degree in Accountancy from the University of Notre Dame.
Mark J. Bachleda, Pharm.D., MBA has served as our Chief Commercial Officer since October 2021. Prior to Galera, Dr. Bachleda served as Vice President & US Business Unit Head of the CAR T Cell Therapy Franchise at Bristol Myers Squibb (BMS) from November 2019 to September 2021, launching Breyanzi® (liso-cel), a CD19 CAR T in large B cell lymphoma, and Abecma® (ide-cel), the first BCMA CAR T in relapsed/refractory
100
multiple myeloma. He held the same role at Celgene Corporation from August 2018 to November 2019 before its acquisition by BMS for $74 billion in 2019. He served as Vice President, Sales & Account Management at Juno Therapeutics from November 2017 to August 2018 before its $9 billion sale to Celgene in 2018. Previously, Dr. Bachleda worked at Amgen for 15 years in commercial operations roles of increasing responsibility up to Country President & General Manager of Amgen Czech Republic where he led an enterprise of 11 commercialized therapies including launches of Kyprolis®, Blincyto®, and Repatha®. Dr. Bachleda is a registered pharmacist and received his Doctor of Pharmacy degree (Pharm.D.) from the University of Illinois at Chicago. He completed a post-doctoral fellowship in Health Policy & Economics at Thomas Jefferson University and holds Master of Business Administration (MBA) degrees from both Columbia University and University of California at Berkeley.
Robert A. Beardsley, Ph.D., a co-founder of the Company, has served as our Chief Operating Officer since 2015, and previously served as our Executive Chair from 2012 to 2017. Prior to this, Dr. Beardsley was the Chief Executive Officer at Galera Therapeutics, LLC from 2010 to 2012, at Metabolic Solutions Development Corporation from 2009 until 2010, and at Kereos from 2003 until 2009, and the acting Chief Executive Officer at Metaphore Pharmaceuticals, Inc. in 2002. He has also served in various management roles at Confluence Life Sciences, bioStrategies Group, Vector Securities International, Enzyme Organics and Mobil Oil. Dr. Beardsley serves on the board of Euclises, a private company, and has served on a number of boards of directors of public and private companies including Epigenetx, KemPharm, Kereos, CollaGenex Pharmaceuticals, Bioseek, and Metaphore Pharmaceuticals. Dr. Beardsley received a B.S. in Chemical Engineering, a Ph.D. in Biochemical Engineering from the University of Iowa and an M.B.A. in Finance from the University of Chicago.
Eugene Kennedy , M.D., FACS has served as our Chief Medical Officer since September 2022. Dr. Kennedy is a renowned Johns Hopkins-trained surgical oncologist and former Chief of Pancreatic and Hepatobiliary Surgery at Thomas Jefferson University with over 15 years’ experience in clinical development and biopharma leadership. Before joining Galera, Dr. Kennedy served as Chief Medical Officer at Innovative Cellular Therapeutics from March 2021 to August 2021, where he was an integral part of developing the Phase 1 clinical protocol and filing a first-in-human IND for a solid tumor CAR T-cell therapy. From March 2020 until March 2021, he served as Chief Medical Officer at Lumos Pharma, where he was responsible for clinical development strategy and execution, as well as investor and patient outreach. Prior to joining Lumos, Dr. Kennedy served as Chief Medical Officer at NewLink Genetics from November 2017 to March 2020, where he oversaw clinical trials across multiple product candidates and indications, having held positions of increasing responsibility since 2013. From 2006 until joining NewLink Genetics, Dr. Kennedy served as Associate Professor of Surgery at Thomas Jefferson University and held leadership positions as Chief of the Section of Pancreatic and Hepatobiliary Surgery and Co-Director of the Jefferson Pancreas, Biliary, and Related Cancers Center. Prior to joining Jefferson, Dr. Kennedy practiced, taught, and held leadership roles at the Johns Hopkins Hospital and Louisiana State University. He has authored over 50 peer-reviewed publications. Dr. Kennedy received a bachelor’s degree from the University of Virginia and an M.D. from the Medical College of Virginia.
Jennifer Evans Stacey, Esq. has served as our Chief Legal & Compliance Officer and Secretary since October 2021. Prior to Galera, Ms. Stacey served as Vice President, General Counsel, Secretary and Government Relations at The Wistar Institute, an international biomedical research institute focused on cancer, vaccines and infectious disease from April 2016 to October 2021. Previously, she served as Senior Vice President, General Counsel, Human Resources and Secretary at Antares Pharma, Inc. from May 2014 to July 2015. Prior to that, Ms. Stacey served as Executive Vice President, General Counsel, Human Resources, and Secretary at Auxilium Pharmaceuticals, Inc., and as Senior Vice President, Corporate Communications, General Counsel and Secretary at Aventis Behring, LLC (now CSL Behring). She began her career in life sciences at Rhône-Poulenc Rorer (now Sanofi) including two years in their Paris office. Ms. Stacey currently serves on the board of directors of Context Therapeutics, Inc. Ms. Stacey graduated with an A.B. from Princeton University and earned her J.D. from the University of Pennsylvania Law School.
Non-Employee Directors
Michael Powell, Ph.D. has served as a member of our board of directors since November 2016 and as its Chair since July 2017, and also serves as Chair of our Nominating and Corporate Governance Committee. In August 2021, Dr. Powell joined Omega Funds as an Executive Partner. Previously, he was a General Partner at
101
Sofinnova Investments, a biopharmaceutical investment firm, from 1997 until June 2021. Dr. Powell was Group Leader of Drug Delivery at Genentech from 1990 until 1997, and Director of Product Development at Cytel from 1987 until 1990. Dr. Powell currently serves on the board of directors of Aerium Therapeutics, a private company. He served on the Washington University board of trustees in St. Louis and was an Adjunct Professor of Pharmaceutical Chemistry at the University of Kansas. Dr. Powell holds a Ph.D. in Physical Chemistry from the University of Toronto and he completed post-doctoral studies in Bioorganic Chemistry at the University of California as a National Science and Engineering Research Council Scholar. We believe Dr. Powell is qualified to serve on our board of directors due to his extensive experience in investing in pharmaceutical companies.
Lawrence Alleva has served as a member of our board of directors since June 2019 and also serves as Chair of our Audit Committee. He is a former partner with PricewaterhouseCoopers LLP (PwC), where he worked for 39 years from 1971 until his retirement in June 2010, including 28 years’ service as a partner. Mr. Alleva worked with numerous pharmaceutical and biotechnology companies as clients and, additionally, served PwC in a variety of office, regional and national practice leadership roles, most recently as the U.S. Ethics and Compliance Leader for the firm’s Assurance Practice from 2006 until 2010. Mr. Alleva currently serves on the boards of directors of Bright Horizons Family Solutions, Inc., Mersana Therapeutics, Inc. and Adaptimmune Therapeutics PLC and chairs the audit committee for those companies. He previously served on the boards of directors and as chair of the audit committees of TESARO, Inc. from March 2012 to the time of its sale to GSK in January 2019, Mirna Therapeutics, Inc. from June 2015 until its merger with another company in September 2017 and of GlobalLogic, Inc. from June 2011 through the sale of the company in June 2014. Mr. Alleva is a Certified Public Accountant (inactive). He received a B.S. degree in Accounting from Ithaca College and attended Columbia University’s Executive M.B.A. non-degree program. We believe Mr. Alleva is qualified to serve on our board of directors due to his finance background and industry experience, including his service on the boards of directors of other public biotechnology companies.
Emmett Cunningham, M.D., Ph.D. has served as a member of our board of directors since September 2018. Dr. Cunningham is an Operating Partner at Blackstone Life Sciences, a life sciences investment firm, having joined Blackstone as part of its acquisition of Clarus in December 2018. He joined Clarus in 2006 as a Principal. From February 2004 to December 2005, he was Senior Vice President, Medical Strategy at Eyetech Pharmaceuticals, Inc., a pharmaceutical company. From April 2002 to February 2004, Dr. Cunningham was Vice President of Clinical Research Development and Licensing. Dr. Cunningham is also Adjunct Clinical Professor of Ophthalmology at Stanford University School of Medicine and the co-founder and Chair of the Ophthalmology Innovation Summit. Dr. Cunningham currently serves on the boards of directors of SFJ Pharmaceuticals Group and Silktech Biopharmaceuticals, both private companies. He previously served on the boards of directors of Annexon Biosciences from December 2014 until February 2021, Graybug Vision from May 2016 to September 2020, and Lumos Pharma from January 2019 until February 2021, and as the Chair of the board of directors of Restoration Robotics from October 2017 to June 2018. Dr. Cunningham received a B.S. from Drexel University, a B.A., M.D. and M.P.H. from Johns Hopkins University and a Ph.D. in neuroscience from the University of California at San Diego. We believe Dr. Cunningham is qualified to serve on our board of directors due to his experience in research and investing in medical companies.
Kevin Lokay has served as a member of our board of directors since March 2019. Mr. Lokay is Head of Change Implementation, U.S. Oncology at AstraZeneca plc, a pharmaceutical company, a position he has held since April 2022. Prior to that, Mr. Lokay was the Head of the U.S. Immuno-oncology Franchise at AstraZeneca plc from November 2019 to April 2022 and the Head of the U.S. Lung Cancer Franchise at AstraZeneca plc from August 2018 until November 2019. Mr. Lokay served as an advisor to AbbVie Inc., a pharmaceutical company, from August 2017 until December 2017. Mr. Lokay was previously Vice President and Business Unit Head, Oncology at Boehringer Ingelheim, a pharmaceutical company, a position he held from December 2009 until December 2016. Prior to joining Boehringer Ingelheim, he was President and Chief Executive Officer of Cytogen Corporation from 2007 until 2008 and served in various positions at GlaxoSmithKline from 1997 until 2007 and at Merck & Co. from 1981 until 1997. Mr. Lokay received a B.A. in Economics from Lafayette College and a M.S. from Purdue University. We believe that Mr. Lokay is qualified to serve on our board of directors due to his extensive experience in the biopharmaceutical industry.
Linda West has served as a member of our board of directors since March 2020 and also serves as Chair of our Compensation Committee. Ms. West served in multiple leadership roles of increasing responsibility for E. I.
102
du Pont de Nemours and Company from August 1981 until her retirement in December 2019. Ms. West most recently served as Vice President, Corporate Planning and Analyses, where she led the execution of transformational transactions from October 2009 until her retirement, including major divestitures, spin-offs, acquisitions, and the merger with The Dow Company followed by simultaneous spin-offs into three independent companies. Throughout her career with DuPont, Ms. West led early and late stage businesses including DuPont Imaging Technologies, DuPont Personal Protection, DuPont Microcircuit Materials, and DuPont Industrial Imaging. Prior to serving as Vice President, Corporate Planning and Analyses, Ms. West was the Chief Financial Officer of multiple DuPont businesses and was the Vice President, General Auditor and Chief Ethics and Compliance Officer for five years during the initial implementation of the Sarbanes-Oxley Act of 2002. Ms. West currently serves on the board of directors of Context Therapeutics. Ms. West holds a B.S. in Accounting with a minor in Business Administration from the University of Delaware. We believe that Ms. West is qualified to serve on our board of directors due to her finance background and extensive experience in business management and corporate transactions.
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
Market Information and Holders
Our common stock has been publicly traded on the Nasdaq Global Market under the symbol “GRTX” since November 7, 2019. Prior to that time, there was no public market for our common stock.
On March 4, 2023, there were 13 holders of record of our common stock.
Dividends
We have never declared or paid any dividends on our common stock. We anticipate that we will retain all of our future earnings, if any, for use in the operation and expansion of our business and do not anticipate paying cash dividends in the foreseeable future.
Purchases of Equity Securities by the Issuer or Affiliated Purchasers
We did not repurchase any of our equity securities during the quarter ended December 31, 2022.
Recent Sales of Unregistered Securities
We did not make any sales of unregistered securities during the year ended December 31, 2022.
Item 6. [Reserved]
103
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
You should read the following discussion and analysis of our financial condition and results of operations together with our consolidated financial statements and the related notes and other financial information included elsewhere in this Form 10-K. Some of the information contained in this discussion and analysis contains forward-looking statements that involve risks and uncertainties. You should review the sections titled “Summary Risk Factors” and Part I, Item 1A. “Risk Factors” in this Form 10-K for a discussion of important factors that could cause actual results to differ materially from the results described below. Our results of operations for the year ended December 31, 2020, including a discussion of the year ended December 31, 2021 compared to the year ended December 31, 2020, has been reported previously in our Annual Report on Form 10-K for the year ended December 31, 2021, filed with the SEC on March 10, 2022, under the heading “Management’s Discussion and Analysis of Financial Condition and Results of Operations.”
Overview
We are a clinical stage biopharmaceutical company focused on developing and commercializing a pipeline of novel, proprietary therapeutics that have the potential to transform radiotherapy in cancer. We leverage our expertise in superoxide dismutase mimetics to design drugs to reduce normal tissue toxicity from radiotherapy and to increase the anti-cancer efficacy of radiotherapy. Avasopasem manganese (avasopasem, or GC4419) is a highly selective small molecule dismutase mimetic in development for the reduction of severe oral mucositis, or SOM, in patients with head and neck cancer, or HNC, and for the reduction of esophagitis in patients with lung cancer. We are also exploring the potential for avasopasem to reduce cisplatin-induced kidney damage. SOM is a common, debilitating complication of radiotherapy in patients with HNC. In February 2018, the U.S. Food and Drug Administration, or FDA, granted Breakthrough Therapy Designation to avasopasem for the reduction of SOM induced by radiotherapy. In February 2023, the FDA accepted and granted Priority Review designation to our New Drug Application, or NDA, for avasopasem for this indication. Our second dismutase mimetic product candidate, rucosopasem manganese (rucosopasem, or GC4711), is in clinical-stage development to augment the anti-cancer efficacy of stereotactic body radiation therapy, or SBRT, in patients with non-small cell lung cancer, or NSCLC, and locally advanced pancreatic cancer, or LAPC.
Radiotherapy-induced SOM can lead to devastating complications for patients. A majority will suffer severe pain which is often managed with the use of opioids. Patients with SOM are at risk of dehydration and malnutrition as a result of the inability to eat or drink, and often require nutrition through an intravenous line or surgical placement of a feeding tube. Each year in the United States, approximately 67,000 patients are diagnosed with HNC, according to the American Cancer Society. In the five largest European markets, approximately 68,000 patients are diagnosed annually with HNC, and an additional 23,000 in Japan. We estimate that approximately 65% of patients diagnosed with HNC will be treated with radiotherapy. All patients with HNC treated with radiotherapy are at risk for developing SOM, which suggests a target patient population of approximately 43,500 patients in the United States alone for our lead indication. We believe that SOM in patients with HNC represents a total market opportunity of more than $1.5 billion in the United States based on branded supportive care price analogs. There are currently no FDA-approved drugs for SOM in these patients and we believe avasopasem, which to date is not approved for any indication, has the potential to become the standard of care for the reduction of SOM in patients with HNC receiving radiotherapy.
In December 2021, we announced topline efficacy results from a Phase 3 trial of avasopasem for the reduction of radiotherapy-induced SOM in patients with locally advanced HNC, which we refer to as the ROMAN trial. The results demonstrated efficacy across multiple SOM endpoints with a statistically significant reduction on the primary endpoint of reduction in the incidence of SOM and a statistically significant reduction on the secondary endpoint of number of days of SOM, with a median of 18 days in the placebo arm versus 8 days in the avasopasem arm. We had previously announced topline results from the ROMAN trial in October 2021 that incorrectly stated the reduction on the primary endpoint was not statistically significant. Upon further analysis, an error by the contract research organization was identified in the statistical program. Correction of this error yielded the correct, statistically significant p-values for the primary and a key secondary endpoint. Exploratory analyses, such as time to SOM onset and SOM incidence at various landmarks of radiotherapy delivered, further demonstrated clinical efficacy of avasopasem in reducing the burden of SOM. Avasopasem appeared to be generally well tolerated compared to placebo.
104
In October 2022, we announced the presentation of the one-year tumor and renal function outcomes data from the ROMAN trial as well as topline results from a recently completed meta-analysis of the ROMAN and GT-201 (Phase 2b) SOM trial results at the 2022 American Society for Radiation Oncology, or ASTRO, Annual Meeting. After one-year follow-up, patients with HNC treated with avasopasem in combination with the standard-of-care regimen (intensity-modulated radiation therapy, or IMRT, plus cisplatin) demonstrated comparable tumor outcomes and overall survival to patients in the placebo arm, showing that avasopasem protected HNC patients from SOM without affecting the treatment benefit of standard-of-care chemoradiotherapy. In addition, after one year of follow-up, patients treated with avasopasem in combination with IMRT plus cisplatin had a 10% incidence of chronic kidney disease, or CKD, compared to 20% of patients in the placebo arm, which was a pre-defined exploratory endpoint evaluating renal function. CKD (eGFR <60) is a known toxicity risk with cisplatin, which can have significant long-term consequences. The prospective exploration of this potential benefit of avasopasem was driven by published preclinical data and a post hoc assessment of patients from the GT-201 trial presented at the 2020 American Society of Clinical Oncology, or ASCO, Annual Meeting. We believe these CKD data suggest another potential benefit of avasopasem for these patients beyond reducing SOM. In addition to the ROMAN long-term endpoints, a meta-analysis of the Company’s two randomized placebo-controlled trials (ROMAN and GT-201; n=551) was included in the ASTRO presentation; these results reinforced that across both trials avasopasem therapy resulted in clinically meaningful improvements in radiotherapy-induced SOM, including reductions in the incidence, number of days, severity, and delay in the onset of SOM compared to placebo.
In December 2022, we submitted an NDA to the FDA for avasopasem for the reduction of radiotherapy-induced SOM in patients with HNC undergoing standard-of-care treatment. The NDA is supported by the data from the two randomized, double-blinded, placebo-controlled trials (ROMAN and GT-201), as well as data from other clinical trials of avasopasem in the proposed indication. In February 2023, the FDA accepted the NDA for filing and granted priority review with a Prescription Drug User Fee Act, or PDUFA, target date of August 9, 2023. The FDA indicated in its acceptance of filing letter that it is not planning to hold an advisory committee meeting on the application.
In December 2021, we also announced topline results from a Phase 2a multi-center trial in Europe assessing the safety and efficacy of avasopasem in patients with HNC undergoing standard-of-care radiotherapy, which we refer to as the EUSOM trial. This trial was conducted in twelve centers across six countries in Europe and enrolled 38 patients, of which 33 completed full treatment. Avasopasem appeared to be generally well tolerated. The incidence of SOM was 54.5% and median number of days of SOM was 9 days in the EUSOM trial, in line with the ROMAN trial in which the incidence of SOM in the avasopasem arm was 54% and the median duration was 8 days. We plan to meet with the European Medicines Agency, or EMA, in 2023 to discuss the potential registration pathway in Europe for avasopasem for radiotherapy-induced SOM.
In May 2022, we announced topline results from an open-label, single-arm Phase 2a trial evaluating avasopasem for its ability to reduce the incidence of radiotherapy-induced esophagitis in patients with lung cancer, which we refer to as the AESOP trial. This multi-center trial enrolled 39 patients (62 screened) of which 35 completed treatment with 60 gray of radiotherapy plus chemotherapy over six weeks. Of these 35 patients, 29 received at least five weeks of 90 mg of avasopasem on the days they underwent radiotherapy. These 29 patients were evaluated as the pre-specified per protocol population. The results demonstrated that avasopasem substantially reduced the incidence of severe esophagitis in patients with lung cancer receiving chemoradiotherapy compared to expectations based on review of historical data in the literature. Avasopasem was generally well tolerated. The adverse events experienced are comparable to those expected with chemoradiotherapy.
There are currently no FDA-approved drugs and no established guidelines for the treatment of radiotherapy-induced esophagitis. We intend to pursue a strategy for avasopasem, if approved for reduction in the incidence of SOM, that involves presenting the AESOP clinical data to entities like the National Comprehensive Cancer Network, or NCCN, to support the use of avasopasem to reduce esophagitis as a medically accepted indication in published drug compendia, notwithstanding that this indication may not be approved by the FDA.
In addition to developing avasopasem for the reduction of normal tissue toxicity from radiotherapy, we are developing our second dismutase mimetic product candidate, rucosopasem, to increase the anti-cancer efficacy of higher daily doses of radiotherapy, or SBRT. SBRT typically involves a patient receiving one to five large doses of radiotherapy, in contrast to the 30 to 35 small daily doses typical of intensity modulated radiation therapy, or
105
IMRT. Clinically, SBRT is increasingly used in patients with certain tumors, such as LAPC and NSCLC, that are less responsive to the small daily doses typical of IMRT. Even with the use of SBRT, there is need for improvement in treatment outcomes for certain tumors.
In September 2021, in support of rucosopasem, we announced final results from our pilot Phase 1/2 safety and anti-cancer efficacy trial of avasopasem in combination with SBRT in patients with unresectable or borderline resectable LAPC. The results included a minimum follow up of one year on all 42 patients enrolled in the trial and were consistent with the positive interim results reported with a minimum follow up of six months. In this proof-of-concept trial, relative improvements were observed in overall survival, progression-free survival, local tumor control and time to distant metastases. 46% of patients in the active arm were alive at last follow-up (11 out of 24) compared to 33% in the placebo arm (6 out of 18). As previously reported, 29% of patients in the active arm achieved a 30% or greater decrease in primary tumor size (partial response) compared to 11% of patients in the placebo arm. Avasopasem was well tolerated, with similar rates of early and late adverse events in the active and placebo arms.
We used our observations from the pilot LAPC trial of avasopasem to inform the design of our rucosopasem clinical trials in combination with SBRT. We have successfully completed Phase 1 trials of intravenous rucosopasem in healthy volunteers and initiated a Phase 1/2 trial in patients with non-small cell lung cancer, or NSCLC, in October 2020, which we refer to as the GRECO-1 trial, and in May 2021, initiated a Phase 2b trial in patients with LAPC, which we refer to as the GRECO-2 trial.
The GRECO-1 trial is supported in part by a Small Business Innovation Research grant from the National Cancer Institute of the National Institutes of Health for the investigation of our dismutase mimetics in combination with SBRT for the treatment of lung cancer. We intend for this trial to assess the anti-cancer efficacy and safety of rucosopasem in combination with SBRT. In June 2022, we reported interim results from the open-label Phase 1 stage of the trial with six months follow-up on all seven patients. Rucosopasem in combination with SBRT appeared to be well tolerated through the cutoff date of June 14, 2022. The most frequent adverse events were fatigue, cough, and nausea, which are common in patients with lung cancer receiving radiotherapy. Through six months, in-field partial responses were observed in three patients and stable disease was observed in three others based on RECIST criteria. These results included target tumor reductions in five patients of 61%, 58%, 33%, 29% and 27% and one patient with an 8% increase. Preservation of pulmonary lung function was also observed compared to our expectations based on review of historical literature evaluating pulmonary function in a similar patient population with SBRT alone. We expect to complete enrollment in the randomized, placebo-controlled Phase 2 stage of this trial in the second half of 2023.
The GRECO-2 trial is designed to assess rucosopasem in combination with SBRT in patients with LAPC, based on our observations from the pilot LAPC trial with avasopasem. The primary endpoint of this trial is overall survival. We expect to complete enrollment in the GRECO-2 trial in the second half of 2023.
Since our inception, we have devoted substantially all of our resources to organizing and staffing our company, business planning, raising capital, acquiring and developing product and technology rights, and conducting research and development. We have incurred recurring losses and negative cash flows from operations and have funded our operations primarily through the sale and issuance of equity and proceeds received under the Amended and Restated Purchase and Sale Agreement, which we refer to as the Royalty Agreement, with Clarus IV Galera Royalty AIV, L.P., Clarus IV-A, L.P., Clarus IV-B, L.P., Clarus IV-C, L.P. and Clarus IV-D, L.P., or collectively, Blackstone or Blackstone Life Sciences (formerly known as Clarus Ventures). On February 17, 2023, we completed a registered direct offering, which resulted in the issuance and sale of 14,320,000 shares of our common stock and warrants to purchase up to 14,320,000 shares of common stock at a combined offering price of $2.095 per share and accompanying warrant, generating gross proceeds of $30.0 million. The warrants have an exercise price of $1.97 per share of common stock, are exercisable immediately following their issuance and will expire five years from the date of issuance. We received net proceeds of approximately $27.7 million from this offering, after deducting placement agent fees and offering expenses.
Our ability to generate product revenue sufficient to achieve profitability will depend heavily on the successful development and eventual commercialization of one or more of our current or future product candidates. Our net loss was $62.2 million and $80.5 million for the years ended December 31, 2022 and 2021, respectively. As
106
of December 31, 2022, we had $31.6 million in cash, cash equivalents and short-term investments and an accumulated deficit of $378.3 million. We expect to continue to incur significant expenses and operating losses for the foreseeable future as we operate as a public company, advance our product candidates through all stages of development and clinical trials, build our commercial infrastructure and, ultimately, seek regulatory approval of our product candidates. In addition, if we obtain marketing approval for any of our product candidates, we expect to incur significant commercialization expenses related to product manufacturing, marketing, sales and distribution.
As a result, we will need to raise substantial additional capital to support our continuing operations and pursue our growth strategy. Until such time as we can generate significant revenue from product sales, if ever, we plan to finance our operations through the sale of equity, debt financings or other capital sources, which may include collaborations with other companies or other strategic transactions. There is no assurance that we will be successful in obtaining an adequate level of financing as and when needed to finance our operations on terms acceptable to us or at all. If we are unable to secure adequate additional funding as and when needed, we may have to significantly delay, scale back or discontinue the development and commercialization of one or more product candidates or delay our pursuit of potential in-licenses or acquisitions.
Because of the numerous risks and uncertainties associated with product development, we are unable to predict the timing or amount of increased expenses or when or if we will be able to achieve or maintain profitability. Even if we are able to generate product sales, we may not become profitable. If we fail to become profitable or are unable to sustain profitability on a continuing basis, then we may be unable to continue our operations at planned levels and be forced to reduce or terminate our operations.
We expect our existing cash, cash equivalents and short-term investments as of December 31, 2022, together with the net proceeds from our February 2023 registered direct offering, will enable us to fund our operating expenses and capital expenditure requirements into the fourth quarter of 2023, but not for more than one year after the date of the filing of this Annual Report on Form 10-K. As a result there is substantial doubt about our ability to continue as a going concern through the 12 months from the date of the filing of this Annual Report on Form 10-K. Our anticipated operating expenses involve significant risks and uncertainties and are dependent on our current assessment of the extent and costs of activities required to advance our product candidates. In the future, we anticipate that we will need to raise substantial additional financing to fund our operations through equity or debt financings, or through strategic transactions. To meet these requirements, we may seek to sell equity or convertible securities in public or private transactions that may result in significant dilution to our stockholders. We may offer and sell shares of our common stock under an existing registration statement or any registration statement we may file in the future. If we raise additional funds through the issuance of convertible securities, these securities could have rights senior to those of our common stock and could contain covenants that restrict our operations. We may also defer certain operating expenses unless and until additional capital is received. However, there can be no assurance that we will be successful in raising additional capital or that such capital, if available, will be on terms that are acceptable to us, or that we will be successful in deferring certain operating expenses. If we are unable to raise sufficient additional capital or defer sufficient operating expenses, we may be compelled to reduce the scope of our operations and planned capital expenditures and may decide to delay or discontinue certain activities, including planned research and development activities, hiring plans, manufacturing activities and commercial preparation efforts.
Nasdaq Listing Notification
On June 8, 2022, we received written notice, or the Notice, from The Nasdaq Stock Market LLC, or Nasdaq, indicating that we were no longer in compliance with the minimum Market Value of Listed Securities, or MVLS, of $50.0 million required for continued listing on The Nasdaq Global Market, as set forth in Nasdaq Listing Rule 5450(b)(2)(A), or the MVLS Requirement. The Notice had no immediate effect on the listing of our common stock, which continued to trade on The Nasdaq Global Market under the symbol “GRTX.”
In accordance with Nasdaq Listing Rule 5810(c)(3)(C), we had a period of 180 calendar days, or until December 5, 2022, or the Compliance Date, to regain compliance with the MVLS Requirement. To regain compliance, our MVLS had to close at $50,000,000 or more for a minimum of 10 consecutive business days prior to the Compliance Date. On December 6, 2022, we received a letter from Nasdaq indicating that we did not regain compliance with the MVLS Requirement by the Compliance Date and that, unless we request a hearing before a
107
Nasdaq hearings panel, or Panel, to appeal Nasdaq’s delisting determination by December 13, 2022, trading of our common stock would be suspended on December 15, 2022, and our common stock would be delisted from The Nasdaq Global Market. On December 13, 2022, we requested a hearing before a Panel. On January 24, 2023, prior to the scheduled hearing date, we received a letter from Nasdaq notifying us that we had regained compliance with the MVLS Requirement, as our MVLS had closed at over $50,000,000 for 10 consecutive business days, and that the hearing had been cancelled.
Delisting from the Nasdaq Global Market or any Nasdaq market could make trading our common stock more difficult for investors, potentially leading to declines in our share price and liquidity. In addition, delisting from Nasdaq could also make it more difficult for us to raise additional capital. See “Risk Factors—Our common stock may be delisted from The Nasdaq Global Market if we cannot maintain compliance with Nasdaq’s continued listing requirements, which could harm our business, the trading price of our common stock, our ability to raise additional capital and the liquidity of the market for our common stock” in Part I, Item 1A of this Annual Report on Form 10-K.
Business Update Regarding COVID-19
The current COVID-19 pandemic may continue to affect our employees, communities, clinical trial sites and business operations, as well as the U.S. economy and international financial markets. The full extent to which the COVID-19 pandemic will directly or indirectly impact our business, results of operations and financial condition will depend on future developments that are highly uncertain and cannot be accurately predicted, including the duration and potential resurgence of the pandemic. See “Risk Factors—Other Risks Related to Our Business—The COVID-19 pandemic has adversely impacted, and could continue to adversely impact, our business, including our preclinical studies and clinical trials, results of operations and financial condition” in Part I, Item 1A of this Annual Report on Form 10-K.
Our third-party contract manufacturing partners continue to operate at or near normal levels. While we currently do not anticipate any material interruptions in our clinical trial supply or manufacturing scale-up activities, it is possible that the COVID-19 pandemic and response efforts may have an impact in the future on our third-party suppliers and contract manufacturing partners' ability to manufacture our clinical trials supply or progress manufacturing scale-up activities.
We have also implemented measures designed to protect the health and safety of our workforce.
Critical Accounting Policies and Estimates
Our management’s discussion and analysis of our financial condition and results of operations are based on our consolidated financial statements, which have been prepared in accordance with U.S. generally accepted accounting principles. The preparation of these consolidated financial statements requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, and expenses and the disclosure of contingent assets and liabilities in our financial statements. On an ongoing basis, we evaluate our estimates and judgments, including those described below. We base our estimates on historical experience, known trends and events, and various other factors that are believed to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions.
While our significant accounting policies are described in more detail in Note 2 to our consolidated financial statements included elsewhere in this Annual Report on Form 10-K, we believe the following accounting policies are the most critical to the judgments and estimates used in the preparation of our financial statements.
Royalty Purchase Liability
Pursuant to our amended Royalty Agreement with Blackstone Life Sciences, we have received cash payments totaling $117.5 million from Blackstone based upon the achievement of specified clinical milestones, which have been recorded as long-term debt obligations. Interest expense on such obligation is imputed by
108
estimating risk adjusted future royalty payments over the term of the amended Royalty Agreement which takes into consideration the probability of obtaining FDA approval. Other significant assumptions include adjustments to estimated gross revenues to arrive at net product sales from which a royalty payment can be estimated. The non-cash interest expense recorded increases the balance of our royalty obligation. The royalty obligation will be reduced when royalty payments are made, if any.
Actual royalty payments, however, are highly uncertain and may change depending on a number of factors, including our ability to obtain FDA approval, successfully commercialize our product candidates and the timing of future royalty payments. We impute interest expense on our royalty purchase obligations based on such factors at each reporting period. As these factors change, we will adjust our estimate of the imputed interest expense accordingly.
Research and Development Expenses
Research and development expenses consist primarily of costs incurred in connection with the development of our product candidates. We expense research and development costs as incurred.
We accrue an expense for manufacturing, preclinical studies and clinical trial activities performed by third parties based upon estimates of the proportion of work completed over the term of the individual trial and patient enrollment rates in accordance with agreements with CMOs, CROs and clinical trial sites. We determine the estimates by reviewing contracts, vendor agreements and purchase orders, and through discussions with our internal research and development personnel and external service providers as to the progress or stage of completion of trials or services and the agreed-upon fee to be paid for such services. However, actual costs and timing of these activities are highly uncertain, subject to risks and may change depending upon a number of factors, including our clinical development plan.
We make estimates of our accrued expenses as of each balance sheet date in our consolidated financial statements based on facts and circumstances known at that time. If the actual timing of the performance of services or the level of effort varies from the estimate, we will adjust the accrual accordingly. Nonrefundable advance payments for goods and services, including fees for process development or manufacturing and distribution of clinical supplies that will be used in future research and development activities, are deferred and recognized as expense in the period that the related goods are consumed or services are performed.
JOBS Act Transition Period
In April 2012, the JOBS Act was enacted. Section 107 of the JOBS Act provides that an “emerging growth company” can take advantage of the extended transition period provided in Section 7(a)(2)(B) of the Securities Act for complying with new or revised accounting standards. Thus, an emerging growth company can delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. However, we have chosen to opt out of such extended transition period and, as a result, we will comply with new or revised accounting standards on the relevant dates on which adoption of such standards is required for non-emerging growth companies. Our decision to opt out of the extended transition period for complying with new or revised accounting standards is irrevocable. However, we may take advantage of the other exemptions discussed below.
Subject to certain conditions, as an emerging growth company we may rely on certain exemptions and reduced reporting requirements, including, without limitation, (1) not being required to provide an auditor’s attestation report on our system of internal control over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act and (2) not being required to comply with any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation or a supplement to the auditor’s report providing additional information about the audit and the financial statements, known as the auditor discussion and analysis. We will remain an emerging growth company until the earlier to occur of (a) the last day of the fiscal year in which we have total annual gross revenues of $1.235 billion or more, (b) the last day of the fiscal year following the fifth anniversary of the date of the completion of our IPO (December 31, 2024), (c) the date on which we have issued more than $1 billion in nonconvertible debt during the previous three years, or (d) the date on which we are deemed to be a large accelerated filer under the rules of the SEC, which means the market value of our
109
common stock that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter.
Components of Results of Operations
Research and Development Expense
Research and development expenses consist primarily of costs incurred in connection with the discovery and development of our product candidates. We expense research and development costs as incurred. These expenses include:
We track our external research and development expenses on a program-by-program basis, such as fees paid to CROs, CMOs and research laboratories in connection with our preclinical development, process development, manufacturing and clinical development activities. However, we do not track our internal research and development expenses on a program-by-program basis as they primarily relate to personnel-related and share-based compensation expense, early-stage research expenses and other costs that are deployed across multiple projects under development.
The following table summarizes our research and development expenses by program for the years ended December 31, 2022 and 2021 (in thousands):
|
|
Year ended |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Avasopasem manganese (GC4419) |
|
$ |
9,086 |
|
|
$ |
28,120 |
|
Rucosopasem manganese (GC4711) |
|
|
9,590 |
|
|
|
6,332 |
|
Other research and development expense |
|
|
3,294 |
|
|
|
7,905 |
|
Personnel related and share-based compensation |
|
|
9,042 |
|
|
|
10,060 |
|
|
|
$ |
31,012 |
|
|
$ |
52,417 |
|
Research and development activities are central to our business model. Product candidates in later stages of clinical development, such as avasopasem, generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of later-stage clinical trials. Our research and development expenses may increase over the next several years as we increase personnel costs, including
110
stock-based compensation, conduct our later-stage clinical trials for avasopasem and rucosopasem, if applicable, conduct other clinical trials for current and future product candidates and prepare regulatory filings for our product candidates.
The successful development of our product candidates is highly uncertain. At this time, we cannot reasonably estimate or know the nature, timing and costs of the efforts that will be necessary to complete the remainder of the development of our product candidates, including the significant costs associated with our ongoing and planned clinical trials, which likely will vary significantly as a result of many factors, including:
Our research and development expenditures are subject to additional uncertainties, including the terms and timing of regulatory approvals. We may never succeed in achieving regulatory approval for our product candidates. We may obtain unexpected results from our clinical trials. We may elect to discontinue, delay or modify clinical trials of our product candidates. A change in the outcome of any of these variables with respect to the development of a product candidate could result in a significant change in the costs of and timing associated with the development of that product candidate. For example, if the FDA or other regulatory authorities were to require us to conduct clinical trials beyond those that we currently anticipate, or if we experience significant delays in enrollment in any of our clinical trials, we could be required to expend significant additional financial resources and time on the completion of clinical development.
General and Administrative Expense
General and administrative expense consists primarily of personnel expenses, including salaries, benefits and share-based compensation expense for employees in executive, finance, accounting, legal, information technology, commercial, business development and human resource functions. General and administrative expense also includes corporate facility costs, including rent, utilities, depreciation and maintenance, not otherwise included in research and development expense, as well as legal fees related to intellectual property and corporate matters and fees for accounting and consulting services.
We expect that our general and administrative expense will increase in the future to support our continued research and development activities, potential commercialization efforts, and to expand our operations
111
and organizational capabilities. These increases will likely include increased costs related to the hiring of additional personnel, fees to outside consultants, lawyers and accountants and expenses related to services associated with maintaining compliance with the requirements of Nasdaq and the SEC, insurance and investor relations costs. Should we commercialize our product candidates, we expect to incur significantly increased expenses associated with building our commercial infrastructure.
Interest Income
Interest income consists of amounts earned on our cash and cash equivalents held with large institutional banks, U.S. Treasury obligations and a money market mutual fund invested in U.S. Treasury obligations, and our short-term investments in U.S. Treasury and government agency obligations.
Interest Expense
Interest expense consists of non-cash interest on proceeds received under the Royalty Agreement with Blackstone and non-cash interest expense associated with the amortization of the debt discount recorded for the Blackstone warrants.
Foreign Currency Losses
Foreign currency losses consist primarily of exchange rate fluctuations on transactions denominated in a currency other than the U.S. dollar.
Income Tax Benefit
In the year ended December 31, 2022, we recognized an income tax benefit for the revaluation of our deferred tax liability as a result of changes to the anticipated effective tax rate in certain state and local jurisdictions in which we have operations.
Net Operating Loss and Research and Development Tax Credit Carryforwards
As of December 31, 2022, we had federal and state tax net operating loss carryforwards of $162.3 million and $184.4 million, respectively, which each begin to expire in 2032 unless previously utilized. We also had foreign net operating loss carryforwards of $1.7 million which do not expire. As of December 31, 2022, we also had federal, state and foreign research and development tax credit carryforwards of $7.3 million. The federal and state research and development tax credit carryforwards will begin to expire in 2032 and 2036, respectively, unless previously utilized. The foreign research and development tax credit carryforwards do not have an expiration date.
Utilization of the federal and state net operating losses and credits may be subject to a substantial annual limitation. The annual limitation may result in the expiration of our net operating losses and credits before we can use them. We have recorded a valuation allowance on substantially all of our deferred tax assets, including our deferred tax assets related to our net operating loss and research and development tax credit carryforwards, given the current uncertainty over our ability to utilize such amounts.
112
Results of Operations for the Years Ended December 31, 2022 and 2021
The following table sets forth our results of operations for the years ended December 31, 2022 and 2021 (in thousands):
|
|
Year ended |
|
|
|
|
||||||
|
|
2022 |
|
|
2021 |
|
|
Change |
|
|||
Operating expenses: |
|
|
|
|
|
|
|
|
|
|||
Research and development |
|
$ |
31,012 |
|
|
$ |
52,417 |
|
|
$ |
(21,405 |
) |
General and administrative |
|
|
20,214 |
|
|
|
20,951 |
|
|
|
(737 |
) |
Loss from operations |
|
|
(51,226 |
) |
|
|
(73,368 |
) |
|
|
22,142 |
|
Other income (expense): |
|
|
|
|
|
|
|
|
|
|||
Interest income |
|
|
506 |
|
|
|
32 |
|
|
|
474 |
|
Interest expense |
|
|
(11,571 |
) |
|
|
(7,194 |
) |
|
|
(4,377 |
) |
Foreign currency loss |
|
|
(1 |
) |
|
|
(4 |
) |
|
|
3 |
|
Loss before income tax benefit |
|
|
(62,292 |
) |
|
|
(80,534 |
) |
|
|
18,242 |
|
Income tax benefit |
|
|
70 |
|
|
|
— |
|
|
|
70 |
|
Net loss |
|
$ |
(62,222 |
) |
|
$ |
(80,534 |
) |
|
$ |
18,312 |
|
Research and Development Expense
Research and development expense decreased by $21.4 million from $52.4 million for the year ended December 31, 2021 to $31.0 million for the year ended December 31, 2022. The decrease was primarily attributable to a decrease of $19.0 million for avasopasem development costs, as the ROMAN, EUSOM, and AESOP trials completed enrollment in 2021, and decreased manufacturing expenses. Other research and development expenses decreased $4.6 million due to reduced costs for independent contractors and consultants, decreased costs for development of additional product candidates, and a reduction in facility and equipment related expenses. Personnel related and share-based compensation expenses decreased $1.0 million due to decreased headcount. Partially offsetting these decreases, rucosopasem development costs increased $3.3 million as enrollment increased in the GRECO-2 trial and we expanded the trial to Europe and Canada, partially offset by decreased manufacturing and preclinical expenses.
General and Administrative Expense
General and administrative expense decreased by $0.8 million from $21.0 million for the year ended December 31, 2021 to $20.2 million for the year ended December 31, 2022, principally due to the timing of spend for avasopasem commercial preparations.
Interest Income
Interest income increased by $0.5 million from $32,000 for the year ended December 31, 2021 to $0.5 million for the year ended December 31, 2022, due to increased interest rates on invested cash and securities.
Interest Expense
We recognized $11.6 million and $7.2 million in non-cash interest expense during the years ended December 31, 2022 and 2021, respectively, in connection with the Royalty Agreement with Blackstone Life Sciences. The increase is primarily attributable to interest on the $57.5 million in milestone payments received in June and July 2021, as well as an increase in the imputed interest rate.
Liquidity and Capital Resources
We do not currently have any approved products and have never generated any revenue from product sales. Through December 31, 2022, we have funded our operations primarily through the sale and issuance of equity
113
and $117.5 million of proceeds received under the Royalty Agreement with Blackstone Life Sciences, receiving aggregate gross proceeds of $342.9 million. In November 2019, we completed our IPO, which resulted in the issuance and sale of 5,000,000 shares of common stock at a public offering price of $12.00 per share, generating net proceeds of $53.0 million after deducting underwriting discounts and other offering costs. On December 9, 2019, in connection with the partial exercise of the over-allotment option granted to the underwriters of our IPO, 445,690 additional shares of common stock were sold at the IPO price of $12.00 per share, generating net proceeds of approximately $5.0 million after deducting underwriting discounts and other offering costs.
In December 2020, we entered into an Open Market Sale Agreement, or the Sales Agreement, with Jefferies LLC, or Jefferies, as sales agent, pursuant to which we may, from time to time, issue and sell common stock with an aggregate value of up to $50.0 million in “at-the-market,” or ATM, offerings under our Registration Statement on Form S-3 (File No. 333-251061) filed with the SEC on December 1, 2020. Sales of common stock, if any, pursuant to the Sales Agreement, may be made in sales deemed to be an “at the market offering” as defined in Rule 415(a) of the Securities Act, including sales made directly through the Nasdaq Global Market or on any other existing trading market for our common stock. During the year ended December 31, 2022, we sold an aggregate of 1,982,773 shares of our common stock under the Sales Agreement at a weighted average price per share of $2.00, generating aggregate net proceeds of $3.8 million after deducting fees, commissions and other expenses. As of December 31, 2022, there was $37.8 million of common stock remaining available for sale under the Sales Agreement.
As of December 31, 2022, we had $31.6 million in cash, cash equivalents and short-term investments and an accumulated deficit of $378.3 million. We have no ongoing material financing commitments, such as lines of credit or guarantees, that are expected to affect our liquidity over the next five years.
On February 17, 2023, we completed a registered direct offering, which resulted in the issuance and sale of 14,320,000 shares of our common stock and warrants to purchase up to 14,320,000 shares of common stock at a combined offering price of $2.095 per share and accompanying warrant, generating gross proceeds of $30.0 million. The warrants have an exercise price of $1.97 per share of common stock, are exercisable immediately following their issuance and will expire five years from the date of issuance. We received net proceeds of approximately $27.7 million from this offering, after deducting placement agent fees and offering expenses.
Cash Flows
The following table shows a summary of our cash flows for the periods indicated (in thousands):
|
|
Year ended |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Net cash used in operating activities |
|
$ |
(43,426 |
) |
|
$ |
(67,958 |
) |
Net cash provided by investing activities |
|
|
23,994 |
|
|
|
5,238 |
|
Net cash provided by financing activities |
|
|
3,889 |
|
|
|
66,707 |
|
Net increase (decrease) in cash, cash equivalents and restricted cash |
|
$ |
(15,543 |
) |
|
$ |
3,987 |
|
Operating Activities
During the year ended December 31, 2022, we used $43.4 million of net cash in operating activities. Cash used in operating activities reflected our net loss of $62.2 million, partially offset by non-cash charges of $18.8 million related to share-based compensation, interest expense on our Royalty Agreement with Blackstone Life Sciences, depreciation and amortization expense and deferred income tax. The primary use of cash was to fund our operations related to the development of our product candidates.
During the year ended December 31, 2021, we used $68.0 million of net cash in operating activities. Cash used in operating activities reflected our net loss of $80.5 million and a $2.6 million net decrease in cash from changes in our operating assets and liabilities, partially offset by non-cash charges of $15.2 million related to share-based compensation, interest expense on our Royalty Agreement with Blackstone Life Sciences and depreciation
114
and amortization expense. The primary use of cash was to fund our operations related to the development of our product candidates.
Investing Activities
During the year ended December 31, 2022, investing activities provided $24.0 million in cash proceeds from net sales of our short-term investments.
During the year ended December 31, 2021, investing activities provided $5.2 million of net cash, primarily attributable to $5.5 million in net proceeds from the purchases and sales of our short-term investments, partially offset by $0.3 million for the purchase of property and equipment.
Financing Activities
During the year ended December 31, 2022, financing activities provided $3.9 million from the sale of our common stock under the Sales Agreement with Jefferies and the exercise of stock options.
During the year ended December 31, 2021, financing activities provided $66.7 million in net cash proceeds, primarily attributable to $57.5 million in proceeds received in connection with the Royalty Agreement with Blackstone Life Sciences, $7.9 million in net proceeds from the sale of our common stock under the ATM Sales Agreement, and $1.3 million in proceeds from the exercise of stock options.
Funding Requirements
Our operating expenses increased substantially in 2021. While our operating expenses decreased in 2022, primarily due to a decrease in avasopasem development costs as the ROMAN, EUSOM, and AESOP trials completed enrollment in 2021, our expenses are expected to increase in 2023 in connection with our ongoing activities, particularly as we continue the research and development of, continue or initiate clinical trials of, seek marketing approval for, and prepare for commercialization of our product candidates. In addition, if we obtain marketing approval for any of our product candidates, we expect to incur significant commercialization expenses related to product sales, marketing, manufacturing and distribution. Furthermore, we expect to continue to incur significant costs associated with operating as a public company. Accordingly, we would need to obtain substantial additional funding in connection with our continuing operations. If we are unable to raise capital when needed or on attractive terms, we would be forced to delay, reduce or eliminate our research and development programs or future commercialization efforts.
We expect our existing cash, cash equivalents and short-term investments as of December 31, 2022, together with the net proceeds from our February 2023, registered direct offering will enable us to fund our operating expenses and capital expenditure requirements into the fourth quarter of 2023, but not for more than one year after the date of the filing of this Annual Report on Form 10-K. As a result there is substantial doubt about our ability to continue as a going concern through the 12 months from the date of the filing of this Annual Report on Form 10-K. Our anticipated operating expenses involve significant risks and uncertainties and are dependent on our current assessment of the extent and costs of activities required to advance our product candidates. In the future, we anticipate that we will need to raise substantial additional financing to fund our operations through equity or debt financings, or through strategic transactions. To meet these requirements, we may seek to sell equity or convertible securities in public or private transactions that may result in significant dilution to our stockholders. We may offer and sell shares of our common stock under an existing registration statement or any registration statement we may file in the future. If we raise additional funds through the issuance of convertible securities, these securities could have rights senior to those of our common stock and could contain covenants that restrict our operations. We may also defer certain operating expenses unless and until additional capital is received. However, there can be no assurance that we will be successful in raising additional capital or that such capital, if available, will be on terms that are acceptable to us, or that we will be successful in deferring certain operating expenses. If we are unable to raise sufficient additional capital or defer sufficient operating expenses, we may be compelled to reduce the scope of our operations and planned capital expenditures and may decide to delay or discontinue certain activities, including planned research and development activities, hiring plans, manufacturing activities and commercial preparation efforts.
115
Because of the numerous risks and uncertainties associated with research, development and commercialization of product candidates, we are unable to estimate the exact amount of our working capital requirements. Our future funding requirements will depend on, and could increase significantly as a result of, many factors, including:
Identifying potential product candidates and conducting preclinical studies and clinical trials is a time-consuming, expensive and uncertain process that takes many years to complete, and we may never generate the necessary data or results required to obtain marketing approval and achieve product sales. In addition, our product candidates, if approved, may not achieve commercial success. Our commercial revenues, if any, will be derived from sales of product candidates that we do not expect to be commercially available for the next couple of years, if at all. Accordingly, we will need to continue to rely on additional financing to achieve our business objectives. Adequate additional financing may not be available to us on acceptable terms, or at all. For example, the trading prices for our and other biopharmaceutical companies’ stock have been highly volatile as a result of disruptions and extreme volatility in the global economy, including rising inflation and interest rates, declines in economic growth, the conflict between Russia and Ukraine, the COVID-19 pandemic and uncertainty about economic stability. As a result, we may face difficulties raising capital through sales of our common stock and any such sales may be on unfavorable terms. See “Risk Factors” in Part I, Item 1A of this Annual Report on Form 10-K.
Until such time, if ever, as we can generate substantial product revenues, we expect to finance our cash needs through a combination of equity offerings, debt financings, collaborations, strategic alliances and licensing arrangements. To the extent that we raise additional capital through the sale of equity or convertible debt securities, our shareholders’ ownership interest will be diluted, and the terms of these securities may include liquidation or other preferences that adversely affect our existing stockholders’ rights. Debt financing, if available, may involve agreements that include covenants limiting or restricting our ability to take specific actions, such as incurring additional debt, making capital expenditures or declaring dividends.
If we raise funds through additional collaborations, strategic alliances or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or to grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds when needed, we may be required to delay, limit, reduce or terminate our product development or future commercialization efforts or grant rights to develop and market product candidates that we would otherwise prefer to develop and market ourselves.
116
Royalty Agreement with Blackstone Life Sciences (Formerly Known as Clarus Ventures)
In November 2018, we entered into the Royalty Agreement with Blackstone Life Sciences. Pursuant to the Royalty Agreement, Blackstone agreed to pay us, in the aggregate, up to $80.0 million, or the Royalty Purchase Price, in four tranches of $20.0 million each upon the achievement of specified clinical milestones in our ROMAN trial. We agreed to apply the proceeds from such payments primarily to support clinical development and regulatory activities for avasopasem, rucosopasem and any pharmaceutical product comprising or containing avasopasem or rucosopasem, or, collectively, the Products, as well as to satisfy working capital obligations and for general corporate expenses. We received the first tranche of the Royalty Purchase Price in November 2018, the second tranche of the Royalty Purchase Price in April 2019, and the third tranche of the Royalty Purchase Price in February 2020, in each case in connection with the achievement of the first three milestones, respectively, under the Royalty Agreement.
In May 2020, we entered into Amendment No. 1 to the Royalty Agreement, or the Amendment, with Clarus IV Galera Royalty AIV, L.P., or the Blackstone Purchaser. The Blackstone Purchaser is affiliated with Blackstone Life Sciences, successor in interest to Clarus Ventures. The Amendment increased the Royalty Purchase Price by $37.5 million to $117.5 million by increasing the fourth tranche from $20.0 million to $37.5 million and adding a new $20.0 million tranche upon the achievement of an additional clinical enrollment milestone. We received the new $20.0 million tranche of the Amendment in June 2021, in connection with the enrollment of the first patient in the GRECO-2 trial. Also in June 2021, we completed enrollment in the ROMAN trial, thereby achieving the milestone associated with the fourth tranche, and received the associated $37.5 million in July 2021.
Pursuant to the amended Royalty Agreement, in connection with the payment of each tranche of the Royalty Purchase Price, we have agreed to sell, convey, transfer and assign to Blackstone all of our right, title and interest in a high single-digit percentage of (i) worldwide net sales of the Products and (ii) all amounts received by us or our affiliates, licensees and sublicensees with respect to Product-related damages (collectively, the Product Payments) during the Royalty Period. The Royalty Period means, on a Product-by-Product and country-by-country basis, the period of time commencing on the commercial launch of such Product in such country and ending on the latest to occur of (i) the 12th anniversary of such commercial launch, (ii) the expiration of all valid claims of our patents covering such Product in such country, and (iii) the expiration of regulatory data protection or market exclusivity or similar regulatory protection afforded by the health authorities in such country, to the extent such protection or exclusivity effectively prevents generic versions of such Product from entering the market in such country.
The amended Royalty Agreement will remain in effect until the date on which the aggregate amount of the Product Payments paid to Blackstone exceeds a fixed single-digit multiple of the actual amount of the Royalty Purchase Price received by us, unless earlier terminated pursuant to the mutual written agreement of us and Blackstone. If no Products are commercialized, we would not have an obligation to make Product Payments to Blackstone, which is the sole mechanism for repaying the liability.
In May 2020, as partial consideration for the Amendment, we issued two warrants to the Blackstone Purchaser to purchase an aggregate of 550,661 shares of our common stock at an exercise price equal to $13.62 per share, each of which became exercisable upon the receipt by us of the applicable specified milestone payment. The issued warrants expire six years after the initial exercise date of each respective warrant.
Patheon Manufacturing Agreements
In August 2021, we entered into a Master Manufacturing Services Agreement with Patheon, or the Master Agreement. The Master Agreement governs the general terms under which Patheon, or one of its affiliates, will provide non-exclusive manufacturing services to us for the drug products specified by us from time to time. Pursuant to the Master Agreement, we have agreed to order from Patheon at least a certain percentage of our commercial requirements for a product under a related product agreement. Each product agreement that we may enter into from time to time will be governed by the terms of the Master Agreement, unless expressly modified in such product agreement.
117
In August 2021, we and Patheon entered into a product agreement for avasopasem, or the Product Agreement, under the Master Agreement to govern the terms and conditions of Patheon’s manufacture and commercial supply to us of avasopasem manganese from Patheon’s Greenville, North Carolina manufacturing site.
The Master Agreement, and any related product agreement, has an initial term that expires on December 31, 2027 and includes renewal terms, as applicable. In addition, each party has the ability to terminate the Product Agreement upon the occurrence of certain customary conditions. The Master Agreement contains representations, warranties and indemnity obligations customary for agreements of this type, and the Product Agreement establishes certain pricing for avasopasem that may be adjusted as set forth in the Master Agreement.
Our obligation to purchase avasopasem under the Product Agreement is subject to certain binding forecast periods at certain established prices, which will be reviewed each year on January 1 by us and Patheon. We currently do not have any contractual commitment to purchase avasopasem under the Product Agreement.
New Operating Lease
In October 2022, we entered into a new operating lease agreement for office space in Malvern, Pennsylvania. The lease commencement date is expected to occur in May 2023. The lease expiration date will be 7.4 years after the lease commencement date. Total rental payments are approximately $1.6 million from the commencement date through the expiration date. We are also required to pay the increase in certain operating expenses over a base year, in accordance with the terms of the lease agreement.
Item 7A. Quantitative and Qualitative Disclosures About Market Risk.
We are a smaller reporting company as defined in Rule 12b-2 of the Exchange Act and are not required to provide the information otherwise required under this Item 7A.
118
Item 8. Financial Statements and Supplementary Data.
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
119
Report of Independent Registered Public Accounting Firm
To the Stockholders and Board of Directors
Galera Therapeutics, Inc.:
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of Galera Therapeutics, Inc. and subsidiaries (the Company) as of December 31, 2022 and 2021, the related consolidated statements of operations, comprehensive loss, changes in stockholders’ equity (deficit), and cash flows for the years then ended, and the related notes (collectively, the consolidated financial statements). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2022 and 2021, and the results of its operations and its cash flows for the years then ended, in conformity with U.S. generally accepted accounting principles.
Going Concern
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note 1 to the consolidated financial statements, the Company has incurred recurring losses and negative cash flows from operations since inception that raise substantial doubt about its ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ KPMG LLP
We have served as the Company’s auditor since 2014.
Philadelphia, Pennsylvania
March 8, 2023
120
GALERA THERAPEUTICS, INC.
CONSOLIDATED BALANCE SHEETS
(IN THOUSANDS EXCEPT SHARE AND PER-SHARE AMOUNTS)
|
|
December 31, 2022 |
|
|
December 31, 2021 |
|
||
Assets |
|
|
|
|
|
|
||
Current assets: |
|
|
|
|
|
|
||
Cash and cash equivalents |
|
$ |
4,266 |
|
|
$ |
19,859 |
|
Short-term investments |
|
|
27,331 |
|
|
|
51,358 |
|
Restricted cash |
|
|
50 |
|
|
|
— |
|
Refundable PDUFA fee |
|
|
3,242 |
|
|
|
— |
|
Prepaid expenses and other current assets |
|
|
3,646 |
|
|
|
6,175 |
|
Total current assets |
|
|
38,535 |
|
|
|
77,392 |
|
Property and equipment, net |
|
|
438 |
|
|
|
527 |
|
Acquired intangible asset |
|
|
2,258 |
|
|
|
2,258 |
|
Goodwill |
|
|
881 |
|
|
|
881 |
|
Right-of-use lease assets |
|
|
43 |
|
|
|
296 |
|
Other assets |
|
|
1,881 |
|
|
|
1,957 |
|
Total assets |
|
$ |
44,036 |
|
|
$ |
83,311 |
|
Liabilities and stockholders’ deficit |
|
|
|
|
|
|
||
Current liabilities: |
|
|
|
|
|
|
||
Accounts payable |
|
$ |
3,581 |
|
|
$ |
5,044 |
|
Accrued expenses |
|
|
9,754 |
|
|
|
7,633 |
|
Lease liabilities |
|
|
44 |
|
|
|
258 |
|
Total current liabilities |
|
|
13,379 |
|
|
|
12,935 |
|
Royalty purchase liability |
|
|
139,635 |
|
|
|
128,063 |
|
Lease liabilities, net of current portion |
|
|
— |
|
|
|
44 |
|
Deferred tax liability |
|
|
203 |
|
|
|
273 |
|
Total liabilities |
|
|
153,217 |
|
|
|
141,315 |
|
|
|
|
|
|
|
|||
Stockholders’ deficit: |
|
|
|
|
|
|
||
Preferred stock, $0.001 par value: 10,000,000 shares authorized; no shares |
|
|
— |
|
|
|
— |
|
Common stock, $0.001 par value: 200,000,000 shares authorized; |
|
|
28 |
|
|
|
26 |
|
Additional paid-in capital |
|
|
269,137 |
|
|
|
258,086 |
|
Accumulated other comprehensive loss |
|
|
(22 |
) |
|
|
(14 |
) |
Accumulated deficit |
|
|
(378,324 |
) |
|
|
(316,102 |
) |
Total stockholders’ deficit |
|
|
(109,181 |
) |
|
|
(58,004 |
) |
Total liabilities and stockholders’ deficit |
|
$ |
44,036 |
|
|
$ |
83,311 |
|
See accompanying notes to consolidated financial statements.
121
GALERA THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF OPERATIONS
(IN THOUSANDS EXCEPT SHARE AND PER SHARE AMOUNTS)
|
|
Year ended |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Operating expenses: |
|
|
|
|
|
|
||
Research and development |
|
$ |
31,012 |
|
|
$ |
52,417 |
|
General and administrative |
|
|
20,214 |
|
|
|
20,951 |
|
Loss from operations |
|
|
(51,226 |
) |
|
|
(73,368 |
) |
Other income (expenses): |
|
|
|
|
|
|
||
Interest income |
|
|
506 |
|
|
|
32 |
|
Interest expense |
|
|
(11,571 |
) |
|
|
(7,194 |
) |
Foreign currency loss |
|
|
(1 |
) |
|
|
(4 |
) |
Loss before income tax benefit |
|
|
(62,292 |
) |
|
|
(80,534 |
) |
Income tax benefit |
|
|
70 |
|
|
|
— |
|
Net loss |
|
|
(62,222 |
) |
|
|
(80,534 |
) |
Net loss per share of common stock, basic and diluted |
|
$ |
(2.30 |
) |
|
$ |
(3.12 |
) |
Weighted-average shares of common stock outstanding, basic and diluted |
|
|
27,086,664 |
|
|
|
25,789,458 |
|
See accompanying notes to consolidated financial statements.
122
GALERA THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF COMPREHENSIVE LOSS
(IN THOUSANDS)
|
|
Year ended |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Net loss |
|
$ |
(62,222 |
) |
|
$ |
(80,534 |
) |
Unrealized loss on short-term investments |
|
|
(8 |
) |
|
|
(26 |
) |
Comprehensive loss |
|
$ |
(62,230 |
) |
|
$ |
(80,560 |
) |
See accompanying notes to consolidated financial statements.
123
GALERA THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF CHANGES IN STOCKHOLDERS’ EQUITY (DEFICIT)
(IN THOUSANDS EXCEPT SHARE AMOUNTS)
|
|
Common stock |
|
|
Additional |
|
|
Accumulated |
|
|
Accumulated |
|
|
Total |
|
|||||||||
|
|
Shares |
|
|
Amount |
|
|
capital |
|
|
loss |
|
|
Deficit |
|
|
Equity (Deficit) |
|
||||||
Balance at December 31, 2020 |
|
|
24,976,142 |
|
|
$ |
25 |
|
|
$ |
241,649 |
|
|
$ |
12 |
|
|
$ |
(235,568 |
) |
|
$ |
6,118 |
|
Share-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
7,231 |
|
|
|
— |
|
|
|
— |
|
|
|
7,231 |
|
Exercise of stock options |
|
|
591,257 |
|
|
|
— |
|
|
|
1,266 |
|
|
|
— |
|
|
|
— |
|
|
|
1,266 |
|
Sale of shares under Open Market Sale |
|
|
891,368 |
|
|
|
1 |
|
|
|
7,940 |
|
|
|
— |
|
|
|
— |
|
|
|
7,941 |
|
Unrealized loss on short-term |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(26 |
) |
|
|
— |
|
|
|
(26 |
) |
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(80,534 |
) |
|
|
(80,534 |
) |
Balance at December 31, 2021 |
|
|
26,458,767 |
|
|
|
26 |
|
|
|
258,086 |
|
|
|
(14 |
) |
|
|
(316,102 |
) |
|
|
(58,004 |
) |
Share-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
7,164 |
|
|
|
— |
|
|
|
— |
|
|
|
7,164 |
|
Exercise of stock options |
|
|
68,526 |
|
|
|
— |
|
|
|
81 |
|
|
|
— |
|
|
|
— |
|
|
|
81 |
|
Sale of shares under Open Market Sale |
|
|
1,982,773 |
|
|
|
2 |
|
|
|
3,806 |
|
|
|
— |
|
|
|
— |
|
|
|
3,808 |
|
Unrealized loss on short-term |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(8 |
) |
|
|
— |
|
|
|
(8 |
) |
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(62,222 |
) |
|
|
(62,222 |
) |
Balance at December 31, 2022 |
|
|
28,510,066 |
|
|
$ |
28 |
|
|
$ |
269,137 |
|
|
$ |
(22 |
) |
|
$ |
(378,324 |
) |
|
$ |
(109,181 |
) |
See accompanying notes to consolidated financial statements.
124
GALERA THERAPEUTICS, INC.
CONSOLIDATED STATEMENTS OF CASH FLOWS
(IN THOUSANDS)
|
|
Year ended |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Operating activities: |
|
|
|
|
|
|
||
Net loss |
|
$ |
(62,222 |
) |
|
$ |
(80,534 |
) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
||
Depreciation and amortization |
|
|
114 |
|
|
|
778 |
|
Noncash interest expense |
|
|
11,571 |
|
|
|
7,194 |
|
Share-based compensation expense |
|
|
7,164 |
|
|
|
7,231 |
|
Deferred tax liability |
|
|
(70 |
) |
|
|
— |
|
Changes in operating assets and liabilities: |
|
|
|
|
|
|
||
Refundable PDUFA fee |
|
|
(3,242 |
) |
|
|
— |
|
Prepaid expenses and other current assets |
|
|
2,529 |
|
|
|
(1,022 |
) |
Other assets |
|
|
330 |
|
|
|
(246 |
) |
Accounts payable |
|
|
(1,463 |
) |
|
|
(101 |
) |
Accrued expenses |
|
|
2,121 |
|
|
|
(951 |
) |
Other liabilities |
|
|
(258 |
) |
|
|
(307 |
) |
Cash used in operating activities |
|
|
(43,426 |
) |
|
|
(67,958 |
) |
Investing activities: |
|
|
|
|
|
|
||
Purchases of short-term investments |
|
|
(59,891 |
) |
|
|
(71,979 |
) |
Proceeds from sales of short-term investments |
|
|
83,910 |
|
|
|
77,500 |
|
Purchase of property and equipment |
|
|
(25 |
) |
|
|
(283 |
) |
Cash provided by investing activities |
|
|
23,994 |
|
|
|
5,238 |
|
Financing activities: |
|
|
|
|
|
|
||
Proceeds from royalty purchase agreement |
|
|
— |
|
|
|
57,500 |
|
Proceeds from the sale of common stock, net of issuance costs |
|
|
3,808 |
|
|
|
7,941 |
|
Proceeds from exercise of stock options |
|
|
81 |
|
|
|
1,266 |
|
Cash provided by financing activities |
|
|
3,889 |
|
|
|
66,707 |
|
Net increase (decrease) in cash, cash equivalents and restricted cash |
|
|
(15,543 |
) |
|
|
3,987 |
|
Cash, cash equivalents and restricted cash at beginning of year |
|
|
19,859 |
|
|
|
15,872 |
|
Cash, cash equivalents and restricted cash at end of year |
|
$ |
4,316 |
|
|
$ |
19,859 |
|
Supplemental schedule of non-cash investing and financing activities: |
|
|
|
|
|
|
||
Purchase of property and equipment included in accounts payable and |
|
$ |
— |
|
|
$ |
9 |
|
See accompanying notes to consolidated financial statements.
125
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
1. Organization and description of business
Galera Therapeutics, Inc. was incorporated as a Delaware corporation on November 19, 2012 (inception) and together with its subsidiaries (the Company, or Galera) is a clinical stage biopharmaceutical company focused on developing and commercializing a pipeline of novel, proprietary therapeutics that have the potential to transform radiotherapy in cancer. Galera's technology consists of selective small molecule dismutase mimetics that are in late-stage development in patients with cancer. Avasopasem manganese (avasopasem, or GC4419) is in development for radiotherapy-induced toxicities, including severe oral mucositis (SOM) in patients with locally advanced head and neck cancer (HNC) and esophagitis in patients with lung cancer. The Company is also exploring the potential for avasopasem to reduce cisplatin-induced kidney damage. In February 2018, the U.S. Food and Drug Administration (FDA) granted Breakthrough Therapy Designation to avasopasem for the reduction of SOM induced by radiotherapy. In February 2023, the FDA accepted and granted Priority Review designation to the Company's New Drug Application (NDA) for avasopasem for this indication. Galera’s second dismutase mimetic product candidate, rucosopasem manganese (rucosopasem, or GC4711), is in clinical-stage development to augment the anti-cancer efficacy of stereotactic body radiation therapy (SBRT) in patients with non-small cell lung cancer (NSCLC) and locally advanced pancreatic cancer (LAPC).
In December 2021, the Company announced corrected topline efficacy results from a Phase 3 trial (referred to as the ROMAN trial) evaluating avasopasem for the reduction of radiotherapy-induced SOM in patients with locally advanced HNC. The results demonstrated efficacy across multiple SOM endpoints with a statistically significant reduction on the primary endpoint of reduction in the incidence of SOM and a statistically significant reduction on the secondary endpoint of number of days of SOM, with a median of 18 days in the placebo arm versus 8 days in the avasopasem arm. The Company had previously announced topline results from the ROMAN trial in October 2021 that incorrectly stated the reduction on the primary endpoint was not statistically significant. Upon further analysis following the October topline data announcement, an error by the contract research organization was identified in the statistical program. Correction of this error yielded the correct, statistically significant p-values for the primary and a key secondary endpoint. The ROMAN trial is the Company’s second randomized trial conducted in patients with HNC to achieve statistical significance and demonstrate improved clinical benefit in reducing SOM. In December 2022, the Company submitted an NDA to the FDA for avasopasem for radiotherapy-induced SOM in patients with HNC undergoing standard-of-care treatment. The NDA is supported by the data from the two randomized, double-blinded, placebo-controlled trials (ROMAN and Phase 2b GT-201), as well as data from other clinical trials of avasopasem in the proposed indication. In February 2023, the FDA accepted the NDA and granted priority review with a Prescription Drug User Fee Act (PDUFA) target date of August 9, 2023. The FDA indicated in its acceptance of filing letter that it is not planning to hold an advisory committee meeting on the application.
In addition to developing avasopasem for the reduction of normal tissue toxicity from radiotherapy, the Company is developing its second dismutase mimetic product candidate, rucosopasem, to increase the anti-cancer efficacy of higher daily doses of radiotherapy, or SBRT. In September 2021, in support of rucosopasem, the Company announced final results from its Phase 1/2 pilot trial of avasopasem in combination with SBRT in patients with unresectable or borderline resectable LAPC. In this proof-of-concept trial, survival and tumor outcome benefits were observed. The Company used its observations from this pilot trial to inform the design of rucosopasem clinical trials in combination with SBRT. The Company has successfully completed Phase 1 trials of intravenous rucosopasem in healthy volunteers and is currently evaluating rucosopasem in combination with SBRT in a Phase 1/2 safety and anti-cancer efficacy trial in NSCLC and a Phase 2b trial of rucosopasem in combination with SBRT in patients with LAPC.
Liquidity
The Company has incurred recurring losses and negative cash flows from operations since inception and has an accumulated deficit of $378.3 million as of December 31, 2022. The Company anticipates incurring additional losses until such time, if ever, that it can generate significant sales of its product candidates currently in development. The Company follows the provisions of Financial Accounting Standards Board, or FASB, Accounting Standards Codification, or ASC, Topic 205-40, Presentation of Financial Statements—Going Concern, which
126
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
requires management to assess the Company’s ability to continue as a going concern for one year after the date the financial statements are issued. The Company expects its existing cash, cash equivalents and short-term investments as of December 31, 2022, together with the net proceeds from its February 2023 registered direct offering, will enable the Company to fund its operating expenses and capital expenditure requirements into the fourth quarter of 2023, but not for more than one year after the date these consolidated financial statements were issued, and therefore management has concluded that substantial doubt exists about the Company’s ability to continue as a going concern. Management’s plans to mitigate this risk include raising additional capital through equity or debt financings, or through strategic transactions. Management’s plans may also include the deferral of certain operating expenses unless and until additional capital is received. However, there can be no assurance that the Company will be successful in raising additional capital or that such capital, if available, will be on terms that are acceptable to the Company, or that the Company will be successful in deferring certain operating expenses. If the Company is unable to raise sufficient additional capital or defer sufficient operating expenses, the Company may be compelled to reduce the scope of its operations and planned capital expenditures. In the future, if the Company is not able to continue to raise sufficient capital to fund its operations, the Company may decide to delay or discontinue certain activities, including planned research and development activities, hiring plans, manufacturing activities and commercial preparation efforts. The consolidated financial statements have been prepared assuming the Company will continue as a going concern, which contemplates the continuity of operations, the realization of assets and the satisfaction of liabilities and commitments in the normal course of business.
In December 2020, the Company filed a registration statement with the Securities and Exchange Commission (SEC) which covers the offering, issuance and sale of up to $200.0 million in Company securities, which includes an Open Market Sale Agreement with Jefferies LLC (the Sales Agreement) covering the offering, issuance and sale of up to a maximum aggregate offering price of $50.0 million of the Company’s common stock, which could be utilized to raise funding for future operating expenses and capital expenditure requirements. During the year ended December 31, 2022, the Company sold approximately 2.0 million shares of common stock and received net proceeds of $3.8 million pursuant to the Sales Agreement. As of December 31, 2022, there remained $37.8 million available under the Sales Agreement.
On February 17, 2023, the Company completed a registered direct offering, which resulted in the issuance and sale of 14,320,000 shares of its common stock and warrants to purchase up to 14,320,000 shares of common stock at a combined offering price of $2.095 per share and accompanying warrant, generating gross proceeds of $30.0 million. The warrants have an exercise price of $1.97 per share of common stock, are exercisable immediately following their issuance and will expire five years from the date of issuance. The Company received net proceeds of approximately $27.7 million from this offering, after deducting placement agent fees and offering expenses.
2. Basis of presentation and significant accounting policies
Basis of presentation and consolidation
The accompanying consolidated financial statements have been prepared in conformity with U.S. generally accepted accounting principles (U.S. GAAP). Any reference in these notes to applicable guidance is meant to refer to U.S. GAAP as found in the Accounting Standards Codification (ASC) and Accounting Standards Updates (ASU) of the Financial Accounting Standards Board (FASB).
The consolidated financial statements include the accounts of Galera Therapeutics, Inc. and its wholly owned subsidiaries, Galera Therapeutics Australia Pty Ltd (Galera Australia) and Galera Labs, LLC. All intercompany accounts and transactions have been eliminated in consolidation.
The Company has determined the functional currency of Galera Australia to be the U.S. dollar. The Company records remeasurement gains and losses on monetary assets and liabilities, such as accounts payable, which are not denominated in U.S. dollars in the statements of operations.
The Company manages its operations as a single reportable segment for the purposes of assessing performance and making operating decisions.
127
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Use of estimates
The preparation of consolidated financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities as of the date of the consolidated financial statements and the reported amounts of expenses during the reporting period. Actual results could differ from those estimates.
Estimates and assumptions are periodically reviewed and the effects of revisions are reflected in the consolidated financial statements in the period they are determined to be necessary. Significant areas that require management’s estimates include the share-based compensation assumptions, royalty purchase liability assumptions and accrued research and development expenses.
Fair value of financial instruments
Management believes that the carrying amounts of the Company’s financial instruments, including accounts payable and accrued expenses, approximate fair value due to the short-term nature of those instruments. Short-term investments are recorded at their estimated fair value. The royalty purchase liability is accounted for as debt and interest is accreted over the expected repayment period which approximates fair value.
Concentration of credit risk
Financial instruments that potentially subject the Company to significant concentrations of credit risk consist primarily of cash and cash equivalents and short-term investments. The Company maintains deposits in federally insured financial institutions in excess of federally insured limits. The Company has not experienced any losses in such accounts and believes it is not exposed to significant risk on its cash and cash equivalents.
Cash and cash equivalents
The Company considers all highly liquid investments that have maturities of three months or less when acquired to be cash equivalents. Cash and cash equivalents as of December 31, 2022 and 2021 consisted of bank deposits, U.S. Treasury obligations and a money market mutual fund invested in U.S. Treasury obligations.
Restricted cash
Restricted cash represents collateral provided under a commercial credit card agreement entered into with TD Bank, N.A. during July 2022. Restricted cash was $50,000 as of December 31, 2022. The Company has recorded this deposit and accumulated interest thereon as restricted cash on its consolidated balance sheet.
Refundable PDUFA fee
In December 2022, the Company paid a $3.2 million PDUFA fee to the FDA in conjunction with the filing of its NDA for avasopasem. The Company requested and has been granted a small business waiver of this PDUFA fee from the FDA, and the amount has been recorded as a Refundable PDUFA fee on the Company’s consolidated balance sheet, as a refund is expected in 2023.
Short-term investments
Short-term investments consist of debt securities with a maturity of greater than three months when acquired. The Company classifies its short-term investments at the time of purchase as available-for-sale securities, which are net of unrealized losses of $8,000 and $26,000 during the years ended December 31, 2022 and 2021, respectively. Available-for-sale securities are carried at fair value. Unrealized gains and losses on available-for-sale securities are reported in accumulated other comprehensive income (loss), a component of stockholders’ equity (deficit), until realized.
128
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Property and equipment
Property and equipment are recorded at cost and depreciated using the straight-line method over their estimated useful lives ranging from to five years. Leasehold improvements are amortized over the shorter of their economic lives or the remaining lease term. The costs of maintenance and repairs are expensed as incurred. Improvements and betterments that add new functionality or extend the useful life of the asset are capitalized.
Impairment of long-lived assets
Long-lived assets are reviewed for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. Recoverability of assets to be held and used is measured by a comparison of the carrying amount of an asset to estimated undiscounted future cash flows expected to be generated by the asset. If the carrying amount of an asset exceeds its estimated undiscounted future cash flows, then an impairment charge is recognized for the amount by which the carrying value of the asset exceeds the estimated fair value of the asset. As of December 31, 2022, the Company believes that no revision of the remaining useful lives or write-down of long-lived assets is required.
Goodwill and acquired intangible asset
In November 2012, the Company completed a Series A redeemable convertible preferred stock (Series A) financing with venture capital investors and simultaneously acquired Galera Therapeutics, LLC (LLC), a limited liability company incorporated in Missouri in 2009. LLC was renamed Galera Labs, LLC in January 2013 and operates as a wholly-owned subsidiary of the Company. The Company applied the purchase method of accounting under which the consideration given to the LLC members and noteholders was allocated to the fair value of the net assets assumed from the LLC at the date of the acquisition. The sole intangible asset acquired represented the fair value of in-process research and development (IPR&D) which has been recorded on the accompanying consolidated balance sheets as an indefinite life intangible asset. A deferred tax liability was recorded for the difference between the fair value of the acquired IPR&D and its tax basis of zero which was recognized as goodwill in applying the purchase method of accounting.
Intangible assets related to IPR&D are considered indefinite-lived intangible assets and, along with goodwill, are not amortized, but are assessed for impairment annually or more frequently if impairment indicators exist. For those compounds that reach commercialization, the IPR&D assets will be amortized over their estimated useful lives. If the associated research and development effort related to IPR&D is abandoned, the related assets will be written-off and the Company will record a noncash impairment loss on its consolidated statements of operations. For the years ended December 31, 2022 and 2021, the Company determined that there was no impairment to goodwill or IPR&D.
Royalty purchase liability
In November 2018, the Company entered into an Amended and Restated Purchase and Sale Agreement (the Royalty Agreement), with Clarus IV Galera Royalty AIV, L.P., Clarus IV-A, L.P., Clarus IV-B, L.P., Clarus IV-C, L.P. and Clarus IV-D, L.P. (collectively, Blackstone or Blackstone Life Sciences). Pursuant to the Royalty Agreement, Blackstone agreed to pay up to $80.0 million (the Royalty Purchase Price) in four tranches of $20.0 million each upon the achievement of specific Phase 3 clinical trial patient enrollment milestones. The Company received the first tranche of the Royalty Purchase Price in November 2018, the second tranche of the Royalty Purchase Price in April 2019, and the third tranche of the Royalty Purchase Price in February 2020, in each case in connection with the achievement of the first three milestones, respectively. The proceeds received have been recorded as long-term debt obligations. Interest expense on such obligation is imputed by estimating risk adjusted future royalty payments over the term of the Royalty Agreement which takes into consideration the probability of obtaining FDA approval. Other significant assumptions include adjustments to estimated gross revenues to arrive at net product sales from which a royalty payment can be estimated. The non-cash interest expense recorded increases the balance of the royalty obligation. The royalty obligation will be reduced when royalty payments are made, if any.
129
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
In May 2020, the Company entered into Amendment No. 1 to the Royalty Agreement (the Amendment) with Clarus IV Galera Royalty AIV, L.P. (the Blackstone Purchaser). The Blackstone Purchaser is affiliated with Blackstone Life Sciences, the successor in interest to Clarus Ventures. The Amendment increased the Royalty Purchase Price by $37.5 million to $117.5 million, by increasing the fourth tranche from $20.0 million to $37.5 million, which was received in July 2021, and adding a new $20.0 million tranche upon the achievement of an additional clinical enrollment milestone, which was received in June 2021. The Company accounted for the Amendment as a debt modification and is amortizing fees paid to the Blackstone Purchaser related to the Amendment over the estimated term of the royalty purchase liability utilizing the effective-interest method.
Actual royalty payments are highly uncertain and may change depending on a number of factors, including the Company’s ability to obtain FDA approval, successfully commercialize the Company’s product candidates and the timing of future royalty payments. The Company imputes interest expense on the royalty purchase obligations based on such factors at each reporting period. As these factors change, the Company will adjust its estimate of the imputed interest expense accordingly.
Leases
At lease commencement, the Company records a lease liability based on the present value of lease payments over the expected lease term including any options to extend the lease that the Company is reasonably certain to exercise. The Company calculates the present value of lease payments using an incremental borrowing rate as the Company’s leases do not provide an implicit interest rate. The Company’s incremental borrowing rate for a lease is the rate of interest it would have to pay on a collateralized basis to borrow an amount equal to the lease payments under similar terms. At the lease commencement date, the Company records a corresponding right-of-use lease asset based on the lease liability, adjusted for any lease incentives received and any initial direct costs paid to the lessor prior to the lease commencement date. The Company may enter into leases with an initial term of 12 months or less (short-term leases). For short-term leases, the Company records the rent expense on a straight-line basis and does not record the leases on the balance sheet. The Company had no short-term leases as of December 31, 2022 and 2021.
After lease commencement, the Company measures its leases as follows: (i) the lease liability based on the present value of the remaining lease payments using the discount rate determined at lease commencement, and (ii) the right-of-use lease asset based on the remeasured lease liability, adjusted for any unamortized lease incentives received, any unamortized initial direct costs and the cumulative difference between rent expense and amounts paid under the lease agreement. Any lease incentives received and any initial direct costs are amortized on a straight-line basis over the expected lease term. Rent expense is recorded on a straight-line basis over the expected lease term.
Research and development expenses
Research and development costs are expensed as incurred and consist primarily of funds paid to third parties for the provision of services for product candidate development, clinical and preclinical development and related supply and manufacturing costs, and regulatory compliance costs. The Company accrues and expenses preclinical studies and clinical trial activities performed by third parties based upon estimates of the proportion of work completed over the term of the individual trial and patient enrollment rates in accordance with agreements with clinical research organizations and clinical trial sites. The Company determines the estimates by reviewing contracts, vendor agreements and purchase orders, and through discussions with internal clinical personnel and external service providers as to the progress or stage of completion of trials or services and the agreed-upon fee to be paid for such services. However, actual costs and timing of clinical trials are highly uncertain, subject to risks and may change depending upon a number of factors, including the Company’s clinical development plan.
Management makes estimates of the Company’s accrued expenses as of each balance sheet date in the Company’s consolidated financial statements based on facts and circumstances known to the Company at that time. If the actual timing of the performance of services or the level of effort varies from the estimate, the Company will adjust the accrual accordingly. Nonrefundable advance payments for goods and services, including fees for process development or manufacturing and distribution of clinical supplies that will be used in future research and development activities, are deferred and recognized as expense in the period that the related goods are consumed or services are performed.
130
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
In September 2020, the Company was awarded a Small Business Innovation Research grant from the National Cancer Institute of the National Institutes of Health, which will partially fund its Phase 1/2 safety and anti-cancer efficacy trial in NSCLC (the Grant). Costs entitled to reimbursement under the Grant are accounted for as a reduction to research and development expenses. During the year ended December 31, 2021, the Company recorded a reduction to research and development expense of $0.4 million for expenses for which it has been reimbursed under the Grant. The Company has fully utilized the $1.1 million of available funding under the Grant and did not receive any reimbursement during the year ended December 31, 2022.
Share-based compensation
The Company measures share-based awards at their grant-date fair value and records compensation expense on a straight-line basis over the vesting period of the awards.
Estimating the fair value of share-based awards requires the input of subjective assumptions, including the expected life of the options and stock price volatility. The Company accounts for forfeitures of stock option awards as they occur. The Company uses the Black-Scholes option pricing model to value its stock option awards. The assumptions used in estimating the fair value of share-based awards represent management’s estimate and involve inherent uncertainties and the application of management’s judgment. As a result, if factors change and management uses different assumptions, share-based compensation expense could be materially different for future awards.
The expected life of the stock options is estimated using the “simplified method,” as the Company has limited historical information from which to develop reasonable expectations about future exercise patterns and post-vesting employment termination behavior for its stock option grants. The simplified method is the midpoint between the vesting period and the contractual term of the option. For stock price volatility, the Company uses comparable public companies as a basis for its expected volatility to calculate the fair value of option grants. The risk-free rate is based on the U.S. Treasury yield curve commensurate with the expected life of the option.
Employee Benefit Plan
The Company sponsors a 401(k) defined contribution plan for its employees. Employee contributions are voluntary. The Company matches employee contributions in an amount equal to 100% of the first 4% of eligible compensation, and such employer contributions are immediately vested. During the year ended December 31, 2022 and 2021, the Company provided matching contributions of $0.3 million and $0.3 million, respectively.
Income taxes
The Company uses the asset and liability method of accounting for income taxes. Deferred tax assets and liabilities are recognized for the estimated future tax consequences attributable to differences between the financial statement carrying amounts of existing assets and liabilities and their respective tax bases, and operating loss and credit carryforwards. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. A valuation allowance is provided when it is more likely than not that some portion or all of a deferred tax asset will not be realized. The Company recognizes the benefit of an uncertain tax position that it has taken or expects to take on its income tax return if such a position is more likely than not to be sustained.
Net loss per share
Basic loss per share of common stock is computed by dividing net loss by the weighted-average number of shares of common stock outstanding during each period. Diluted loss per share of common stock includes the effect, if any, from the potential exercise or conversion of securities, such as stock options and common stock warrants, which would result in the issuance of incremental shares of common stock. For diluted net loss per share, the weighted-average number of shares of common stock is the same for basic net loss per share due to the fact that when a net loss exists, dilutive securities are not included in the calculation as the impact is anti-dilutive.
131
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
The following potentially dilutive securities have been excluded from the computation of diluted weighted-average shares of common stock outstanding, as they would be anti-dilutive:
|
|
December 31, |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Stock options |
|
|
5,783,185 |
|
|
|
4,970,975 |
|
Common stock warrants |
|
|
550,661 |
|
|
|
550,661 |
|
|
|
|
6,333,846 |
|
|
|
5,521,636 |
|
3. Fair value measurements
The Company utilizes valuation techniques that maximize the use of observable inputs and minimize the use of unobservable inputs to the extent possible. The Company determines fair value based on assumptions that market participants would use in pricing an asset or liability in the principal or most advantageous market. When considering market participant assumptions in fair value measurements, the following fair value hierarchy distinguishes between observable and unobservable inputs, which are categorized in one of the following levels:
The following table presents the Company’s assets and liabilities that are measured at fair value on a recurring basis (amounts in thousands):
|
|
December 31, 2022 |
|
|||||||||
|
|
(Level 1) |
|
|
(Level 2) |
|
|
(Level 3) |
|
|||
Assets |
|
|
|
|
|
|
|
|
|
|||
Money market funds and U.S. Treasury obligations |
|
$ |
3,467 |
|
|
$ |
— |
|
|
$ |
— |
|
|
|
|
|
|
|
|
|
|
|
|||
Short-term investments |
|
|
|
|
|
|
|
|
|
|||
U.S. government agency securities |
|
$ |
— |
|
|
$ |
8,172 |
|
|
$ |
— |
|
U.S. Treasury obligations |
|
|
19,159 |
|
|
|
— |
|
|
|
— |
|
Total short-term investments |
|
$ |
19,159 |
|
|
$ |
8,172 |
|
|
$ |
— |
|
|
|
December 31, 2021 |
|
|||||||||
|
|
(Level 1) |
|
|
(Level 2) |
|
|
(Level 3) |
|
|||
Assets |
|
|
|
|
|
|
|
|
|
|||
Money market funds and U.S. Treasury obligations |
|
$ |
12,346 |
|
|
$ |
— |
|
|
$ |
— |
|
|
|
|
|
|
|
|
|
|
|
|||
Short-term investments |
|
|
|
|
|
|
|
|
|
|||
U.S. government agency securities |
|
$ |
— |
|
|
$ |
5,413 |
|
|
$ |
— |
|
U.S. Treasury obligations |
|
|
45,945 |
|
|
|
— |
|
|
|
— |
|
Total short-term investments |
|
$ |
45,945 |
|
|
$ |
5,413 |
|
|
$ |
— |
|
132
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
There were no changes in valuation techniques during the years ended December 31, 2022 and 2021. The Company’s short-term investment instruments classified using Level 1 inputs within the fair value hierarchy are classified as such because they are valued using quoted market prices, broker or dealer quotations, or alternative pricing sources with reasonable levels of price transparency. The fair value of Level 2 securities is estimated based on observable inputs other than quoted prices in active markets for identical assets and liabilities, quoted prices for identical or similar assets or liabilities in inactive markets, or other inputs that are observable or can be corroborated by observable market data for substantially the full term on the assets or liabilities.
4. Property and equipment
Property and equipment consist of (amounts in thousands):
|
|
December 31, |
|
|
December 31, |
|
||
|
|
2022 |
|
|
2021 |
|
||
Laboratory equipment |
|
$ |
1,398 |
|
|
$ |
1,379 |
|
Computer hardware and software |
|
|
292 |
|
|
|
292 |
|
Leasehold improvements |
|
|
270 |
|
|
|
264 |
|
Furniture and fixtures |
|
|
179 |
|
|
|
179 |
|
Property and equipment, gross |
|
|
2,139 |
|
|
|
2,114 |
|
Less: Accumulated depreciation and amortization |
|
|
(1,701 |
) |
|
|
(1,587 |
) |
Property and equipment, net |
|
$ |
438 |
|
|
$ |
527 |
|
Depreciation and amortization expense was $0.1 million and $0.8 million for the years ended December 31, 2022 and 2021, respectively.
5. Accrued expenses
Accrued expenses consist of (amounts in thousands):
|
|
December 31, |
|
|
December 31, |
|
||
|
|
2022 |
|
|
2021 |
|
||
Compensation and related benefits |
|
$ |
2,655 |
|
|
$ |
2,038 |
|
Research and development expenses |
|
|
6,764 |
|
|
|
5,360 |
|
Professional fees and other expenses |
|
|
335 |
|
|
|
235 |
|
|
|
$ |
9,754 |
|
|
$ |
7,633 |
|
6. Royalty purchase liability
Pursuant to the Royalty Agreement, Blackstone agreed to pay up to $80.0 million (the Royalty Purchase Price) in four tranches of $20.0 million each upon the achievement of specific Phase 3 clinical trial patient enrollment milestones. The Company received the first tranche of the Royalty Purchase Price in November 2018, the second tranche of the Royalty Purchase Price in April 2019, and the third tranche of the Royalty Purchase Price in February 2020, in each case in connection with the achievement of the first three milestones, respectively.
In May 2020, the Company entered into Amendment No. 1 to the Royalty Agreement (the Amendment) with Clarus IV Galera Royalty AIV, L.P. (the Blackstone Purchaser). The Blackstone Purchaser is affiliated with Blackstone Life Sciences, the successor in interest to Clarus Ventures. The Amendment increased the Royalty Purchase Price by $37.5 million, to $117.5 million by increasing the fourth tranche from $20.0 million to $37.5 million and adding a new $20.0 million tranche upon the achievement of an additional clinical enrollment milestone. The Company accounted for the Amendment as a debt modification and is amortizing fees paid to the Blackstone Purchaser related to the Amendment over the estimated term of the royalty purchase liability utilizing the effective-interest method. In June 2021, the Company received the new tranche ($20.0 million) under the Amendment in connection with the enrollment of the first patient in a Phase 2b trial of rucosopasem in combination with SBRT in patients with locally advanced pancreatic cancer, which the Company refers to as the GRECO-2 trial. Also in June
133
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
2021, the Company completed enrollment in the ROMAN trial, thereby achieving the milestone associated with the fourth tranche ($37.5 million) under the Amendment, which was received in July 2021.
The Company accounts for the Royalty Agreement as a debt instrument. The $117.5 million in proceeds received as of December 31, 2022 have been recorded as a liability on the accompanying consolidated balance sheets. Interest expense is imputed based on the estimated royalty repayment period described below, which takes into consideration the probability and timing of obtaining FDA approval and the potential future revenue from commercializing its product candidates, and which results in a corresponding increase in the liability balance. As a result of the NDA submission and market research conducted in 2022 to estimate the potential opportunity for avasopasem for radiation-induced SOM among key stakeholders, the Company updated the assumptions underlying the calculation of interest expense on the royalty purchase liability. The Company recognized $11.6 million and $7.2 million in noncash interest expense during the years ended December 31, 2022 and 2021, respectively. As of December 31, 2022, the effective interest rate was 9.6%.
Pursuant to the Royalty Agreement and the Amendment, in connection with the payment of each tranche of the Royalty Purchase Price, the Company has agreed to sell, convey, transfer and assign to Blackstone all of its right, title and interest in a high single-digit percentage of (i) worldwide net sales of avasopasem and rucosopasem (collectively, the Products) and (ii) all amounts received by the Company or its affiliates, licensees and sublicensees with respect to Product-related damages (collectively, the Product Payments) during the Royalty Period. The Royalty Period means, on a Product-by-Product and country-by-country basis, the period of time commencing on the commercial launch of such Product in such country and ending on the latest to occur of (i) the 12th anniversary of such commercial launch, (ii) the expiration of all valid claims of the Company’s patents covering such Product in such country, and (iii) the expiration of regulatory data protection or market exclusivity or similar regulatory protection afforded by the health authorities in such country, to the extent such protection or exclusivity effectively prevents generic versions of such Product from entering the market in such country.
The Royalty Agreement and the Amendment will remain in effect until the date on which the aggregate amount of the Product Payments paid to Blackstone exceeds a fixed single-digit multiple of the actual amount of the Royalty Purchase Price received by the Company, unless earlier terminated pursuant to the mutual written agreement of the Company and Blackstone. If no Products are commercialized, the Company would not have an obligation to make Product Payments to Blackstone, which is the sole mechanism for repaying the liability.
Upon execution of the Amendment, the Company issued common stock warrants to the Blackstone Purchaser, each of which became exercisable upon the receipt by the Company of the applicable specified milestone payment. The issued warrants expire six years after the initial exercise dates, as follows:
|
|
Shares |
|
|
Exercise Price |
|
|
Initial Exercise Date |
|
Expiration Date |
||
New Milestone Warrant |
|
|
293,686 |
|
|
$ |
13.62 |
|
|
6/7/2021 |
|
6/6/2027 |
Fourth Milestone Warrant |
|
|
256,975 |
|
|
$ |
13.62 |
|
|
7/19/2021 |
|
7/18/2027 |
The warrants are equity-classified and were valued at $4.7 million at the time of issuance using the Black-Scholes option pricing model. The warrants were recorded as a discount to the royalty purchase liability. The Company amortizes the debt discount to interest expense over the estimated term of the royalty purchase liability utilizing the effective-interest method.
7. Leases
The Company had a non-cancelable operating lease for office and laboratory space in Malvern, Pennsylvania which ended in February 2023. The discount rate used to account for the Company’s operating lease is the Company’s estimated incremental borrowing rate of 5.3%.
134
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Supplemental balance sheet information related to leases was as follows:
|
|
December 31, |
|
|
December 31, |
|
||
|
|
2022 |
|
|
2021 |
|
||
Operating Leases |
|
|
|
|
|
|
||
Right-of-use lease assets |
|
$ |
43 |
|
|
$ |
296 |
|
|
|
|
|
|
|
|
||
Lease liabilities, current |
|
|
44 |
|
|
|
258 |
|
Lease liabilities, net of current portion |
|
|
— |
|
|
|
44 |
|
Total operating lease liabilities |
|
$ |
44 |
|
|
$ |
302 |
|
|
|
|
|
|
|
|
||
The components of lease expense were as follows:
|
|
Year ended |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Operating lease costs |
|
|
|
|
|
|
||
Operating lease rental expense |
|
$ |
259 |
|
|
$ |
303 |
|
Interest on lease liabilities |
|
|
8 |
|
|
|
22 |
|
Total operating lease expense |
|
$ |
267 |
|
|
$ |
325 |
|
Supplemental cash flow information related to leases was as follows:
|
|
Year ended |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Cash paid for amounts included in the measurement of lease liabilities |
|
|
|
|
|
|
||
Operating cash flows for operating leases |
|
$ |
266 |
|
|
$ |
325 |
|
Right-of-use assets obtained in exchange for lease obligation |
|
|
|
|
|
|
||
Operating leases |
|
|
— |
|
|
|
70 |
|
Our operating lease liabilities as of December 31, 2022 will mature, as follows (amounts in thousands):
2023 |
|
|
44 |
|
Total |
|
|
44 |
|
Less: imputed interest |
|
|
0 |
|
|
|
$ |
44 |
|
Additionally, in October 2022 the Company entered into a new operating lease agreement for office space in Malvern, Pennsylvania. The lease commencement date is expected to occur in May 2023. The lease expiration date will be 7.4 years after the lease commencement date. Total rental payments are approximately $1.6 million from the commencement date through the expiration date. The Company is also required to pay the increase in certain operating expenses over a base year, in accordance with the terms of the lease agreement.
8. Commitments
Executive employment agreements
The Company has entered into employment agreements with certain key executives, providing for compensation and severance in certain circumstances, as described in the respective agreements.
Legal matters
The Company is subject from time to time to various claims and legal actions arising during the ordinary course of its business. Management believes that there are currently no claims or legal actions that would reasonably be expected to have a material adverse effect on the Company’s results of operations, financial condition or cash flows.
135
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
9. Stockholders' Equity (Deficit)
Equity offerings
In December 2020, the Company entered into the Sales Agreement with Jefferies LLC (Jefferies) as sales agent, pursuant to which it may, from time to time, issue and sell common stock with an aggregate value of up to $50.0 million in “at-the-market” (ATM) offerings under the Company’s Registration Statement on Form S-3 (File No. 333-251061) filed with the SEC on December 1, 2020. Sales of common stock, if any, pursuant to the Sales Agreement, may be made in sales deemed to be an “at the market offering” as defined in Rule 415(a) of the Securities Act, including sales made directly through the Nasdaq Global Market or on any other existing trading market for the Company’s common stock. The Company is required to pay Jefferies a commission equal to three percent of the gross sales proceeds and has provided Jefferies with customary indemnification rights. During the year ended December 31, 2022, 1,982,773 shares were sold under the Sales Agreement at a weighted average price per share of $2.00. Net proceeds to the Company after deducting fees, commissions and other expenses related to the offering were $3.8 million for the year ended December 31, 2022. As of December 31, 2022, there was $37.8 million of available capacity under the Sales Agreement.
Share-based compensation
In connection with the Company’s Initial Public Offering, or IPO, in November 2019, the Company’s board of directors adopted and the Company’s stockholders approved the Galera Therapeutics, Inc. 2019 Incentive Award Plan (the 2019 Plan), which became effective upon the effectiveness of the registration statement on Form S-1 for the IPO. Upon effectiveness of the 2019 Plan, the Company ceased granting new awards under the Prior Plan (as defined herein).
The 2019 Plan provides for the grant of incentive stock options, nonstatutory stock options, restricted stock awards, restricted stock units, stock appreciation rights and other stock-based awards. The number of shares of common stock initially available for issuance under the 2019 Plan was 1,948,970 shares of common stock plus the number of shares subject to awards outstanding under the Prior Plan that expire, terminate or are otherwise surrendered, cancelled, forfeited or repurchased by the Company on or after the effective date of the 2019 Plan. In addition, the number of shares of common stock available for issuance under the 2019 Plan is subject to an annual increase on the first day of each calendar year beginning on January 1, 2020 and ending on and including January 1, 2029 equal to the lesser of (i) 4% of the Company’s outstanding shares of common stock on the final day of the immediately preceding calendar year, and (ii) such smaller number of shares of common stock as determined by the Company’s board of directors. As of December 31, 2022, there were 1,429,544 shares available for future issuance under the 2019 Plan, including 1,058,350 shares added pursuant to this provision effective January 1, 2022. Pursuant to this provision, the Company added an additional 1,140,402 shares to the total shares available for issuance under the 2019 Plan effective January 1, 2023. The maximum number of shares of common stock that may be issued under the 2019 Plan upon the exercise of incentive stock options is 14,130,029.
In November 2019, the Company’s board of directors adopted and the Company’s stockholders approved the Galera Therapeutics, Inc. 2019 Employee Stock Purchase Plan (the ESPP). The ESPP allows employees to buy Company stock through after-tax payroll deductions at a discount from market value. The number of shares of common stock initially available for issuance under the ESPP was 243,621 shares of common stock. In addition, the number of shares of common stock available for issuance under the ESPP is subject to an annual increase on the first day of each calendar year beginning on January 1, 2020 and ending on and including January 1, 2029 equal to the lesser of (i) 1% of the Company’s outstanding shares of common stock on the final day of the immediately preceding calendar year and (ii) such smaller number of shares of common stock as determined by the Company’s board of directors, provided that not more than 3,288,886 shares of common stock may be issued under the ESPP. As of December 31, 2022, there were 1,006,084 shares available for issuance under the ESPP, including 264,587 shares added pursuant to this provision effective January 1, 2022. Pursuant to this provision, the Company added an additional 285,100 shares to the total shares available for issuance under the ESPP effective January 1, 2023.
In November 2012, the Company adopted the Equity Incentive Plan (the Prior Plan). The total number of shares subject to outstanding awards under the Prior Plan as of December 31, 2022 was 1,888,574. No shares
136
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
remain available for issuance under the Prior Plan and no further grants will be made under the Prior Plan; however, the Prior Plan continues to govern awards that are outstanding under it.
The Company's stock option awards vest based on the terms in the governing agreements and generally vest over four years and have a term of 10 years.
Share-based compensation expense was as follows for the years ended December 31, 2022 and 2021 (in thousands):
|
|
|
Year ended |
|
|||||
|
|
|
2022 |
|
|
2021 |
|
||
Research and development |
|
|
$ |
2,596 |
|
|
$ |
2,919 |
|
General and administrative |
|
|
|
4,568 |
|
|
|
4,312 |
|
|
|
|
$ |
7,164 |
|
|
$ |
7,231 |
|
The following table summarizes the activity related to stock option grants for the year ended December 31, 2022:
|
|
Shares |
|
|
Weighted |
|
|
Weighted- |
|
|||
Outstanding at January 1, 2022 |
|
|
4,970,975 |
|
|
$ |
8.45 |
|
|
7.1 |
|
|
Granted |
|
|
1,608,646 |
|
|
|
2.05 |
|
|
|
|
|
Exercised |
|
|
(68,526 |
) |
|
|
1.20 |
|
|
|
|
|
Forfeited |
|
|
(727,910 |
) |
|
|
7.63 |
|
|
|
|
|
Outstanding at December 31, 2022 |
|
|
5,783,185 |
|
|
$ |
6.86 |
|
|
|
6.8 |
|
Vested and exercisable at December 31, 2022 |
|
|
3,593,449 |
|
|
$ |
7.41 |
|
|
|
5.7 |
|
Vested and expected to vest at December 31, 2022 |
|
|
5,783,185 |
|
|
$ |
6.86 |
|
|
|
6.8 |
|
As of December 31, 2022, the unrecognized compensation cost was $9.5 million and will be recognized over an estimated weighted-average remaining amortization period of 2.1 years. The aggregate intrinsic value of options outstanding and options exercisable as of December 31, 2022 was $83,000 and $34,000, respectively. Options granted during the years ended December 31, 2022 and 2021 had weighted-average grant-date fair values of $1.57 and $7.73 per share, respectively.
The fair value of options is estimated using the Black-Scholes option pricing model, which takes into account inputs such as the exercise price, the estimated fair value of the underlying common stock at the grant date, expected term, expected stock price volatility, risk-free interest rate and dividend yield. The fair value of stock options granted during the years ended December 31, 2022 and 2021 was determined using the methods and assumptions discussed below.
137
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
The grant date fair value of each option grant was estimated throughout the year using the Black-Scholes option-pricing model using the following weighted-average assumptions:
|
|
Year ended |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Expected term (in years) |
|
|
6.2 |
|
|
|
6.2 |
|
Expected stock price volatility |
|
|
92.7 |
% |
|
|
89.5 |
% |
Risk-free interest rate |
|
|
2.07 |
% |
|
|
0.79 |
% |
Expected dividend yield |
|
|
0 |
% |
|
|
0 |
% |
10. Income Taxes
The Company’s loss before income taxes for the years ended December 31, 2022 and 2021 is as follows (in thousands):
|
|
Year ended December 31, |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Domestic |
|
$ |
(62,281 |
) |
|
$ |
(80,587 |
) |
Foreign |
|
|
(11 |
) |
|
|
53 |
|
|
|
$ |
(62,292 |
) |
|
$ |
(80,534 |
) |
The Company’s tax benefit for the years ended December 31, 2022 and 2021 is summarized as follows (in thousands):
|
|
Year ended December 31, |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Current |
|
|
|
|
|
|
||
Federal |
|
$ |
— |
|
|
$ |
— |
|
State |
|
|
— |
|
|
|
— |
|
Foreign |
|
|
— |
|
|
|
— |
|
|
|
|
— |
|
|
|
— |
|
Deferred: |
|
|
|
|
|
|
||
Federal |
|
|
— |
|
|
|
— |
|
State |
|
|
(70 |
) |
|
|
— |
|
Foreign |
|
|
— |
|
|
|
— |
|
|
|
|
(70 |
) |
|
|
— |
|
Total income tax benefit |
|
$ |
(70 |
) |
|
$ |
— |
|
138
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
A reconciliation of the federal income tax rate to the Company’s effective tax rate is as follows:
|
|
Year ended December 31, |
|
|
|||||
|
|
2022 |
|
|
2021 |
|
|
||
Rate reconciliation: |
|
|
|
|
|
|
|
||
Federal tax benefit at statutory rate |
|
|
21.0 |
|
% |
|
21.0 |
|
% |
State tax, net of federal benefit |
|
|
3.1 |
|
|
|
1.5 |
|
|
Net operating loss carryforwards |
|
|
0.1 |
|
|
|
— |
|
|
Change in tax rate |
|
|
(9.6 |
) |
|
|
— |
|
|
Sale of royalty interest |
|
|
(3.9 |
) |
|
|
(16.9 |
) |
|
Research and development |
|
|
(0.8 |
) |
|
|
0.4 |
|
|
Change in valuation allowance |
|
|
(9.1 |
) |
|
|
(5.6 |
) |
|
Share-based compensation |
|
|
(0.6 |
) |
|
|
(0.3 |
) |
|
Other |
|
|
(0.1 |
) |
|
|
(0.1 |
) |
|
Total provision |
|
|
0.1 |
|
% |
|
— |
|
% |
The tax effects of temporary differences that give rise to significant portions of the deferred tax assets and liabilities were as follows (in thousands):
|
|
December 31, |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Deferred tax assets |
|
|
|
|
|
|
||
Net operating loss carryforwards |
|
$ |
41,697 |
|
|
$ |
43,111 |
|
Share-based compensation |
|
|
3,731 |
|
|
|
2,874 |
|
Research and development credits |
|
|
7,287 |
|
|
|
7,827 |
|
Capitalized research and development expenses |
|
|
6,354 |
|
|
|
— |
|
Accrued expenses and other |
|
|
82 |
|
|
|
77 |
|
Gross deferred tax assets |
|
|
59,151 |
|
|
|
53,889 |
|
Valuation allowance |
|
|
(58,424 |
) |
|
|
(52,787 |
) |
Net deferred tax asset |
|
|
727 |
|
|
|
1,102 |
|
Deferred tax liabilities |
|
|
|
|
|
|
||
Accrued expenses and other |
|
|
(348 |
) |
|
|
(722 |
) |
Acquired in-process research and development |
|
|
(582 |
) |
|
|
(653 |
) |
Net deferred tax liabilities |
|
$ |
(203 |
) |
|
$ |
(273 |
) |
In assessing the need for a valuation allowance, the Company may utilize indefinite-lived deferred tax liabilities from an intangible asset as a future source of income. The Company’s acquired IPR&D intangible asset can be utilized as a source of income arising from the future reversal of temporary difference that can be offset against post 2017 indefinite-lived net operating losses (NOLs). Therefore, the Company is permitted to offset the indefinite-lived deferred tax liability up to the 80 percent limitation for NOLs generated subsequent to January 1, 2018.
Beginning in 2022, the 2017 Tax Cuts and Jobs Act requires taxpayers to capitalize research and development expenses with amortization periods over and fifteen years, depending on where the research is conducted. The Company has $28.3 million of research and development costs being capitalized in 2022. However, given the Company has a valuation allowance against its deferred tax assets, including the capitalized research and development costs, the enacted provision does not have a material impact on the consolidated financial statements.
The valuation allowance increased by $5.6 million and $4.5 million for the years ended December 31, 2022 and 2021, respectively.
139
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
The following table summarizes carryforwards of federal, state and foreign NOLs as of December 31, 2022 and 2021, respectively (in thousands):
|
|
December 31, |
|
|||||
|
|
2022 |
|
|
2021 |
|
||
Combined NOL Carryforwards: |
|
|
|
|
|
|
||
Federal |
|
$ |
162,335 |
|
|
$ |
145,265 |
|
State |
|
|
184,382 |
|
|
|
167,252 |
|
Foreign |
|
|
1,716 |
|
|
|
1,516 |
|
The NOL carryforwards begin expiring in 2032 for both federal and state income tax purposes. As of December 31, 2022, the Company also has federal, state and foreign research and development tax credit carryforwards of $7.3 million that will begin to expire in 2032, unless previously utilized.
The NOL and tax credit carryforwards are subject to review and possible adjustment by the Internal Revenue Service and state tax authorities. In general, under Section 382 of the Code, a corporation that undergoes an “ownership change,” generally defined as a greater than 50% change by value in its equity ownership over a three-year period, is subject to limitations on its ability to utilize its pre change tax credits as well as its NOLs to offset future taxable income. During 2021, the Company conducted a Section 382 study and determined that approximately $63.7 million in NOLs and $2.7 million in research and development tax credits were limited by Section 382 as of December 31, 2021. As a result of the Section 382 analysis, approximately $1.4 million of research and development tax credits are scheduled to expire unused due to the annual Section 382 limitation and therefore were written off in 2021.
The Company will recognize interest and penalties related to uncertain tax positions as income tax expense. As of December 31, 2022, the Company had no accrued interest and penalties related to uncertain tax positions and no amounts have been recognized in the Company’s statements of operations. Due to NOL and tax credit carryforwards that remain unutilized, income tax returns from 2019 through 2021 remain subject to examination by the taxing jurisdictions. The NOLs remain subject to review until utilized.
11. Related Party Transactions
IntellectMap provides IT-advisory services to the Company. The chief executive officer of IntellectMap is the brother of the Company’s chief executive officer. Fees incurred by the Company with respect to IntellectMap during each of the years ended December 31, 2022 and 2021 were $0.2 million.
12. Subsequent Events
In February 2023, the FDA accepted for filing and granted priority review to the NDA for avasopasem for radiotherapy-induced SOM in patients with HNC, with a PDUFA target date of August 9, 2023. This may have an impact on the amounts the Company records for interest on the royalty purchase liability, and such impact may be material.
On February 17, 2023, the Company completed a registered direct offering, which resulted in the issuance and sale of 14,320,000 shares of its common stock and warrants to purchase up to 14,320,000 shares of common stock at a combined offering price of $2.095 per share and accompanying warrant, generating gross proceeds of $30 million. The warrants have an exercise price of $1.97 per share of common stock, are exercisable immediately following their issuance and will expire five years from the date of issuance. The Company received net proceeds of approximately $27.7 million from this offering, after deducting placement agent fees and offering expenses.
140
GALERA THERAPEUTICS, INC.
NOTES TO CONSOLIDATED FINANCIAL STATEMENTS
Item 9. Changes in and Disagreements with Accountants on Accounting and Financial Disclosure.
None.
Item 9A. Controls and Procedures.
Limitations on Effectiveness of Controls and Procedures
In designing and evaluating our disclosure controls and procedures, management recognizes that any controls and procedures, no matter how well designed and operated, can provide only reasonable assurance of achieving the desired control objectives. In addition, the design of disclosure controls and procedures must reflect the fact that there are resource constraints, and that management is required to apply judgment in evaluating the benefits of possible controls and procedures relative to their costs.
Evaluation of Disclosure Controls and Procedures
Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated, as of the end of the period covered by this Annual Report on Form 10-K, the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15d-15(e) under the Exchange Act). Based on that evaluation, our Chief Executive Officer and Chief Financial Officer concluded that our disclosure controls and procedures were effective at the reasonable assurance level as of December 31, 2022.
Management’s Annual Report on Internal Control over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over our financial reporting, as such term is defined in Rule 13a-15(f) under the Exchange Act. Our management conducted an assessment of the effectiveness of our internal control over financial reporting based on the criteria set forth in “Internal Control - Integrated Framework (2013)” issued by the Committee of Sponsoring Organizations of the Treadway Commission. Based on this assessment, our management concluded that, as of December 31, 2022, our internal control over financial reporting was effective.
Attestation Report of the Independent Registered Public Accounting Firm
This Annual Report on Form 10-K does not include an attestation report of our independent registered public accounting firm due to an exemption established by the JOBS Act for "emerging growth companies."
Changes in Internal Control over Financial Reporting
There were no changes in our internal control over financial reporting identified in management’s evaluation pursuant to Rules 13a-15(d) or 15d-15(d) of the Exchange Act during the quarter ended December 31, 2022 that materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information.
None.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
Not applicable.
141
PART III
Item 10. Directors, Executive Officers and Corporate Governance.
Code of Ethics
Our board of directors has adopted a written Code of Business Conduct and Ethics applicable to all officers, directors and employees, including our principal executive officer, principal financial officer, principal accounting officer or controller, or persons performing similar functions. We have posted a current copy of our Code of Business Conduct and Ethics on our website at www.galeratx.com in the “Investors” section under “Corporate Governance.” We intend to satisfy the disclosure requirement under Item 5.05 of Form 8-K regarding amendment to, or waiver from, a provision of our Code of Business Conduct and Ethics, as well as Nasdaq’s requirement to disclose waivers with respect to directors and executive officers, by posting such information on our website at the address and location specified above. The information contained on our website is not incorporated by reference into this Annual Report on Form 10-K.
Executive Officers and Directors
The information concerning our executive offers and directors required by this Item 10 is contained under the caption “Information about our Executive Officers and Directors” at the end of Part I of this Annual Report on Form 10-K. The remainder of the information required to be disclosed by this Item 10 will be included in our definitive Proxy Statement for the 2023 Annual Meeting of Stockholders under the headings “Corporate Governance,” “Delinquent Section 16(a) Reports” (if applicable) and “Committees of the Board” and is incorporated herein by reference.
Item 11. Executive Compensation.
The information required by this Item 11 will be included in our definitive Proxy Statement for the 2023 Annual Meeting of Stockholders under the headings “Executive and Director Compensation” and “Compensation Committee Interlocks and Insider Participation” (if applicable) and is incorporated herein by reference.
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
Securities Authorized for Issuance under Equity Compensation Plans
Plan Category |
|
Number of |
|
|
Weighted-Average |
|
|
Number of |
|
|||
Equity compensation plans approved by |
|
5,783,185(2) |
|
|
$6.86(3) |
|
|
|
2,435,628 |
|
||
Equity compensation plans not approved by |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Total |
|
|
5,783,185 |
|
|
$ |
6.86 |
|
|
|
2,435,628 |
|
142
Other
The remaining information required by this Item 12 will be included in our definitive Proxy Statement for the 2023 Annual Meeting of Stockholders under the headings “Security Ownership of Certain Beneficial Owners and Management” and is incorporated herein by reference.
The information required by this Item 13 will be included in our definitive Proxy Statement for the 2023 Annual Meeting of Stockholders under the headings “Corporate Governance” and “Certain Relationships and Related Person Transactions” and is incorporated herein by reference.
Item 14. Principal Accountant Fees and Services.
Our independent registered public accounting firm is KPMG LLP, Philadelphia, PA, Auditor Firm ID: 185.
The information required by this Item 14 will be included in our definitive Proxy Statement for the 2023 Annual Meeting of Stockholders under the heading “Independent Registered Public Accounting Firm Fees and Other Matters” and is incorporated herein by reference.
143
PART IV
Item 15. Exhibits and Financial Statement Schedules.
(a)(1) Financial Statements.
The financial statements required by this item are listed in Item 8, “Financial Statements and Supplementary Data” herein.
(a)(2) Financial Statement Schedules.
All financial statement schedules have been omitted because they are not applicable, not required or the information required is shown in the financial statements or the notes thereto.
(a)(3) Exhibits.
The following is a list of exhibits filed as part of this Annual Report on Form 10-K.
144
Exhibit Index
Exhibit Number |
|
Description |
|
Form |
|
File No. |
|
Exhibit |
|
Filing Date |
|
Filed/ Furnished Herewith |
|
|
|
|
|
|
|
|
|
|
|
|
|
3.1 |
|
Restated Certificate of Incorporation of Galera Therapeutics, Inc. |
|
|
001-39114 |
|
|
11/12/2019 |
|
|
||
3.2 |
|
Amended and Restated Bylaws of Galera Therapeutics, Inc. |
|
|
001-39114 |
|
3.1 |
|
09/25/2020 |
|
|
|
4.1 |
|
Form of Certificate of Common Stock |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
4.2 |
|
Description of Securities |
|
|
001-39114 |
|
|
03/11/2021 |
|
|
||
4.3 |
|
Second Amended and Restated Investors’ Rights Agreement, dated as of August 30, 2018, by and among Galera Therapeutics, Inc. and the investors party thereto, as amended |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
4.4 |
|
Form of Warrant to Purchase Stock, dated May 11, 2020, issued by Galera Therapeutics, Inc. to Clarus IV Galera Royalty AIV, L.P., together with a schedule of warrantholders |
|
|
001-39114 |
|
|
08/10/2020 |
|
|
||
4.5 |
|
Form of Warrant to Purchase Common Stock, dated February 17, 2023, issued by Galera Therapeutics, Inc. |
|
|
001-39114 |
|
|
02/16/2023 |
|
|
||
10.1# |
|
Employment Agreement, dated October 25, 2019, by and between Galera Therapeutics, Inc. and J. Mel Sorensen, M.D. |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.2# |
|
Employment Agreement, dated October 25, 2019, by and between Galera Therapeutics, Inc. and Robert A. Beardsley, Ph.D. |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.3# |
|
Employment Agreement, dated October 25, 2019 by and between Galera Therapeutics, Inc. and Christopher Degnan |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.4# |
|
Employment Agreement, dated October 25, 2019, by and between Galera Therapeutics, Inc. and Jon T. Holmlund, M.D. |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.5# |
|
Employment Agreement, dated October 7, 2021, by and between Galera Therapeutics, Inc. and Jennifer Evans Stacey |
|
|
001-39114 |
|
|
11/10/2021 |
|
|
||
10.6# |
|
October 7, 2021, by and between Galera Therapeutics, Inc. and Mark Bachleda and amendments to Employment Agreement, dated January 31, 2022 and September 19, 2022, by and between Galera Therapeutics, Inc. and Mark Bachleda |
|
|
001-39114 |
|
|
11/09/2022 |
|
|
||
10.7# |
|
Employment Agreement, dated July 25, 2022, by and between Galera Therapeutics, Inc. and Eugene Kennedy, M.D. |
|
|
001-39114 |
|
|
11/09/2022 |
|
|
||
10.8# |
|
Form of Indemnification Agreement between Galera Therapeutics, Inc. and its directors and officers |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.9.1# |
|
Galera Therapeutics, Inc. 2019 Incentive Award Plan |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.9.2# |
|
Form of Stock Option Award Agreement under the Galera Therapeutics, Inc. 2019 Incentive Award Plan |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.9.3# |
|
Form of Restricted Stock Award Agreement under the Galera Therapeutics, Inc. 2019 Incentive Award Plan |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
145
10.9.4# |
|
Form of Restricted Stock Unit Award Agreement under the Galera Therapeutics, Inc. 2019 Incentive Award Plan |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.10# |
|
Galera Therapeutics, Inc. 2019 Employee Stock Purchase Plan |
|
|
333-234184 |
|
|
10/28/2019 |
|
|
||
10.11# |
|
Galera Therapeutics, Inc. Equity Incentive Plan, as amended |
|
|
333-234184 |
|
|
10/11/2019 |
|
|
||
10.12# |
|
Galera Therapeutics, Inc. Non-Employee Director Compensation Policy, as amended May 5, 2022 |
|
|
001-39114 |
|
|
08/09/2022 |
|
|
||
10.13.1† |
|
Amended and Restated Purchase and Sale Agreement, dated as of November 14, 2018, by and among Galera Therapeutics, Inc. and Clarus IV Galera Royalty AIV, L.P., Clarus IV-A, L.P., Clarus IV-B, L.P., Clarus IV-C, L.P., and Clarus IV-D, L.P. |
|
|
333-234184 |
|
|
10/11/2019 |
|
|
||
10.13.2† |
|
Amendment No. 1 Amended and Restated Purchase and Sale Agreement, dated May 11, 2020, by and between Galera Therapeutics, Inc. and Clarus IV Galera Royalty AIV, L.P. |
|
|
001-39114 |
|
|
08/10/2020 |
|
|
||
10.14† |
|
Warrant Purchase Agreement, dated May 11, 2020, by and between Galera Therapeutics, Inc. and Clarus IV Galera Royalty AIV, L.P. |
|
|
001-39114 |
|
|
08/10/2020 |
|
|
||
10.15† |
|
Master Manufacturing Services Agreement between Patheon Manufacturing Services LLC and Galera Therapeutics, Inc., dated August 13, 2021 |
|
|
001-39114 |
|
|
08/18/2021 |
|
|
||
10.16 |
|
Placement Agency Agreement dated February 15, 2023, by and between Galera Therapeutics, Inc. and Piper Sandler & Co. |
|
|
001-39114 |
|
|
02/16/2023 |
|
|
||
10.17 |
|
Securities Purchase Agreement dated February 15, 2023 by and among Galera Therapeutics, Inc. and the purchasers named therein |
|
|
001-39114 |
|
|
02/16/2023 |
|
|
||
21.1 |
|
Subsidiaries of Galera Therapeutics, Inc. |
|
|
333-234184 |
|
|
10/11/2019 |
|
|
||
23.1 |
|
|
|
|
|
|
|
|
|
|
* |
|
31.1 |
|
|
|
|
|
|
|
|
|
|
* |
|
31.2 |
|
|
|
|
|
|
|
|
|
|
* |
|
32.1 |
|
|
|
|
|
|
|
|
|
|
** |
|
32.2 |
|
|
|
|
|
|
|
|
|
|
** |
|
101.INS |
|
Inline XBRL Instance Document - the Instance Document does not appear in the interactive data file because its XBRL tags are embedded within the Inline XBRL document |
|
|
|
|
|
|
|
|
|
* |
101.SCH |
|
Inline XBRL Taxonomy Extension Schema Document |
|
|
|
|
|
|
|
|
|
* |
101.CAL |
|
Inline XBRL Taxonomy Extension Calculation Linkbase Document |
|
|
|
|
|
|
|
|
|
* |
146
101.DEF |
|
Inline XBRL Taxonomy Extension Definition Linkbase Document |
|
|
|
|
|
|
|
|
|
* |
101.LAB |
|
Inline XBRL Taxonomy Extension Label Linkbase Document |
|
|
|
|
|
|
|
|
|
* |
101.PRE |
|
Inline XBRL Taxonomy Extension Presentation Linkbase Document |
|
|
|
|
|
|
|
|
|
* |
104 |
|
Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) |
|
|
|
|
|
|
|
|
|
* |
* Filed herewith.
** Furnished herewith.
# Indicates management contract or compensatory plan.
† |
Portions of this exhibit have been redacted in compliance with Regulation S-K Item 601(b)(10)(iv). |
Item 16. Form 10-K Summary
None.
147
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
Galera Therapeutics, Inc. |
|
|
|
|
|
Date: March 8, 2023 |
|
By: |
/s/ J. Mel Sorensen, M.D. |
|
|
|
J. Mel Sorensen, M.D. |
|
|
|
Chief Executive Officer and President |
|
|
|
|
Date: March 8, 2023 |
|
By: |
/s/ Christopher Degnan |
|
|
|
Christopher Degnan |
|
|
|
Chief Financial Officer |
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, this Report has been signed below by the following persons on behalf of the Registrant in the capacities and on the dates indicated.
Name |
|
Title |
|
Date |
|
|
|
|
|
/s/ J. Mel Sorensen, M.D. |
|
Chief Executive Officer, President and Director |
|
March 8, 2023 |
J. Mel Sorensen, M.D. |
|
(principal executive officer) |
|
|
|
|
|
|
|
/s/ Christopher Degnan |
|
Chief Financial Officer |
|
March 8, 2023 |
Christopher Degnan |
|
(principal financial and accounting officer) |
|
|
|
|
|
|
|
/s/ Mike Powell, Ph.D. |
|
Chairman of the Board of Directors |
|
March 8, 2023 |
Michael Powell, Ph.D. |
|
|
|
|
|
|
|
|
|
/s/ Lawrence Alleva |
|
Director |
|
March 8, 2023 |
Lawrence Alleva |
|
|
|
|
|
|
|
|
|
/s/ Emmett Cunningham, M.D., Ph.D., MPH |
|
Director |
|
March 8, 2023 |
Emmett Cunningham, M.D., Ph.D., MPH |
|
|
|
|
|
|
|
|
|
/s/ Linda West |
|
Director |
|
March 8, 2023 |
Linda West |
|
|
|
|
|
|
|
|
|
/s/ Kevin Lokay |
|
Director |
|
March 8, 2023 |
Kevin Lokay |
|
|
|
|
|
|
|
|
|
148
Similar companies
See also JOHNSON & JOHNSON - Annual report 2024 (10-K 2024-12-29) Annual report 2025 (10-Q 2025-06-29)See also ELI LILLY & Co - Annual report 2024 (10-K 2024-12-31) Annual report 2025 (10-Q 2025-06-30)
See also PFIZER INC - Annual report 2024 (10-K 2024-12-31) Annual report 2025 (10-Q 2025-03-30)
See also AbbVie Inc. - Annual report 2024 (10-K 2024-12-31) Annual report 2025 (10-Q 2025-06-30)
See also NOVO NORDISK A S
