Ginkgo Bioworks Holdings, Inc. - Annual Report: 2021 (Form 10-K)
Table of Contents
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 10-K
(Mark One)
☒ |
ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
For the fiscal year ended December 31, 2021
OR
☐ |
TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 FOR THE TRANSITION PERIOD FROM TO |
Commission File Number 001-40097
GINKGO BIOWORKS HOLDINGS, INC.
(Exact name of Registrant as specified in its Charter)
Delaware |
87-2652913 |
(State or other jurisdiction of incorporation or organization) |
(I.R.S. Employer Identification No.) |
27 Drydock Avenue 8th Floor Boston, MA |
02210 |
(Address of principal executive offices) |
(Zip Code) |
Registrant’s telephone number, including area code: (877) 422-5362
Securities registered pursuant to Section 12(b) of the Act:
Title of each class |
|
Trading Symbol(s) |
|
Name of each exchange on which registered |
Class A common stock, par value $0.0001 per share |
|
DNA |
|
NYSE |
Warrants to purchase one share of Class A common stock, each at an exercise price of $11.50 per share |
|
DNA.WS |
|
NYSE |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark if the Registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. YES ☐ No ☒
Indicate by check mark if the Registrant is not required to file reports pursuant to Section 13 or 15(d) of the Act. YES ☐ No ☒
Indicate by check mark whether the Registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the Registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ NO ☐
Indicate by check mark whether the Registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the Registrant was required to submit such files). Yes ☒ NO ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer |
|
☐ |
|
Accelerated filer |
|
☐ |
Non-accelerated filer |
|
☒ |
|
Smaller reporting company |
|
☐ |
|
|
|
|
Emerging growth company |
|
☒ |
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☐
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report. ☐
Indicate by check mark whether the Registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). YES ☐ NO ☒
The aggregate market value of the voting and non-voting common equity held by non-affiliates of Soaring Eagle Acquisition Corp. (SRNG), our predecessor, on June 30, 2021, based on a closing price of $9.96 for SRNG’s Class A common stock, was approximately $250.5 million.
As of March 17, 2022, there were 1,080,456,818 shares of Class A common stock, 391,147,458 shares of Class B common stock, and 288,000,000 shares of non-voting Class C common stock outstanding.
Table of Contents
Table of Contents
|
|
Page |
|
|
|
Item 1. |
1 |
|
Item 1A. |
51 |
|
Item 1B. |
90 |
|
Item 2. |
90 |
|
Item 3. |
90 |
|
Item 4. |
90 |
|
|
|
|
|
|
|
Item 5. |
91 |
|
Item 6. |
92 |
|
Item 7. |
Management’s Discussion and Analysis of Financial Condition and Results of Operations |
93 |
Item 7A. |
112 |
|
Item 8. |
112 |
|
Item 9. |
Changes in and Disagreements With Accountants on Accounting and Financial Disclosure |
112 |
Item 9A. |
112 |
|
Item 9B. |
113 |
|
Item 9C. |
Disclosure Regarding Foreign Jurisdictions that Prevent Inspections |
113 |
|
|
|
|
|
|
Item 10. |
114 |
|
Item 11. |
118 |
|
Item 12. |
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters |
123 |
Item 13. |
Certain Relationships and Related Transactions, and Director Independence |
127 |
Item 14. |
132 |
|
|
|
|
|
|
|
Item 15. |
133 |
|
Item 16. |
137 |
Table of Contents
Cautionary Note Regarding Forward Looking Statements
This report includes forward-looking statements regarding, among other things, the plans, strategies and prospects, both business and financial, of Ginkgo. These statements are based on the beliefs and assumptions of the management of Ginkgo. Although Ginkgo believes that its plans, intentions and expectations reflected in or suggested by these forward-looking statements are reasonable, Ginkgo cannot assure you that it will achieve or realize these plans, intentions or expectations. Forward-looking statements are inherently subject to risks, uncertainties and assumptions. Generally, statements that are not historical facts, including statements concerning possible or assumed future actions, business strategies, events or results of operations, are forward-looking statements. These statements may be preceded by, followed by or include the words “believes”, “estimates”, “expects”, “projects”, “forecasts”, “may”, “will”, “should”, “seeks”, “plans”, “scheduled”, “anticipates” or “intends” or similar expressions. Forward-looking statements contained in this annual report on Form 10-K (“Annual Report”) include, but are not limited to, statements about:
These and other factors that could cause actual results to differ from those implied by the forward-looking statements in this Annual Report are more fully described under the heading “Risk Factors” and elsewhere in this report. The risks described under the heading “Risk Factors” are not exhaustive. Other sections of this Annual Report describe additional factors that could adversely affect the business, financial condition or results of Ginkgo. New risk factors emerge from time to time and it is not possible to predict all such risk factors, nor can Ginkgo assess the impact of all such risk factors on the business of Ginkgo, or the extent to which any factor or combination of factors may cause actual results to differ materially from those contained in any forward-looking statements. Forward-looking statements are not guarantees of performance. You should not put undue reliance on these statements, which speak only as of the date hereof. All forward-looking statements attributable to Ginkgo or persons acting on its behalf are expressly qualified in their entirety by the foregoing cautionary statements. Ginkgo undertakes no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law.
Risk Factors Summary
Investing in our securities involves risks. You should carefully consider the risks described in “Risk Factors” beginning on page 51 before making a decision to invest in our Class A common stock. If any of these risks actually occur, our business,
i
Table of Contents
financial condition and results of operations would likely be materially adversely affected. Some of the risks related to Ginkgo’s business and industry are summarized below. References in the summary below to “we,” “us,” “our” and “the Company” generally refer to Ginkgo.
ii
Table of Contents
iii
Table of Contents
PART I
Item 1. Business.
Unless the context otherwise requires, all references in this section to the “Company,” “Ginkgo,” “we,” “us,” or “our” refer to the business of Ginkgo Bioworks Holdings, Inc. and its subsidiaries.
Mission
Our mission is to make biology easier to engineer. That has never changed. Every choice we’ve made with respect to our business model, our platform, our people and our culture is grounded in whether it will advance our mission. Biology inherently offers incredible capabilities that we can only imagine in human-made technologies—self-assembly, self-repair, self-replication—capabilities that can enable more renewable and innovative approaches for nearly every industry. To realize this potential, we are building a platform for cell programming by bringing together unparalleled scale, software, automation, data science and reusable biological knowledge, enabling responsible solutions for the next generation of foods, pharmaceuticals, materials and more.
Overview
Ginkgo is building the industry-standard horizontal platform for cell programming. Our founders are engineers from diverse fields who, more than 20 years ago, were inspired by an astonishing feature of biology: it runs on digital code. It’s just A, T, C, and G rather than 0 and 1. But where computer bits are used to communicate information, genetic code is inherently physical and as it is read, physical structures are made. We program computers to manipulate bits, but we program cells to manipulate atoms. Cells are the building blocks of our food, our environment and even ourselves.
We use our platform to program cells on behalf of our customers. These “cell programs” (or “programs”) are designed to enable biological production of products as diverse as novel therapeutics, key food ingredients, and chemicals currently derived from petroleum. Through 2021, we have worked on 105 Cumulative Programs, which represent the cumulative number of unique programs Ginkgo has commenced in end markets as diverse as specialty chemicals, agriculture, food, consumer products, and pharmaceuticals. Biology did not evolve by end market. All of these applications run on cells which have a common code—DNA—and a common programming platform can enable all of them. Because of this shared platform, we are able to drive scale and learning efficiencies while maintaining flexibility and diversity in our program areas. Ultimately, customers come to us because they believe we maximize the probability of successfully developing their products.
Customers look to Ginkgo to overhaul their manufacturing processes or develop new products through biology. They might, for example, be looking to produce a particular chemical via fermentation, at a lower cost, with enhanced supply chain reliability or sustainability. Or perhaps the customer needs a microbe that will live and grow on the roots of corn and convert nitrogen in the air into usable fertilizer for a plant, resulting in improved plant growth. Or a customer might need an antibody that binds to and neutralizes a certain target, along with a way to produce those antibodies at scale. All of these programs and more run on a common platform at Ginkgo.
We care deeply how our platform is used and recognize biosecurity as a necessary complement to our cell programming work. Biosecurity is ingrained in our platform and in our ethos—we work collaboratively to help ensure responsible research conduct and deployment within our own work and within the broader synthetic biology community. We have additionally invested in developing targeted biosecurity and public health capabilities, including an initiative for pathogen biomonitoring at scale. Through this initiative, known as Concentric by Ginkgo, we serve as a system integrator to implement customizable and accessible biosecurity programs for monitoring and mitigating the spread of COVID-19 in thousands of communities across the country. We are dedicated to setting the industry standard for biosecurity as we continue to scale our platform.
The foundation of our platform includes two core assets that execute a wide variety of cell programs for customers according to their specifications: our Foundry and our Codebase.
1
Table of Contents

Figure 1: Our platform is used to design, write, and debug DNA code in engineered organisms to execute programs for our customers. Our Foundry leverages proprietary software, automation, and data analytics to reduce the cost of cell programming. Our Codebase consists of reusable biological assets that helps accelerate the engineering process.
As the platform scales, we have observed a virtuous cycle between our Foundry, our Codebase, and the value we deliver to customers. Sketched below, we believe this virtuous cycle sustains Ginkgo’s growth and differentiated value proposition.
Put simply: we believe that as the platform improves with scale, it drives more scale, which drives further platform improvements, and so on. We believe this positive feedback loop has the potential to drive compounding value creation in the future, as every new program we add contributes to both near-term revenues and has the potential to add significant downstream economics.
2
Table of Contents

Figure 2: Ginkgo’s virtuous cycle: as we scale, we see greater efficiency and higher odds of technical success, which helps drive further scaling as our value proposition improves.
Our business model mirrors the structure of our platform and we are compensated in two primary ways. First, we charge usage fees for Foundry services, in much the same way that cloud computing companies charge usage fees for utilization of computing capacity or contract research organizations (“CROs”) charge for services. The total addressable market for biological research and development (“R&D”) services, including labor and tools, was estimated to be over $40 billion in 2021 and is growing strongly. Additionally, we negotiate a value share with our customers (typically in the form of royalties, milestones, and/or equity interests) in order to align our economics with the success of the programs enabled by our platform. As we add new programs, our portfolio of programs with this “downstream” value potential grows. Through these value shares, we are tapping into what industry sources expect to be a $2 to $4 trillion market for bioengineered products in the next 10 to 20 years.
We believe that cell programming has the potential to be as ubiquitous in the physical world as computer programming has become in the digital world. We believe products in the future will be grown rather than made. To enable that vision, we are building a horizontal platform to make biology easier to engineer. Our business model is aligned with this strategy and with the success of our customers, setting us on what we believe is a path towards sustainable innovation for years to come.
An Introduction to Synthetic Biology
To fully tell the story of cell programming, we have to start four billion years ago. All living things evolved from a single cell, a tiny bubble containing the code that enabled it to assemble and reproduce itself. But, importantly, that process of reproduction wasn’t perfect; each copy introduced new mutations in the code. These changes are responsible for one of the most powerful and defining features of biology: evolution. Over eons, that first cell and all its progeny copied themselves, and their DNA evolved to create new functions: to eat new kinds of foods and to produce new kinds of chemicals, structures, and behaviors. As reproduction became more, well, interactive, organisms developed tools to borrow DNA from each other, accelerating the pace of evolution. These functions, and thus the genetic code programming the functions, stuck around when they helped the organisms survive and create more descendants. This went on and on for four billion years, leaving us the wild codebase of DNA that enables the diversity of life forms we see on the planet today.
3
Table of Contents
Synthetic biology’s story begins mere decades ago, as biologists began to decode the molecular secrets of DNA. The billions-year-old tools of cells—enzymes that cut, copy, and paste sequences of DNA code—are now being leveraged by humans to read, write, and edit DNA in the lab. Polymerases that copy DNA are used to enable polymerase chain reaction (“PCR”) tests for COVID-19 and the CRISPR/Cas system from bacteria now enables editing of human genomes to potentially cure genetic diseases.
Today we are using these tools to learn from the full breadth of evolution and biodiversity to write new biological code. Simple soil bacteria produce everything from vital antibiotics to the smell of fresh rain. We can reuse elements of these DNA programs to make new products. Biochemistry is extraordinarily versatile; we’ve reused the same genetic code libraries across applications as diverse as fine fragrances, baking, and consumer electronics. We may be able to develop programs that can digest human-made “forever chemicals” that biology never encountered before.
As cell programmers, we operate with humility and respect for biology. Our tools are simply borrowed, and the history of biotechnology is a mere blink of an eye compared to the history of living things. Today, we write rudimentary code. We believe that someday our children will write poetry in DNA.
Programming life

Figure 3: DNA strands are sequences composed of four chemical bases, or nucleotides, represented by the letters A, T, C and G.
Like computers, cells run on digital code. DNA strands are sequences composed of four chemical bases, or nucleotides, represented by the letters A, T, C and G. The letters along the strand encode the proteins that make up the cell and perform biochemical functions. The translation of DNA to RNA to Protein is known as the “central dogma” of molecular biology.
The Central Dogma of Molecular Biology

Figure 4: The translation of DNA to RNA to Protein is known as the “central dogma” of molecular biology.
“Traditional” genetic engineering uses special types of proteins from bacteria that can cut and paste DNA to move sequences from one organism to another. In 1982, Genentech Inc. partnered with Eli Lilly and Company to bring these techniques to market, producing human insulin inside the bacteria E. coli. Genetic engineers were able to cut the code for the human insulin protein and paste it into the genome of E. coli and “boot up” the sequence: the bacteria could now produce the human protein, which could then be extracted, purified, and used by diabetics. This life-saving development replaced a vastly more expensive and supply-constrained method of extracting insulin from animal pancreases.
4
Table of Contents
Relatively simple proteins like insulin can be produced by transferring one gene sequence into a simple microorganism. Many other biochemicals require much more complex cell programming and are produced by a series of special proteins, called enzymes, working together. These enzymes transform a starting material, or “feedstock,” such as sugar, into a final product, such as an antibiotic, vitamin, or other valuable small molecule. In this way, biology also programs chemistry. Cell programmers can design such multi-enzyme “pathways” and transfer them into a cell to boot up. For example, the cell programs we’re writing for Cronos Group, Inc. to produce cannabinoids require many different enzymes to convert feedstock into cannabinoids such as cannabidiol.
Once the cell is programmed to produce a new molecule, it can produce the molecule and also replicate itself, creating an exponentially growing number of product-producing cells. Many products of genetic engineering are manufactured in facilities that look like breweries, taking advantage of the centuries old process of industrial fermentation to grow cells at high density, and transforming simple sugars into valuable products that can be extracted and commercialized.
Improved tools for cell programming, including automation, miniaturization, and data science, alongside the decreasing cost of DNA synthesis—writing DNA—are opening up new possibilities for cell programming. For each new program, Ginkgo’s organism engineers design, print, and test hundreds or thousands of different sequences for each step of a pathway, exploring the breadth of biological design space and improving the probability of success. We provide more details about our platform in the sections that follow.
5
Table of Contents

Figure 5: An overview of a simple cell program.
The Impact of Cell Programming
The power of biology has never been more apparent. Synthetic biology was featured on the cover of The Economist in April of 2019. A few years later, billions of doses of a novel type of vaccine have been distributed, created by companies such as Pfizer and Moderna and made up of a form of biological code, mRNA. Our own cells read that code to produce viral proteins and stimulate our immune response to fight back against the SARS-CoV-2 virus. We no longer question if biotechnology will transform a given industry, we simply question whether we are creative enough to imagine how, and whether we are ready to utilize biology responsibly.
ESG is in our DNA
Biology affects all of us, and we believe cell programming will change the world. Our customers are developing products with far reaching implications in health and the environment. This potential for extraordinary impact, which reaches to the core of who we are and everything about our natural world, requires extraordinary care in how the tools of cell programming
6
Table of Contents
are built and used. Technologies reflect the values of the organizations that build them, so our commitment to Environmental, Social, and Governance (“ESG”) priorities and care must underscore everything we do.
We also must recognize that biotechnologies have not always reflected the values necessary for sustainable and equitable impact and, as a result, remain controversial. Indeed, companies that produce genetically modified organisms (“GMOs”) for human consumption are restricted from certain ESG indices, placing genetic engineering as a major ESG risk alongside the production of weapons, tobacco products, and fossil fuels. We hope to chart a new course built on care so that the world can benefit from the power of biological engineering while avoiding potential risks.
Environmental
We face an urgent environmental crisis that is forcing us to reconsider how we make everything, from our homes, to our food, to our clothing. For centuries, we’ve treated nature as an infinite resource and infinite trash can, extracting raw materials, shaping them through industrial processes that spew out greenhouse gases, and then throwing them away. But these resources are not infinite and there is no “away.” The results have been disastrous—climate change, loss of biodiversity, and pollution have impacted every corner of our world and continue to threaten our way of life.
Cell programming and biological manufacturing are working to address some of the issues that are most contributing to climate change today, from fossil fuel dependency to agricultural emissions, and land use to plastic pollution. Ultimately, biology offers a fundamental shift in how things are made and disposed of: a world where things grow and decay, creating circular, regenerative processes.
There is significant concern that genetic engineering itself creates a form of genetic “pollution” in the environment, with genes from one context introduced into another. This is a concern we take seriously and consider deeply throughout the lifecycle of our programs to ensure that genes introduced will not cause damage—for example, by spreading antibiotic resistance or toxins. We care because the environmental release of certain genetically engineered microbes can also offer tremendous environmental benefit. For example:
And we are just getting started… we believe biology is our best tool to reverse the damage to our planet and chart us on a path towards sustainability in the future.
Social
Technology isn’t neutral. Our values and biases are embedded in the technologies we make, in the applications we consider, and in the ways we address problems. Inclusion of those who have historically been left out of the development of new technologies is essential to building equitable and positive outcomes. Just as biological ecosystems thrive with more diversity, the inclusion of many different voices is essential to growing our company and to ensuring that the viewpoints of historically marginalized people are included in the development of our platform. We have many active efforts in recruiting and retaining diverse talent and will continue to invest in this work (see “—Our People & Culture”).
Marginalized people who have been left out of the development of technologies are also the groups most likely to bear the greatest harm, whether from climate change, pollution, or health disparities. The COVID-19 pandemic has made this inequality starkly clear—in the United States, it has been communities of color that have been disproportionately impacted by the pandemic and have had the least access to testing, treatment, and vaccination.
In March of 2020, we committed to $25 million of pro bono work to help accelerate novel diagnostics, therapeutics, and vaccines to help fight COVID-19. Our early work included efforts to improve the manufacturing of vaccines, with a goal to lower costs and increase accessibility of vaccines worldwide. Shortly thereafter, we launched Concentric by Ginkgo, a service to provide public health testing infrastructure for communities. Our pooled testing service was designed with accessibility and privacy as core design principles, to bring low-cost, easy-to-use testing to K-12 schools in the places that have been most affected by the pandemic. We partnered with school districts such as Baltimore City Public Schools to make
7
Table of Contents
sure that our program was designed to serve the community and to build trust with groups who have been excluded, exploited and mistreated by biomedical research in the past.
These values and initiatives are not just a top-down corporate policy, they are an intrinsic part of our culture. Grassroots fundraising challenges to support local and international aid organizations are a regular feature of our internal messaging channels. One of our software engineers even programmed a free tool, @vaccinetime on Twitter, that helped thousands of Massachusetts residents find vaccine appointments.
Governance
Our culture is built on care, transparency, diversity, employee ownership and engagement, and a deep, humble respect for biology. Transparency is essential to how we operate, to enable sharing of the insights and tools that enable our platform to grow, as well as to build trust and accountability with all of our stakeholders. We have advocated for more transparency in our industry, including supporting GMO labeling, and seek to educate policymakers and the general public about the benefits and risks of synthetic biology through our advocacy efforts.
The individuals who work at Ginkgo and build our platform care deeply about how that platform is used and the impact our company will have in the world. We believe a workforce with strong equity ownership will make the wise decisions needed to build long-term value for our company, and a company whose long-term impacts make them proud. That is why we have implemented a multi-class stock structure that permits all employees (current and future), not just founders, to hold high-vote (10 votes per share) common stock. We believe that our multi-class stock structure will help maintain the long-term mentality we have benefited from as a founder-led company.
For more information, see “Risk Factors—Risks Relating to our Organizational Structure and Governance—Only our employees and directors are entitled to hold shares of Ginkgo Class B common stock (including shares of our Class B common stock granted or otherwise issued to our employees and directors in the future), which shares have 10 votes per share. This limits or precludes other stockholders’ ability to influence the outcome of matters submitted to stockholders for approval, including the election of directors, the approval of certain employee compensation plans, the adoption of amendments to our organizational documents and the approval of any merger, consolidation, sale of all or substantially all of our assets, or other major corporate transaction requiring stockholder approval.”
We have selected directors with decades of experience serving as leaders in the life sciences and technology industries. Our board of directors and management team will leverage that experience and consider the interests of stockholders, customers, employees, suppliers, academic researchers, governments, communities, and other stakeholders to pursue long-term value for our company and drive the sustained health of our global community. For more information, see “Risk Factors—Risks Relating to our Organizational Structure and Governance—Our focus on the long-term best interests of our company and our consideration of all of our stakeholders, including our stockholders, workforce, customers, suppliers, academic researchers, governments, communities and other stakeholders that we may identify from time to time, may conflict with short-term or medium-term financial interests and business performance, which may adversely impact the value of our common stock.”
Cell programming is expected to transform all industries
Biology grows. Biology adapts and evolves. Biology heals itself and regenerates. Biology is also, remarkably, programmable, offering us the tools to work with biology to transform how we make stuff. With cell programming, we help our customers across industries grow better products. What does “better” mean? Better products might be more sustainable, have more stable and resilient supply chains, be more accessible, have higher quality and more consistency, and come with lower economic and environmental costs of manufacturing. They can also be truly transformative, fundamentally changing the field of possibilities for what products can do. We have supported many companies that are leveraging our cell programming platform to address some of the world’s most challenging environmental and social issues.
Pharma & Biotech
Biopharma has been a nexus of tremendous innovation in cell programming and synthetic biology. Just in the past year, we have seen the creation and broad adoption of a novel form of biological prophylactic in the form of nucleic acid vaccines. These vaccines contain genetic code that our bodies read to produce viral proteins and stimulate an immune response and antibody production. New nucleic acid vaccines can be programmed quickly, such as the booster vaccines being developed against emerging SARS-CoV-2 variants, offering the potential for rapid response to other future pathogens. They can also be programmed to target a number of other diseases. In the wake of the success of nucleic acid vaccines during the COVID-19 pandemic, new programs for HIV and cancer vaccines, among others, are accelerating.
8
Table of Contents
Biologic medicines like insulin and other protein drugs and antibodies are also produced via cell programming, making a difference in the treatment of countless diseases. Over 20% of the therapies approved by the U.S. Food and Drug Administration (“FDA”) in 2021 were biologics. In addition, new modalities enabled by cell programming, such as cell and gene therapies, microbiome therapies, and regenerative medicines, are beginning to come online. We believe human health and the ways we treat disease will be transformed by improvements in cell programming technology.
Ginkgo has been active in this field in recent years and we expect to significantly expand our support of therapeutic applications over coming years. From companies developing “living medicines” (Synlogic, Inc. (“Synlogic”)) to those involved in COVID-19 vaccine production (Moderna and others) to those developing novel antibiotics (Roche), we are using our platform to deliver transformational innovations across a range of disease areas.
Industrials & Environment
Since the industrial revolution, manufacturing techniques have been extractive, wasteful, and unsustainable. Not only must we innovate new manufacturing methods in order to keep up with growing demand, we must also work to remediate issues we have caused historically, by cleaning up our environment and addressing climate change.
Ginkgo is not only working with customers to create cell programs that enable cost-efficient, renewable, and sustainable production of chemicals and materials, such as our work with Genomatica, Inc. (“Genomatica”), but we have also participated in the formation of Allonnia, a company focused on environmental remediation. Plastic waste and many of the pollutants that plague industrial manufacturing and extraction sites are novel in the course of evolutionary history, so biology has not yet evolved to degrade them efficiently. Cell programming can enable the discovery and development of new enzymes capable of degrading recalcitrant pollutants and recycling waste while entirely reimagining manufacturing for the future.
Food & Agriculture
Food is inherently biological: it comes from life and sustains life. Cell programming can be leveraged to improve the availability of essential food and nutrition to a growing population, decrease the environmental impact and cost of food production, and provide consumers with increased choice.
We are working with some of the largest multinational agriculture companies, including Bayer (through our joint venture, Joyn Bio) and Corteva, to develop cell programs that would make crop production more efficient and sustainable, reducing synthetic nitrogen fertilizer and pesticide usage. In food, we have been active in flavors and sweeteners, and we are the principal cell programming platform for Motif FoodWorks, Inc. (“Motif”), a company that is making animal proteins without the need for industrial farming of animals, which launched its first product, HEMAMI™, in 2021.
Consumer & Technology
Most physical goods have biological origins—from the petrochemicals in our fabrics to fine chemicals extracted from plants—but industry does not necessarily leverage biology, or leverage biology efficiently, to produce these items. Petrochemicals, for example, are used in everything from our fabrics to our cosmetics to our paints. These chemicals and polymers are generally created in complex chemical and physical reactions from crude oil, but crude oil is just the result of millions of years of decomposition of previously living matter (they are fossil fuels after all). These biological building blocks can instead be programmed in a living organism to produce these items sustainably, without extracting natural resources. Even in areas where industry does leverage biology, such as extracting raw materials or fine chemicals from plants, we believe the current approaches are woefully inefficient or rife with social consequences.
We have helped some of the world’s largest fragrance companies such as Givaudan use fermentation to much more efficiently produce rare molecules typically extracted from plants. In a related field, we are also supporting Cronos in their effort to biosynthesize cannabinoids, with the goal of reducing cost, improving purity and predictability, and enabling production of rare molecules. We also helped launch a new company in 2021, Arcaea (formerly Kalo Ingredients, LLC), which is focusing on leveraging biology, from proteins to the microbiome, to build a suite of innovative and efficacious personal care products.
Market Opportunity
For several decades in the computing industry, software ran entirely in local environments: companies built and ran their own servers and customized their applications. The dominance of software-as-a-service (“SaaS”) software and cloud computing over the past decade has demonstrated the value in having common architectures and enabling horizontal platforms. What
9
Table of Contents
users may have sacrificed in customizability, they more than gained in innovation, efficiency, and scalability. We believe Ginkgo is ushering in a similar transition in cell programming, a programming discipline with the power to shape living things and grow applications across the physical world.
The value of these applications will measure in the trillions of dollars
Given the breadth of application areas and the potential of biology (see “—The Impact of Cell Programming”), we believe that the end markets for bioengineered products will be enormous. Industry sources estimate that in the next 10 to 20 years, there will be approximately $2 to $4 trillion of annual direct economic impact from these products, with significant secondary effects. But these applications reflect only what we can already imagine. As we develop a greater ability to program biology and direct it towards novel and more challenging applications, the spectrum of possibilities will undoubtedly grow. Computers were used for little more than counting for decades; we firmly believe the most valuable applications of cell programming are not yet apparent.

Figure 6: Industry sources estimate a $2 to $4 trillion total addressable market for bioengineered products between 2030 and 2040.
Large existing market for “on prem” cell programming research and development
Cell programming today is done in a highly inefficient, distributed manner reminiscent of the early days of computing. Essentially every organization looking to innovate in biology builds its own biology labs in the same way that individual tech companies used to set up their own servers instead of using cloud computing. Scientists spend hours moving liquids around rather than designing novel experiments just as computer programmers once spent most of their time physically writing and debugging code (by punching cards, for example) rather than designing new applications.
Because of the way cell programming is done today, intellectual property that could be useful for multiple applications is tied up by exclusivities that delay the progress of the field overall. Ginkgo’s platform breaks down these silos and democratizes access to the most advanced technologies in the field, enabling customers of all sizes to more efficiently drive innovation.
According to industry sources, the market for cell programming R&D is approximately $40 billion and growing rapidly. This work is being done in a distributed manner, sacrificing benefits from scale and learning economies. Approximately 60% of the spend today is on labor—scientists designing and executing experiments—while the remaining 40% of this is spent on “tools”—things like DNA synthesis, reagents, and equipment. Ginkgo brings efficiencies to both elements of this existing market.
10
Table of Contents
As the cost of computing power declined exponentially in computer programming, the demand for computing power increased exponentially as developers dreamed up more and more sophisticated applications. We expect the same to be true in cell programming: as our platform scales in capability and capacity, we hope that the range of applications accessible to cell programming will likewise expand in breadth and sophistication.
Industry Overview
We believe that Ginkgo is changing the structure of the biotechnology industry. In much the same way that cloud computing centralized hosting services and ushered in a wave of SaaS software companies, Ginkgo is scaling the capabilities needed to program cells. By making these tools more accessible, we hope to usher in a wave of innovation in both “hardware” (life science tools) and “software” (cell programs).
At Ginkgo, we have always admired the symbiotic and regenerative nature of biology, which sits in stark contrast to the often extractive nature of existing technologies. We are often asked who we think the “winners” and “losers” in the industry will be as Ginkgo scales, as if it is a given that our growth must come at the expense of others in the ecosystem. We reject that notion. As our platform scales, we seek to drive benefits for all existing players in this ecosystem:
11
Table of Contents

Figure 7: Schematic of the synthetic biology industry structure. Ginkgo connects and integrates the hardware and tools provided in the technology layer, creating a platform that can be used by cell programming customers who are building products for end market use.
Program Layer: Ginkgo enables and accelerates product companies, which historically have had to vertically integrate
Ginkgo is not a product company; we are an enabling platform for product companies in a range of end markets. We do not seek to “pick winners” and focus instead on building our platform rather than investing in product-specific risk. Platforms require scale and a relentless focus on innovation while taking a product to market requires many specialized functions that vary depending on the product:
Once the product is developed, major investments are also needed to manufacture, distribute, and market the product. These are the jobs of our customers, the product companies.
12
Table of Contents
Historically, product companies have had to invest in their own R&D capabilities, building their own labs and hiring their own scientists. This investment is inefficient due to lack of scale and drains resources away from application testing and product development. Ginkgo’s platform is not application-specific. The same engineering tools can be used for programs in completely different application areas: cells all run on the same genetic code. As product companies develop their products on Ginkgo’s platform, they gain efficiencies and increase their probability of success. New companies that build on our platform never need to make the fixed capital investments to start a lab from scratch; they are able to leapfrog and compete effectively against established companies.
Technology Layer: Ginkgo collaborates with life science tools companies to drive technology advancements
Because we’re constantly thinking about how to enable the next several years of exponential scaling of our platform, we have good insights into future bottlenecks and welcome the opportunity to collaborate to build technologies that will break through those barriers. We are the largest customer for many of our strategic suppliers and, as such, play an important role in advancing new technologies. As a result, we are often able to secure preferred access, often including custom development and leading economic terms, to next-generation technologies and pass those benefits along to customers.
We expect to continue to invest in and support the development of emerging technologies in this space. In certain areas where Ginkgo has unique needs, we may acquire technologies directly, as we did with Gen9, Inc.’s DNA assembly platform, which was particularly valuable for more complex DNA synthesis needs. In many other areas, we will support new and existing technology companies by placing anchor orders and partnering to develop technology roadmaps that break new ground.
By acting as a horizontal platform, Ginkgo can focus on what we do best (cell programming), our customers can focus on what they do best (bringing products to market in their industry), and our suppliers can focus on what they do best (building great hardware and tools). Biology did not evolve by industry and so cell programming is able to benefit from the scale and efficiency of a horizontal platform. Vertical integration is no longer required, allowing each layer of the ecosystem to flourish as we collectively enable more rapid growth across the industry.
Enabling Customer Success
Ginkgo serves diverse customers across a variety of end markets. Some of these customers may have in-house biological R&D teams and others may have never thought biotechnology applied to their business. In either case, they come to us with a challenge—whether it is supply chain volatility, a race to develop an innovative new product, or an existential threat facing an industry on the wrong side of history—and we partner to enable a biological solution. We begin our relationship by working collaboratively to design the set of specifications for the end product(s) our customer desires. Our cell programmers then take that set of specifications and design an engineering plan to create a cell program that meets or exceeds that set of specifications. When we finish, our customers receive the final engineered organism (which either produces or is their product of interest) and a full “tech transfer” package for manufacturing and downstream processing (which they can implement themselves or pass to a contract manufacturer with our support). Our customers then take these organisms and/or purified products through the final stages of product development (e.g., formulations, clinical trials, field trials, etc.).
Our commercial team is organized to both establish new relationships with potential customers (traditional business development) as well as maintain and expand relationships with our existing customers (which we call “alliance management”).
Our business development team has both expertise in relevant industries (Consumer & Technology, Industrial & Environment, Agriculture, Food & Nutrition, Pharma & Biotech and Government & Defense) as well as expertise in our Foundry capabilities and synthetic biology. With this background we are able to identify industry or consumer challenges where biology can serve as a solution. Our categories of customers, independent of industry, include potential customers who have R&D teams with some synthetic biology capabilities where choosing Ginkgo can bring automation, scale, and codebase beyond their own; potential customers who are considering but have not yet built lab-scale capabilities where a partnership with Ginkgo allows them to spend their capital on commercialization efforts; and potential customers who are not yet working in synthetic biology whose industries or products stand to be disrupted by biological solutions. Our business development team, with support from our Codebase and Foundry team members, crafts solutions for each of these types of customers through a strategic discussion of customer needs and fit with Ginkgo capabilities.
To grow existing customers, our alliance management team, through close collaboration on our existing programs, seeks technical and business opportunities for our customers that serve as the basis for consideration of future programs. As our programs demonstrate technical success, our existing customers often bring their next strategic R&D needs to our attention.
13
Table of Contents
Over 100 Cumulative Programs across diverse industries have run on our platform
While most biotechnology companies focus on building products within a fairly narrow scope, Ginkgo has uniquely pursued a partnered strategy across all end markets. This was not easy. For many years, our platform was less efficient than the status quo of an expert scientist working by-hand at a lab bench. In the early days, the only end markets willing to take a chance on our platform were those without in-house biotechnology capabilities. But as Ginkgo’s platform improved over time and with scale, we were able to win contracts in increasingly sophisticated end markets with more in-house biotechnology expertise. Today, our platform is diversified across all major end markets with marquee customers and a range of focus areas within each.

Figure 8: Cumulative Programs run by third-party customers on Ginkgo’s platform (excluding proof of concept projects and other exploratory work). Today, Ginkgo has a diverse set of programs across all major end markets.
Our customers include large multinational organizations with multibillion dollar R&D budgets as well as startups who are depending on us for essentially all of their bioengineering needs. While these customers and their focus areas may look very different, they are all important and valuable to Ginkgo. All of these programs leverage a common infrastructure, and as we demonstrate the value of this platform, we have the ability to grow significantly with our customers.
Ability to grow with our customers and increasingly complement existing R&D budgets
Ginkgo has grown substantially through inside sales with our existing customers. Some of our customers, such as Motif FoodWorks, never needed to build in-house cell engineering capabilities and so as they grow and expand their product pipeline, we expect their demand for our platform will increase and they will benefit from our improving scale efficiencies over time as well. The relative value of our platform compared to the next best option (building a lab, bioengineering team, and intellectual property from scratch) is immense, which yields extremely high retention rates for customers in this category.
Other customers may already have in-house cell programming capabilities. As Ginkgo demonstrates the value-add of our platform by successfully delivering on programs, we have the opportunity to grow our collaborations with them, complementing their core R&D capabilities. We don’t view this as a “replacement” of customer scientists with Ginkgo’s platform. Rather, we hope to expand our customers’ capacity and need for innovation—giving them more “shots on goal” and enabling them to invest more heavily in R&D as the return on investment of each dollar spent increases.
We have demonstrated this with several customers. With one customer, an initial proof of concept program has turned into a broader strategic relationship with over nine programs today. With another, we launched a relationship with two programs, quickly expanding it to five by the end of the following year. The growth we have seen with our oldest customers means we continue to have significant customer concentration as it takes time for new customers to ramp up their use of Ginkgo’s platform. During 2021, two of our customers each contributed greater than 10% of revenue and collectively they accounted for 28% of total revenue, down from 39% in 2020. We believe customer concentration will decline over time even as we expect to continue to grow our relationships with existing large customers. However, our ability to grow with our customers requires us to maintain satisfied customers, and program or other operational setbacks could impede our ability to meet customer expectations and grow our business.
14
Table of Contents
Powerful proof points across categories
Our platform has now been validated by sophisticated customers across a range of industries. As we launch programs in new areas, those provide a toehold for future sales in that space. As an example, our pro bono project for Moderna, Inc. at the start of the COVID-19 pandemic to enhance production of a key raw ingredient through process engineering provided a proof point and initiated us into this emerging segment, leading to a commercial relationship with another nucleic acid vaccine company, as well as a program to produce a key processing enzyme for mRNA vaccines. Similarly, the technical success of programs with Motif FoodWorks, Aldevron, and Cronos Group in 2021 attracted inbound interest from other potential customers.
It is still incredibly challenging to break into new industries and our ability to expand into new sectors may be harder than we expect. However, our recent progress in therapeutics has been a significant milestone given that we are ultimately competing against very strong in-house capabilities. We believe that as more proof points emerge across industries, the barriers to adoption will diminish.
Our Platform
Ginkgo’s platform combines a strong technical foundation with an ecosystem of supporting resources to maximize our partners’ odds of technical and commercial success. In the nucleus of our platform are our Foundry and Codebase, which our scientists leverage to complete customer programs. The Foundry is, in its simplest form, a very large, highly efficient biology lab, enabled by over a decade of investment in proprietary workflows, custom software, robotic automation, and data science and analytics. It is paired with our Codebase, a collection of biological “parts” and a database of biological data, which helps our scientists program cells. But great technology alone is not enough, and we are building a community and ecosystem around our technical platform that provides our partners with end-to-end support.
Our Foundry brings economies of scale to cell programming
Cell programming projects involve a conceptually similar engineering cycle regardless of the specific product or market. Based on customer specifications, Ginkgo’s program team develops designs of proteins, pathways and gene networks (see Figure 5) that might meet the specification, leveraging public and proprietary biological knowledge bases (see “—Our Codebase—organizing the world’s biological code”). Those conceptual designs are developed using computer-aided design tools until the exact DNA sequences for those designs have been determined. Those DNA sequences are then “printed,” assembled and inserted into a cell to execute the new DNA code. These prototype cells are then studied and the output or performance of each is measured and compared to the customer’s desired specification. Learnings using data analytics and data science tools inform a new round of prototypes and this cycle is repeated until either the specification has been met or the customer decides to end the program.
The likelihood of technical success increases with each iterative engineering cycle and with the number of prototypes that are explored per cycle. However, with traditional tools for genetic engineering, each of these cycles can be slow, expensive and error prone. Many projects across the industry run out of budget or time. Conventional R&D teams often look to stay within budget by running rapid engineering cycles using largely manual tools and small numbers of prototypes per cycle. However, the inability to broadly explore the potential design space (there are more possible sequences of a 200 amino acid protein encoded in 600 DNA letters than there are stars in the observable universe) and the reliance on manual tools is a difficult handicap to overcome. Since people can only work so hard and since engineering cycles can’t be shortened beyond the duration of the physical steps, this approach has limited potential to improve in the future.
At Ginkgo, we invest in improving the tools and technology for programming cells in order to maximize program success within the constraints of customer timelines and budgets. We do so by scaling the number of prototypes that can be evaluated in each engineering cycle in an effort to reduce the number of cycles required to meet the customer’s specification and ultimately shorten project timelines. A typical screen for one enzyme step in a program might evaluate 1,000 to 2,000 variants to optimize function, of which the top 10 to 100 might be short-listed for further study. A relatively basic program might have 3 to 5 enzymes working in concert, and so in the process of optimizing the entire pathway, thousands or tens of thousands of enzymes and pathway combinations might be designed, built, and tested in the Foundry. The methods we use to increase scale also tend to reduce the average cost per prototype, which means that more prototypes can be evaluated for a given program budget.
Because diverse cell programs share similarities in process and code, many programs can be run simultaneously in a carefully designed centralized facility. This facility, where we use our investments in advanced cell programming technologies to manage diverse programs, is what we call our Foundry.
15
Table of Contents
We make it possible to centralize many cell programming projects in our Foundry by deconstructing programs into a set of common steps and then standardizing those steps. For each step, we have built a specialized functional team that performs that step for all programs. Those teams define a set of standardized services that can be used in concert to execute an end-to-end cell programming process. Each team has access to scientific, software, and robotic engineering resources to replace manual ad hoc operations with standardized, automated, and optimized services. In addition to enabling scale, this approach ensures standard operating procedures, know-how, and human skill become encoded in software that can be more effectively debugged, monitored, controlled, and optimized.

Figure 9: A non-exhaustive summary of the functions performed throughout the lifecycle of a program in the Foundry. At each stage, learnings are generated, driving improved design cycles and functional optimizations.
While the engineering strategies described above have historically been relatively uncommon in the life sciences, they are obviously not our invention. Rather, we are inspired by the lessons from other engineering disciplines and seek to apply those to biology. Automotive manufacturing, semiconductor fabrication, and data centers, among many other industries, demonstrate how automation, data, economies of scale, and continuous improvement can produce compounding gains in scale, costs, and quality. Critically, routine performance of these strategies across dozens of projects gives us the data and experience needed to drive continuous improvement.
As described above, a key strategy in our Foundry is to increase the scale of our operations so that we can run more programs and more prototypes in parallel (i.e., large batch sizes). This approach benefits from operational efficiencies and economies of scale across many dimensions:
These efficiencies and economies of scale can be observed empirically from a relationship we refer to as “Knight’s Law,” named after Tom Knight, one of our co-founders, and loosely inspired by Moore’s Law for semiconductors. As shown below, we have seen an exponential increase in the output of the Foundry over time alongside an exponential decline in the average cost per unit of output. While this trend was interrupted by temporary lab shutdowns during the COVID-19 pandemic and reduced capacity due to social distancing, we have since continued to drive our business towards achieving these metrics.
16
Table of Contents
While Knight’s Law does not provide the full story on our development, it is a useful tool that allows us to continue to build efficiencies of scale. We continue to build our internal metrics around Knight’s Law and believe we can continue to drive this kind of capacity growth in the foreseeable future, though it is dependent on the development of new technologies, which inherently carries risk, and, like Moore’s Law, we will likely hit a limit over time. This feature compares to a conventional facility, where scaling is driven predominantly by the addition of employees, an exponential increase in work would be infeasible and the cost per unit of work would decline little, if at all.

Figure 10: The output of the platform increased by over 3X per year for 5 years (with the exception of 2020), and while we expect that kind of scaling to continue, there is no guarantee that we will be able to do so.
17
Table of Contents

Figure 11: As the output of the platform has increased, our total R&D / operational costs per unit of output has decreased by approximately 50% per year.
We are frequently asked, and spend much time thinking about, whether it will be possible for compounding gains in output and productivity to continue for many years in the future. It is important to note that given significantly advanced tools, most steps in cell programming could be miniaturized to a point where single molecules of DNA and single cells are being manipulated and monitored. At that ultimate degree of miniaturization, the costs and timelines of cell programming could be reduced orders of magnitude from where they are today. Newly available microfluidic technologies, such as those developed by our partner, Berkeley Lights, Inc. (“Berkeley Lights”), point to the reality of this future of cell programming at the single-cell level. Additionally, because many of the enabling tools of cell programming are biological in nature (e.g., polymerases and CRISPR), we are able to point the platform at itself, developing new biological tools to reduce the number of steps or the complexity of a certain operation. For example, we could develop better gene editing enzymes or novel ways to screen cells in a multiplexed format using biological sensors. It is easy to theorize about these types of developments, however they are hard to execute, we will undoubtedly run into roadblocks along the way and we will have to invest significantly in developing new technologies in order to enable the types of improvements we seek to achieve.
Recent advances in machine learning, molecular simulation, and other computational techniques also hold great promise to improve our ability to program cells. We believe our Foundry is well-positioned to build the kind of large, well-structured datasets that such computational approaches need to succeed. In time, we believe computational approaches will reduce the need for certain kinds of experiments (for example, we already use machine learning to make protein and enzyme design projects more efficient). If computational approaches can replace certain sets of experiments, we expect to use the recovered Foundry capacity to work on ever-more complex cell programming challenges. The reality is that the cells that we program today accomplish relatively simple functions, such as: “produce as much of molecule X as possible.” Programming cells for complex functions, such as live-cell therapeutics, responsive building materials, multicellular organisms, etc., will require sophisticated sub-systems for environmental sensing, intracellular information processing and feedback, and a multidimensional program that responds to such environmental stimuli. Only when we can deliver such sophisticated programmed cells will we have truly unlocked the potential of biology, and we see the Foundry as being an integral part of the platform for doing so.
Our Codebase—organizing the world’s biological code
Codebase is a familiar term to software developers but is a new concept in biology. Modern software firms develop their own (typically proprietary) codebase of source code and code libraries that can be leveraged by their software developers to more easily create new applications than they could starting from scratch. Additionally, vast repositories of debugged code are
18
Table of Contents
shared publicly so that programmers across application areas can leverage prior art in order to innovate faster. This allows software developers to focus their time and effort on developing new features rather than recreating existing logic. Ginkgo’s Codebase is comprised of reusable genetic parts and strains that can be repurposed in new cell programs. We are continually investing in better ways to characterize functional biological code to drive increased reusability. In addition to the raw performance data we generate through our Foundry experiments (more than 24.8 million strain tests run as of December 31, 2021), we have also incorporated many public databases for genetic sequences and have a proprietary data set of over 3.8 billion additional sequences that we leverage in our designs.
Engineering biology is complex—one of the reasons that Foundry scale is important is that it remains highly difficult to predict the performance of a biological “part” in a given context from a DNA sequence alone. The genomics revolution has outpaced biologists’ ability to test the functionality of each DNA sequence as it was discovered, particularly because most of the community is still performing biological experiments by hand without the benefit of automation. Each program performed at Ginkgo involves testing thousands or millions of DNA sequences; with a small fraction of those ending up in our final engineered cells. For that reason, high-performance biological sequences—the handful of designs from thousands of candidate designs that meet our performance goals for an experiment—are hard-won assets and form a key component of Ginkgo’s Codebase. Not to be discounted, the “losing” designs are still valuable, helping inform more effective campaigns in the future that avoid known failure modes.

19
Table of Contents
Figure 12: Our Codebase incorporates both biological assets from nature as well as engineered assets and data from our Foundry experiments. Because the Foundry enables us to test many thousands of prototype enzymes, pathways, and strains in individual engineering cycles, we are able to quickly expand the range of characterized biological assets in our Codebase.
In some ways Codebase is a “parts catalog” that we can draw from when developing a new organism. As Ginkgo performs more projects, we contribute new parts to our Codebase that can be reused in new contexts. For example, we developed novel synthetic promoters (DNA sequences that can turn on the expression of a gene of interest) that allowed us to increase production of proteins in yeast. Initially, we tested thousands of designs to arrive at a select number of promoters with high performance. Now those high-performing promoters can be reused in any program that involves producing a protein in yeast; they are a modular piece of genetic code. Over the past 20 years, our team has supported efforts to build these kinds of parts libraries—the International Genetically Engineered Machine (“iGEM”) Parts Registry and AddGene are two notable examples of initiatives to make reusable parts available to researchers in the community. But despite these efforts, we continue to see intellectual property siloed within organizations across the biotechnology industry, leaving many without the additional intellectual property they need to develop their programs. Ginkgo’s Codebase allows our customers to draw from a broader set of biological assets than any single company would develop for a given application. The scale and diversity of our programs have allowed us to develop a large Codebase that grows with the addition of each new program and can be opened to the broad swath of partners and cell programmers using our platform.
Cell programmers must consider not only the genes in the programs that they design, but also the ways that they interact with the cell that “runs” the program. Therefore, Codebase is more than just the individual modular parts we use to design biological programs. The organisms that have been optimized to run the programs, whether because they have been engineered for robust growth or because they are particularly adept at producing certain classes of products, are known as “chassis” strains. These strains can be reused across multiple programs, significantly reducing the amount of work needed to optimize a program and engineer a commercially viable organism. The breadth of Ginkgo’s customer base allows us to use these chassis strains in many more contexts than traditional industrial biotech players.
For example:
Our Foundry and Codebase are inextricably linked. Our Foundry scale allows us to generate unparalleled Codebase assets. These Codebase assets help us improve our designs and provide reusable parts and chassis strains that improve the efficiency
20
Table of Contents
and probability of success of our cell programming efforts in the Foundry. As the capabilities of the platform improve, it drives further demand, which increases the rate of learning in our Codebase. The continuous learning and improvements inherent in this relationship is one of the key features of our platform.

Figure 13: We believe our initial CDKs can help cell programmers build proof-of-concept strains faster.
An ecosystem to support cell programmers
Ginkgo has long recognized that it is critical to build a true ecosystem around our technical platform. We have been inspired by the leading horizontal platforms in information technology, such as Microsoft Windows and Amazon Web Services (“AWS”), which built real developer communities and provided a range of value-added services on top of their core technology. Like these pathbreakers, who set the stage for a generation of computer developers, we too are trying to ensure that the cell programmers who build applications on our platform have the tools they need to succeed beyond the lab.
21
Table of Contents

Figure 14: Ginkgo strives to create an ecosystem to ensure that cell programmers have the tools they need to succeed.
Access to capital
As in the early days of computer programming, it is still extremely expensive to program biology. For that reason, it can be easier for larger companies to make investments in innovation around this space. But Ginkgo’s platform gives small companies and innovators access to the same horsepower as larger players and obviates the need to invest in fixed laboratory assets, providing an even greater strategic benefit. To help address this discrepancy, Ginkgo has assisted in launching new companies (such as Motif and Allonnia) by bringing together strategic and financial investors to secure funding for these early stage companies. Going forward, we intend to leverage our own balance sheet and to partner with investors, enabling companies at all stages to benefit from our platform. We believe that, as Ginkgo’s customers demonstrate increasing success, there will be an explosion of capital for cell programming applications and a recognition of Ginkgo’s platform as setting the industry standard and providing the backbone for these development efforts.
Manufacturing support
Our job is to ensure that our cell programs can be executed at scale and we support our customers to ensure successful commercial scale manufacturing. We have built relationships with a number of leading contract manufacturing organizations and have demonstrated that we can transfer our lab-developed protocols to commercial scale (e.g., 50,000+ L fermentation tanks) with predictable performance. We have an in-house deployment team dedicated to supporting our customers’ scale-up and downstream processing needs. We have even helped certain customers, such as Cronos Group, Inc., acquire and build out their own in-house manufacturing capabilities and certain programs, such as our work with Moderna, Inc., focus on manufacturing process optimization.
Intellectual property protection and regulatory support
Ginkgo takes responsibility for the cell engineering intellectual property generated through customer collaborations. Our scientific team works continuously with our intellectual property team to file patent applications and monitor for freedom to operate. We are also active in helping shape and influence the evolving regulatory landscape for biological engineering. While our customers are responsible for handling their own regulatory procedures on a product-by-product basis, our broader view and sphere of influence can help build understanding of and support for novel product classes.
22
Table of Contents
Building a community of cell programmers
We launched Ferment, our annual conference, in 2018. The conference highlights developments and thought leadership in the field and brings together scientists, entrepreneurs, investors, and suppliers. Our conference in 2021 brought over 700 participants to our headquarters in Boston. Even prior to launching Ginkgo, our founders focused on building community within the emerging field of cell programming. Tom Knight, one of our founders, was among the professors who launched the iGEM Competition in 2004, which has now had over 70,000 participants from over 40 countries take part in the competition (including dozens of Ginkgo employees and all five founders!).
Facilitating partnerships within our community
Because Ginkgo serves both large market incumbents and smaller startups, our community also serves to facilitate introductions between innovators and those looking to invest in innovation. We believe that investors and large strategic companies have come to recognize Ginkgo’s platform as a key enabler of innovation and are keen to get to know the companies that are building with us. Those relationships can be the source of funding and go-to-market support for the earlier stage companies building on the platform, increasing the odds that they develop successful products.
We invest in building trust and credibility for the entire industry
The most powerful technologies require the most care. Biology is too powerful for us to not care about how our platform is used. We have and will continue to invest heavily to build and maintain trust in bioengineering as a technology platform across all layers of the industry. At the platform layer, we have focused on building robust biosecurity measures. At the application layer, we are proud to enable a diverse set of programs that drive towards environmental sustainability. We are committed to ESG practices and broad stakeholder engagement at a corporate level. We are also engaged in deep conversations around the implications and ethics of biotechnologies through many public forums, helping shape our platform to promote sustainability in our global community.
Biosecurity: A complement to our platform and demonstrated source of value
With a mission to make biology easier to engineer, we have always recognized the imperative to invest in biosecurity as a key component of our platform. We care how our platform is used, and biosecurity is a necessary complement to synthetic biology that helps us ensure our cell programming work is conducted and deployed responsibly. Consideration for biosecurity is ingrained into our cell engineering platform tools, as evidenced through our membership in the International Gene Synthesis Consortium (“IGSC”), and our application of their harmonized protocols to screen synthetic DNA for concerning sequences, as well as our ongoing work with national defense agencies aiming to develop experimental and computational tools that detect or prevent misuse of bioengineering.
During the COVID-19 pandemic, we collectively witnessed the disruptive effects of a biological threat. The pandemic created a renewed sense of urgency for the need to counter biological risk, and triggered a paradigm shift for biosecurity in public and private institutions. Here at Ginkgo, this pandemic has deepened our resolve to advance global biosecurity and accelerated our capabilities to do so. At the start of the pandemic, we made a commitment of $25 million of free access to our platform for partner COVID-19 projects and launched a partnership with Moderna on process optimization for mRNA vaccine manufacturing. Since then, we’ve worked with Aldevron to deliver a breakthrough in the manufacturing of vaccinia capping enzyme for mRNA vaccines, and we’ve advanced discovery of therapeutics. We’ve also participated in the largest wastewater sequencing effort in the United States, supporting Biobot Analytics and the U.S. Department for Health and Human Services (“HHS”) with genomic sequencing capacity.
Most strikingly within the biosecurity space, we’ve grown Concentric by Ginkgo into a trusted brand and successful business as a large-scale systems integrator to facilitate COVID-19 testing in communities across the country. We tackled a moment of global crisis head-on through the growth of an innovative, scalable, community-centered apparatus for robust health security.
23
Table of Contents

Figure 15: Concentric’s pathogen monitoring network has conducted testing across most of the U.S. Map shading reflects all states in which Concentric has delivered testing since launching in 2020. Not all states have active testing programs as of March 2022.
Through this pathogen monitoring network, we've empowered communities to make informed public health decisions through the many waves of the pandemic. Our tools provide early warning of infection and provide communities with tools that can mitigate outbreaks. We’ve worked closely with national, state, and local public health authorities and educational leaders to make sure that our programs are aligned with broader COVID-19 guidance and mitigation strategies, and are capable of delivering timely data to inform public health decision-making. As a systems integrator, we developed customizable, low-burden programs based on the particular needs of educators, students, parents, guardians, and community members, and grew a world-class customer service system to support each local program in context. This work with educational institutions—organizations that represent the centers of our community—is a meaningful first step in building the pathogen monitoring capabilities critical to the prevention and mitigation of biological risks.
As we sought to evolve our COVID-19 monitoring platform to serve communities in other contexts, we developed strategic partnerships to complement our operational and technical expertise.
We are continually exploring where public and private partnerships can help us scale our platform and further innovate in pathogen biomonitoring.
Our dedication to biosecurity is deeper than our emergency response to the current global pandemic. SARS-CoV-2 will not be the last pathogen we face with pandemic potential, but if we take the necessary steps, it may be the last that catches us
24
Table of Contents
unprepared. Ginkgo has been supporting and engaging with domestic and international organizations and governments to help shape the understanding of a robust biosecurity program beyond the COVID-19 pandemic, and we believe there is a meaningful commercial opportunity in such a program driven by increased awareness of the need for prevention and response systems. Given our experience to date, we believe we are well placed to take a leadership position as the biosecurity platform of choice. If we are to harness biology to engineer products for our health, food, energy, and environment as we drive the growth of a sustainable bioeconomy, we must also move with the same speed and vigor to secure biology.
Our Business Model
The key input into our unit economics is a cell program. For each of these cell programs, we generate economic value in two primary ways. First, we charge usage fees for Foundry services, in much the same way that cloud computing companies charge usage fees for utilization of computing capacity or contract research organizations (CROs) charge for services. Additionally, we negotiate a value share with our customers (typically in the form of royalties, milestones, and/or equity interests) in order to align our economics with the success of the programs enabled by our platform. As we add new programs, our portfolio of programs with this “downstream” value potential grows. Because we typically incur no downstream costs (e.g., manufacturing or product development, which our customers manage), these value share payments flow through with approximately 100% contribution margin. This flexible business model allows for more predictable near-term revenue while not sacrificing our ability to create long-term value with asymmetric upside.
Foundry Revenue
Illustrative Program Economics
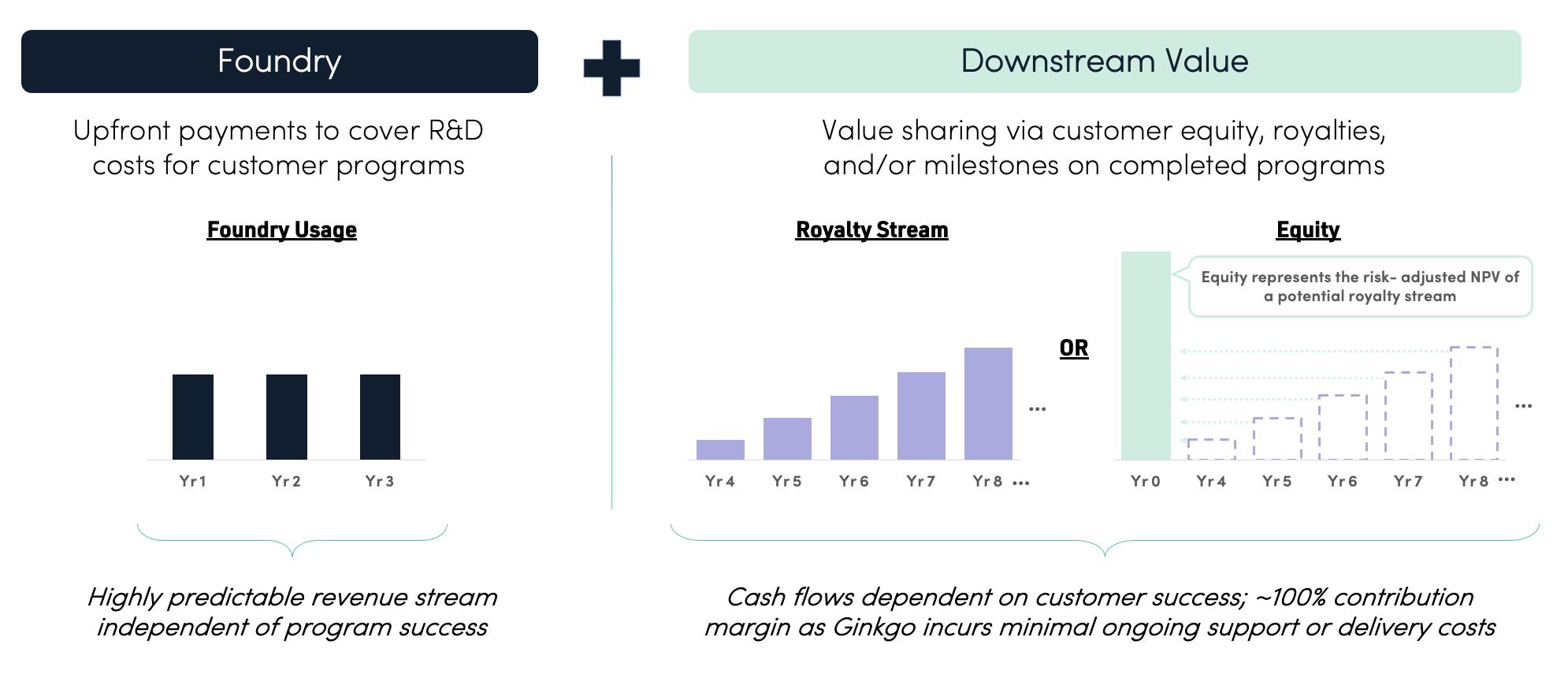
Figure 16: Ginkgo generates economics from programs in two primary ways. First, customers pay upfront fees to cover initial R&D costs for a program. Second, Ginkgo shares in the downstream value (typically in the form of a royalty stream, milestone, and/or equity share) generated by programs.
Foundry Usage Fees
The first stage of a cell program consists of R&D work being performed on Ginkgo’s platform, leveraging our Foundry and Codebase. R&D is inherently risky and our customers recognize that this is a cost they will incur regardless of success and whether they are working on the program in-house or with a partner. Ginkgo provides a much more efficient platform to conduct this R&D work, encouraging companies to build on or adopt our platform.
We estimate that the unit costs of our Foundry cell engineering services are several times less expensive on average than the status quo (a customer doing equivalent R&D in-house, by-hand) and we expect that cost advantage to grow over time. We typically earn usage fees tied to the units of work that we perform on behalf of our customers’ programs. Initially, as we were building and validating the platform, these fees covered less than 20% of the costs incurred to execute a program as the platform was less efficient than the status quo. As our platform has matured and efficiency improved, we have steadily increased the portion of program R&D costs that are covered upfront by customers and we now expect that new programs are
25
Table of Contents
structured to fully cover our direct costs, which will eventually enable us to earn a modest margin. Foundry usage fees provide a strong foundation of predictable revenue that is independent of any commercialization efforts by our partners.
As we continue to scale the Foundry and build Codebase, we expect to drive further efficiencies and decrease our average unit costs. This presents us with a strategic choice going forward. We could retain these efficiencies and increase our margins or we could pass these efficiencies on to our customers, increasing the number of shots on goal and, therefore, the likelihood of program success given a fixed budget. We believe the right choice for long-term value creation is to pass the savings to our customers, reducing the barriers to adoption and driving increased demand for our platform. Our Foundry usage fees are thus impacted by a number of drivers:
The expected impact of these drivers is represented below:

Figure 17: Illustrative drivers of Ginkgo’s long-term financial model and customer value proposition.
The multi-year nature of an average cell programming project means that our usage fees are predictable and recurring in nature. Additionally, given the lead times inherent in developing technical plans as part of a sales process, we have good visibility into new Foundry usage fee bookings. This provides a strong foundation for the business and allows us to be patient while we wait for downstream economics.
Downstream Value Share
As the key enabling technology for our customers’ products, we are able to earn a share of the value of the products that are created using our platform. We are quite flexible and have structured a variety of value sharing mechanisms, including royalties, equity, and lump-sum commercial milestone payments. Because the economics to us should be roughly equivalent, we are generally agnostic on which form of downstream value capture we receive and the decision is typically based on customer size and preference, with archetypes described below.
26
Table of Contents

Figure 18: Illustrative of the forms of downstream value capture received by Ginkgo, based on customer size and preference.
Because Ginkgo typically will have completed the program (and received associated usage fees) prior to realizing downstream value, cash flows from the downstream value capture component generally fall straight to the bottom line as we incur minimal to no ongoing support or delivery costs once the strain is commercialized. This dynamic creates opportunities for outsized returns as our clients successfully commercialize products built on our platform. As we add more programs to the platform over time, we expect downstream value share to contribute income, and therefore we believe our overall margins and cash flow profile will grow significantly.
Biosecurity Revenue
In the second quarter of 2020, in response to the COVID-19 pandemic, we launched our commercial offering of COVID-19 testing products and services for businesses, academic institutions, and other organizations in which we generate product and service revenue. We generate product revenue through the sale of lateral flow assay (“LFA”) diagnostic test kits, polymerase chain reaction (“PCR”) sample collection kits and pooled test kits, all of which we sell to our customers on a standalone basis. We generate service revenue primarily through the sale of our end-to-end COVID-19 testing services which consist of multiple promised goods and services including sample collection kits, physician authorizations, onsite test administration, outsourced laboratory PCR analysis, and access to results reported through a web-based portal.
Beginning in the first quarter of 2021, we launched our pooled testing initiative which focuses on providing end-to-end COVID-19 testing and reporting services to public health authorities. We are currently offering pooled testing and reporting services for K-12 schools across the United States, at airports through our partnership with XpresCheck and the CDC, as well as through other congregate settings such as our partnership with Eurofins. In the future, we believe that testing services may have a value proposition internationally and in other use cases including wastewater monitoring and air monitoring.
Our Sustainable Advantage
We have defined a unique business model over the past 13 years. The biotechnology industry has been product-centric for decades, with early horizontal platforms in life sciences frequently vertically integrating upon the development of the first successful product on their platform. As Ginkgo has embarked on this journey, we have studied and learned from innovators and established platform companies in other industries as we built our platform and business. We now benefit from significant historical investments, a virtuous cycle that grows with scale, and a strong business model that is aligned with our customers’ outcomes. These establish a strong sustainable advantage that we believe will help establish Ginkgo as a true industry standard.
Decade-plus head start in creating an industry standard platform
Hardware, software and biological tools need to be tightly integrated to replicate our platform. We have spent over 13 years building the software, automation and data science to best support a high throughput, generalized platform and expect to continue investing in this area. Our software, automation and data infrastructure cannot be easily replicated without bringing together a number of rare, specialized skillsets. In addition, without the scale and demand to stress test a high throughput platform, we expect any newly developed platform would be suboptimal. We estimate that it took us over eight years of investment and iteration to reach cost parity with “by hand” cell programming. We believe competitors will find it difficult to
27
Table of Contents
justify the investment in the software, automation and data science needed for high throughput operations before they acquire matching high demand.
Scale economics provide a structural cost advantage
As the only scaled horizontal platform in this space, we have the broadest number of programs that can be run on our platform, providing the highest potential for scale economics. Other companies choose to target specific markets and vertically integrate into products with high expected value. This has a tendency to overfit the capabilities of their R&D team to their targets. As discussed above, our continued scaling and investment in flexible tools that can apply to a broad range of end markets helps us drive efficiencies in the Foundry and Codebase across our diverse programs. Furthermore, as we scale, we are able to leverage advanced technologies that are only practical at scale and also may obtain preferred pricing with a number of suppliers. Competitors may be unable to source equivalent technology or negotiate similar pricing without first achieving scale, a feat that is difficult to do with a narrowly focused R&D platform.
Strong network and learning effects
In addition to a raw scale economic, we also accumulate knowledge and reusable Codebase from each program that runs on the platform. Every program benefits from the programs that came before and generates benefits for other current and future programs. These learnings and reusable assets are cumulative, extremely hard to replicate, and increasingly valuable to our customers. Because our learnings are generated by the work we execute in our Foundry, the scaling in our Foundry drives a scaling in our rate of learning. Thus, there is a recursive element to our platform: as the platform gets better, it also improves faster—we are excited to make this advantage of our platform available to our ecosystem of cell programmers.
Ginkgo’s value creation is aligned closely with customer success
Our platform drives value for customers along two dimensions: reducing the cost of laboratory work via automation and increasing the probability of technical success due to cumulative data and learnings. Our financial model is aligned with those factors. As we gain efficiency, we drive further demand for cell programming, which drives our Foundry revenue up. As both demand and probability of success increase, our risk-adjusted value share also increases. Our model only requires we share in a small fraction of the downstream value created by our programs, providing our customers the opportunity to generate and retain significant value. Ultimately, this encourages broader adoption of our platform across industries.
Furthermore, we seek to maintain close relationships with our customers, supporting their work, and earning their loyalty and satisfaction. The breadth and highly integrated nature of our platform makes it inefficient for a customer to simultaneously work with Ginkgo and any theoretical competitor. As there is not yet a standard interface for cell programming, it requires an upfront investment to learn how to choose and design programs to make the best use of our platform. Thus, customer retention is high and there are substantial switching costs.
We are uniquely positioned to attract the top cell programmers
Just as the top software programmers want to work with the latest technologies, we believe the top cell programmers will be attracted to our industry leading platform and access to its unique capabilities. Our ability to hire and retain the best cell programmers as internal users and developers of our platform pushes us to continually improve and also builds a base of Ginkgo-trained experts. If these Ginkgo trained cell programmers move on to roles and opportunities in product-specific companies, we expect they will become ambassadors for the Ginkgo approach in their next role, expanding our reach into potential customers.
History of investing in credibility and trust
Let’s face it, GMOs have an image problem. This image problem has led to activities by the first generation of genetic engineering companies that backfired: lobbying against transparency in labeling laws, trying to “rebrand” GMOs with different terminology, and other efforts that have failed to build trust and engagement with stakeholders. We have taken a different approach. Rather than avoid the term, we’ve championed transparent labeling, sought to engage and build trust through open dialog, and enthusiastically embraced the potential for GMOs to do great things. We don’t seek to make GMOs acceptable through branding; we aim to make GMOs that people love.
28
Table of Contents

Figure 19: Ginkgo seeks to make GMOs that people love.
Doing so requires care and attention to both the technical and social aspects of our platform and its impacts. This means investing in biosecurity and, as noted above, embedding it into our platform and how we operate. This also means engaging with the social complexities of science and technology with a diverse group of people. We strive for a company culture based on a foundation of Diversity, Equity and Inclusion (see also the sections titled “—The Impact of Cell Programming—ESG is in our DNA” and “—Our People & Culture”), and aim to engage different perspectives through our creative residency and through our magazine, Grow. Through both our internal and external efforts, we seek to engage with the realities of what has made genetic engineering an ESG risk historically, and work towards equitable and positive impact.
Our Growth Strategy
We are seeking to usher in a new paradigm for cell programming. It took us over eight years of basic research and investment in software, automation, data science and scale to reach parity with the status quo of individual scientists conducting experiments by hand at a lab bench. It took us several more years to demonstrate business model maturity: delivering a platform with enough value-add to customers that we could cover the cost of cell engineering R&D programs while building Codebase and sharing in the downstream value of our programs. We believe that we are now at an inflection point where we believe we have the opportunity to become the industry standard. We see several drivers of this evolution and growth.
Scale our platform and continue to drive efficiencies and improvements
As discussed above, our platform improves with scale and to date we have observed a positive feedback loop between our Foundry and Codebase. As we scale capacity and demand on the Foundry, we expect our average unit costs to fall, creating a better value proposition for our customers as their program budgets stretch further and drive more demand. Similarly, Foundry output also grows our Codebase, which supports better program execution, creating a better value proposition for our customers as well.
We occupy over 300,000 square feet and maintain state-of-the-art machinery and laboratory equipment. We have built more than 50 custom integrated work cells, consisting of robotic automation systems, mass spectrometry, fermenters, sequencers, and more. We have the capabilities to engineer dozens of species of organisms from bacteria to fungi to mammalian cells. We have worked on enabling products as varied as polymers, bacterial therapeutics, bulk protein production, novel antibiotics, fine chemicals, and more.
We have been able to work on a diversity of programs while consistently driving efficiencies in the Foundry with scale. We expect to accelerate growth in capacity by integrating new technologies across our existing footprint, building new Foundry space, and investing in software, automation and data to increase utilization.
Leveraging our proof points to grow within all industries
We have now established proof points of success in a diverse set of end markets, in several cases far exceeding our customers’ specifications. When engaging with existing customers or potential new customers in similar or adjacent industry
29
Table of Contents
verticals, we can point to these case studies of success to demonstrate the value of our platform. This reduces the barriers to adoption, helps us grow our customer base, and increases the number of new programs under contract. Importantly, the reusable Codebase we generate from these new programs enables us to stay ahead of vertically focused competitors.
Grow with existing customers
Once we establish a relationship with a customer, there is significant room to expand the scope of our program engagements. We are able to grow with our customers and/or expand into other existing pockets of R&D spending. We have seen customers expand from one early program to five or ten programs a few years later and each new logo we add has the potential to become a true platform partner.
When we work with companies from their inception (or at least from the inception of their biotech investments), we enable them to avoid significant fixed cost investments and benefit from our economies of scale. Our relationship with these customers is extremely strong, as we are the core technology powering their R&D efforts. As a result, when these customers scale, their usage of our platform typically scales commensurately. For companies with existing, established biological capabilities, as we demonstrate the value of our flexible platform, we are able to grow our relationships to complement their core capabilities and increase the probability of success.
Reduce barriers to adoption by integrating with external R&D teams
It can be easy to fall into the trap of assuming that new disruptive technologies must subsume existing ways of working. When hosted servers and SaaS started rising in prominence, corporate IT teams had to wrestle with changing integrations and demands. Some information technology departments were resistant to moving “off-prem” because they felt they were effectively outsourcing their jobs. In response, the leaders in this field, such as Dell, would sometimes hire their customers’ information technology departments and find them jobs within Dell simply to get past this internal resistance. The reality was that these technologies were ushering in a much more substantial era for information technology, which dramatically increased the demand for this type of talent. This centralization of the model (from every company having large information technology departments building customized code to a broader array of specialized software vendors) didn’t come at the expense of information technology and digital technologies, but enabled its flourishing across all industries. We see something similar happening in biotechnology today. Internal R&D teams are typically both very excited to learn about the power of our platform but are also understandably nervous about what “outsourcing” work to Ginkgo might mean for the future of their teams. We have the opportunity to help them see the benefit in a true partnership with Ginkgo.
The vast majority of programs being run on the platform today are being run and managed by Ginkgo program teams—in-house scientists and engineers who are managing the R&D project to meet a customer’s specifications. But we now have a couple early examples of certain customers, those with more in-house biotech expertise, interacting directly with our platform. Over time, we would like to build in enough standardized interfaces that a distributed network of scientists could access the platform directly through a well-defined integration and self-service layer. This transition will allow our program teams to devote more of their efforts to developing Codebase assets, enabling more rapid scaling, and reducing the barriers to adoption by our customers. There are significant technical hurdles for us to overcome in developing this technology, but it is on our near-term roadmap and we are constantly thinking about how to “productize” individual workflows on the platform.
Build an ecosystem
As described above in “—Our Platform,” we believe we are building the industry standard developer platform for cell programming. In much the same way that early computing platforms and operating systems built real communities around their platforms in the 80s and 90s, we intend to build a community of developers building on the Ginkgo platform. As we invest to expand this ecosystem of services for cell programmers building on the Ginkgo platform, our value proposition to cell programmers increases and we become more ubiquitous.
Our People & Culture
A company is made of people. We have sought to bring together a diverse and multidisciplinary group of people who share in our mission to make biology easier to engineer. Today, our extensive cross-functional team is collaborating to build our ecosystem, from organism designers to automation engineers, software developers to people operations, business development to facilities management, finance to molecular biology.
30
Table of Contents
A culture built on care
We’ve strived to grow a culture based on care. As engineers, it is easy to fall into the trap of thinking of ourselves simply as tool builders. Tools can be used in many different ways, both good and bad, and engineers often discuss their tools as value neutral. But tools reflect the social beliefs and biases of the people who make them: today this is becoming increasingly apparent, with more and more evidence of algorithmic bias being built into AI systems, facial recognition, and much more.
As designers of the largest horizontal platform for cell programming, we are keenly aware of the need to care about how our platform is used. More significant than the impacts we have seen from digital platforms on our social world, biology is our health, our bodies, our food, and our environment. As we build the tools for programming biology, we must also care how those tools are used, and ensure that the risks and benefits are transparently and equitably shared.
A diverse, world-class team
As of December 31, 2021 we had 641 full time employees. Building a horizontal platform for cell engineering and a biosecurity offering requires collaboration between diverse skills and functions. It also requires deep technical expertise. Our employees are dedicated to the following functions:
In addition to our full-time employees, our success would not be possible without the collaboration and support of the broad network of partners, contractors, contingent workers and temporary staff who make up the Ginkgo team.
Technologies reflect the values of the people who build them. Diversity, Equity, and Inclusion are valuable and necessary in their own right, but we believe that it is essential to build a diverse team where people from different backgrounds are included and empowered to speak up and shape the growth of this technology. We are committed to growing a diverse team and continuing to empower an inclusive culture with strong employee ownership and engagement.
The full breadth of Ginkgo’s diversity and inclusion cannot be captured in demographic statistics, just as demographic categories cannot capture the full spectrum of diversity of human experience; however, we collect and report these numbers for transparency and as a lagging indicator of our efforts. As of December 31, 2021, 43% of our full time employees self-identify as an underrepresented gender (not cis male) and 12% self-identify as coming from an underrepresented racial or ethnic group in science and engineering (Black or African American, Hispanic or Latino, American Indian or Alaska Native, and Native Hawaiian and other Pacific Islander). We are not yet satisfied with these numbers and all teams have objectives around increasing diversity and building a culture of inclusion to ensure that diverse perspectives thrive.
Laying the groundwork for strong employee engagement in the future
As a private, founder-led company, we were able to infuse the organization with long-term strategic thinking. The long-term engagement and mentality of our employees can be seen in our turnover: voluntary attrition is well below the industry average.
The individuals who work at Ginkgo and build our platform care deeply about how that platform is used and the impact our company will have in the world. As of December 31, 2021, our team owned over 30% of Ginkgo shares outstanding, and we hope to maintain the long-term mentality we have benefited from as a founder-led company now that Ginkgo is a public
31
Table of Contents
company. We believe a workforce with strong equity ownership will make the wise decisions needed to build long-term value for our company and build a company whose long-term impacts make them proud. That is why we have implemented a multi-class stock structure that permits all employees (current and future), not just founders, to hold high-vote (10 votes per share) common stock. We believe that our multi-class stock structure will help maintain this long-term mentality and encourage long-term equity ownership by our employees, thereby resulting in increasing employee ownership over time. For more information, see “Risk Factors—Risks Relating to our Organizational Structure and Governance—Only our employees and directors are entitled to hold shares of Ginkgo Class B common stock (including shares of Ginkgo Class B common stock granted or otherwise issued to our employees and directors in the future), which shares have ten votes per share. This limits or precludes other stockholders’ ability to influence the outcome of matters submitted to stockholders for approval, including the election of directors, the approval of certain employee compensation plans, the adoption of amendments to our organizational documents and the approval of any merger, consolidation, sale of all or substantially all of our assets, or other major corporate transaction requiring stockholder approval.”
Competition
To our knowledge, there are currently no other companies that serve all industries covered by our horizontal cell programming platform. The solutions and applications offered by potential competitors vary in size, breadth, and scope, and given our broad set of application areas, we could face competition in many different forms. We also face competition from customers’ internal R&D departments and other research solution providers that largely conduct genetic engineering by-hand. We also compete against companies that seek to utilize synthetic biology technologies to develop specific products or target certain end markets. Additionally, competing platforms may emerge from various sources, including from joint ventures and partnerships between well-capitalized technology and life sciences companies. We identify the following three groups as our principal set of competitors:
The Status Quo: “on prem” cell programming efforts
The main source of competition we encounter is from potential customers choosing to build or maintain in-house cell engineering teams and capabilities. This status quo includes building out laboratory space and then hiring a team of highly trained scientists to conduct research “by-hand” with limited scale efficiencies. Some internal R&D operations maintain a full suite of capabilities and can design, build and test relatively complex pathways while others may have certain internal capabilities and need to outsource other elements to CROs. We believe this is far less efficient for the customer and likely to yield worse outcomes as customers get fewer shots on goal for a given program budget.
That said, it can still be very difficult for companies to choose to trust Ginkgo with their R&D efforts versus building more traditional “on prem” labs. Smaller companies may feel like they’re “betting the farm” on Ginkgo, while larger companies may be sensitive to displacing existing R&D teams. As such, a key focus area for us is reducing the barriers to adoption for the platform by de-risking the upfront investment for earlier-stage companies and by helping larger companies integrate their scientists closely into our workflows and empower their scientists to manage requests directly so we feel more like a resource and partner than a fully outsourced provider. Investing in these areas is a key focus area for us going forward.
Examples of traditional “synthetic biology” companies that have been vertically integrated from their founding with a focus on building products using synthetic biology include Amyris, Inc. (“Amyris”), Zymergen, Genomatica, Novozymes, DuPont, and DSM. Additionally, the vast majority of therapeutics companies that are leveraging genetic engineering have in-house capabilities, including Biogen, Novo Nordisk, Vertex, Regeneron, Bayer, and many others. These companies may be viewed as competitors to Ginkgo because they are creating products, using cell programming, that may compete with the products Ginkgo is enabling for our customers. However, as a horizontal platform, we view these companies not as competitors but as potential customers and focus not on “beating” them but rather on demonstrating our value proposition.
Verticalized cell engineering platforms
Within certain end markets, Ginkgo may compete against vertically-focused biotechnology companies providing cell engineering R&D capabilities to customers within a narrow set of end markets. While we believe the siloed nature of these companies limits their long-term potential, in the near-term, we may have a harder time penetrating those end markets given the incumbent vertical specialists in that space. The vast majority of these companies exist within therapeutic end markets given the history of cell engineering in that field. In theory, the expertise and learnings they develop from work in one field could be leveraged into neighboring end markets if these companies decided to adopt (and invest in) a more horizontal strategy. Examples of these vertically-focused platforms include AbCellera (antibody discovery), Codexis (enzymes), Senti Bio (cell therapy for oncology applications) and WuXi biologics (therapeutics).
32
Table of Contents
Other possible entrants
We may also face competition from new entrants in the market, including well-capitalized technology companies with possible strategic interests in synthetic biology and its capabilities. Such companies may emerge as competitors given their access to capital, capacity to create multi-disciplinary teams across biology, chemistry, computer science and engineering, and flexibility to enter strategic ventures with life sciences companies.
Intellectual Property
Overview: Foundry and Codebase
As discussed above, Ginkgo’s two core platform assets include:
Ginkgo protects each of these core assets—the Foundry and the Codebase—through a combination of patents and trade secret protections.
Patents
As of December 31, 2021, we had approximately 63 patent “families,” including patents held by the Company as well as by its wholly owned subsidiary Gen9, Inc. Some of these are represented by a Patent Cooperation Treaty (“PCT”) application with related national applications, as well as 16 pending provisional applications. We have over 58 issued U.S. and over 202 issued foreign patents, which includes European nationalizations, and approximately 69 pending U.S. non-provisional and approximately 150 pending foreign patent applications, including patents and patent applications acquired from third parties.
In addition to our proprietary methods and technologies, we also non-exclusively in-license certain intellectual property assets from third parties.
We intend to pursue additional intellectual property protection to the extent that we believe that it would be beneficial and cost-effective. We may abandon applications that are no longer relevant to our business. We cannot provide any assurance that any of our current or future patent applications will result in the issuance of patents. We also cannot assure the scope of any of our future issued patents or warrant that any of our patents will prevent others from commercializing infringing products or technology.
Our patent portfolio is detailed in the chart below:
Patent Family
|
Description
|
Application/
|
Filing Date
|
Issue Date/
|
Earliest Date1
|
Owned by Gen9, Inc. |
|||||
Methods and Devices for High Fidelity Polynucleotide Synthesis |
Microfluidic devices and methods for assembling oligonucleotides by merging droplets containing oligonucleotide fragments with regions of complementarity |
PCT/US2009/055267; WO/2010/025310 |
08/27/2009 |
Nationalized in: US |
01/16/2030 |
|
|
|
|
|
|
Methods and Apparatuses for Chip-Based DNA Error Reduction |
High-fidelity polynucleotide synthesis by generating complementary oligonucleotides to support bound single-stranded oligo (ss-oligo) in a microdroplet using enzymatic processes |
PCT/US2010/057405; WO/2011/066186 |
11/19/2010 |
Nationalized in: EP, FR, DE, LT, NL, ES, SE, CH, GB, LI, and US |
11/19/2030 |
|
|
|
|
|
|
33
Table of Contents
Methods and Microfluidic Devices for the Manipulation of Droplets in High Fidelity Polynucleotide Assembly |
Methods and devices utilizing droplet-based liquid manipulation on a substrate for assembling nucleic acids including steps of sequence error removal |
PCT/US2010/055298; WO/2011/056872 |
11/03/2010 |
Nationalized in: US |
11/03/2030 |
|
|
|
|
|
|
Assembly of High Fidelity Polynucleotides |
Methods and apparatuses for preparing and/or assembling high fidelity nucleic acids on a solid support |
PCT/US2011/020335; WO/2011/085075 |
01/06/2011 |
Nationalized in: US |
01/06/2031 |
|
|
|
|
|
|
Methods and Devices for Oligonucleotide Synthesis |
Devices and methods for the synthesis of polynucleotides and libraries of polynucleotides using manipulation of oligo-containing droplets on a support |
US 8,716,467 |
03/02/2011 |
Issued 5/6/2014 |
05/12/2031 |
US 9,388,407 |
03/31/2014 |
Issued 7/12/2016 |
03/02/2031 |
||
US 9,938,553 |
04/08/2016 |
Issued 4/10/2018 |
03/13/2031 |
||
US 15/908,726; US 2018/0195100 |
02/28/2018 |
Published |
|
||
|
|
|
|
|
|
Methods for Nucleotide Sequencing and High Fidelity Polynucleotide Synthesis |
Methods of obtaining sequence information of target polynucleotides by performing sequencing by ligation and sequencing by polymerase-based reactions |
PCT/US2011/036433; WO/2011/143556 |
05/13/2011 |
Nationalized in: US |
05/13/2031 |
|
|
|
|
|
|
Microarray Synthesis and Assembly of Gene-Length Polynucleotides |
Processes for in vitro synthesis and on-device assembly of long, gene-length polynucleotides based upon assembly of multiple shorter oligos synthesized in situ on a microarray platform |
US 7,563,600; 7,323,320; 8,058,004; 9,023,601; 9,051,666; 10,450,560; 10,640,764; 10,774,325 |
09/12/2002- 02/18/2020 |
Issued 07/21/2009 -09/15/2020 |
09/12/2022 |
US 17/019,448; US 2021/0062185 |
09/14/2020 |
Published |
|
||
PCT/US2003/028946; WO/2004/024886 |
09/12/2003 |
Nationalized in: AU, CA, CH, EP, FR, DE, DK, GB, JP, LI, NL |
09/12/2023 |
||
|
|
|
|
|
|
Compositions, Methods, and Apparatus for Oligonucleotides Synthesis |
Compositions and methods for high-fidelity polynucleotide assembly on solid support from oligos by adding variable length padding sequences to the ends of the oligos |
PCT/US2014/025610; WO/2014/160004 |
03/13/2014 |
Nationalized in: EP, US, DE, GB |
03/13/2034 |
|
|
|
|
|
|
Compositions and Methods for Multiplex Nucleic Acids Synthesis |
Methods of producing target nucleic acid using pluralities of oligos with overhangs, in which overhangs of one plurality are designed to be complementary to overhangs of another plurality |
PCT/US2014/026261; WO/2014/151696 |
03/13/2014 |
Nationalized in: AU, CA, CN, EP, IL, US |
03/13/2034 |
|
|
|
|
|
|
34
Table of Contents
Methods for the Production of Long Length Clonal Sequence Verified Nucleic Acid Constructs |
Methods and compositions for the production and isolation of high fidelity nucleic acids using high throughput sequencing of fragmented oligos which are tagged with unique barcodes at the 5’ and/or 3’ ends |
PCT/US2014/048867; WO/2015/017527 |
07/30/2014 |
Nationalized in: EP, CH, DE, FR, GB, LI, NL |
07/30/2034 |
|
|
|
|
|
|
Protein Arrays and Methods of Making and Using the Same |
Methods and devices for preparing a protein array to generate and express a plurality of proteins from a plurality of nucleic acids on an array |
PCT/US2011/060217; WO/2012/064975 |
11/10/2011 |
Nationalized in: EP, US |
11/10/2031 |
|
|
|
|
|
|
Libraries of Nucleic Acids and Methods for Making the Same (Nucleic Acid Library and its Manufacturing Method) |
Methods for designing and producing non-random libraries of nucleic acids using multiplexed polynucleotide synthesis in which complementary overhangs attached to specific sequences are hybridized and ligated to each other |
PCT/US2014/067444; WO/2015/081114 |
11/25/2014 |
Nationalized in: AU, CA, CN, EP, IL, US |
11/25/2034 |
|
|
|
|
|
|
Iterative Nucleic Acid Assembly Using Activation of Vector-Encoded Traits |
Nucleic acid configurations and cloning strategies for progressively assembling a long nucleic acid product using a plurality of assembly cycles that each include assembling a vector and two or more inserts containing one or more regulatory sequences that activate vector-encoded traits when assembled in a predetermined configuration |
PCT/US2007/019209; WO/2008/027558 |
08/31/2007 |
Nationalized in: US |
08/31/2027 |
|
|
|
|
|
|
Methods and Devices for Nucleic Acid Synthesis |
Methods and apparatus for the synthesis of polynucleotides on a support using primer extension to generate overlapping construction oligonucleotides and assembly of the polynucleotides of interest by hybridizing construction oligos onto anchor support- bound oligonucleotides |
PCT/US2011/060243; WO/2012/078312 |
11/10/2011 |
Nationalized in: AU, CA, EP, BE, DE, GB, IE, LT, NL, CH, CN, DE, ES, FR, GB, IL, JP, LI, SE, US |
11/10/2031 |
|
|
|
|
|
|
Methods for Preparative in Vitro Cloning |
Methods and devices for the isolation of nucleic acids from libraries by tagging a population of nucleic acids |
US 9,752,176 |
06/15/2012 |
Issued 09/05/2017 |
06/15/2032 |
35
Table of Contents
|
with unique oligonucleotide tags |
|
|
|
|
US 15/666,345; US 2018/0023120 |
08/01/2017 |
Published |
|
||
US 17/536,828 |
11/29/2021 |
Pending |
|
||
PCT/US2012/042597; WO/2012/174337 |
06/15/2012 |
Nationalized in: AU, CA, CN, EP, IL, CH, DE, FR, GB, LI, LT, NL, US |
06/15/2032 |
||
|
|
|
|
|
|
Compositions and Methods for High Fidelity Assembly of Nucleic Acids |
Methods, compositions and algorithms for designing and producing a target nucleic acid from blunt-end double stranded nucleic acids generated by digesting the same to create cohesive-end fragments with unique cohesive ends that anneal and are ligated in a predetermined order |
US 13/592,827; US 2013/0059296 |
08/23/2012 |
Published |
|
US 17/373,324; US 2021/0380991 |
07/12/2021 |
Published |
|
||
PCT/US2012/052036; WO/2013/032850 |
08/23/2012 |
Nationalized in: AU, CA, CH, CN, DE, EP, LI, EP, FR, GB, IL, JP, LT, NL, SE, IE, BE, ES, HK, IS |
08/23/2032 |
||
|
|
|
|
|
|
Device and Method for Nucleic Acid Manipulation |
High precision, high selectivity nucleic acid singulation and assembly techniques using mechanical force generated piezoelectrically or acoustically to selectively expel or transfer one or more volumes of nucleic acids from a solid support |
PCT/US2018/033823; WO/2018/217702 |
05/22/2018 |
Nationalized in: AU, CA, CN, EP, IL, JP, US |
05/22/2038 |
|
|
|
|
|
|
Compositions and Methods for Site-Directed DNA Nicking and Cleaving |
Compositions and methods for site-directed DNA nicking and/or cleaving, and use thereof in, for example, polynucleotide assembly to create sticky-end breaks in DNA so that the resulting fragments can be used for DNA assembly |
PCT/US2015/039517; WO/2016/007604 |
07/08/2015 |
Nationalized in: EP, DE, GB, US |
07/08/2035 |
|
|
|
|
|
|
Methods for Nucleic Acid Assembly and High Throughput Sequencing |
Hierarchical assembly of target polynucleotides from construction oligonucleotides |
PCT/US2013/047370; WO/2014/004393 |
06/24/2013 |
Nationalized in: AU, CA, CN, EP, CH, DE, FR, GB, LT, NL, SE, IL, JP, US |
06/24/2033 |
|
|
|
|
|
|
Methods for Sorting Nucleic Acids and Preparative in Vitro Cloning |
Compositions and methods for sorting and cloning of high fidelity nucleic acids by high throughput sequencing using unique barcode pairs (tag oligos) that may be sequenced |
US 10,081,807 |
04/24/2013 |
Issued 09/25/2018 |
04/09/2035 |
36
Table of Contents
|
to identify a nucleic acid of interest |
|
|
|
|
US 10,927,369 |
07/18/2018 |
Issued 02/23/2021 |
10/17/2033 |
||
US 17/152,202; US 2021/0139888 |
01/19/2021 |
Published |
|
||
PCT/US2013/037921; WO/2013/163263 |
04/24/2013 |
Nationalized in: AU, CA, CN, EP, CH, DE, FR, GB, LI, LT, NL, SE, IL |
04/24/2033 |
||
|
|
|
|
|
|
Methods for Screening Proteins Using DNA Encoded Chemical Libraries as Templates for Enzyme Catalysis |
Methods, compositions and devices for screening a protein library for proteins having a desired activity |
US 9,150,853 |
03/13/2013 |
Issued 10/06/2015 |
03/13/2033 |
US 10,308,931 |
08/31/2015 |
Issued 06/04/2019 |
07/27/2033 |
||
US 16/397,314; US 2019/0249169 |
04/29/2019 |
Published |
|
||
|
|||||
Owned by Ginkgo Bioworks, Inc. |
|||||
|
|
|
|
|
|
Methods and Systems for Chemoautotrophic Production of Organic Compounds |
Engineered chemoautotrophs (and methods for engineering such chemoautotrophs) including three metabolic modules: energy conversion pathways allowing use of energy from an inorganic energy source, carbon fixation |
US 8,349,587 |
10/31/2011 |
Issued 01/08/2013 |
10/31/2031 |
|
|
|
|
|
|
|
pathways, and product biosynthetic pathways to convert central metabolites into desired products, such as carbon-based products of interest |
|
|
|
|
|
|
|
|
|
|
|
|
PCT/US2012/62540; WO/2013/066848 |
10/30/2012 |
Nationalized in: US |
10/31/2031 |
|
|
|
|
|
|
Methods and Systems for Methylotrophic Production of Organic Compounds |
Engineered methylotrophs (and methods for selecting such cells) for efficiently converting C1 compounds into various carbon-based products of interest, including systems, mechanisms and methods to confer pathways for energy conversion, methylotrophy, or carbon fixation |
PCT/US2013/073582; WO/2014/089436 |
12/06/2013 |
Nationalized in: US |
12/06/2033 |
|
|
|
|
|
|
Methods and Genetic Systems for Cell Engineering |
Engineered probiotics comprising a nuclease module designed to specifically target and degrade a nucleic acid, a synthetic mobile genetic element module capable of dispersing the system from one host cell to another, and an antibiotic-free maintenance module |
PCT/US2015/022508; WO/2015/148680 |
03/25/15 |
Nationalized in: AU, CA, EP, JP, US |
03/25/2035 |
|
|
|
|
|
|
37
Table of Contents
Methods and Molecules for Yield Improvement Involving Metabolic Engineering |
Methods and compositions relating to cells that have been engineered to reduce or eliminate proteins having enzymatic activity that interferes with the expression of a metabolic product |
PCT/US2010/036902; WO/2010/141468 |
06/01/2010 |
Nationalized in: US |
07/10/2030 |
|
|
|
|
|
|
Methods and Systems for Cell State Quantification* *(Co-Owned with R. Rettberg) |
Engineered cells, and methods for engineering such cells, for genomic, transcriptomic, or proteomic analysis, using multiple peptide tags |
US 9,506,167 |
07/27/2012 |
Issued 11/29/2016 |
01/07/2034 |
|
|
|
|
|
|
|
|
US 10,119,975 |
11/29/2016 |
Issued 11/06/2018 |
07/27/2032 |
|
|
|
|
|
|
Protective Enzymes |
Enzymes for protecting polymers from damage caused by fatty acids from secreted biological fluids such as sebum or sweat |
PCT/US2018/050718; WO2019/055541 |
09/12/2018 |
Nationalized in CN, EP, HK, US |
09/12/2038 |
|
|
|
|
|
|
Chimeric Terpene Synthases |
Cells, enzymes, and methods for production of terpenes (which can be used as fragrances, pheromones, or antimicrobials, among other things) that are partially derived from sequences reconstructed from rare or extinct plants |
PCT/US2019/018122; WO2019/161141 |
02/14/2019 |
Nationalized in: EP, HK, JP, KR, US |
02/14/2039 |
|
|
|
|
|
|
Biosynthesis of Mogrosides |
Cells, enzymes, and methods for producing mogrosides (high-intensity natural sweeteners) by fermentation |
PCT/US2019/060652; WO 2020/097588 |
11/09/2019 |
Nationalized in: CA, CN, EP, JP, US |
11/09/2039 |
|
|
|
|
|
|
Biosynthesis of Mogrosides |
Cells, enzymes, and methods for producing mogrosides (high-intensity natural sweeteners) by fermentation |
PCT/US2020/057067; WO 2021/081327 |
10/23/2020 |
Published |
10/23/2040 |
|
|
|
|
|
|
Biosynthesis of Mogrosides |
Cells, enzymes, and methods for producing mogrosides (high-intensity natural sweeteners) by fermentation |
PCT/US2021/032251; WO2021/231728 |
05/13/2021 |
Published |
05/13/2041 |
|
|
|
|
|
|
Biosynthesis of Cannabinoids and Cannabinoid Precursors |
Cells, enzymes, and methods for producing cannabinoid compounds by fermentation |
PCT/US2020/019760; WO2020/176547 |
02/25/2020 |
Nationalized in CA, EP, IL, US |
02/25/2040 |
|
|
|
|
|
|
Biosynthesis of Cannabinoids and Cannabinoid Precursors |
Cells, enzymes, and methods for producing cannabinoid compounds by fermentation |
PCT/US2020/046838; WO2021/034848 |
08/18/2020 |
Published |
08/18/2040 |
|
|
|
|
|
|
Biosynthesis of Cannabinoids and Cannabinoid Precursors |
Cells, enzymes, and methods for producing cannabinoid compounds by fermentation |
PCT/US2021/024398; WO2021/195520 |
03/26/2021 |
Published |
03/26/2041 |
|
|
|
|
|
|
38
Table of Contents
Biosynthesis of Cannabinoids and Cannabinoid Precursors |
Cells, enzymes, and methods for producing cannabinoid compounds by fermentation |
PCT/US2021/037954; WO2021/257915 |
06/17/2021 |
Published |
06/17/2041 |
|
|
|
|
|
|
Biosynthesis of Cannabinoids and Cannabinoid Precursors |
Cells, enzymes, and methods for producing cannabinoid compounds by fermentation |
PCT/US2021/040941 |
07/08/2021 |
Pending |
07/08/2041 |
Biosynthesis of Cannabinoids and Cannabinoid Precursors |
Cells, enzymes, and methods for producing cannabinoid compounds by fermentation |
PCT/US2021/054641
|
10/12/2021
|
Pending
|
10/12/2041
|
|
|
|
|
|
|
Rare Earth Element (REE)-Binding Proteins |
Cells, binding proteins, and methods for recovering rare earth elements, including lanthanides |
PCT/US2020/038808; WO2020/257702 |
06/19/2020 |
Nationalized in AU, CA, CN, EP, JP, KR, US |
06/19/2040 |
|
|
|
|
|
|
Biosynthesis of Enzymes for use in Treatment of Maple Syrup Urine Disease (MSUD)* *(Co-Owned with Synlogic, Inc.) |
Methods, enzymes, cells, and compositions for treating maple syrup urine disease (MSUD) and other conditions characterized by excessive branched-chain amino acids |
PCT/US2020/038813; WO2020/257707 |
06/19/2020 |
Nationalized in IL, JP, US |
06/19/2040 |
|
|
|
|
|
|
Optimized Bacteria Engineered to Treat Disorders Involving the Catabolism of Leucine, Isoleucine, and/or Valine* *(Co-Owned with Synlogic, Inc.) |
Methods, enzymes, cells, and compositions engineered to improve leucine catabolism and treat disorders involving the catabolism of leucine, isoleucine, or valine |
PCT/US2020/038675; WO 2020/257610 |
06/19/2020 |
Nationalized in CA, US |
06/19/2040 |
|
|
|
|
|
|
Production of Oligosaccharides |
Compositions and methods for producing fructans using sucrose:sucrose 1-fructosyltransferase (1-SST), fructan:fructan |
PCT/US2020/052390; WO 2021/061910 |
09/24/2020 |
Published |
09/24/2040 |
|
|
|
|
|
|
Biosynthesis of Histidine/Enhanced Production of Histidine, Purine Pathway Metabolites, and Plasmid DNA |
Methods and genetically modified cells for the biosynthetic production of histidine, plasmid DNA, or purine pathway metabolites, including synthetic promoters and genes encoding modified ribose phosphate pyrophosphokinase |
PCT/US2020/065286; WO 2021/126961 |
12/16/2020 |
Published |
12/16/2040 |
|
|
|
|
|
|
39
Table of Contents
|
(RPPK) and/or modified 5,10-methylene-tetrahydrofolate dehydrogenase/5,10-methylene-tetrahydrofolate cyclohydrolase (MTHFDC) enzymes |
|
|
|
|
|
|
|
|
|
|
Variant SARS-Cov -2 Proteins and Uses Thereof |
Variant proteins of SARS-CoV-2 nucleocapsid, spike protein, and spike protein receptor binding domain; nucleic acids encoding such variants; and compositions, cells, diagnostic kits containing such variants or its coding nucleic acids; as well as methods of detecting, treating and/or preventing SARS-CoV-2 infection |
PCT/US2021/030875 |
05/05/2021 |
Pending |
05/05/2041 |
Methanol Utilization |
Methods and genetically modified cells for the biosynthetic production of amino acids such as lysine using methanol as a feedstock. |
PCT/US2020/028746; WO 2020/214940 |
04/17/2020 |
Nationalized in: CA, CN, EP, JP, KR, US |
04/17/2040 |
Use of Bone Morphogenetic Proteins and Their Receptors for Aesthetics and Cosmetics |
Methods for preventing or reducing skin wrinkles and/or enhancing or preserving facial contours using one or more Bone Morphogenetic Proteins (BMPs), or an associated BMP protein |
PCT/US2021/049768 |
9/10/2021
|
Pending |
9/10/2041
|
|
|
|
|
|
|
Compositions and Methods for the Production of Compounds |
Host cells, vectors, and nucleic acids encoding recombinant LALs (Large ATP-binding regulators of the LuxR family of transcriptional activators) and LAL binding sites for the production of compounds such as polyketides, and methods for producing such compounds |
PCT/US2017/027215; WO 2017/180748 |
04/12/2017 |
Nationalized in: US, AU, CA, CN, EP, JP, KR |
04/12/2037 |
|
|
|
|
|
|
Compositions and Methods for the Production of Compounds |
Compositions and methods to facilitate combinatorial biosynthesis of polyketides, with engineered polyketide synthases that include modified domains with altered enzymatic activity |
PCT/US2017/058805; WO 2018/081592 |
10/27/2017 |
Nationalized in: US, AU, CA, CN, EP, JP, KR |
10/27/2037 |
|
|
|
|
|
|
40
Table of Contents
Compositions and Methods for the Production of Compounds |
Compositions and methods for use in combinatorial biosynthesis of polyketides by module swapping between polyketide synthase genes, with engineered polyketide synthases that include heterologous modules with altered enzymatic activity |
PCT/US2017/058800; WO 2018/081590 |
10/27/2017 |
Nationalized in: AU, CA, CN, EP, JP, KR, US |
10/27/2037 |
|
|
|
|
|
|
Enhanced Production of Core Lipids in Oleaginous Yeasts |
Engineered cells having genetic modification(s) that increase lipid yield and methods of increasing lipid yield in a cell |
PCT/US2015/067805; WO 2016/109494 |
12/29/2015 |
Nationalized in: BR, CN, EP, IN, US |
12/29/2035 |
|
|
|
|
|
|
Heterologous Production of 10-Methylstearic Acid |
Engineered gene sequences, cells, and methods for producing branched methyl lipids including 10-methylstearate |
PCT/US2017/052491; WO 2018/057607 |
09/20/2017 |
Nationalized in: BR, CA, CN, EP, US |
09/20/2037 |
|
|
|
|
|
|
Heterologous Production of 10-Methylstearic Acid by Cells Expressing Recombinant Methyltransferase |
Engineered methyltransferase gene sequences, cells, and methods for producing branched methyl-lipids or exomethylene-substituted lipids |
PCT/US2018/051919; WO 2019/060527 |
09/20/2018 |
Nationalized in: BR, CA, EP, US |
09/20/2038 |
|
|
|
|
|
|
Methods and Compositions Involving Promoters Derived From Yarrowia lipolytica |
Promoters, recombinant nucleic acids, cells and methods for modulating lipid production in oleaginous microorganisms such as yeasts |
16/942,509; US2021-0032604A1 |
07/29/2020 |
Pending |
07/29/2040 |
|
|
|
|
|
|
Microorganisms Engineered to Use Unconventional Sources of Nitrogen |
Microorganisms engineered to grow on an atypical nitrogen source and their use in fermentation to produce a variety of compounds including commodities, fine chemicals, and pharmaceuticals |
PCT/US2014/010332; WO 2014/107660 |
01/06/2014 |
Nationalized in: AU, CA, BR, IN, US |
01/06/2034 |
|
|
|
|
|
|
Microorganisms Engineered to Use Unconventional Sources of Phosphorous or Sulfur |
Microorganisms engineered to grow on an atypical phosphorus or sulfur source and their use in fermentation to produce a variety of compounds including commodities, fine chemicals, and pharmaceuticals |
PCT/US2014/52841; WO 2015/031441 |
08/27/2014 |
Nationalized in: CN, AU, CA, BR, IN, EP, FR, DE, GB, US |
08/27/2034 |
|
|
|
|
|
|
41
Table of Contents
Diacylglycerol Acyltransferase (DGA1) Polynucleotides, and Methods of Increasing Yeast Cell Lipid Production by Overexpression of Heterologous DGA1 |
Cells engineered to express heterologous DGA1 enzyme(s) that confer increased lipid production and/or enhanced efficiency of glucose consumption, as well as methods of lipid production using these cells |
PCT/US2015/17227; WO 2015/127421 |
02/24/2015 |
Nationalized in: CN, AU, IN, FI, EP, BE, DK, FR, DE, LU, SE, CH, GB, US |
02/24/2035 |
|
|
|
|
|
|
Selective Advantage in Fermentation |
Microorganisms engineered to grow on an atypical nitrogen, phosphorus, and/or sulfur source; fermentation compositions composed of such microorganisms and a fermentation medium containing an atypical nitrogen, phosphorus, and/or sulfur source; and fermentation processes thereof |
PCT/US2015/024943; WO 2015/157431 |
04/08/2015 |
Nationalized in: AU, IN, US |
04/08/2035 |
|
|
|
|
|
|
Increasing Cellular Lipid Production by Increasing the Activity of Diacylglycerol Acyltransferase and Decreasing the Activity of Triacylglycerol Lipase |
Engineered cells having genetic modification(s) that increase lipid yield and methods of increasing lipid yield in a cell |
PCT/US15/28760; WO 2015/168531 |
05/01/2015 |
Nationalized in: AU, IN, US |
05/01/2035 |
|
|
|
|
|
|
Increasing Lipid Production in Oleaginous Yeast |
Engineered cells with genetic modification(s) that increase lipid yields including modifications that increase type 1, type 2, and/or type 3 diacylglycerol acyltransferase activity and modifications that decrease lipase activity, as well as methods of increasing lipid yield |
PCT/US2015/033251; WO 2015/184303 |
05/29/2015 |
Nationalized in: AU, CN, IN, EP, US |
05/29/2035 |
|
|
|
|
|
|
Increasing Lipid Production and Optimizing Lipid Composition |
Recombinant nucleic acids, engineered cells, and methods for increasing lipid production that involve increasing or decreasing the activity of one or more selected genes |
PCT/US2015/033211; WO 2015/184277 |
05/29/2015 |
Nationalized in: AU, CN, EP, IN, US |
05/29/2035 |
|
|
|
|
|
|
Oleic Acid Production in Yeast |
Engineered cells having genetic modification(s) that increase oleic acid yield and methods of increasing oleic acid yield in a cell |
PCT/US2015/64710; WO 2016/094520 |
12/09/2015 |
Nationalized in: CN, BR, IN, EP, US |
12/09/2035 |
|
|
|
|
|
|
42
Table of Contents
Derivatives of 10-Methylene Lipids, Process for Preparing Such Derivatives and Use Thereof |
Tuberculostearic acid (10-methylstearic acid) derivatives, processes for producing such compounds, and their use in processes for preparing polyamides, polyesters, lactams, and lactones |
PCT/EP2020/058484; WO 2020/0193681 |
03/26/2020 |
Nationalized in: EP |
03/26/2040 |
|
|
|
|
|
|
Synthetic Expression Systems |
Transcriptional units, synthetic expression systems, and host cells capable of expressing a gene of interest; methods for the production of bioproducts in methanol-free culture conditions. |
PCT/US2021/049180 |
09/05/2021 |
Pending |
09/05/2041 |
|
|
|
|
|
|
Synthetic Methanol Inducible Promoters and Uses Thereof |
Synthetic promoters capable of facilitating the high-yield synthesis of proteins and molecules; transcriptional units and host cells comprising such synthetic promoters; and methods of use thereof |
PCT/US2021/059135 |
11/12/2021 |
Pending |
11/12/2041 |
1. The expiration date of a United States patent may be earlier or later than as listed in this table due to patent term adjustment and/or the existence of a terminal disclaimer.
Trade secrets
Ginkgo’s technology-related intellectual property that is not patent-protected is maintained as trade secrets. We employ a variety of safeguards to protect our trade secrets, including contractual arrangements with our employees, consultants, contractors and other advisors that impose obligations of confidentiality, assignment of inventions, and security; digital security measures; and physical security precautions.
We require confidentiality and material transfer agreements from third parties that receive our confidential data or materials, and we also incorporate confidentiality and material transfer precautions into our collaboration agreements. For example, in the course of a cell program, we might transfer samples of intermediate strains to the customer for testing and scale-up work and then transfer a final commercial strain upon completion of our work. To protect both intermediate and final strains, we use strain transfer agreements that document the contractual restrictions and controls we have put into place, typically including, in the case of intermediate strains, covenants requiring the customer to return or destroy all strain samples after testing.
Trademarks and domain names
Although our business is directed at sophisticated corporate customers rather than end consumers, we have trademark rights and registrations in our name, logo, and other brand indicia in the United States and other jurisdictions around the world. We also have registered domain names for websites that we use in our business, such as www.ginkgobioworks.com.
Intellectual property transaction structure
We earn revenue from collaboration agreements with customers under which we perform cell programming activities. Through our cell programs, we develop cells that produce or are products for our customers, which they market in their verticals.
With respect to intellectual property, we have relatively standard transaction structures that apply to cell programs for a customer. In this situation, our collaboration agreements typically provide that Ginkgo will own all collaboration-related intellectual property (“Foreground IP”) concerning cell programming. To protect our collaboration partners’ investment in
43
Table of Contents
the collaboration and to provide them with a competitive advantage from working with Ginkgo, Ginkgo provides a limited exclusive license to patents within the Foreground IP that cover the product, usually within a specified field. However, our terms may vary.
We typically do not provide exclusive licenses to unpatented Foreground IP (i.e., trade secrets and other know-how) that results from a collaboration. In our typical deal structure, we also do not provide exclusive licenses to our “background” intellectual property — i.e., the intellectual property, whether patented or unpatented, that we developed before entering into a collaboration or develop independently from our work in the collaboration. We believe that our transaction structures allow us to maximize the reuse of Codebase across programs and ensure that technology we develop does not lie fallow.
In-License Agreements
Amyris Partnership Agreement
On October 20, 2017, we entered into a partnership agreement (the “Partnership Agreement”) with Amyris, which, as amended from time to time, terminated all prior agreements between Ginkgo and Amyris. In the Partnership Agreement, Amyris, among other things, granted us a non-exclusive license effective as of June 28, 2016 (the date of an earlier agreement between the parties) under all of Amyris’s rights in and to certain specified microbial strains, and under all patents and applications associated with such microbial strains, to make, have made, use, sell, offer to sell and import any products other than farnesene and/or farnesene derivatives that are chemically produced from farnesene. The license is subject to any previous exclusive licenses provided to third parties and is royalty-free, fully paid-up, sublicensable, non-exclusive and perpetual (i.e., it survives termination or expiration of the Partnership Agreement except in the case of our insolvency).
Strateos Collaboration Agreement
On October 2, 2017, we entered into a collaboration agreement with Strateos, Inc. f/k/a Transcriptic, Inc. (“Strateos”), which was amended and restated on April 20, 2021 (the “Strateos Collaboration Agreement”). Under the Strateos Collaboration Agreement, Strateos granted us a non-exclusive, perpetual, irrevocable, fully paid-up, royalty-free license under certain intellectual property rights to use its software platform in a range of activities relating to our business, including, among other things, developing and commercializing cell lines, developing data packages, providing foundry and analytical services and performing diagnostic testing. In connection with the Strateos Collaboration Agreement, we paid Strateos an up-front fee of $3.0 million and agreed to pay an additional $9.0 million in fees during the five-year term of the agreement in consideration for services provided by Strateos under the agreement, of which more than $7.7 million had already been paid as of December 31, 2021. Either party may terminate the Strateos Collaboration Agreement without cause upon six months’ written notice to the other party. Either party may also terminate the agreement for the other party’s material breach, insolvency or change of control to a direct competitor of the terminating party. After expiration of the agreement, either party’s termination of the agreement for convenience or the other party’s insolvency, or our termination of the agreement for Strateos’ material breach or Strateos’ change of control to one of our direct competitors, we will retain a license to use Strateos’ software. We retain such rights for an 18-month period in the event the agreement is terminated by Strateos for certain material breaches of the agreement, but we do not retain such rights in the event of Strateos’ termination due to our change of control to a Strateos direct competitor, our leak or other unauthorized disclosure of Strateos’ code, or a material breach of our obligations involving payment, intellectual property or confidentiality.
Suppliers
Ginkgo’s suppliers for cell programming operations comprise primarily manufacturers and distributors of life science tools, consumables and equipment as well as certain specific providers of contract research, development and manufacturing services. We will sometimes enter into long-term, strategic partnerships with innovative suppliers. Because of the significant scale of our Foundry’s operations, we believe we are often an early adopter and the largest customer at scale of certain new life science tools and technologies. Our supply agreements with Twist Bioscience Corporation (“Twist”) and Berkeley Lights, as further described below, are examples of such strategic supplier relationships. We will also occasionally acquire technology or Codebase assets for strategic reasons and because we can integrate the technology effectively into our platform — Gen9, Inc. and Warp Drive Bio, Inc. are two examples.
Our suppliers for our biosecurity offering include multiple manufacturers and distributors of lateral flow assay (“LFA”) test kits and COVID-19 sample collection kits. We have developed a national network of third party labs for provision of COVID-19 molecular testing services. We also utilize third parties for certain other services, including physician authorizations and on-site test administration, in the provision of our end-to-end COVID-19 testing offering.
44
Table of Contents
Our software, automation, data, information technology, DevOps and information security functions utilize various third party software and information technology service providers, including AWS, for data storage and processing. We also routinely engage a variety of third parties for professional services, contract employment services and consulting services.
Twist
In March 2018, we entered into a non-cancelable supply agreement, as amended in February 2022 (the “2018 Agreement”), with Twist, which requires us to purchase synthetic DNA at specified volumes on a quarterly basis over a four-year term. To the extent we fail to meet our quarterly minimum purchase obligations, we are required to pay a fee per unit of shortfall. The products we may purchase that contribute toward achieving our quarterly minimum purchase obligation can vary based on our discretion, subject to advance notice provided to Twist.
Our quarterly minimum purchase obligation may be adjusted for the following reasons: (i) due to a lack of availability of certain products for purchase in a given quarter; (ii) due to a lack of certain features available; (iii) delays in shipments over two consecutive quarters beyond the agreed upon lead times; and (iv) if the average yield of certain products measured over two consecutive quarters is greater than a specified yield. We receive volume discounts on purchases based on specified volume thresholds over the term of the 2018 Agreement.
The 2018 Agreement can only be terminated (i) upon mutual agreement of both parties, (ii) by us upon a specified change of control, (iii) upon a material breach of the contract by either party, or (iv) by Twist in the event that we fail to place orders for more than a certain percentage of our required quarterly minimums under the 2018 Agreement for two consecutive quarters. The purchase minimums in the 2018 Agreement create an enforceable obligation only in conjunction with each purchase order.
Berkeley Lights
In September 2019, we signed a collaboration agreement (the “Berkeley Collaboration”) with Berkeley Lights, a cell biology company focused on enabling and accelerating the rapid development and commercialization of microbial biotherapeutics and other cell-based products for its customers. Under the Berkeley Collaboration, we incorporate Berkeley Lights’ platform into the Foundry to accelerate the engineering of biotherapeutics and cell-based products. Under the Berkeley Collaboration, both parties agree to use diligent efforts to jointly develop certain workflow plans.
We are obligated to pay Berkeley Lights at least $109.0 million, and up to $150.0 million, over the term period of the Berkeley Collaboration from October 2019 through September 2026 for (i) payments for Berkeley Lights’ efforts under the workflow development plans and (ii) payments for purchases of certain equipment, associated consumables, and other goods and services. We have the option to buy down our purchase obligations after the second contract year by making a one-time payment to Berkeley Lights. We are also required to pay to Berkeley Lights certain license fees for the use of Berkeley Lights’ platform and certain milestone payments of up to $11.5 million payable when a therapeutic discovered using certain workflows reaches specified development and regulatory milestones. For more details on the minimum purchase commitments under the Berkeley Collaboration, see Note 14, “Commitments and Contingencies,” of our audited consolidated financial statements included elsewhere in Annual Report.
Under the Berkeley Collaboration, we are granted an exclusivity period for each workflow developed for us by Berkeley Lights, but Berkeley Lights has the option to buy down the exclusivity period, after which the parties will equally share the development costs of the associated workflow.
The Berkeley Collaboration will continue until the seventh anniversary of its effective date, subject to certain automatic extension provisions, including for delays resulting from Berkeley Lights’ failure to supply products or services conforming with the Berkeley Collaboration. The collaboration will automatically terminate if we, at any time after the second contract year, elect to exercise our buy down right. In addition, either party may terminate the Berkeley Collaboration (i) for the material breach by the other party (including, with respect to Ginkgo, a material supply failure), (ii) upon the occurrence of certain insolvency related events of the other party, and (iii) for certain force majeure events.
Government Contracts
We have entered into agreements with governmental entities and contractors in the past to serve as a U.S. government contractor or subcontractor and may do so again in the future. See “Risk Factors—We have pursued in the past and may pursue additional U.S. Government contracting and subcontracting opportunities in the future and as a U.S. Government contractor and subcontractor, we are subject to a number of procurement rules and regulations”.
45
Table of Contents
Government Regulations
Our business, or the business of our customers, may be regulated by the FDA and other federal authorities in the United States, including the U.S. Federal Trade Commission (“FTC”), U.S. Department of Agriculture (“USDA”), U.S. Drug Enforcement Administration (“DEA”) and Environmental Protection Agency (“EPA”), as well as comparable authorities in foreign jurisdictions and various state and local authorities in the United States. Failure to comply with applicable regulations may result in enforcement actions, civil or criminal sanctions, and adverse publicity.
FDA regulation
We provide cell engineering and product discovery services to customers engaged in the manufacture of foods, cosmetics and pharmaceutical products. The FDA regulates the research, development, testing, quality control, import, export, safety, effectiveness, storage, recordkeeping, premarket review, approval or licensure, processing, formulation, manufacturing, packaging, labeling, advertising, promotion, marketing, distribution, sale, post-market monitoring and reporting of our customers’ pharmaceuticals, cosmetics and food products, and the FTC also regulates the advertising and promotion of these products.
We also act as a systems integrator and authorized distributor of certain COVID-19 diagnostic test and collection kits manufactured by independent third parties, and we work with laboratory partners that provide clinical laboratory testing services as part of the COVID-19 testing services we offer, and these tests and test kits may be subject to regulation by the FDA. In particular, the tests and test kits used in our COVID-19 testing services may be subject to regulation by the FDA as medical devices, and may be required to comply with the requirement that such products have obtained clearance, approval, or other marketing authorizations, such as an Emergency Use Authorization (“EUA”), before they can be commercialized, as well as post-market requirements such as adverse event reporting and restrictions on labeling, marketing, and distribution.
The HHS and FDA issued several policy statements in November 2021 governing the regulation of COVID-19 Laboratory Developed Tests (“LDTs”) that resume FDA premarket review of COVID-19 LDTs that HHS halted in August 2020. Pursuant to these new policy statements, FDA expects laboratories to seek FDA marketing authorization and otherwise comply with FDA device regulations when marketing COVID-19 LDTs. An LDT is an in vitro diagnostic test that is intended for clinical use and is designed, manufactured, and used within a single laboratory. LDTs are classified as medical devices, but the FDA has historically exercised enforcement discretion and has generally not enforced FDA requirements, including premarket review, with respect to laboratories that offer LDTs. While HHS and FDA have announced their intention to require premarket review of COVID-19 LDTs, either agency may change its position in the future.
Medical products, including COVID-19 tests, that are granted an EUA or other marketing authorization must comply fully with the terms and conditions provided in the EUA or other marketing authorization. For example, EUAs for COVID-19 tests may include conditions of authorization applicable to the EUA holder, authorized distributors and authorized laboratories. Noncompliance with applicable requirements could result in negative consequences, including adverse publicity, judicial or administrative enforcement, warning letters or untitled letters from the FDA, mandated corrective promotional materials, advertising or communications with doctors, and civil or criminal penalties, among others. The FDA can also withdraw marketing authorization for the applicable product, and in the case of a product subject to an EUA, the authorization to market the product under the EUA lasts only as long as the declared public health emergency.
DEA regulation
We are engaged in the research, development, and export of certain products that may be regulated as controlled substances, including microbes designed to generate precursors to cannabinoids or other chemical intermediates. The Controlled Substances Act of 1970 (“CSA”), as amended from time to time, establishes registration, security, recordkeeping, reporting, storage, distribution and other requirements administered by the DEA. The DEA is concerned with the control of handlers of controlled substances, and with the equipment and raw materials used in their manufacture and packaging, in order to prevent loss and diversion into illicit channels of commerce. The DEA regulates controlled substances as Schedule I, II, III, IV or V substances. Schedule I substances by definition have no established medicinal use, and may not be marketed or sold in the United States. Schedule I substances are considered to present the highest risk of abuse, and Schedule V substances the lowest relative risk of abuse among controlled substances. Marijuana is classified as a Schedule I controlled substance. However, the term does not include “hemp,” which means the cannabis plant and any part of that plant, including the seeds and all derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a delta-9 tetrahydrocannabinol (“THC”) concentration of not more than 0.3% on a dry weight basis.
Annual registration is required for any facility that manufactures, distributes, dispenses, imports or exports any controlled substance. The registration is specific to the particular location, business activity and controlled substance schedule. For
46
Table of Contents
example, separate registrations are needed for import and manufacturing, and each registration will specify which controlled substance schedule is authorized for that activity.
The DEA typically inspects a facility to review its security measures prior to issuing a registration. The DEA requires “effective controls and procedures” to guard against theft and diversion of controlled substances. Security requirements vary by controlled substance schedule (with the most stringent requirements applying to Schedule I and Schedule II substances), type of business activity conducted, quantity of substances handled, and a variety of other factors. Required security measures include background checks on employees and physical control of inventory. While the specific means by which effective controls and procedures are achieved may vary, security practices may include use of cages, surveillance cameras and inventory reconciliations. Records must be maintained for the handling of all controlled substances, and, in certain scenarios, periodic reports made to the DEA. Reports must also be made for thefts or losses of any controlled substance, and disposal of controlled substances must adhere to various methods authorized by the regulations. In addition, special authorization and notification requirements apply to imports and exports.
Failure by registered establishments to maintain compliance with applicable requirements, particularly as manifested in loss or diversion, can result in enforcement action. The DEA may seek civil penalties, refuse to renew necessary registrations, or initiate proceedings to revoke those registrations. In certain circumstances, violations could eventuate in criminal proceedings. Individual states also regulate controlled substances.
Laboratory Licensing and Certification Requirements
The clinical laboratories we partner with for our COVID-19 testing program are subject to federal oversight under Clinical Laboratory Improvement Amendments of 1988 (“CLIA”), which requires all clinical laboratories to meet certain quality assurance, quality control and personnel standards. Laboratories also must undergo proficiency testing and are subject to inspections. Standards for testing under CLIA are based on the complexity of the tests performed by the laboratory, with tests classified as “high complexity,” “moderate complexity,” or “waived.” Laboratories performing high complexity testing are required to meet more stringent requirements than moderate complexity laboratories. Certain of our partner laboratories must undergo on-site surveys at least every two years, which may be conducted by the Centers for Medicare and Medicaid Services (“CMS”) under the CLIA program or by a private CMS approved accrediting agency. In addition, we hold CLIA Certificates of Waiver and may perform certain CLIA-waived tests on behalf of our clients, which subjects us to certain CLIA requirements. The sanction for failure to comply with CLIA requirements may be suspension, revocation or limitation of a laboratory’s CLIA certificate, which is necessary to conduct business, as well as significant fines and criminal penalties.
The operations of our partner laboratories and our laboratories holding CLIA Certificates of Waiver are also subject to state and local laboratory regulation. CLIA provides that a state may adopt laboratory regulations different from or more stringent than those under federal law, and a number of states have implemented their own laboratory regulatory requirements. State laws may require that laboratory personnel meet certain qualifications, specify certain quality controls, or require maintenance of certain records. No assurances can be given that we or our partner laboratories will pass all future licensure or certification inspections.
Federal Select Agent Regulations
Our research facilities that synthesize DNA sequences or perform other activities could become subject to the Federal Select Agent Program (“FSAP”), which involves rules administered by the Centers for Disease Control and Prevention (“CDC”) and the USDA Animal and Plant Health Inspection Service (“APHIS”). The FSAP regulates the possession, use, and transfer of biological select agents and toxins that have the potential to pose a severe threat to public health, animal or plant health, or animal or plant products. FSAP regulatory requirements include: (i) registration with the CDC and/or APHIS for research facilities that deal with the select agents and toxins; (ii) submission to periodic biosafety and security inspections; and (iii) reporting of theft, loss or release of select agents. Federal agency enforcement actions for violations of FSAP regulations can include the initiation of corrective actions, complete or partial suspension or revocation of select agent registrations or civil or criminal liability.
Genetically Modified Materials Regulations
Our technologies and the technologies of our customers involve the use of genetically modified cells, organisms and biomaterials, including, without limitation, GMOs and genetically modified microorganisms (“GMMs”), Genetically Modified Materials”), and their respective products. In the United States, the FDA, APHIS and the EPA are the primary agencies that regulate the use of GMOs, GMMs and potential products derived from GMOs or GMMs or Genetically Modified Materials, pursuant to the Coordinated Framework for the Regulation of Biotechnology.
47
Table of Contents
The FDA reviews the safety of food consumed by humans and of feed consumed by animals under the Federal Food, Drug and Cosmetic Act (“FDCA”). Under the FDCA, food and feed manufacturers are responsible for ensuring that the products they market, including those developed through genetic engineering, are safe and properly labeled. In addition, the FDA must approve the use of any food additives, including GMOs, before marketing.
APHIS examines whether a plant itself presents a “plant pest” risk under the Plant Protection Act (“PPA”). Specifically, APHIS is responsible for regulating the introduction (i.e., importation, interstate movement or release into the environment) of certain GMOs and plants under the plant pest provisions in the PPA to ensure that they do not pose a plant pest risk. APHIS finalized changes to the PPA’s implementing regulations with respect to certain GMOs in May 2020. A person or organization may request a regulatory status review from APHIS to determine whether a GMO is unlikely to pose a plant pest risk and, therefore, is not regulated under the plant pest provisions of the PPA or the regulations codified at 7 C.F.R. Part 340; requesting a regulatory status review tends to assume the GMO at issue does not otherwise fall within a regulatory exemption. If the GMO does not qualify for an exemption or if the APHIS regulatory status review process finds that the plant poses a plausible plant pest risk, then the GMO may require an APHIS permit to clarify the process. A regulated article may be subject to APHIS for the environmental release, importation, or interstate movement of the GMO or its progeny.
EPA regulates, under the Federal Insecticide, Fungicide and Rodenticide Act (“FIFRA”), the pesticides (including plant incorporated protectants) that are used with crops, including GMO herbicide-tolerant crops. FIFRA generally requires all pesticides to be registered before distribution or sale, unless they are exempted. Under FIFRA, a pesticide registrant must demonstrate that the pesticide at issue, when used pursuant to its specifications, “will not generally cause unreasonable adverse effects on the environment” to secure a registration. EPA must approve each distinct pesticide product, each distinct use pattern, and each distinct use site. In addition to EPA’s FIFRA authority, EPA also regulates potential human health impacts from pesticides under the FDCA. EPA does so by establishing “tolerance levels” (i.e., “the amount of pesticide that may remain on food products”) under the FDCA.
Telehealth regulation
Our telehealth provider partner is subject to various federal, state and local certification and licensing laws, regulations and approvals, relating to, among other things, the adequacy of health care, the practice of medicine and other health professions (including the provision of remote care and cross-coverage practice), equipment, personnel, operating policies and procedures and the prerequisites for ordering laboratory tests. Some states have enacted regulations specific to providing services to patients via telehealth. Such regulations include, among other things, informed consent requirements that some states require providers to obtain from their patients before providing telehealth services. Health professionals who provide professional services using telehealth modalities must, in most instances, hold a valid license to practice the applicable health profession in the state in which the patient is located. In addition, certain states require a physician providing telehealth to be physically located in the same state as the patient. Any failure to comply with these laws and regulations could result in civil or criminal penalties against telehealth providers.
State corporate practice of medicine and fee splitting laws
Our relationship with our telehealth provider partner, who provides physician oversight and support to individuals seeking COVID-19 diagnostic or screening testing, including evaluating each request for testing, communicating and providing consultation services for certain test results, is subject to various state laws, which are intended to prevent unlicensed persons from interfering with or influencing a physician’s professional judgment, and prohibiting the sharing of professional services income with non-professional or business interests. These laws vary from state to state and are subject to broad interpretation and enforcement by state regulators. A determination of non-compliance could lead to adverse judicial or administrative action against us and/or our telehealth provider partner, civil or criminal penalties, receipt of cease and desist orders from state regulators, loss of provider licenses, or a restructuring of our arrangement with our telehealth provider partner.
Healthcare fraud and abuse laws
Although none of our COVID-testing offerings are currently billed to any third-party payor, including any commercial payor or government healthcare program, by us or any of our laboratory or telehealth provider partners, we may nonetheless be subject to a number of federal and state healthcare regulatory laws that restrict business practices in the healthcare industry. These laws include, but are not limited to, federal and state anti-kickback, false claims, and other healthcare fraud and abuse laws.
The federal Anti-Kickback Statute prohibits, among other things, individuals or entities from knowingly and willfully offering, paying, soliciting or receiving remuneration, directly or indirectly, overtly or covertly, in cash or in kind to induce or in return for purchasing, leasing, ordering or arranging for or recommending the purchase, lease or order of any item or
48
Table of Contents
service reimbursable under Medicare, Medicaid or other federal healthcare programs. A person or entity does not need to have actual knowledge of this statute or specific intent to violate it in order to have committed a violation.
The federal physician self-referral prohibition, commonly known as the Stark Law, prohibits a physician, in the absence of an applicable exception, from making a referral for certain designated health services covered by the Medicare or Medicaid program, including clinical laboratory services, if the physician or an immediate family member of the physician has a financial relationship with the entity providing the designated health services. The Stark Law also prohibits the entity furnishing the designated health services from billing, presenting or causing to be presented a claim for the designated health services furnished pursuant to the prohibited referral.
The federal civil and criminal false claims laws, including the civil False Claims Act, and civil monetary penalties laws prohibit, among other things, any individual or entity from knowingly presenting, or causing to be presented, a false claim for payment to the federal government, knowingly making, using or causing to be made or used a false record or statement material to a false or fraudulent claim to the federal government, or from knowingly making a false statement to avoid, decrease or conceal an obligation to pay money to the federal government. In addition, the government may assert that a claim including items or services resulting from a violation of the federal Anti-Kickback Statute or the Stark Law, constitutes a false or fraudulent claim for purposes of the civil False Claims Act.
In addition to the Anti-Kickback Statute and the Stark Law, the United States recently enacted a law known as the Eliminating Kickbacks in Recovery Act (“EKRA”), which created a new federal crime for knowingly and willfully: (1) soliciting or receiving any remuneration in return for referring a patient to a recovery home, clinical treatment facility, or laboratory; or (2) paying or offering any remuneration to induce such a referral or in exchange for an individual using the services of a recovery home, clinical treatment facility, or laboratory. Unlike the Anti-Kickback Statute, EKRA is not limited to services reimbursable under a government health care program, but instead extends to all services reimbursed by “health care benefit programs.”
The federal Health Insurance Portability and Accountability Act of 1996, as amended by Health Information Technology for Economic and Clinical Health Act (“HIPAA”) created additional federal criminal statutes that prohibit, among other things, knowingly and willfully executing a scheme to defraud any healthcare benefit program, including private third-party payors and knowingly and willfully falsifying, concealing or covering up a material fact or making any materially false, fictitious or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items or services. Similar to the U.S. federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation.
Similar state and local laws and regulations may also restrict business practices in the medical device and clinical laboratory industries, such as state anti-kickback and false claims laws, which may apply to business practices, including but not limited to, research, distribution, sales and marketing arrangements and claims involving healthcare items or services reimbursed by non-governmental third-party payors, including private insurers, or by patients themselves; and state laws that require companies to comply with the industry’s voluntary compliance guidelines and the relevant compliance guidance promulgated by the federal government, or otherwise restrict payments that may be made to healthcare providers and other potential referral sources.
Violation of any of such laws or any other governmental regulations that apply may result in significant criminal, civil and administrative penalties including damages, fines, imprisonment, disgorgement, additional reporting requirements and oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of non-compliance with these laws, contractual damages, reputational harm, diminished profits and future earnings, disgorgement, exclusion from participation in government healthcare programs and the curtailment or restructuring of our operations.
Federal and state data privacy and security regulations
Numerous state, federal and foreign laws, including consumer protection laws and regulations, govern the collection, dissemination, use, access to, confidentiality and security of personal information, including health-related information. In the United States, numerous federal and state laws and regulations, including data breach notification laws, health information privacy and security laws, including HIPAA, and federal and state consumer protection laws and regulations (e.g., Section 5 of the Federal Trade Commission Act (“FTC Act”)), that govern the collection, use, disclosure, and protection of health-related and other personal information could apply to our operations or the operations of our partners. HIPAA, and its respective implementing regulations, imposes obligations on “covered entities,” including certain health care providers, health plans, and health care clearinghouses, and their respective “business associates” that create, receive, maintain or transmit individually identifiable health information for or on behalf of a covered entity, as well as their covered subcontractors with respect to safeguarding the privacy, security and transmission of individually identifiable health
49
Table of Contents
information. Violations of the HIPAA privacy and security regulations may result in civil and criminal penalties. The U.S. Department of Health and Human Services (“HHS”) is required to conduct periodic compliance audits of covered entities and their business associates. HIPAA also authorizes State Attorneys General to bring civil actions seeking either an injunction or damages in response to violations of HIPAA privacy and security regulations.
In addition, certain state laws, such as the California Confidentiality of Medical Information Act, the California Consumer Privacy Act of 2018 (“CCPA”) and the California Privacy Rights Act (“CPRA”) govern the privacy and security of personal information, including health-related information in certain circumstances, some of which are more stringent than HIPAA and many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. Failure to comply with these laws, where applicable, can result in the imposition of significant civil and/or criminal penalties and private litigation. Privacy and security laws, regulations, and other obligations are constantly evolving, may conflict with each other (thus complicating compliance efforts), and can result in investigations, proceedings, or actions that lead to significant civil or criminal penalties and restrictions on data processing.
Ginkgo Corporate Information
Prior to consummation of the Business Combination on September 16, 2021, Ginkgo Bioworks Holdings, Inc., a Delaware corporation, was known as Soaring Eagle Acquisition Corp., a blank check company incorporated as a Cayman Islands exempted company on October 22, 2020. Ginkgo’s operating subsidiary was incorporated in 2008 as Ginkgo Bioworks, Inc., a Delaware corporation.
Ginkgo’s principal executive office is located at 27 Drydock Avenue, Boston, Massachusetts 02210, and Ginkgo’s telephone number is (877) 422-5362. Ginkgo’s corporate website address is www.ginkgobioworks.com. We make available on the Investor Relations section of our website, free of charge, our annual reports on Form 10-K, quarterly reports on Form 10-Q, current reports on Form 8-K, proxy statements, and Forms 3, 4 and 5, and amendments to those reports as soon as reasonably practicable after filing such documents with, or furnishing such documents to, the U.S. Securities and Exchange Commission (the “SEC”). The SEC maintains a website (www.sec.gov) that contains reports, proxy and information statements and other information regarding issuers that file electronically with the SEC.
The information contained on, or accessible through, our corporate website is not incorporated into this Annual Report and should not be considered part of this Annual Report. The inclusion of the corporate website address is an inactive textual reference only.
50
Table of Contents
Item 1A. Risk Factors.
An investment in our securities involves a high degree of risk. You should carefully consider the following risk factors, together with all of the other information included in this Annual Report, before making an investment decision. Our business, prospects, financial condition or operating results could decline due to any of these risks and, as a result, you may lose all or part of your investment.
Unless the context otherwise requires, all references in this section to the “Company,” “we,” “us” or “our” refer to the business of Ginkgo and its subsidiaries.
Risks Related to Ginkgo’s Business
We have a history of net losses. We expect to continue to incur losses for the foreseeable future, and we may never achieve or maintain profitability.
We have incurred significant operating losses since our inception. Our net loss attributable to our stockholders was approximately $1,830.0 million, $126.6 million and $119.3 million for fiscal years ended December 31, 2021, 2020 and 2019, respectively. As of December 31, 2021, we had an accumulated deficit of approximately $2,297.9 million. We may incur losses and negative cash flow from operating activities for the foreseeable future as we continue to invest significant additional funds toward further developing our platform, the cell programs we perform on behalf of our customers and otherwise growing our business. Our operating expenses have increased as a result of becoming a public company, and we expect that our operating expenses will continue to increase as we grow our business. We have derived a significant portion of our revenues from fees and milestone payments from technical development services provided to customers to advance programs. Historically, these fees have not been sufficient to cover the full cost of our operations. Additionally, if our customers terminate their agreements or development plans with us, our near-term revenues could be adversely affected. In addition, certain of our customer agreements provide for milestone payments, future royalties and other forms of contingent consideration, the payment of which are uncertain, as they are dependent on our ability to successfully develop engineered cells, bioprocesses, or other deliverables and our customers’ ability and willingness to successfully develop and commercialize products and processes.
Our expenses may exceed revenues for the foreseeable future and we may not achieve profitability. If we fail to achieve profitability, or if the time required to achieve profitability is longer than we anticipate, we may not be able to expand or continue our business, and the value of our common stock could be negatively impacted. Our ability to achieve or sustain profitability is based on numerous factors, many of which are beyond our control, including the development of our platform, the initiation of new programs with new and existing customers, the commercial terms of our programs, our ability to advance cell engineering programs in a timely and cost-effective manner, our ability to extend new offerings to customers, our customers’ ability to scale up bioprocesses, the ability of our customers to produce and sell products, the impact of market acceptance of our customers’ products, and our and our customers’ market penetration and margins. Even if we do achieve profitability, we may not be able to sustain or increase profitability on a quarterly or annual basis.
We may need substantial additional capital in the future in order to fund our business.
We have consumed considerable amounts of capital to date, and we expect to incur continued net losses over the next several years as we continue to develop our business, advance our programs, expand and enhance our platform, and make the capital investments necessary to scale up our Foundry operations and Codebase assets. We may also use additional capital for strategic investments and acquisitions. We believe that our cash and cash equivalents, short-term investments, and interest earned on investments will be sufficient to meet our projected operating requirements for several years and until we reach profitability. However, these assumptions may prove to be incorrect and we could exhaust our available capital resources sooner than we currently expect. Because of the numerous risks and uncertainties associated with our programs, including risks and uncertainties that could impact the rate of progress of our programs, we are unable to estimate with certainty the amounts of capital outlays and operating expenditures associated with these activities.
We do not currently have any commitments for future funding. We may receive fees, milestones, and royalty payments under our customer agreements, but these are not guaranteed. Additionally, we may be able to sell our equity interests in certain subsidiaries or collaborations but most of these equity stakes are illiquid (e.g. in private companies) and we may not be able to find a buyer or may incur significant impairment if forced to sell these positions for liquidity. We may not receive any further funds under those agreements, the funds we receive may be lower than projected, or our program costs may be higher than projected. In addition, we may not be able to sign new customer agreements or enter into new development plans with existing customers with adequate funds to cover program development expenses. As a result of these and other factors, we do
51
Table of Contents
not know whether additional financing will be available when needed, or, if available, whether such financing would be on terms favorable to our stockholders or us.
If future financings involve the issuance of equity securities, our existing stockholders would suffer dilution. If we raise debt financing in the future, we may be subject to restrictive covenants that limit our ability to conduct our business. Our ability to raise funds may be adversely impacted by current or future economic conditions. If we fail to raise sufficient funds and continue to incur losses, our ability to fund our operations, take advantage of strategic opportunities, or otherwise respond to competitive pressures could be significantly limited. If adequate funds are not available, we may not be able to successfully execute our business plan or continue our business.
We have experienced rapid growth and expect our growth to continue, and if we fail to effectively manage our growth, then our business, results of operations, and financial condition could be adversely affected.
We have experienced substantial growth in our business since inception, which has placed and may continue to place significant demands on our company culture, operational infrastructure, and management. We believe that our culture has been a critical component of our success. We have invested substantial time and resources in building our team and nurturing a culture of empowerment of, and active engagement by, our employees. As we expand our business and mature as a public company, we may find it difficult to maintain our culture while managing this growth. Any failure to manage our anticipated growth and organizational changes in a manner that preserves the key aspects of our culture could be detrimental to future success, including our ability to recruit and retain personnel, and effectively focus on and pursue our objectives. This, in turn, could adversely affect our business, results of operations, and financial condition.
In addition, in order to successfully manage our rapid growth, our organizational structure has become more complex and is likely to continue to become more complex. In order to manage these increasing complexities, we will need to continue to scale and adapt our operational, financial, and management controls, as well as our reporting systems and procedures. The expansion of our systems and infrastructure will require us to commit substantial financial, operational, and management resources before our revenue increases and without any assurances that our revenue will increase.
Finally, continued growth could strain our ability to maintain reliable service levels and offerings for our customers. If we fail to achieve the necessary level of capacity, quality and efficiency in performing services and other development activities, or the necessary level of efficiency in our organizational structure as we grow, then our business, results of operations and financial condition could be adversely affected.
Our limited operating history makes it difficult to evaluate our current business and future prospects.
We have a portfolio of cell engineering programs which vary in start date, duration, complexity, and revenue potential. Additionally, our downstream economics in the form of equity interests, milestone payments, or royalty streams add an additional level of uncertainty to our possible future performance. Consequently, predictions about our future success or viability are highly uncertain and may not be as accurate as they could be if we had a longer company history of successfully developing, commercializing and generating revenue from our programs and/or downstream economic participation. With respect to our biosecurity offering, prior to 2020, we had no experience developing or commercializing testing services. Moreover, as described above, given the limited operating history of our biosecurity offering, our reliance on school funding for testing, potential disruptions from vaccine rollout generally, the availability of COVID-19 therapeutics, the impact of summer vacation, and the increased availability of over-the-counter testing options, the future performance of our COVID-19 offerings is unpredictable. Moreover, we cannot predict how long the COVID-19 pandemic will continue and, therefore, we cannot predict the duration of the revenue stream from our COVID-19 testing products and services.
Our long-term objective is to generate free cash flow from the commercialization of programs by customers across a variety of industries, as well as, from our biosecurity-focused offerings. Our estimated costs and timelines for the completion of programs are based on our experiences to date and our expectations for each stage of the program in development. Given the variety of types of programs we support and the continued growth of our platform, there is variability in timelines and costs for launching and executing programs, and completion dates can change over the course of a customer engagement. In addition, our costs and timelines may be greater or subject to variability where regulatory requirements lead to longer timelines, such as in agriculture, food, and therapeutics. In addition, we have equity interests in certain companies and there is and will continue to be variability in the financial performance of these other companies or future companies in which we may have equity interests.
As a business with a limited operating history, we may encounter unforeseen expenses, difficulties, complications, delays, and other known and unknown obstacles. We have encountered in the past, and will encounter in the future, risks and uncertainties frequently experienced by growing companies with limited operating histories in emerging and rapidly
52
Table of Contents
changing industries. If our assumptions regarding these risks and uncertainties, which we use to plan and operate our business, are incorrect or change, or if we do not address these risks successfully, our results of operations could differ materially from our expectations, and our business, financial condition, and results of operations could be adversely affected.
If we cannot maintain and expand current customer partnerships and enter into new customer partnerships, our business could be adversely affected.
We do not generate substantial revenue from our own products, and instead generate revenue from customer collaborations in which we provide services, and also receive downstream value in the form of royalties, equity, or milestone payments. As a result, our success depends on our ability to expand the number, size and scope of our customer collaborations. Our ability to win new business depends on many factors, including our reputation in the market, the quality of our service offerings relative to alternatives, the pricing and efficiency of our services relative to alternatives, and our technical capabilities. If we fail to maintain a position of strength in any of these factors, our ability to either sign new customer collaborations or launch new programs with existing customers may suffer and this could adversely affect our prospects. Additionally, in the process of developing programs, we generate Foundry know-how and accumulate meaningful biological and data assets, including optimized proteins and organisms, characterized genetic parts, enhanced understanding of metabolic pathways, biological, chemical, and genetic libraries, and other elements of biological data. Data and know-how generated from our programs provide the basis for expanded capabilities that we believe further supports our customer collaborations. As a result, in addition to reducing our revenue or delaying the development of our programs, the loss of one or more of our customer relationships or the failure to add new customers or programs may hinder our accumulation of such information, thus hindering our efforts to advance our technological differentiation and improve our platform.
We engage in conversations with companies regarding potential customer collaborations on an ongoing basis. We may spend considerable time and money engaging in these conversations and feasibility assessments, including understanding the technical approach to a program, customer concerns and limitations, and legal or regulatory landscape of a potential program or offering, which may not result in a commercial agreement. Even if an agreement is reached, the resulting relationship may not be successful for many reasons, including our inability to complete a program to our customers’ specifications or within our customers’ time frames, or unsuccessful development or commercialization of products or processes by our customers. In such circumstances, our revenues and downstream value potential from such a collaboration might be meaningfully reduced.
We currently own and may in the future own equity interests in other operating companies, including certain of our customers; consequently, we have exposure to the volatility and liquidity risks inherent in holding their equity and overall operational and financial performance of these businesses.
We currently own equity interests in several of our customers. In the future, we may also own equity interests in other companies. The process by which we receive equity interests and the factors we consider in deciding whether to accept, hold or dispose of these equity positions may differ significantly from those that an independent investor would evaluate when considering equity interests in a company. Owning equity increases our exposure to the risks of the other company and, in the case of customers, beyond the products of our collaborations. Our equity ownership positions expose us to market volatility and the potential for negative returns. We may have restrictions on resale or limited markets to sell our equity ownership. In many cases, our equity position is a minority position which exposes us to further risk, as we are not able to exert control over the companies in which we hold securities.
In connection with future collaborations or joint ventures, we may, from time to time, receive warrants or options, all of which involve special risks. To the extent we receive warrants or options in connection with future collaborations or joint ventures, we would be exposed to risks involving pricing differences between the market value of underlying securities and our exercise price for the warrants or options, a possible lack of liquidity, and the related inability to close a warrant or option position, all of which could ultimately have an adverse effect on our financial position.
We leverage our own resources and partner with strategic and financial investors in order to help early stage companies and innovators secure funding and benefit from our platform, which exposes us to a number of risks.
Since our founding, we have helped to launch new companies (such as Motif, Allonnia LLC, Arcaea LLC (formerly known as Kalo), Ayana Bio, LLC and Verb Biotics, LLC) by bringing together strategic and financial investors to secure funding for these early stage and small companies. Going forward, we intend to continue to leverage our own balance sheet and partner with investors to enable companies at all stages to benefit from our platform.
Partnering with and investing in early stage and small companies may expose us to a number of risks, including that early stage and small companies may have:
53
Table of Contents
Any of these factors or changes thereto could impair an early stage or small company’s financial condition, results of operation, cash flow or result in other adverse events, such as bankruptcy. This, in turn, could result in losses in our investments and a change in our income (loss) on investments.
We may pursue strategic acquisitions and investments that are dilutive to our stockholders and that could have an adverse impact on our business if they are unsuccessful.
We have made acquisitions in the past and, as appropriate opportunities become available, we may acquire additional businesses, assets, technologies, or products to enhance our business in the future, but our ability to do so successfully cannot be ensured. We have also made investments in companies that we view as synergistic with our business. Although we conduct due diligence on these acquisitions and investments, such processes may fail to reveal significant liabilities and we could incur losses resulting from undiscovered liabilities of the acquired business that are not covered by indemnification we may obtain from the seller. Even if we identify suitable opportunities, we may not be able to complete such acquisitions on favorable terms or at all. Any acquisitions we make may not strengthen our competitive position, and these transactions may be viewed negatively by customers or investors. We may decide to incur debt or spend cash in connection with a strategic acquisition, which may cause us to face liquidity concerns or be subject to restrictive covenants in the future. We have, and in the future may, also issue common stock or other equity securities to the stockholders of the acquired company, which could constitute a material portion of our then-outstanding shares of common stock and may reduce the percentage ownership of our existing stockholders.
In addition, we may not be able to successfully integrate the acquired personnel, assets, technologies, products and/or operations into our existing business in an effective, timely, and non-disruptive manner or retain acquired personnel following an acquisition. Acquisitions may also divert management’s attention from day-to-day responsibilities, increase our expenses and reduce our cash available for operations and other uses. In addition, we may not be able to fully recover the costs of such acquisitions or be successful in leveraging any such strategic transactions into increased business, revenue, or profitability. We also cannot predict the number, timing, or size of any future acquisitions or the effect that any such transactions might have on our operating results.
Accordingly, although there can be no assurance that we will undertake or successfully complete any acquisitions, any transactions that we do complete may not yield the anticipated benefits and may be subject to the foregoing or other risks and have a material and adverse effect on our business, financial condition, results of operations, and prospects. Conversely, any failure to pursue or delay in completing any acquisition or other strategic transaction that would be beneficial to us could delay the development of our platform or advancement of our programs and, thus, potential commercialization of our customer’s products.
54
Table of Contents
Our programs may not achieve milestones and other anticipated key events on the expected timelines or at all, which could have an adverse impact on our business and could cause the price of our common stock to decline.
We may adopt various technical, manufacturing, regulatory, commercial, and other objectives for our programs. These milestones may include our or our customer’s expectations regarding the commencement or completion of technical development, the achievement of manufacturing targets, the submission of regulatory filings, or the realization of other development, regulatory, or commercialization objectives by us or our customers. The achievement of many of these milestones may be outside of our control. All of these milestones are based on a variety of assumptions, including assumptions regarding capital resources, constraints, and priorities, progress of and results from R&D activities, and other factors, including impacts resulting from the COVID-19 pandemic, any of which may cause the timing of achievement of the milestones to vary considerably. If we, our collaborators, or our customers fail to achieve milestones in the expected timeframes, the commercialization of our programs may be delayed, our credibility may be undermined, our business and results of operations may be harmed, and the trading price of our common stock may decline.
We must continue to secure and maintain sufficient and stable supplies of laboratory reagents, consumables, equipment, and laboratory services. We depend on a limited number of suppliers, some of which are single- source suppliers, and contract manufacturers for critical supplies, equipment, and services for research, development, and manufacturing of our products and processes. Our reliance on these third parties exposes us to risks relating to costs, contractual terms, supply, and logistics, and the loss of any one or more of these suppliers or contract manufacturers or their failure to supply us with the necessary supplies, equipment, or services on a timely basis, could cause delays in our research, development, or production capacity and adversely affect our business.
The COVID-19 pandemic has caused substantial disruption in global supply chains and the ability of third parties to provide us services on a timely basis or at all. As a result, we have experienced shortages in some of our key equipment and supplies, including those required in our labs, as well as, disruptions in services provided by third parties, and may continue to do so in the future as a result of the pandemic, or otherwise. We may also experience price increases, quality issues and longer lead times due to unexpected material shortages, service disruptions, and other unanticipated events, which may adversely affect our supply of lab equipment, lab supplies, chemicals, reagents, supplies, and lab services. For some suppliers, we do not enter into long-term agreements and instead secure our materials and services on a purchase order basis. Our suppliers may reduce or cease their supply of materials or services to us at any time in the future. If the supply of materials or services is interrupted, our programs may be delayed.
We depend on a limited number of suppliers for critical items, including lab consumables and equipment, for the development of our programs. Some of these suppliers are single-source suppliers. We do not currently have the infrastructure or capability internally to manufacture these items at the necessary scale or at all. Although we have a reserve of supplies and although alternative suppliers exist for some of these critical products, services, and equipment, our existing processes used in our Foundries have been designed based on the functions, limitations, features, and specifications of the products, services, and equipment that we currently utilize. While we work with a variety of domestic and international suppliers, our suppliers may not be obligated to supply products or services or our arrangements may be terminated with relatively short notice periods. Additionally, we do not have any control over the process or timing of the acquisition or manufacture of materials by our manufacturers and cannot ensure that they will deliver to us the items we order on time, or at all.
In particular, we rely on Twist for custom DNA synthesis and Thermo Fisher Scientific Inc. and others for certain instruments and consumables. The price and availability of DNA, chemicals, reagents, equipment, consumables, and instruments have a material impact on our ability to provide Foundry services. We also rely on third parties, such as Berkeley Lights, to develop workflows to use the equipment they provide to us. We may rely on contract manufacturers like Fermic, s.a. de.c.v for scale-up fermentation development, fermentation, and manufacturing of products for some customers.
The loss of the products, services, and equipment provided by one or more of our suppliers could require us to change the design of our research, development, and manufacturing processes based on the functions, limitations, features, and specifications of the replacement items or seek out a new supplier to provide these items. Additionally, as we grow, our existing suppliers may not be able to meet our increasing demand, and we may need to find additional suppliers. We may not be able to secure suppliers who provide lab supplies at, or equipment and services to, the specification, quantity, and quality levels that we demand (or at all) or be able to negotiate acceptable fees and terms of services with any such suppliers.
As described above, some lab equipment, lab consumables, and other services and materials that we purchase are purchased from single-source or preferred suppliers, which limits our negotiating leverage and our ability to rely on additional or
55
Table of Contents
alternative suppliers for these items. Our dependence on these single-source and preferred suppliers exposes us to certain risks, including the following:
Moreover, to meet anticipated market demand, our suppliers may need to increase manufacturing capacity, which could involve significant challenges. This may require us and our suppliers to invest substantial additional funds and hire and retain the technical personnel who have the necessary experience. Neither we nor our suppliers may successfully complete any required increase to existing research, development, or manufacturing capacity in a timely manner, or at all.
For the year ended December 31, 2021, our cost of lab equipment, lab supplies, and lab services accounted for a significant portion of our total R&D expenses. In the event of price increases by suppliers, we may attempt to pass the increased costs to our customers. However, we may not be able to raise the prices of our Foundry services sufficiently to cover increased costs resulting from increases in the cost of our materials and services, or the interruption of a sufficient supply of materials or services. As a result, materials and services costs, including any price increase for our materials and services, may negatively impact our business, financial condition, and results of operations.
Some of our suppliers and contract manufacturers are foreign entities. We may face disruptions due to the inability to obtain customs clearances in a timely manner or restrictions on shipping or international travel due to the COVID-19 pandemic. As a result of ongoing global supply chain challenges resulting in very long lead times for certain products and equipment, we may order in larger volumes in order to secure the supplies we require for our future operations, which may negatively impact our financial conditions, especially if we are unable to use the supplies ordered.
We use biological, hazardous, flammable and/or regulated materials that require considerable training, expertise and expense for handling, storage and disposal and may result in claims against us.
We work with biological and chemical materials that could be hazardous to human, animal, or plant health and safety or the environment. Our operations produce hazardous and biological waste products, and we largely contract with third parties for the disposal of these products. Federal, state, and local laws and regulations govern the use, generation, manufacture, storage, handling, and disposal of these materials and wastes. Compliance with applicable laws and regulations is expensive, and current or future laws and regulations may restrict our operations. If we do not comply with applicable laws and regulations, we may be subject to fines and penalties.
In addition, we cannot eliminate the risk of (a) accidental or intentional injury or (b) release, or contamination from these materials or wastes, which could expose us to liability. Furthermore, laws and regulations are complex, change frequently, and have tended to become more stringent. We cannot predict the impact of such changes and cannot be certain of our future compliance. Accordingly, in the event of release, contamination, or injury, we could be liable for the resulting harm or penalized with fines in an amount exceeding our resources and our operations could be suspended or otherwise adversely affected. These liabilities could also include regulatory actions, litigation, investigations, remediation obligations, damage to our reputation and brand, supplemental disclosure obligations, loss of customer, consumer, and partner confidence in the safety of our laboratory operations, impairment to our business, and corresponding fees, costs, expenses, loss of revenues, and other potential liabilities, as well as, increased costs or loss of revenue or other harm to our business.
56
Table of Contents
The release of GMOs or Genetically Modified Materials, whether inadvertent or purposeful, into uncontrolled environments could have unintended consequences, which may result in increased regulatory scrutiny and otherwise harm our business and financial condition.
The genetically engineered organisms and materials that we develop may have significantly altered characteristics compared to those found in the wild, and the full effects of deployment or release of our genetically engineered organisms and materials into uncontrolled environments may be unknown. In particular, such deployment or release, including an unauthorized release, could impact the environment or community generally or the health and safety of our employees, our customers’ employees, and the consumers of our customers’ products.
In addition, if a high profile biosecurity breach or unauthorized release of a biological agent occurs within our industry, our customers and potential customers may lose trust in the security of the laboratory environments in which we produce genetically modified organisms (“GMOs”) and genetically modified plant or animal cells and genetically modified proteins and biomaterials (collectively, “Genetically Modified Materials”), even if we are not directly affected. Any adverse effect resulting from such a release, by us or others, could have a material adverse effect on the public acceptance of products from engineered cells and our business and financial condition. Such a release could result in increased regulatory scrutiny of our facilities, platform, and programs, and could require us to implement additional costly measures to maintain our regulatory permits, licenses, authorizations and approvals. To the extent such regulatory scrutiny or changes impact our ability to execute on existing or new programs for our customers, or make doing so more costly or difficult, our business, financial condition, or results of operations may be adversely affected. In addition, we could have exposure to liability for any resulting harm, as well as to regulatory actions, litigation, investigations, remediation obligations, damage to our reputation and brand, supplemental disclosure obligations, loss of customer, consumer, and partner confidence in the safety of engineered cells materials, and organisms, impairment to our business, and corresponding fees, costs, expenses, loss of revenues, and other potential liabilities, as well as, increased costs or loss of revenue or other harm to our business.
We could synthesize DNA sequences or engage in other activity that contravenes biosecurity requirements, or regulatory authorities could promulgate more far-reaching biosecurity requirements that our standard business practices cannot accommodate, which could give rise to substantial legal liability, impede our business, and damage our reputation.
The Federal Select Agent Program (“FSAP”) involves rules administered by the Centers for Disease Control and Prevention and the Animal and Plant Health Inspection Service that regulate possession, use, and transfer of biological select agents and toxins that have the potential to pose a severe threat to public, animal, or plant health or to animal or plant products. In accordance with the International Gene Synthesis Consortium’s (“IGSC”) Harmonized Screening Protocol for screening of synthetic DNA sequence orders, we follow biosafety and biosecurity industry practices and avoid DNA synthesis activities that implicate FSAP rules by screening synthetic DNA sequence orders against the IGSC’s Regulated Pathogen Database; however, we could err in our observance of compliance program requirements in a manner that leaves us in noncompliance with FSAP or other biosecurity rules. In addition, authorities could promulgate new biosecurity requirements that restrict our operations. One or more resulting legal penalties, restraints on our business or reputational damage could have material adverse effects on our business, financial condition, or results of operations.
Third parties may use our engineered cells, materials, and organisms and accompanying production processes in ways that could damage our reputation.
After our customers have received our engineered cells, materials, and organisms and accompanying production processes, we do not have any control over their use and our customers may use them in ways that are harmful to our reputation. In addition, while we have established a biosecurity program designed to comply with biosafety and biosecurity requirements and export control requirements in an effort to ensure that third parties do not obtain our engineered cells or other biomaterials for malevolent purposes, we cannot guarantee that these preventative measures will eliminate or reduce the risk of the domestic and global opportunities for the misuse or negligent use of our engineered cells materials, and organisms and production processes. Accordingly, in the event of such misuse or negligent use, our reputation, future revenue, and operating results may suffer.
International expansion of our business exposes us to business, regulatory, political, operational, financial, and economic risks associated with doing business outside of the United States.
We currently market our services and deliver our programs, materials, and processes outside of the United States and may market future offerings outside of the United States. We, and our suppliers, collaborators, and customers, currently conduct business outside of the United States. From time to time, our services may include the hiring or secondment of our employees outside the United States at third party facilities or require the hiring or secondment of foreign persons within our facilities.
57
Table of Contents
Accordingly, we are subject to a variety of risks inherent in doing business internationally, and our exposure to these risks will increase as we continue to expand our operations and customer base. These risks include:
Additionally, as part of our growth strategy, we will continue to evaluate potential opportunities for international expansion. Operating in international markets requires significant resources and management attention and will subject us to regulatory, economic and political risks in addition to those we face in the United States. However, our international expansion efforts may not be successful, which could limit the size of our market or the ability to provide services or programs internationally.
In addition, due to potential costs from any international expansion efforts and potentially higher supplier costs outside of the United States, our international operations may operate with a lower margin profile. As a result, our margins may fluctuate as we expand our operations and customer base internationally.
Any of these factors could significantly harm our future international expansion and operations and, consequently, our revenue and results of operations.
58
Table of Contents
Risks Related to Our Customers
We rely on our customers to develop, produce and manufacture products using the engineered cells and/or biomanufacturing processes that we develop. If these initiatives by our customers are not successful or do not achieve commercial success, or if our customers discontinue their development, production and manufacturing efforts using our engineered cells and/or biomanufacturing processes, our future financial position may be adversely impacted.
We operate as a platform company. As such, we rely on our customers to commercialize products that may be enabled by our engineered cells and/or biomanufacturing processes. A portion of the value in our customer collaborations is earned through downstream value sharing in the form of equity, royalty streams, or milestone payments. If our customers are not successful in bringing these products to market, the downstream portion of our value will be adversely impacted. Because we do not directly control manufacturing, product or downstream process development or commercialization, we have limited ability to impact the quality of our partners’ production processes and ultimate commercial success.
In addition, our customers may simply choose not to develop or commercialize a product we have enabled in which we are entitled to downstream value sharing. In our current relationships, we would have limited or no recourse to find alternative methods to monetize these products without the original customer. Because this industry is still nascent and the regulatory environment is evolving, we have limited historical information on the probability of commercial success for bioengineered products or biomanufacturing processes in the market and have limited ability to underwrite the likelihood that our customers will be able to create valuable products or processes in their market using the results of their programs with us. If we overestimate the probability of commercial success, the price of our common stock may be adversely impacted as a result of lower expectations for future cash flows from customer collaborations.
Our revenue is concentrated in a limited number of customers, some of which are related parties, and our revenue, results of operations, cash flows and reputation in the marketplace may suffer upon the loss of a significant customer.
We have derived, and may continue to derive, a significant portion of our revenue from a limited number of large customers. During the year ended December 31, 2021, two customers each represented more than 10% of our total revenue and cumulatively represented 28% of our total revenue. Due to the significant time required to acquire new customers, to plan and develop new programs for customers, and to satisfactorily execute on existing programs, the loss of one or both of these customers, or the loss of any other significant customer or a significant reduction in the amount of demand from a significant customer would adversely affect our revenue, results of operations, cash flows and reputation in the marketplace. There is always a risk that existing customers will not elect to do business with us in the future or will experience financial difficulties. If our customers experience financial difficulties or business reversals which reduce or eliminate the need for our services, they may be unable or unwilling to fulfill their contracts with us. There is also the risk that our customers will attempt to impose new or additional requirements on us that reduce the profitability of the services performed by Ginkgo. Our customer concentration also increases the concentration of our accounts receivable and our exposure to payment defaults by key customers, which could expose us to substantial and potentially unrecoverable costs if we do not receive payment from key customers. Additionally, the loss of any significant customer could pose reputational harm to Ginkgo and make it more challenging to acquire new customers.
In addition, while our customer collaborations are typically multi-year, we generally do not require our customers to generate a minimum amount of annual demand and without such contracts, our customers are not obligated to use our services beyond the amounts they choose to incur. Our customers may choose to use less of our services depending on program progress, their own technological capabilities, market demand for their products and/or their own internal budget cycles. As a result, we cannot accurately predict our customers’ decisions to reduce or cease utilizing our services. Even where we enter into long-term contracts with our customers, there is no guarantee that such agreements will be negotiated on terms that are commercially favorable to us in the long-term. In addition, existing customers may choose to perform some or all of the services they expect from us internally, with another third-party partner or by using capabilities from acquisitions of assets.
In certain cases, our business partners may have discretion in determining when and whether to make announcements about the status of our collaborations, including about developments and timelines for advancing programs, and the price of our common stock may decline as a result of announcements of unexpected results or developments.
Generally, we and our customers must mutually agree on determining when and whether to make announcements about the status of our collaborations, including developments in our programs and timelines for commercialization of or improvements to products using engineered cells developed using our platform. However, in some cases our customers may report or otherwise may be obligated to disclose certain matters without our consent. Our partners may also wish to report such information more or less frequently than we intend to or may not wish to report such information at all. We or our partners may announce a collaboration or partnership even if there is no guarantee that we will recognize program fees. The
59
Table of Contents
price of our common stock may decline as a result of a public announcement of unexpected results or developments in our partnerships, or as a result of our partners not consenting to an announcement or withholding information.
Risks Related to the COVID-19 Pandemic
The recent COVID-19 pandemic and the global attempt to contain it may harm our business and results of operations.
The full impact of the continuing COVID-19 pandemic and related public health measures on our business will depend largely on future developments, including the duration and severity of the pandemic, which remains highly uncertain. Extraordinary actions have been taken by international, federal, state and local public health and governmental authorities to contain and combat the outbreak and spread of COVID-19 throughout the world, including travel bans, quarantines, capacity limitations at facilities, “stay-at-home” orders and similar mandates for many individuals to substantially restrict daily activities and for many businesses to curtail or cease normal operations. Additionally, our operations rely on the availability of laboratory scientists, engineers and facility, safety, quality and compliance personnel to work on-site. If a critical team member falls ill or needs to quarantine, or if a critical mass of our personnel falls ill or needs to quarantine, we may not be able to continue operations. The COVID-19 pandemic has also had an adverse effect on our ability to attract, recruit, interview and hire at the pace we would typically expect to support our rapidly expanding operations, as well as on our ability to build out facilities to accommodate expanding operations.
The COVID-19 pandemic has had, and is expected to continue to have, an adverse impact on our operations, particularly as a result of preventive and precautionary measures that we, other businesses, and governments are taking. For example, as part of these efforts and in accordance with applicable government directives, we initially temporarily suspended some programs at our facilities in Boston, Massachusetts in late March 2020. We have continued to operate within the rules and guidance applicable to our business during the pandemic, including by requiring physical distancing, quarantining our personnel and reducing capacity limits in our facilities, and operations at third-party facilities have been similarly impacted by governmental mandates and guidelines; however, a continuing implementation of these restrictions, or the implementation of additional restrictions, could further impact our ability to operate effectively and conduct ongoing R&D, laboratory operations, sales and marketing activities or other activities or operations, or lead to further compliance costs.
We have also incurred expenses associated with our efforts to accommodate personnel during the COVID-19 pandemic, including costs associated with the provision of COVID-19 testing to our personnel, safety accommodations, providing on-site amenities and enhanced on-site cleaning efforts, and we will continue to incur such expenses associated with our operations.
The pandemic has also caused substantial disruption in global supply chains. These interruptions may require us to suspend operations or delay programs. If we continually delay programs with existing customers, we may be in breach of our contracts with existing customers or customers may decide to cease doing business with us or have decreased demand for our products. We may also experience a slow-down in our pipeline of new programs or a termination of existing programs if our customers or potential customers face disruptions during the pandemic. Difficulties and delays such as those we have experienced and may experience in the future may prevent us from meeting our operating and financial goals, both in general and within our targeted timelines, and may cause our revenues and operating results to fluctuate from period to period.
Uncertainty regarding the ongoing demand and/or capacity (including capacity at third party clinical testing laboratories) of our COVID-19 individual and pooled sample tests could materially adversely affect our business.
Our COVID-19 testing programs are subject to inherent risks of commercial viability, such as demand for tests, price or market share erosion due to competition and the duration of the COVID-19 pandemic. We are in a highly competitive market – many companies have launched or are seeking to launch COVID-19 testing products and many of these companies already have an existing commercial and technical infrastructure to market and commercialize such offerings. We have limited experience marketing or commercializing diagnostic or pooled sample testing programs and may not be able to sufficiently support operations with our current base of personnel or recruit enough personnel to effectively commercialize COVID-19 testing programs, particularly during a pandemic, at which time the pipeline for experienced personnel will be in high demand. Moreover, as vaccines for COVID-19 and at-home or over-the-counter COVID-19 tests become more widely available, and as infection rates decrease, demand for COVID-19 testing may also decrease.
Our COVID-19 testing business relies heavily on the adoption of pooled testing in schools, which may be hesitant to adopt COVID-19 testing without positive support from parents or teachers. Although we make test validation results and protocols available to parents and teachers, they may not trust the accuracy of the tests or may have concerns about how the tests are performed, how samples are used or tracked and whether appropriate privacy measures are being taken with respect to individually identifiable health information, including genetic information. The ability for schools to pay for COVID-19
60
Table of Contents
testing relies heavily on the availability of federal, state or local funding for testing. If such funding is not available or if there are restrictions on the use of such funding for our pooled sample test offerings, our COVID-19 testing business may not be commercially viable. In addition, as a result of the recent approval of a COVID-19 vaccine for children younger than sixteen years of age, the demand for COVID-19 testing in schools could diminish significantly or be eliminated.
Creating the commercial and technical infrastructure to test on a mass scale is expensive. We may also be limited in our ability to scale up based on expense or unavailability of the required materials, equipment, personnel and infrastructure necessary to deliver diagnostic or pooled sample tests on a mass scale. We may not be able to recover our investment expenses with sufficient revenue generated by our diagnostic and pooled sample testing efforts.
Our ability to commercialize our testing programs is also subject to regulatory or governmental controls, decisions or actions. If the U.S. Department of Health and Human Services (“HHS”) terminates its Declaration Justifying Emergency Use of Medical Countermeasures because the circumstances justifying emergency use no longer exist and, if the third-party COVID-19 tests that are used in our testing services are not able to obtain premarket approval, clearance or other marketing authorization from the U.S. Food and Drug Administration (“FDA”), we may be unable to market or distribute these COVID-19 tests, fulfill our contractual testing requirements or generate revenues from our test offerings. We may also experience price erosion if federal or state governments implement price controls.
Finally, the sale of each test is dependent on the supply of the appropriate collection devices authorized for use with the COVID-19 tests we utilize in our testing programs. Disruptions in this supply chain will have a material adverse effect on our ability to sell tests.
Uncertainty regarding the sales and delivery of our COVID-19 individual and pooled sample tests could materially adversely affect our business.
Although we have partnerships with third party clinical testing laboratories to support a high volume of pooled sample testing for COVID-19 nationally, pooled testing has not yet been adopted by all states nor have we established partnerships with clinical testing laboratories in all states. We are continuing to develop processes to scale capacity of COVID-19 pooled sample collection and testing. However, we can give no assurance that we will be able to successfully scale the pooled sample collection and test capacity or that we will be able to establish or maintain the collaborative third party relationships that support such testing capacity. In addition, even if we are able to scale to high volume testing nationwide, there can be no assurance that the testing capacity will be used.
We may be subject to tort liability if the COVID-19 tests we utilize in our testing programs provide inaccurate results.
The Public Readiness and Emergency Preparedness Act (the “PREP Act”) provides immunity for manufacturers, distributors, program planners, qualified persons, and their officials, agents, and employees from certain claims under state or federal law for a “loss” arising out of the administration or use of a “covered countermeasure” in the United States. Distributors are certain persons or entities engaged in the distribution of drugs, biologics, or devices. Program planners include persons who supervise or administer a program with respect to the administration, distribution, provision, or use of a Covered Countermeasure (as defined in the PREP Act). Covered Countermeasures include security countermeasures and “qualified pandemic or epidemic products,” including products intended to diagnose or treat pandemic or epidemic disease, such as COVID-19 diagnostic tests, as well as treatments intended to address conditions caused by such products. Covered Countermeasures must also be approved, cleared, or authorized for emergency use, or otherwise authorized for investigational use, by the FDA in order to be considered Covered Countermeasures under the PREP Act.
For these immunities to apply, the Secretary of HHS must issue a declaration in cases of public health emergency or “credible risk” of a future public health emergency. On March 10, 2020, the Secretary of HHS issued a declaration under the PREP Act and has issued subsequent amendments thereto to provide liability immunity for activities related to certain countermeasures against the ongoing COVID-19 pandemic.
We act as the authorized distributor of certain third-party COVID-19 tests and collection kits that have received Emergency Use Authorization (“EUA”) and supervise testing programs for our COVID-19 testing customers. While we believe our test distribution and program planning activities with respect to these programs would be covered under the provisions of the PREP Act, this cannot be assured. Also, there can be no assurance that the U.S. Congress will not act in the future to reduce coverage under the PREP Act or to repeal it altogether.
Furthermore, some of the third-party tests used as part of our pooled testing program are not covered by an EUA and, at this time, we do not believe that such testing services, administration, or program planning related to our pooled testing program will qualify for PREP Act immunity. If product liability lawsuits are brought against us in connection with allegations of
61
Table of Contents
harm connected to our COVID-19 testing services, we may incur substantial liabilities and may be required to limit our testing services. The PREP Act is a complex law with limited judicial precedent, and thus even for the third-party COVID-19 tests and collection kits used in our testing services that are subject to EUAs, we may have to expend significant time and legal resources to obtain dismissal of a lawsuit on the basis of PREP Act immunity.
If we cannot successfully defend ourselves against claims that our COVID-19 testing services caused injuries and if we are not entitled to immunity under the PREP Act, or the U.S. Congress limits or eliminates coverage under the PREP Act, or if the liability protections under the PREP Act are not adequate to cover all claims, we may incur substantial liabilities. Regardless of merit or eventual outcome, product liability claims may result in decreased demand for our services, injury to our reputation, costs to defend litigation, loss of revenue, and substantial money awards to customers.
We are dependent on our relationships with our telehealth partner to provide healthcare services, and our business would be adversely affected if those relationships were disrupted.
Our contractual relationships with our telehealth partner who provides physician authorization for COVID-19 diagnostic and screening testing may implicate certain state laws in the United States that generally prohibit non-physician entities from practicing medicine, exercising control over physicians or engaging in certain practices such as fee-splitting with physicians. There can be no assurance that these laws will be interpreted in a manner consistent with our practices or that other laws or regulations will not be enacted in the future that could have a material and adverse effect on our business, financial condition and results of operations. Regulatory authorities, state medical boards of medicine, state attorneys general and other parties, including our telehealth partner, may assert that we are engaged in the prohibited corporate practice of medicine, and/or that its arrangements with its telehealth partner constitutes unlawful fee-splitting. If a state’s prohibition on the corporate practice of medicine or fee-splitting law is interpreted in a manner that is inconsistent with our practices, we would be required to restructure or terminate our relationship with our telehealth partner to bring our activities into compliance with such laws. A determination of non-compliance, or the termination of or failure to successfully restructure these relationships could result in disciplinary action, penalties, damages, fines, and/or a loss of revenue, any of which could have a material and adverse effect on our business, financial condition and results of operations. State corporate practice of medicine doctrines and fee-splitting prohibitions also often impose penalties on healthcare professionals for aiding the corporate practice of medicine, which could discourage our telehealth partner from providing services to us.
Risks Related to the Synthetic Biology Industry
Rapidly changing technology and emerging competition in the synthetic biology industry could make the platform, programs, and products we and our customers are developing obsolete or non-competitive unless we continue to develop our platform and pursue new market opportunities.
The synthetic biology industry is still emerging and is characterized by rapid and significant technological changes, frequent new product introductions and enhancements, and evolving industry demands and standards. Our future success will depend on our ability to sign and initiate new programs that address the evolving needs of our customers on a timely and cost-effective basis, to advance existing programs and to pursue new market opportunities that develop as a result of technological and scientific advances. Additionally, our customers may face significant competition or other risks which may adversely impact our business and results of operations.
There are a number of companies in the broader synthetic biology industry, and our future success will depend on our ability to maintain a competitive position with respect to technological advances. Technological development by others may result in our platform becoming obsolete. Our ability to compete successfully will depend on our ability to develop proprietary technologies that enable our customers to develop products using our platform in a manner that is either less expensive, faster, superior or otherwise differentiated from what a competitor’s technologies and products might enable. If we are unable to continue to successfully advance our platform or the services it provides at scale, or if our customers are unable to commercialize the products or processes made or improved upon by using our platform, our business and results of operations will be adversely impacted.
Due to the significant lead time involved in launching a new program or developing a new product or process using our platform, our customers are required to make a number of assumptions and estimates regarding the commercial feasibility of a new program, including assumptions and estimates regarding the size of an emerging product category and demand for those end-products and processes which will use our technology, the ability to scale-up manufacturing processes to produce a product on a commercial scale, the ability to penetrate that emerging product category, customer adoption of a downstream product, the existence or non-existence of products being simultaneously developed by competitors, potential market penetration and obsolescence, planned or unplanned. As a result, it is possible that we may commence a new program with a customer who wishes to develop a product or process that has been displaced by the time of launch, addresses a market that
62
Table of Contents
no longer exists or is smaller than previously thought, that end-consumers do not like or otherwise is not competitive at the time of launch, in each case, after the incurrence of significant opportunity costs on our part to develop such product. The ultimate success of the products developed by our customers using our services may be dependent on the success of other markets in which we or our customers do not operate in or have knowledge or expertise or which, in each case, may not reach the size anticipated by us or our customers or may be replaced by another emerging product category or eliminated entirely.
The market, including customers and potential investors, may be skeptical of our ability to deliver on programs because they are based on a relatively novel and complex technology.
The market, including customers and potential investors, may be skeptical of the viability and benefits of bioengineered products as well as our enabling abilities, including our platform and programs, because they are based on a relatively novel approach and the adoption of complex technology. There can be no assurance that our platform and programs will be understood, approved, or accepted by customers, regulators and potential investors or that we will be able to sell our services profitably at competitive prices and with features sufficient to establish demand.
In addition, in order for novel products from our programs to be successfully commercialized, support from the entire relevant supply chain is needed. Relationships with all parts of the supply chain are important in order to gain visibility into market trends and feature and specification requirements and in order to ensure customers are able to successfully manufacture their products, obtain regulatory approval and gain access to key distribution channels. If we are unable to convince these potential customers, their suppliers, or the consumers who purchase products containing or made or developed using engineered cells and/or biomanufacturing processes, of the utility and value of such products or that such products are superior to the products they currently use, we will not be successful in entering these markets and our business and results of operations will be adversely affected. If potential investors are skeptical of the success of our platform or cell programs, our ability to raise capital and the value of our common stock may be adversely affected.
Ethical, legal and social concerns about GMOs and Genetically Modified Materials and their resulting products could limit or prevent the use of products or processes using our technologies, limit public acceptance of such products or processes and limit our revenues.
Our technologies and the technologies of our customers involve the use of genetically modified cells, organisms and biomaterials, including, without limitation, GMOs, genetically modified microorganisms (“GMMs”), Genetically Modified Materials and their respective products. The use, production and marketing of Genetically Modified Materials, are subject to laws and regulations in many countries, some of which are new and some of which are still evolving. In the United States, the FDA, the Environmental Protection Agency (“EPA”) and the U.S. Department of Agriculture (“USDA”) are the primary agencies that regulate the use of GMOs, GMMs and potential products derived from GMOs or GMMs. If regulatory approval of the Genetically Modified Materials or resulting products is not secured, our business operations, financial condition and our ability to grow as a business could be adversely affected. We expect to encounter regulations regarding Genetically Modified Materials in most if not all of the countries in which our customers may seek to establish production capabilities or sell their products and the scope and nature of these regulations will likely be different from country to country. Governmental authorities could, for safety, social or other purposes, impose limits on, or implement regulation of, the use, production or marketing of Genetically Modified Materials. If our customers cannot meet the applicable requirements in other countries in which they intend to produce or sell their products, or if it takes longer than anticipated to obtain such approvals, our business could be adversely affected.
In addition, public perception regarding the safety and environmental hazards of, and ethical concerns over, Genetically Modified Materials or the processes used to create them, including gene editing or gene regulating technologies, could influence public acceptance of our and our customers’ technologies, products and processes. For instance, certain advocacy groups engage in efforts that include regulatory legal challenges and labeling campaigns for genetically modified products, as well as application of pressure to consumer retail outlets seeking a commitment not to carry genetically modified foods. These groups in the past have pressured retail food outlets and grocery store chains to publicly state that they will not carry genetically modified foods and have pressed food brands to publicly state that they will not use ingredients produced by genetically modified microbes. In addition, certain labeling-related initiatives have heightened consumer awareness of GMOs, which may make consumers less likely to purchase products containing GMO ingredients, which could have a negative impact on the commercial success of our customers’ products and programs. These concerns could result in increased expenses, regulatory scrutiny, delays or other impediments to our programs. The subject of Genetically Modified Materials has received negative publicity, which has aroused public debate. This adverse publicity has led to, and could continue to lead to, greater regulation and trade restrictions on imports of Genetically Modified Materials or their resulting products. In addition, with the acquisition of Dutch DNA Biotech B.V. (“DDNA”), we are expanding into the European Union market, which has increased government regulation and scrutiny over genetically modified products. There is a risk
63
Table of Contents
that products produced using our technologies could cause adverse health effects or other adverse events, which could also lead to negative publicity, regulatory action or private litigation. If we are unable to overcome the ethical, legal and social concerns relating to genetic engineering, our programs could face increased expenses, regulatory scrutiny, delays or other impediments to deliver our programs or the commercialization of resulting products and processes.
Finally, the COVID-19 pandemic may increase biosecurity concerns by public and/or governmental stakeholders regarding genetic engineering technologies and risks around engineered viruses, microbes and organisms. Such concerns, restrictions, or governmental restrictions could limit the use of Genetically Modified Materials in our customers’ products, which could have a material adverse effect on our business, financial condition and results of operations.
Risks Related to Intellectual Property
If we are unable to obtain, maintain and defend patents protecting our intellectual property, our competitive position will be harmed.
Our success depends in part on our ability to obtain and maintain intellectual property protection for our proprietary technologies. We protect our proprietary technologies through patents and trade secrets, both of which entail risk. If we are unable to obtain, maintain or protect intellectual property rights related to our technology, or if our intellectual property rights are inadequate, our competitive position, business, financial conditions, results of operations and prospects may be harmed.
Because of the volume and nature of our inventions, patent protection may not be practicable, available, or appropriate for some aspects of our proprietary technologies. While we own patents and pending patent applications in the United States and in foreign jurisdictions, these applications do not ensure the protection of our intellectual property. There may be prior art of which we are not aware. Additionally, obtaining, maintaining, defending and enforcing patents is costly, time consuming and complex, and we may not be able to file and prosecute all necessary or desirable patent applications, or maintain and enforce any patents that may issue from such patent applications at a reasonable cost or in a timely manner. It is also possible that we will fail to identify patentable aspects of our technologies before it is too late to obtain patent protection. Although we enter into confidentiality agreements with parties who have access to confidential or patentable aspects of our R&D output, such as our employees, collaborators, consultants, advisors and other third parties, any of these parties may breach the agreements and disclose such output before a patent application is filed, thereby jeopardizing our ability to seek patent protection.
Further, pending applications may not be issued or may be issued with claims significantly narrower than we currently seek. Patents for which claims have been allowed may be successfully challenged and invalidated. Unless and until our pending applications issue, their protective scope is impossible to determine and, even after issuance, their protective scope may be limited.
Recent changes in patent law have made patents covering life science inventions more difficult to obtain and enforce. Further legislative changes or changes in the interpretation of existing patent law could increase the uncertainty and cost surrounding the prosecution of our owned patent applications and the maintenance, enforcement or defense of our owned patents. The Leahy-Smith America Invents Act (“the Leahy-Smith Act”) included changes that affect the way patent applications are prosecuted; redefine prior art; enable third-party submission of prior art to the United States Patent and Trademark Office (“USPTO”) during patent prosecution; and provide cost-effective avenues for competitors and other third parties to challenge the validity of patents at USPTO-administered post-grant proceedings, including post-grant review, inter partes review and derivation proceedings. Thus, the Leahy-Smith Act and its continued implementation could increase the uncertainties and costs surrounding the prosecution of our patent applications and the enforcement or defense of our issued patents, all of which could have a material adverse effect on our business, financial condition, results of operations, and prospects.
Other changes in the law may further detract from the value of life science patents and facilitate challenges to our patents. In some cases, we use genetic sequence information from naturally occurring organisms, which may not be patentable. Recent U.S. Supreme Court rulings have narrowed the scope of patent protection for naturally occurring sequences and for inventions based on the observation and exploitation of natural phenomena. These decisions have weakened the rights of patent owners in certain situations. The U.S. Court of Appeals for the Federal Circuit has also issued a series of rulings that create obstacles to the patenting of groups of genetic sequences that share functional characteristics, making it more difficult to obtain claims to certain genetic constructs, particularly antibodies. These changes in the law have created uncertainty with respect to the validity and enforceability of patents covering natural and engineered sequences. Depending on future actions by the U.S. Congress, the federal courts and the USPTO, the laws and regulations governing patents could change in unpredictable ways that could have a further material adverse effect on our patent rights and our ability to protect, defend and enforce our patent rights in the future.
64
Table of Contents
Further, the issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, and our patents may be challenged in the courts or patent offices in the United States and abroad. An adverse determination in any such challenge could result in loss of exclusivity, or patent claims being narrowed, invalidated or held unenforceable, in whole or in part. Any of these results could limit our ability to stop others from using or commercializing similar or identical technology to compete directly with us. In addition, if the breadth or strength of protection provided by our patents and patent applications is threatened, regardless of the outcome, it could dissuade companies from collaborating with us. Such proceedings also may result in substantial cost and require significant time from our scientists and management, even if the eventual outcome is favorable to us.
The laws of some foreign countries do not protect intellectual property rights to the same extent as do the laws of the United States or may apply different rules concerning the assignment of intellectual property rights. Many companies have encountered significant problems in protecting and defending intellectual property rights in certain foreign jurisdictions. We may encounter similar difficulties, particularly as we expand to work with foreign employees and contractors and expand our collaboration activities into foreign markets. The legal systems of certain countries, particularly certain developing countries, do not favor the enforcement of patents by foreign holders and, in some cases, do not favor the enforcement of patents at all, particularly patents in the life sciences. This could make it difficult for us to stop the infringement of our patents. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial costs and divert our efforts and attention from other aspects of our business and could be unsuccessful.
Reductions in the scope or enforceability of our patent protection may adversely affect our customers’ ability to commercialize their products and may thus reduce our downstream value from royalties, equity, or commercial milestone payments.
If we are unable to protect the confidentiality of our trade secrets, our business and competitive position will be harmed.
Because patent protection may not be available or appropriate for significant aspects of the technology we are developing, our success may depend in large part on our proprietary information, including genetic and other chemical and biological data, processes, know-how, and other trade secrets developed over years of R&D, some of which are embodied in proprietary software. We rely heavily on trade secret protections, especially in cases where we believe patents or other forms of registered intellectual property protection may not be appropriate or obtainable. However, trade secrets are difficult to protect. The secrecy of the Company’s trade secrets must be maintained for them to retain their status and protection as trade secrets. While we strive to protect the secrecy of our trade secrets and other proprietary information, including by requiring our employees, customers, consultants, and contractors to enter into confidentiality agreements and instituting multilayered protections covering our digital environment and biomaterials, we may not be able to adequately protect our trade secrets or other proprietary information. We cannot guarantee that we have entered into such agreements with every party that may have or has had access to our trade secrets, biomaterials or proprietary technology and processes. Further, despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches.
We seek to preserve the integrity and confidentiality of our information by maintaining physical security of our premises and physical and electronic security of our information technology systems, but it is possible that these security measures could be breached. We also rely on systems provided by third parties, which may suffer security breaches or incidents. Such security breaches may be inadvertent or may come about due to intentional misconduct or other malfeasance or by human error or technical malfunctions, including those caused by hackers, employees, contractors, or vendors. It may be difficult or impossible to recover trade secrets or other confidential information once it is hacked, and hackers may operate from jurisdictions that will not cooperate with such efforts. Enforcing any claim that a third party unlawfully obtained and is using any of our trade secrets is expensive and time consuming, and the outcome is unpredictable. In addition, courts in some jurisdictions are less willing or unwilling to protect trade secrets even when a hacker or thief can be identified.
Our competitors may lawfully obtain or independently develop knowledge that is equivalent to one or more of our trade secrets. Were they to do so, we would be unable to prevent them from using that independently developed knowledge. Such a competitor could claim that we had learned the trade secret from them and bring an action against us on that basis. If any of our trade secrets were to be disclosed to or independently developed by a competitor or other third party, our competitive position could be materially and adversely harmed. Moreover, a competitor could file for patent protection covering intellectual property that we have chosen to protect as a trade secret. In such a case, we might be restricted or excluded from using that intellectual property even if we had developed it before our competitor did.
Our facilities hold large collections of microbial strains, cell lines and other biomaterials. Failure to implement adequate controls and protections, failure to implement adequate disposal procedures, unauthorized visitors in the labs, or customers’
65
Table of Contents
failure to adequately protect biological materials can put us and our customers at risk of losing valuable assets through negligence or theft and enabling the use of those lost materials by our competitors. While we believe that we take reasonable measures to protect the security of biomaterials owned by us or our customers, it is possible that our security controls and practices may not prevent unauthorized or other improper access to such genetic material. Any unauthorized access, acquisition, use, destruction, or release of the GMOs we engineer could result in our having exposure to significant liability under our contracts, as well as to regulatory actions, litigation, investigations, remediation obligations, damage to our reputation and brand, supplemental disclosure obligations, loss of customer, consumer, or partner confidence in the security of our platform, impairment to our business, and corresponding fees, costs, expenses, loss of revenues, and other potential liabilities.
Our customers sometimes provide organisms, genetic material and/or data to us in connection with our collaborations. In the event that we fail to protect customer materials or data or inadvertently use such materials or data for unauthorized purposes, we could be liable to our customers under trade secret laws or contractual provisions.
There could be unintended consequences to the environment generally or the health and safety of our employees or the public as a result of an unauthorized release of Genetically Modified Materials into uncontrolled environments. In addition, if a biosecurity breach or unauthorized release of genetic material were to occur within our industry, our customers and potential customers might lose trust in the security of the laboratory environments in which we produce GMOs, even if we are not directly affected. Any adverse effect resulting from such a release, by us or others, could have a material adverse effect on the public acceptance of our products and business and our financial condition. Such a release could result in enhanced regulatory activity and we could have exposure to liability for any resulting harm.
We may be subject to claims challenging the inventorship or ownership of our patents, biomaterials and other intellectual property.
Certain of our employees, consultants and contractors were previously employed at universities or other software or biotechnology companies, including our competitors or potential competitors. Additionally, some of our consultants or contractors may have ongoing relationships with universities. Although we try to ensure that our employees, consultants and contractors do not use the intellectual property of others in their work for us, we may be subject to claims that these individuals or other contractors have used or disclosed intellectual property, including trade secrets or other proprietary information, of another. Litigation may result from these claims.
While it is our policy to require that our employees, consultants and contractors who may be involved in the development of intellectual property execute agreements assigning such intellectual property to us, we may be unsuccessful in executing such an agreement with each party who in fact develops intellectual property that we regard as our own. Our intellectual property assignment agreements with them may not be self-executing or may be breached, and we may be forced to bring claims against third parties, or defend claims they may bring against us, to determine the ownership of what we regard as our intellectual property. Such claims could have a material adverse effect on our business, financial condition, results of operations and prospects.
If we are unsuccessful in litigating any such claims, in addition to paying monetary damages, we may lose valuable intellectual property rights or personnel, which could have a material adverse effect on our competitive business position and prospects. Such intellectual property rights could be awarded to a third party, and we could be required to obtain a license from such third party to use or commercialize our technology or products, which license might not be available on commercially reasonable terms, or at all. Even if we are successful in prosecuting or defending against such claims, litigation could result in substantial costs and be a distraction to our management and employees.
The life science academic and research community has abided by norms of free exchange of biomaterials, but recently, norms have begun to change so that parties may assert ownership and control over biomaterials that they permitted to be freely disseminated in the past. Thus, despite our best efforts to confirm our right to use biomaterials in our possession, we may use organisms that we believe to be free of encumbrance that are, in fact, subject to claims of title by others. In such a situation, litigation may be required to clear title, if it can be cleared at all. Similarly, we may be subject to claims that we have used biomaterials obtained from licensors or repositories for unauthorized purposes, or purposes not consistent with the licensing terms of the providing organization.
We may become involved in lawsuits or other enforcement proceedings to protect or enforce our patents or other intellectual property, which could be expensive, time consuming and potentially unsuccessful.
Competitors and other third parties may infringe or otherwise violate our issued patents or other intellectual property. In addition, our patents may become involved in inventorship, ownership, or priority disputes. We may also become subject to
66
Table of Contents
claims by collaboration partners that intellectual property or biomaterials that we believe to be owned by us is actually owned by them. Any litigation concerning any of these issues would be expensive, time consuming and uncertain. There can be no assurances that we would prevail in any suit brought by us or against us by third parties, or successfully settle or otherwise resolve those claims. Significant litigation would have substantial costs, even if the eventual outcome were favorable to us, and would divert management’s attention from our business objectives.
Under certain circumstances, we may share or lose rights to intellectual property developed under U.S. federally funded research grants and contracts.
Some of our inventions, data, or other intellectual property have been or may be developed during the course of research funded by the U.S. government. The U.S. government may have the right to take title to government-funded inventions if we fail to disclose the inventions to the government in a timely manner or fail to file a patent for the intellectual property within specified time limits. Further, in consequence of our receiving government funding, the U.S. government may have certain rights to intellectual property that we use in our platform or programs pursuant to the Bayh-Dole Act of 1980, as amended (the “Bayh-Dole Act”). Under the Bayh-Dole Act, U.S. government rights in certain “subject inventions” developed under a government-funded program may include a non-exclusive, irrevocable worldwide license to use inventions for any governmental purpose. In some circumstances, the U.S. government may acquire unlimited rights in data we generate. In addition, the U.S. government has the right to require us, or an assignee or exclusive licensee to U.S. Government-funded inventions, to grant licenses to any of these inventions to the government or a third party if the government determines that: (i) adequate steps have not been taken to commercialize the invention; (ii) government action is necessary to meet public health or safety needs; (iii) government action is necessary to meet requirements for public use under federal regulations; or (iv) the right to use or sell such inventions is exclusively licensed to an entity within the United States and substantially manufactured outside the United States without the U.S. government’s prior approval. Additionally, we may be restricted from granting exclusive licenses for the right to use or sell such inventions unless the licensee agrees to comply with relevant Bayh-Dole Act restrictions (e.g., manufacturing substantially all of the invention in the United States) and reporting requirements. In addition, the U.S. government may acquire title in any country in which a patent application is not filed. Certain technology and inventions are also subject to transfer restrictions during the term of these agreements with the U.S. government and for a period thereafter. These restrictions may limit sales of products or components, transfers to foreign subsidiaries for the purpose of the relevant agreements, and transfers to certain foreign third parties. If any of our intellectual property becomes subject to any of the rights or remedies available to the U.S. government or third parties pursuant to the Bayh-Dole Act, this could impair the value of our intellectual property and could adversely affect our business.
The use of digital genetic sequence information may be subject to the Nagoya Protocol, which could increase our costs and adversely affect our business.
The Nagoya Protocol is a supplemental agreement to the Convention on Biological Diversity (“CBD”). The Protocol is designed to provide for equitable sharing of benefits arising from the utilization of genetic resources and traditional knowledge. Under the Nagoya Protocol, countries possessing genetic resources (“source countries”) are tasked with setting up procedures and institutional infrastructure for researchers to obtain prior (not post-hoc) informed consent, both from the source country and from any relevant indigenous or traditional communities, for biological research. Many have been slow to adopt workable institutions permitting the rational negotiation of benefit-sharing agreements. Many source countries are now asserting that the use of digital genetic sequence information is subject to the constraints of the Nagoya Protocol or national- or local-level benefit-sharing requirements. It is unclear whether this position will ultimately be adopted or what the implications of such adoption might be. It is unclear what a source country might assert if we used genetic sequences (i) extracted by a third party from a natural resource that was removed from its source country before that source country ratified the CBD or signed the Nagoya Protocol (ii) extracted by a third party and uploaded to public sequence databases after the source country ratified the CBD; (iii) in a heterologous host organism; or (iv) as a base for further engineering, so that the sequence we use no longer conforms to the natural sequence on which it was based.
We make extensive use of public and proprietary sequence databases to support our work. While we undertake efforts to identify and comply with laws and international protocols relating to the use of genetic resources, the uncertainty surrounding the use of digital sequence information and the lack of workable institutions in many source countries for the efficient negotiation of benefit-sharing agreements may limit our use or cause uncertainty in our use of certain sequences that we obtain from public access databases or natural sources. New financial obligations may arise regarding our use of sequence information. Customers that must certify their compliance with Nagoya Protocol obligations may be reluctant to do business with us unless we engage in expensive and time-consuming benefit-sharing negotiations with source countries of publicly available genetic sequences. These changes could increase our R&D costs and adversely affect our business, financial condition, and results.
67
Table of Contents
Third party patents may limit our freedom to operate in certain areas, which may adversely affect our business.
There may be patents that affect our freedom to operate in certain areas, and we may as a result choose to design around or license such patents from third parties. If we must spend significant time and money designing around or licensing patents held by others, our business and financial prospects may be harmed. We may be restricted from carrying out certain operations in our Foundry, or we may be limited in our ability to design new products for our customers. We may become subject to claims by third parties alleging that we are infringing, misappropriating, or otherwise violating their intellectual property rights.
If we are sued for infringing intellectual property rights of third parties, such litigation could be costly and time consuming and could prevent or delay us from using our platform and technologies.
Any litigation arising from any dispute relating to the intellectual property of third parties would be expensive, time-consuming, and uncertain. There can be no assurance that we would prevail in any such dispute. Parties making claims against us might be able to obtain injunctive or other relief, which could block our or our customers’ ability to develop, commercialize and sell products or use our technologies, and could result in the award of substantial damages against us, including treble damages, attorney’s fees, costs and expenses if we were found to have willfully infringed. In the event of a successful claim against us, we or our customers might be required to pay damages and ongoing royalties, and obtain licenses from third parties, or be prohibited from selling certain products or using certain technologies. We may not be able to obtain these licenses on acceptable or commercially reasonable terms, if at all. In addition, we or our customers could encounter delays in product or service introductions while we attempt to develop alternative or redesign existing products or technologies to avoid or resolve these claims. Our loss in any lawsuit or failure to obtain a license could prevent us from using our platform and technologies. Such a loss or failure could materially affect our business and reputation. Any litigation pertaining to these issues would have substantial costs, even if the eventual outcome is favorable to us, and would divert management’s attention from our business objectives.
If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be adversely affected.
Our registered or unregistered trademarks or trade names may be challenged, infringed, diluted, tarnished, circumvented or declared generic or determined to be infringing on other marks. We may not be able to protect our rights to these trademarks and trade names, which we need to build name recognition among potential collaborators or customers in our markets of interest. At times, competitors may adopt trade names or trademarks similar to ours, thereby impeding our ability to build brand identity and possibly leading to market confusion. In addition, there could be potential trade name or trademark infringement, dilution or tarnishment claims brought by owners of other trademarks or trademarks that incorporate variations of our registered or unregistered trademarks or trade names. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, then we may not be able to compete effectively and our business may be adversely affected. Our efforts to enforce or protect our proprietary rights related to trademarks, trade names, trade secrets, domain names, copyrights or other intellectual property may be ineffective and could result in substantial costs and diversion of resources and could adversely affect our business, financial condition, results of operations and prospects.
Any claims or lawsuits relating to infringement of intellectual property rights brought by or against us will be costly and time consuming and may adversely affect our business, financial condition and results of operations.
Any of the risks identified above could result in significant litigation. In addition to the specific litigation-related risks identified above, litigation of any kind carries certain inherent risks. Because of the substantial amount of discovery required in connection with litigation in U.S. courts, there is a risk that some of our confidential information could be compromised in the discovery process. There could be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on our share price.
Further, our agreements with some of our customers, suppliers or other entities require us to defend or indemnify these parties if they become involved in infringement claims that target our products, services or technologies, or in certain other situations. If we must defend or indemnify third parties, we could incur significant costs and expenses that could adversely affect our business, operating results or financial condition.
68
Table of Contents
Intellectual property rights do not necessarily address all potential threats to our business.
The degree of future protection afforded by our intellectual property rights is uncertain because intellectual property rights have limitations and may not adequately protect our business or permit us to maintain our competitive advantage. The following examples are illustrative:
Should any of these events occur, they could harm our business, financial condition, results of operations and prospects.
We rely on licenses from third parties for certain services and intellectual property. If we lose any of these licenses, it could have a material adverse effect on our business, financial condition and results of operations.
We rely, and expect to continue to rely on, certain services and intellectual property that we license from third parties for use in our operations, platform, products, services and offerings. We cannot be certain that our licensors are not infringing upon the intellectual property rights of others or that our suppliers and licensors have sufficient rights to the third-party technology used in our business in all jurisdictions in which we may operate. Disputes with licensors over uses or terms could result in the payment of additional royalties or penalties by us, cancellation or non-renewal of the underlying license, or litigation. In the event that we cannot renew and/or expand existing licenses, we may be required to discontinue or limit our use of the operations, platform, products, services or offerings that include or incorporate the licensed intellectual property. Any such discontinuation or limitation could have a material and adverse impact on our business, financial condition and results of operation.
Our use of “open-source” software could negatively affect our ability to market or provide our services and could subject us to possible litigation.
We have used “open-source” software in connection with the development and deployment of our software platform, and we expect to continue to use open-source software in the future. Open-source software is licensed by its authors or other third parties under open-source licenses, which in some instances may subject us to certain unfavorable conditions, including requirements that we offer our products that incorporate the open-source software for no cost, that we make publicly available all or part of the source code for any modifications or derivative works we create based upon, incorporating or using the open-source software, or that we license such modifications or derivative works under the terms of the particular open-source license.
Companies that incorporate open-source software into their products have, from time to time, faced claims challenging the use of open-source software and compliance with open-source license terms. We could be subject to similar suits by parties claiming ownership of what we believe to be open-source software or claiming noncompliance with open-source licensing terms. While we monitor our use of open-source software and try to ensure that none is used in a manner that would require us to disclose our proprietary source code or that would otherwise breach the terms of an open-source agreement, we cannot guarantee that we will be successful, that all open-source software is reviewed prior to use in our platform, that our developers have not incorporated open- source software into our products that we are unaware of or that they will not do so in the future.
Furthermore, there are an increasing number of open-source software license types, almost none of which have been interpreted by U.S. or foreign courts, resulting in a dearth of guidance regarding the proper legal interpretation of such licenses. As a result, there is a risk that open-source software licenses could be construed in a manner that imposes
69
Table of Contents
unanticipated conditions or restrictions on our ability to market or provide our products and services. If we are held to have breached or failed to fully comply with all the terms and conditions of an open-source software license, we could face infringement claims or other liability, or be required to seek costly licenses from third parties to continue providing our offerings on terms that are not economically feasible, if at all, to re-engineer all or a portion of our platform, to discontinue or delay the provision of our offerings if re-engineering could not be accomplished on a timely basis or to make generally available, in source code form, our proprietary code. Further, in addition to risks related to license requirements, use of certain open-source software carries greater technical and legal risks than does the use of third-party commercial software. For example, open-source software is generally provided without any support or warranties or other contractual protections regarding infringement or the quality of the code, including the existence of security vulnerabilities. To the extent that our platform depends upon the successful operation of open-source software, any undetected errors or defects in open-source software that we use could prevent the deployment or impair the functionality of our systems and injure our reputation. In addition, the public availability of such software may make it easier for others to compromise our platform. Any of the foregoing risks could materially and adversely affect our business, financial condition and results of operations.
Risks Related to Personnel, IT and Physical Infrastructure
Loss of key personnel, including our founders and senior executives, and/or failure to attract, train and retain additional key personnel could delay our cell engineering programs and harm our platform development efforts and our ability to meet our business objectives, particularly given the substantial investment required to train certain of our employees.
Our business involves complex, global operations across a variety of markets and requires a management team and employee workforce that is knowledgeable in the many areas in which we operate. Our future success depends upon our ability to attract, train, retain and motivate highly qualified management, scientific, engineering, information technology, operations, business development and marketing personnel, among others. In addition, the market for qualified personnel is very competitive because of (a) the limited number of people available who have the necessary technical skills and understanding of our technology and products and (b) the nature of our industry which requires certain of our technical personnel to be on-site in our facilities. We compete for qualified technical personnel with other life sciences and information technology companies, as well as academic institutions and research institutions in the markets in which we operate, including Boston, Massachusetts, Cambridge, Massachusetts and Emeryville, California. In addition, as we add international operations, we will increasingly need to recruit qualified personnel outside the United States. However, doing so may also require us to comply with laws to which we are not currently subject, which could cause us to allocate or divert capital, personnel and other resources from our organization, which could adversely affect our business, financial condition, results of operations, prospects and reputation. Establishing international operations and recruiting personnel has and may continue to be impacted by COVID-19 travel and operational restrictions. Our senior leadership team is critical to our vision, strategic direction, platform development, operations and commercial efforts. Our employees, including members of our leadership team, could leave our company with little or no prior notice and would be free to work for a competitor. We also do not maintain “key man” life insurance on any of our employees. The departure of one or more of our founders, senior leadership team members or other key employees could be disruptive to our business until we are able to hire qualified successors.
Our continued platform development, growth and commercial success depends, in part, on recruiting and retaining highly-trained personnel across our various target industries and markets with the necessary background and ability to develop and use our platform and to effectively identify and sell to current and new customers. New hires require significant training and, in most cases, take significant time before they achieve full productivity. Our failure to successfully hire and integrate these key personnel into our business could adversely affect our business. To attract top talent, we believe we will need to offer competitive compensation and benefits packages, including equity incentive programs, which may require significant investment. If we are unable to offer competitive compensation this may make it more difficult for us to attract and retain key employees. Moreover, if the perceived value of our equity awards declines, it may adversely affect our ability to attract and retain key employees. If we do not maintain the necessary personnel to accomplish our business objectives, we may experience staffing constraints that adversely affect our ability to support our programs and operations.
In addition, some of our personnel are qualified foreign nationals whose ability to live and work in the U.S. is contingent upon the continued availability of appropriate visas and whose ability to work on some of our technologies may require the procurement of appropriate export licenses. Due to the competition for qualified personnel in the key markets in which we operate, we expect to continue to utilize foreign nationals to fill part of our recruiting needs. As a result, changes to United States immigration policies have and could further restrain the flow of technical and professional talent into the United States and adversely affect our ability to hire and retain qualified personnel.
70
Table of Contents
Our business and results of operations are dependent on adequate access to laboratory and office space and suitable physical infrastructure, including electrical, plumbing, HVAC and network infrastructure, to conduct our operations. Our headquarters and laboratories are located in a flood zone in Boston’s Seaport District. If we are unable to access enough space or we experience failures of our physical infrastructure, our business and results of operations could be adversely affected.
Our business depends on providing customers with technical services. In order to properly conduct our business, we need access to sufficient laboratory space and equipment to perform the activities necessary to advance and complete our programs. Additionally, we need to ensure that our laboratories and corporate offices remain operational at all times, which includes maintaining suitable physical infrastructure, including electrical, plumbing and HVAC, logistics and transportation systems and network infrastructure. We lease our laboratories and office spaces and we rely on the landlords for basic maintenance of our leased laboratories and office buildings. If one of our landlords has not maintained a leased property sufficiently, we may be forced into an early exit from the facility, which could be disruptive to our business. Furthermore, we may continue to acquire laboratories not built by us in order to sufficiently scale and expand our output capacity. If we discover that these buildings and their infrastructure assets are not in the condition we expected when they were acquired, we may be required to incur substantial additional costs to repair or upgrade the laboratories.
Problems in and around one or more of our laboratories or corporate offices, whether or not within our control, could result in service interruptions or significant infrastructure or equipment damage. These could result from numerous factors, including:
We have timeline obligations to certain customers with respect to their programs. As a result, service interruptions or significant equipment damage in our laboratories could result in difficulty maintaining program timelines for these customers and potential claims related to such failures. Because the services we provide in our laboratories are critical to many of our customers’ businesses, service interruptions or significant equipment damage in our laboratories could also result in lost revenue or other indirect or consequential damages to our customers. We cannot guarantee that a court would enforce any contractual limitations on our liability in the event that one of our customers brings a lawsuit against us as a result of a problem at one of our laboratories and we may decide to reach settlements with affected customers irrespective of any such contractual limitations. In addition, any loss of service, equipment damage or inability to meet our service obligations could reduce the confidence of our customers and could consequently impair our ability to obtain and retain customers, which would adversely affect both our ability to generate revenues and our operating results.
Furthermore, we are dependent upon internet service providers, telecommunications carriers and other website operators, some of which have experienced significant system failures and electrical outages in the past.
Our customers may in the future experience difficulties due to system failures unrelated to our systems and offerings. If, for any reason, these providers fail to provide the required services, our business, financial condition and results of operations could be materially and adversely impacted.
71
Table of Contents
Risks Related to Financial Reporting
We rely on our customers, joint venturers, equity investees and other third parties to deliver timely and accurate information in order to accurately report our financial results in the time frame and manner required by law.
We need to receive timely, accurate and complete information from a number of third parties in order to accurately report our financial results on a timely basis. If the information that we receive is not accurate, our consolidated financial statements may be materially incorrect and may require restatement. Although we have audit rights with these parties, performing such an audit could be expensive and time consuming and may not be adequate to reveal any discrepancies in a time frame consistent with our reporting requirements. As a result, we may have difficulty completing accurate and timely financial disclosures, which could have an adverse effect on our business.
We use estimates in determining the fair value of certain assets and liabilities. If our estimates prove to be incorrect, we may be required to write down the value of these assets or write up the value of these liabilities, which could adversely affect our financial position.
Our ability to measure and report our financial position and operating results is influenced by the need to estimate the fair value of an asset or liability. Fair value is estimated based on a hierarchy that maximizes the use of observable inputs and minimizes the use of unobservable inputs. Observable inputs are inputs that reflect the assumptions that market participants would use in pricing the asset or liability developed based on market data obtained from sources independent of the reporting entity. Unobservable inputs are inputs that reflect the reporting entity’s own assumptions about the assumptions market participants would use in pricing the asset or liability developed based on the best information available in the circumstances. We estimate the impact or outcome of future events on the basis of information available at the time of the financial statements. An accounting estimate is considered critical if it requires that management make assumptions about matters that were highly uncertain at the time the accounting estimate was made. If actual results differ from management’s judgments and assumptions, then they may have an adverse impact on our results of operations and cash flows.
Our ability to use our net operating loss carryforwards and certain other tax attributes may be limited.
We have incurred net losses since our inception and we may never achieve or sustain profitability. Generally, for U.S. federal income tax purposes, net operating losses incurred will carry forward. However, net operating loss carryforwards generated prior to January 1, 2018 are subject to expiration for U.S. federal income tax purposes. As of December 31, 2021, we had federal net operating loss carryforwards of approximately $665.2 million, of which $139.2 million begin to expire in 2029. We have approximately $526.0 million of federal net operating losses as of December 31, 2021 that can be carried forward indefinitely. As of December 31, 2021, we had state net operating loss carryforwards of approximately $529.3 million, of which $485.9 million begin to expire in 2029. We have approximately $43.4 million of state net operating losses as of December 31, 2021 that can be carried forward indefinitely. As of December 31, 2021, we had federal research and development tax credit carryforwards of approximately $23.3 million which begin to expire in 2029. As of December 31, 2021, we also had state research and development and investment tax credit carryforwards of approximately $18.0 million which begin to expire in 2030.
Under Sections 382 and 383 of the Internal Revenue Code of 1986, as amended, if a corporation undergoes an “ownership change,” generally defined as a greater than 50% change by value in its equity ownership by certain stockholders over a three-year period, the corporation’s ability to use its pre-ownership change net operating loss carryforwards and other pre-ownership change tax attributes, such as research tax credits, to offset its post- ownership change income or taxes may be limited. Similar provisions of state tax law may also apply to limit the use of our state net operating loss carryforwards and other state tax attributes. We have not performed an analysis to determine whether our past issuances of stock and other changes in our stock ownership may have resulted in one or more ownership changes. If it is determined that we have in the past experienced an ownership change, or if we undergo one or more ownership changes as a result of future transactions in our stock, which may be outside our control, then our ability to utilize our net operating loss carryforwards and other tax attributes may be materially limited. As a result, even if we earn taxable income, we may be unable to use a material portion of our net operating loss carryforwards and other tax attributes, which could adversely affect our future cash flows. There is also a risk that regulatory changes, such as suspensions on the use of net operating losses or other unforeseen reasons, may result in our existing net operating loss carryforwards expiring or otherwise becoming unavailable to offset future taxable income. For these reasons, we may not be able to utilize a material portion of our net operating loss carryforwards and other tax attributes even if we attain profitability.
72
Table of Contents
If we fail to maintain an effective system of internal control over financial reporting, we may not be able to accurately report our financial results or prevent fraud. As a result, stockholders could lose confidence in our financial and other public reporting, which would harm our business and the trading price of our common stock.
As a public reporting company, we are subject to the rules and regulations established by the SEC and the New York Stock Exchange (“NYSE”). These rules and regulations require, among other things, that we establish and periodically evaluate procedures with respect to our internal control over financial reporting. Reporting obligations as a public company are likely to place a considerable strain on our financial and management systems, processes and controls, as well as on our personnel, including senior management. In addition, as a public company, we are required to document and test our internal control over financial reporting pursuant to Section 404 of the Sarbanes-Oxley Act of 2002 so that our management can certify as to the effectiveness of our internal control over financial reporting. Management’s initial certification under Section 404 of the Sarbanes-Oxley Act of 2002 will be required with our Annual Report for the year ending December 31, 2022. In support of such certifications, we are required to document and make significant changes and enhancements, including potentially hiring additional personnel, to our internal control over financial reporting. Likewise, our independent registered public accounting firm is not required to attest to the effectiveness of our internal control over financial reporting until our first annual report is required to be filed with the SEC following the date we are no longer an emerging growth company (“EGC”). At such time as we are required to obtain auditor attestation, if we then have a material weakness, we would receive an adverse opinion regarding our internal control over financial reporting from our independent registered accounting firm.
To achieve compliance with Section 404 within the prescribed period, we will need to continue to dedicate internal resources, including hiring additional financial and accounting personnel and potentially engaging outside consultants. During our evaluation of our internal control, if we identify one or more material weaknesses in our internal control over financial reporting, we will be unable to assert that our internal control over financial reporting is effective. We have identified material weaknesses in our internal control environment in the past and cannot provide assurances that there will not be material weaknesses or significant deficiencies in our internal control over financial reporting in the future. Any failure to maintain internal control over financial reporting could severely inhibit our ability to accurately report our financial condition, or results of operations. If we are unable to conclude that our internal control over financial reporting is effective or if our independent registered public accounting firm determines we have a material weakness or significant deficiency in our internal control over financial reporting, we could lose investor confidence in the accuracy and completeness of our financial reports, the market price of shares of our common stock could decline, and we could be subject to sanctions or investigations by NYSE, the SEC or other regulatory authorities. Failure to remedy any material weakness in our internal control over financial reporting or to implement or maintain other effective control systems required of public companies, could also restrict our future access to the capital markets.
We have identified material weaknesses in our internal control over financial reporting in the past. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of our annual or interim financial statements will not be prevented or detected on a timely basis. If we are unable to remediate these material weaknesses, or if we identify additional material weaknesses in the future or otherwise fail to maintain an effective system of internal controls, we may not be able to accurately or timely report our financial condition or results of operations, which may adversely affect investor confidence in us and, as a result, our stock price.
Our cash and cash equivalents could be adversely affected if the financial institutions in which we hold our cash and cash equivalents fail.
We regularly maintain cash balances at third-party financial institutions in excess of the Federal Deposit Insurance Corporation insurance limit. While we monitor the cash balances in our operating accounts on a daily basis and adjust the balances as appropriate, these balances could be impacted, and there could be a material adverse effect on our business, if one or more of the financial institutions with which we deposit cash fails or is subject to other adverse conditions in the financial or credit markets. To date, we have experienced no loss or lack of access to our invested cash or cash equivalents; however, we can provide no assurance that access to our invested cash and cash equivalents will not be impacted by adverse conditions in the financial and credit markets.
Risks Related to Governmental Regulation and Litigation
Failure to comply with federal, state, local and international laws and regulations could adversely affect our business and our financial condition.
A variety of federal, state, local and international laws and regulations govern certain aspects of our business. For example, we maintain a registration from the U.S. Drug Enforcement Administration (“DEA”) for the research of certain controlled
73
Table of Contents
substances and permits from the Boston Public Health Commission to conduct work with recombinant DNA. Some of our programs or products made or developed using our engineered cells and/or biomanufacturing processes are subject to regulations, including those promulgated by the FDA, DEA or USDA. Products utilized in our COVID-19 testing services are subject to regulations promulgated by the FDA, the Centers for Medicare and Medicaid Services, and certain state governments. In addition, we are subject to laws relating to, among other things, anti-bribery, insider trading, sourcing of biological materials and data privacy. The legal and regulatory requirements that apply to our business may be interpreted and applied in a manner that is inconsistent from one jurisdiction to another or may conflict with other rules or our practices. As a result, our practices may not comply, or may not comply in the future with all such laws, regulations, requirements and obligations. Any failure, or perceived failure, by us to comply with any federal, state, local or international laws, regulations, industry self-regulatory principles, industry standards or codes of conduct, regulatory guidance, orders to which we may be subject or other legal obligations could adversely affect our reputation, brand and business, and may result in claims, proceedings or actions against us by governmental entities or others or other liabilities or require us to change our operations. We may also be contractually required to indemnify and hold harmless third parties from the costs or consequences of non-compliance with any laws, regulations or other legal obligations.
We may also become subject to increasing regulation in the future as we expand our business. We have limited experience operating a business located outside of Massachusetts. As we continue to expand our operations and offerings domestically and globally, we will have to expend significant management and financial resources to maintain compliant practices in those locations. Non-compliance could lead to litigation, which would require substantial management and financial resources.
We may incur significant costs complying with environmental, health and safety laws and regulations, and failure to comply with these laws and regulations could expose us to significant liabilities.
We use hazardous chemical and biological materials in our business and are subject to a variety of federal, state, local and international laws and regulations governing, among other matters, the use, generation, manufacture, transportation, storage, handling, disposal of, and human exposure to these materials, including regulation by governmental regulatory agencies, such as the Occupational Safety and Health Administration and the EPA. We have incurred, and will continue to incur, capital and operating expenditures and other costs in the ordinary course of our business in complying with these laws and regulations.
Although we have implemented safety procedures for storing, handling and disposing of these materials and waste products in an effort to comply with these laws and regulations, we cannot be sure that our safety measures will be compliant or capable of eliminating the risk of injury or contamination from the generation, manufacturing, use, storage, transportation, handling, disposal of and human exposure to hazardous materials and/or flammable chemicals. Failure to comply with environmental, health and safety laws could subject us to liability and resulting damages. There can be no assurance that violations of environmental, health and safety laws will not occur as a result of human error, accident, equipment failure, contamination, intentional misconduct or other causes. Compliance with applicable environmental laws and regulations may be expensive, and the failure to comply with past, present or future laws could result in the imposition of fines, regulatory oversight costs, third party property damage, product liability and personal injury claims, investigation and remediation costs, the suspension of production or a cessation of operations, and our liability may exceed our total assets. Liability under environmental laws can be imposed for the full amount of damages without regard to comparative fault for the investigation and cleanup of contamination and impacts to human health and for damages to natural resources. Contamination at properties we may own and operate and at properties to which we send hazardous materials, may result in liability for us under environmental laws and regulations.
Our business and operations may be affected by other new environmental, health and safety laws and regulations, which may require us to change our operations, or result in greater compliance costs and increasing risks and penalties associated with violations, which could impair our research, development or production efforts and harm our business.
If we fail to comply with healthcare and other governmental regulations, we could face substantial penalties and our business, financial condition and results of operations could be adversely affected.
Our business activities may be subject to regulation and enforcement by the FDA, U.S. Department of Justice, HHS, Office of Inspector General, and other federal and state governmental authorities. Although our offerings are not currently billed to any third-party payor, including any commercial payor or government healthcare program, we may, in the future, submit claims for our COVID-19 testing services to third-party payors, including government healthcare programs. If we submit claims to third-party payors, such activity will expand the scope of federal and state healthcare laws applicable to us.
Federal and state healthcare laws and regulations that may affect our ability to conduct business include, without limitation:
74
Table of Contents
Because of the breadth of these laws and the narrowness of available statutory and regulatory exemptions, exceptions, and safe harbors, it is possible that some of our activities could be subject to challenge under one or more of such laws. We may face claims and proceedings by private parties, and claims, investigations and other proceedings by governmental authorities, relating to allegations that our business practices do not comply with current or future laws or regulations involving applicable fraud and abuse or other healthcare laws and regulations, and it is possible that courts or governmental authorities may conclude that we or any of our partners have not complied with them, or that we may find it necessary or appropriate to settle any such claims or other proceedings. Any action brought against us for violation of these or other laws or regulations, even if we successfully defend against it, could cause us to incur significant legal expenses and divert our management’s attention from the operation of our business. If our operations are found to be in violation of any federal or state laws described above or any other current or future fraud and abuse or other healthcare laws and regulations that apply to us, we may be subject to claims and proceedings by private parties, investigations and other proceedings by governmental authorities, as well as penalties, including significant criminal, civil and administrative penalties, damages and fines, disgorgement, additional reporting requirements and oversight if we become subject to a corporate integrity agreement or similar agreement to resolve allegations of noncompliance with these laws or regulations, imprisonment for individuals and
75
Table of Contents
exclusion from participation in government programs, such as Medicare and Medicaid, as well as contractual damages and reputational harm. We could also be required to curtail or cease our operations. In addition, if any customers, healthcare professionals we engage, laboratory partners or other entities with whom we do business are found not to be in compliance with applicable laws, they may be subject to the same criminal, civil or administrative sanctions, including exclusion from government-funded healthcare programs. Any of the foregoing could seriously harm our business and financial results.
We may become subject to the comprehensive laws and rules governing billing and payment, noncompliance with which could result in non-payment or recoupment of overpayments for our services or other sanctions.
We may, in the future, submit claims for our COVID-19 testing services to third-party payors. Payors typically have differing and complex billing and documentation requirements. If we fail to comply with these payor-specific requirements, we may not be paid for our services or payment may be substantially delayed or reduced. Numerous state and federal laws would also apply to our claims for payment, including but not limited to (i) “coordination of benefits” rules that dictate which payor must be billed first when a patient has coverage from multiple payors, (ii) requirements that overpayments be refunded within a specified period of time, (iii) “reassignment” rules governing the ability to bill and collect professional fees on behalf of other providers, (iv) requirements that electronic claims for payment be submitted using certain standardized transaction codes and formats, and (v) laws requiring all health and financial information of patients to be maintained in a manner that complies with stringent security and privacy standards.
Audits, inquiries and investigations from government agencies and health network partners can occur from time to time in the ordinary course of our business, and could result in costs to us and a diversion of management’s time and attention. New regulations and heightened enforcement activity also could negatively affect our cost of doing business and our risk of becoming the subject of an audit or investigation. If we bill for our service in the future, our failure to comply with rules related to billing or adverse findings from audits by government and private payors could result in, among other penalties, non-payment for services rendered or recoupments or refunds of amounts previously paid for such services. We cannot predict whether any future audits, inquiries or investigations, or the public disclosure of such matters, likely would negatively impact our business, financial condition, results of operations, cash flows and the trading price of our securities. See also “Risk Factors—Risks Related to Governmental Regulation and Litigation—If we fail to comply with healthcare and other governmental regulations, we could face substantial penalties and our business, financial condition and results of operations could be adversely affected.”
We and our laboratory partners are subject to a variety of laboratory testing standards, compliance with which is an expensive and time-consuming process, and any failure to comply could result in substantial penalties and disruptions to our business.
We and the third-party laboratories that we partner with are subject to the Clinical Laboratory Improvement Amendments of 1988 (“CLIA”). CLIA is a federal law that regulates clinical laboratories that perform testing on specimens derived from humans for the purpose of providing information for the diagnosis, prevention or treatment of disease. CLIA requires virtually all laboratories to be certified by the federal government and mandates compliance with various operational, personnel, facilities administration, quality and proficiency testing requirements depending on the level of complexity for which the laboratory is certified. CLIA certification is also a prerequisite to be eligible to bill state and federal healthcare programs, as well as many private third-party payors, for laboratory testing services. Our partner laboratories hold CLIA certifications for high complexity testing, which mandate compliance with various operational, personnel, facilities administration, quality and proficiency testing requirements depending on the level of complexity for which the laboratory is certified. In addition, we hold CLIA high complexity testing certifications and perform certain CLIA-waived tests on behalf of our clients, which subjects us directly to certain CLIA requirements. Sanctions for failure to comply with CLIA requirements may include suspension, revocation, or limitation of a laboratory’s CLIA certificate, as well as the imposition of significant fines or criminal penalties. Any sanction imposed under CLIA, its implementing regulations, or state or foreign laws or regulations governing licensure, or our partner laboratories’ failure to renew a CLIA certificate, a state or foreign license, or accreditation, could have a material adverse effect on our business.
In addition, our partner laboratories and our laboratories holding CLIA Certificates of Waiver are subject to state laws and regulations governing laboratory licensure. Some states have enacted state licensure laws that are more stringent than CLIA. Our ability to successfully deploy COVID-19 testing at large scale may be adversely impacted if our partner laboratories do not maintain the required regulatory licensure and operate in accordance with CLIA standards. In certain markets such as California, New York, and Pennsylvania, we or our partner laboratories may also need to obtain and maintain additional licensure from such states. It is uncertain that our partner laboratories will be granted such licensure and, in such case, we cannot offer testing to patients located in those states, which could limit our ability to offer testing on a wide scale.
76
Table of Contents
It is possible that additional states may enact laboratory licensure requirements in the future, which could further limit our ability to expand our services.
We rely on third-party laboratories in the conduct of our biosecurity business offering. If any of our partners cease working with us, or face supply chain disruptions or other difficulties, our business could be harmed. Specifically, if any of our partners were to lose or fail to obtain or renew their CLIA certifications or state laboratory licenses, whether as a result of a revocation, suspension or limitation, such laboratories would no longer be able to run the COVID-19 tests we offer to our customers, and our ability to successfully deploy a COVID-19 pooled sample testing program nationwide may be adversely impacted.
The testing industry is subject to complex and costly regulation and if government regulations are interpreted or enforced in a manner adverse to us, we may be subject to enforcement actions, penalties, exclusion, and other material limitations on our operations.
We offer COVID-19 testing services by partnering with third-party laboratories, diagnostic test manufacturers and manufacturers of collection kits, which are subject to extensive and frequently changing federal, state and local laws and regulations governing various aspects of our business, including significant governmental certification and licensing regulations. New laws, regulations and judicial decisions, or new interpretations of existing laws, regulations and decisions, may also limit our potential revenues, and we may need to revise our R&D or commercialization programs. The costs of defending claims associated with violations, as well as any sanctions imposed, could significantly adversely affect our financial performance.
We are required to comply with federal and state genetic testing and privacy laws. We have measures in place to collect clinical data and genetic and other biological samples, and disclose test results, from subjects who have provided appropriate informed consents. However, informed consents could be challenged in the future, and those informed consents could prove invalid, unlawful or otherwise inadequate for our purposes. Any legal challenges could consume our management and financial resources.
Current regulations governing the testing services we offer are shifting and in some cases unclear. In addition, our laboratory partners may be unsuccessful in validating, or obtaining or maintaining authorizations for, the tests we rely on to provide our COVID-19 testing services. If any third-party manufacturers or laboratories offering tests that we use in our testing services are deemed by the FDA or other regulatory authorities to have violated applicable law or if the tests or test components are marketed, processed or distributed in violation of applicable law, we may be subject to enforcement action or litigation, or we may be required to find alternative tests to support our testing services, which could increase our costs and prevent us from successfully commercializing our COVID-19 testing services.
In addition, we are required to comply with applicable FDA regulations with respect to our distribution of certain COVID-19 diagnostic test kits and collection kits, including, for certain kits, compliance with applicable terms and conditions of an EUA. Such conditions may include requirements related to collection of information on the performance of the product, reporting of adverse events, recordkeeping requirements, and labeling and promotional activities. To the extent that we market or promote third-party tests or test kits outside of the uses authorized for these products or in a false or misleading manner, the tests or collection kits could be considered misbranded or adulterated and in violation of applicable law.
Advertising for any of the tests or collection kits we distribute or the testing services we offer is also subject to regulation by the Federal Trade Commission (“FTC”), under the FTC Act. The FTC may take enforcement action for advertising claims that are not adequately substantiated or that are false or misleading. Violations of applicable FDA requirements could result in enforcement actions, such as warning or “untitled” letters, revocation of EUAs, seizures, injunctions, civil penalties and criminal prosecutions and fines, and violation of the FTC Act could result in injunctions and other associated remedies, all of which could have a material adverse effect on our business. Most states also have similar regulatory and enforcement authority for laboratory testing and distribution of related collection kits. For example, many state laws require us to hold a specific form of license to distribute COVID-19 diagnostic test kits and collection kits into such states. These requirements vary from one state to another and frequently change. Complying with state laws and regulations may subject us to similar risks and delays as those we could experience under federal regulation.
We are subject to federal and state laws and regulations governing the protection, use, and disclosure of health information and other types of personal information, and our failure to comply with those laws and regulations or to adequately secure the information we hold could result in significant liability or reputational harm.
Numerous state and federal laws, regulations, standards and other legal obligations, including consumer protection laws and regulations, which govern the collection, dissemination, use, access to, confidentiality, security and processing of personal
77
Table of Contents
information, including health-related information, could apply to our operations or the operations of our partners. For example, HIPAA imposes privacy, security and breach notification obligations on certain healthcare providers, health plans, and healthcare clearinghouses, known as covered entities, as well as their business associates that perform certain services that involve creating, receiving, maintaining or transmitting individually identifiable health information for or on behalf of such covered entities, and their covered subcontractors. HIPAA requires covered entities and business associates to develop and maintain policies with respect to the protection of, use and disclosure of protected health information (“PHI”), including the adoption of administrative, physical and technical safeguards to protect such information, and certain notification requirements in the event of a breach of unsecured PHI. If in the future we engage in certain types of standard electronic transactions involving payors, including billing the Medicare or Medicaid programs or commercial health plans, we will be subject to HIPAA as a “covered entity.” We are currently subject to HIPAA as a “business associate” because we perform certain services involving the use or disclosure of PHI on behalf of covered entity customers with respect to our COVID-19 testing service offerings. Implementation of the infrastructure necessary to meet HIPAA standards requires substantial investment. Being subject to HIPAA as a covered entity or business associate exposes us to significant fines and penalties, including criminal fines and penalties.
Additionally, under HIPAA, covered entities must report breaches of unsecured PHI to affected individuals without unreasonable delay, not to exceed 60 days following discovery of the breach by a covered entity or its agents. Notification also must be made to the HHS Office for Civil Rights and, in certain circumstances involving large breaches, to the media. Business associates must report breaches of unsecured PHI to covered entities within 60 days of discovery of the breach by the business associate or its agents. A non-permitted use or disclosure of PHI is presumed to be a breach under HIPAA unless the Covered Entity or Business Associate establishes that there is a low probability the information has been compromised consistent with requirements enumerated in HIPAA.
Entities that are found to be in violation of HIPAA as the result of a breach of unsecured PHI, a complaint about privacy practices or an audit by HHS, may be subject to significant civil, criminal and administrative fines and penalties and/or additional reporting and oversight obligations if required to enter into a resolution agreement and corrective action plan with HHS to settle allegations of HIPAA non-compliance. HIPAA also authorizes state Attorneys General to file suit on behalf of their residents. Courts may award damages, costs and attorneys’ fees related to violations of HIPAA in such cases. While HIPAA does not create a private right of action allowing individuals to sue us in civil court for violations of HIPAA, its standards have been used as the basis for duty of care in state civil suits such as those for negligence or recklessness in the misuse or breach of PHI.
Even when HIPAA or a state law does not apply, according to the FTC, violating consumers’ privacy rights or failing to take appropriate steps to keep consumers’ personal information secure may constitute unfair and/or deceptive acts or practices in violation of Section 5(a) of the FTC Act. The FTC expects a company’s data security measures to be reasonable and appropriate in light of the sensitivity and volume of consumer information it holds, the size and complexity of its business, and the cost of available tools to improve security and reduce vulnerabilities.
Several states have enacted privacy laws governing the use and disclosure of health information, such as the California Confidentiality of Medical Information Act; these laws are not preempted by HIPAA to the extent they are more stringent than HIPAA. Such laws and regulations will be subject to interpretation by various courts and other governmental authorities, thus creating potentially complex compliance issues for us and our partners. Further, in recent years, there have been a number of well-publicized data breaches involving the improper dissemination of personal information of individuals both within and outside of the healthcare industry. Laws in all 50 states require businesses to provide notice to individuals whose personally identifiable information has been disclosed as a result of a data breach. The laws are not consistent, and compliance in the event of a widespread data breach is costly. States are also constantly amending existing laws, and creating new data privacy and security laws, requiring attention to frequently changing regulatory requirements. For example, the California Consumer Privacy Act of 2018 (“CCPA”) went into effect on January 1, 2020. The CCPA creates new transparency requirements and grants California residents several new rights with respect to their personal information. Failure to comply with the CCPA may result in, among other things, significant civil penalties and injunctive relief, or potential statutory or actual damages. On November 3, 2020, California voters passed a ballot initiative for the California Privacy Rights Act (“CPRA”), which will significantly expand the CCPA. Most CPRA provisions will take effect on January 1, 2023, though the obligations will apply to any personal information collected after January 1, 2022. The CPRA will impose additional data protection obligations on covered businesses, including additional consumer rights processes, limitations on data uses, new audit requirements for higher risk data, and opt outs for certain uses of sensitive data. It will also create a new California data protection agency authorized to issue substantive regulations and could result in increased privacy and information security enforcement. The majority of the provisions will go into effect on January 1, 2023, and additional compliance investment and potential business process changes may be required. Similar laws have been proposed
78
Table of Contents
or passed in other states, including the Virginia Consumer Data Protection Act, which will take effect on January 1, 2023. We will need to invest substantial resources in putting in place policies and procedures to comply with these evolving state laws.
As our operations and business grow, we may become subject to or affected by new or additional data protection laws and regulations and face increased scrutiny or attention from regulatory authorities. For example, the European Union General Data Protection Regulation (“GDPR”), which went into effect in May 2018, imposes strict requirements for processing the personal data of individuals within the European Economic Area (“EEA”). Companies that must comply with the GDPR face increased compliance obligations and risk, including more robust regulatory enforcement of data protection requirements and potential fines for noncompliance of up to €20 million or 4% of the annual global revenues of the noncompliant company, whichever is greater. Among other requirements, the GDPR regulates transfers of personal data subject to the GDPR to third countries that have not been found to provide adequate protection to such personal data, including the United States, and the efficacy and longevity of current transfer mechanisms between the EU and the United States remains uncertain. For example, in 2016, the EU and United States agreed to a transfer framework for data transferred from the EU to the United States, called the Privacy Shield, but the Privacy Shield was invalidated in July 2020 by the Court of Justice of the European Union. Further, from January 1, 2021, companies have to comply with the GDPR and also the United Kingdom GDPR (the “UK GDPR”), which, together with the amended UK Data Protection Act 2018, retains the GDPR in UK national law. The UK GDPR mirrors the fines under the GDPR, i.e., fines up to the greater of €20 million (£17.5 million) or 4% of global turnover. The relationship between the United Kingdom and the European Union in relation to certain aspects of data protection law remains unclear, and it is unclear how United Kingdom data protection laws and regulations will develop in the medium to longer term. On June 28, 2021, the European Commission adopted an adequacy decision in favor of the United Kingdom, enabling data transfers from EU member states to the United Kingdom without additional safeguards. However, the United Kingdom adequacy decision will automatically expire in June 2025 unless the European Commission renews or extends that decision and remains under review by the Commission during this period. These changes may lead to additional costs and increase our overall risk exposure.
Although we work to comply with applicable laws, regulations and standards, contractual obligations and other legal obligations, these requirements are evolving and may be modified, interpreted and applied in an inconsistent manner from one jurisdiction to another, and may conflict with one another or other legal obligations with which Ginkgo must comply. Recently, there has been an increase in public awareness of privacy issues in the wake of revelations about the data-collection activities of various government agencies and in the number of private privacy-related lawsuits filed against companies. Any failure or perceived failure by us or our employees, representatives, contractors, consultants, collaborators, or other third parties to comply with such requirements or adequately address privacy and security concerns, even if unfounded, could result in additional cost and liability to us, damage our reputation, and adversely affect our business and results of operations.
We have pursued in the past and may pursue additional U.S. Government contracting and subcontracting opportunities in the future and as a U.S. Government contractor and subcontractor, we are subject to a number of procurement rules and regulations.
We have entered into agreements with governmental entities and contractors in the past to serve as a U.S. government contractor or subcontractor and may do so again in the future. U.S. government procurement contractors and subcontractors must comply with specific procurement regulations and other requirements. These requirements, although customary in U.S. government contracts, could impact our performance and compliance costs, including by limiting or delaying our ability to share information with business partners, customers and investors. The U.S. government has in the past and may in the future demand contract terms that are less favorable than standard arrangements with private sector customers and may have statutory, contractual, or other legal rights to terminate contracts with us for convenience or for other reasons. Generally, U.S. government contracts contain provisions permitting unilateral termination or modification, in whole or in part, at the government’s convenience. Under general principles of government contracting law, if the government terminates a contract for convenience, the government contractor may recover only its incurred or committed costs, settlement expenses and profit on work completed prior to the termination. If the government terminates a contract for default, the government contractor is entitled to recover costs incurred and associated profits on accepted items only and may be liable for excess costs incurred by the government in procuring undelivered items from another source. Any termination for default may also adversely affect our ability to contract with other government customers, as well as our reputation, business, financial condition and results of operations. In addition, changes in U.S. government budgetary priorities could lead to changes in the procurement environment, affecting availability of U.S. government contracting, subcontracting or funding opportunities, which could lead to modification, reduction or termination of our U.S. government contracts or subcontracts. If and to the extent such changes occur, they could impact our results and potential growth opportunities.
Furthermore, our U.S. government contracts grant the government the right to use technologies developed by us under the government contract or the right to share data related to our technologies, for or on behalf of the government. Under our
79
Table of Contents
government contracts, we may not be able to limit third parties, including our competitors, from accessing certain of these technology or data rights, including intellectual property, in providing products and services to the government.
In addition, failure by us, our employees, representatives, contractors, partners, agents, intermediaries, other customers or other third parties to comply with these regulations and requirements could result in reductions of the value of contracts, contract modifications or termination, claims for damages, refund obligations, the assessment of civil or criminal penalties and fines, loss of rights in our intellectual property and temporary suspension or permanent debarment from government contracting, all of which could negatively impact our results of operations and financial condition. Any such damages, penalties, disruptions or limitations in our ability to do business with the public sector could result in reduced sales of our products, reputational damage, penalties and other sanctions, any of which could harm our business, reputation and results of operations.
We are engaged in certain research activities involving controlled substances, including cannabinoids and other chemical intermediates, the making, use, sale, importation, exportation, and distribution of which may be subject to significant regulation by the DEA and other regulatory agencies.
We are engaged in certain research activities involving the development of microbes designed to generate cannabinoids, their precursors and other chemical intermediaries, some of which may be regulated as controlled substances in the United States. Controlled substances are subject to state, federal, and foreign laws and regulations regarding their manufacture, use, sale, importation, exportation, and distribution. Among other things, controlled substances are regulated under the federal Controlled Substances Act of 1970 (“CSA”) and implementing regulations of the DEA. The DEA regulates controlled substances as Schedule I, II, III, IV or V substances. Schedule I substances by definition have no established medicinal use and may generally not be marketed or sold in the United States. Schedule I substances are subject to the most stringent controls and Schedule V the least controls of the five schedules, based on their relative risk of abuse.
Cannabinoids are naturally occurring compounds found in the cannabis plant. The cannabis plant and its derivatives are highly regulated by the DEA and the USDA. Specifically, marihuana, which is defined as all parts of the plant Cannabis sativa L., whether growing or not, the seeds thereof, the resin extracted therefrom, and every compound, manufacture, salt, derivative, mixture, or preparation, is classified as a Schedule I controlled substance. However, the term does not include “hemp,” which means the cannabis plant and any part of that plant, including the seeds and all derivatives, extracts, cannabinoids, isomers, acids, salts, and salts of isomers, whether growing or not, with a delta-9 tetrahydrocannabinol (“THC”) concentration of not more than 0.3% on a dry weight basis. Thus, depending on the THC concentration of the product, the product may or may not be regulated as a controlled substance. The DEA has historically regulated synthetic cannabinoids similarly to naturally-derived cannabinoids. Consequently, even though our cannabinoids that could be produced from microbes may not be derived from the cannabis plant, the DEA may consider them to be controlled substances subject to stringent regulatory controls.
Regulations associated with controlled substances govern manufacturing, labeling, packaging, testing, dispensing, production and procurement quotas, recordkeeping, reporting, handling, shipment and disposal. These regulations include required security measures, such as background checks on employees and physical control of inventory and increase the personnel needs and the expense associated with development and commercialization of products or product candidates including controlled substances. Regulators conduct periodic inspections of entities involved in handling, manufacturing, or otherwise distributing controlled substances, and have broad enforcement authorities. If we are found to be non-compliant with applicable controlled substance registrations and related requirements, we may need to modify its business activities and/or stop handling or producing the products regulated as controlled substances, and could be subject to enforcement action, significant fines or penalties, and/or adverse publicity, among other consequences.
Various states also independently regulate controlled substances. Though state-controlled substances laws often mirror federal law, because the states are separate jurisdictions, they may separately schedule substances, as well. The failure to comply with applicable regulatory requirements could lead to enforcement actions and sanctions from the states in addition to those from the DEA or otherwise arising under federal law.
Changes in government regulations may materially and adversely affect our sales and results of operations.
The markets where we provide our services are heavily influenced by foreign, federal, state and local government regulations and policies. The U.S. or foreign governments may take administrative, legislative, or regulatory action that could materially interfere with our customer’s ability to sell products derived from engineered cells in certain countries and/or to certain customers. The uncertainty regarding future standards and policies may also affect our ability to develop our programs or to license engineered cells to customers and to initiate new programs with our customers, which could have a material adverse effect on our business, financial condition and results of operations.
80
Table of Contents
Changes in U.S. trade policy more generally could trigger retaliatory actions by affected countries, which could impose restrictions on our ability to do business in or with affected countries or prohibit, reduce or discourage purchases of our services by foreign customers, leading to increased program costs, increased costs of developing or manufacturing our customers’ products and higher prices for their products in foreign markets. Changes in, and responses to, U.S. trade policy could reduce the competitiveness of our services or our customers’ products, cause our services to be less in demand and our sales to decline and adversely impact our ability to compete, which could materially and adversely impact our business, financial condition and results of operations.
We are subject to certain U.S. and foreign anti-corruption, anti-bribery and anti-money laundering laws and regulations. We can face serious consequences for violations.
We are subject to the U.S. Foreign Corrupt Practices Act of 1977, as amended (the “FCPA”), the U.S. domestic bribery statute contained in 18 U.S.C. § 201, the U.S. Travel Act, the U.K. Bribery Act and possibly other anti-corruption, anti-bribery and anti-money laundering laws and regulations in the jurisdictions in which we do business, both domestic and abroad. Anti-corruption and anti-bribery laws have been enforced aggressively in recent years. The FCPA and other anti-corruption laws generally prohibit companies, their employees, agents, representatives, business partners and third-party intermediaries from corruptly promising, authorizing, offering, or providing, directly or indirectly, anything of value to government officials, political parties, or candidates for public office for the purpose of obtaining or retaining business or securing an improper business advantage. The UK Bribery Act and other anti-corruption laws also prohibit commercial bribery not involving government officials, and requesting or accepting bribes; and anti-money laundering laws prohibit engaging in certain transactions involving criminally-derived property or the proceeds of criminal activity.
We and our third-party business partners, representatives and agents may have direct or indirect interactions with officials and employees of government agencies or state-owned or -affiliated universities or other entities (for example, to obtain necessary permits, licenses, patent registrations and other regulatory approvals), which increases our risks under the FCPA and other anti-corruption laws. We also engage contractors, consultants and other third parties from time to time to conduct business development activities abroad. We may be held liable for the corrupt or other illegal activities of our employees or third parties even if we do not explicitly authorize such activities. We expect our non-U.S. activities to increase over time, which may also increase our exposure under these laws.
The FCPA also requires that we keep accurate books and records and maintain a system of adequate internal controls. While we have controls to address compliance with such laws, and will continue to review and enhance our compliance program, we cannot assure you that our employees, agents, representatives, business partners or third-party intermediaries will always comply with our policies and applicable law, for which we may be ultimately held responsible.
Any allegations or violation of the FCPA or other applicable anti-bribery, anti-corruption laws and anti-money laundering laws may result in whistleblower complaints, sanctions, settlements, investigations, prosecution, enforcement actions, substantial criminal fines and civil penalties, disgorgement of profits, imprisonment, debarment, tax reassessments, breach of contract and fraud litigation, loss of export privileges, suspension or debarment from U.S. government contracts, adverse media coverage, reputational harm and other consequences, all of which may have an adverse effect on our reputation, business, financial condition, results of operations and prospects. Responding to an investigation or action can also result in a materially significant diversion of management’s attention and resources and significant defense costs and other professional fees.
Significant disruptions to our and our service providers’ information technology systems or data security incidents could result in significant financial, legal, regulatory, business and reputational harm to us.
We are increasingly dependent on information technology systems and infrastructure, including services licensed, leased or purchased from third parties such as cloud computing infrastructure and operating systems, to operate its business. In the ordinary course of business, we collect, store, process and transmit large amounts of sensitive information, including intellectual property, proprietary business information, personal information and other confidential information. It is critical that we do so in a secure manner to maintain the confidentiality, integrity and availability of such sensitive information. We have also outsourced elements of our operations (including elements of its information technology infrastructure) to third parties, and as a result, we manage a number of third-party vendors who may have access to our networks or our confidential information. While we take measures to safeguard and protect this information, threats to network and data security are increasingly diverse and sophisticated. As a result of the COVID-19 pandemic, we may also face increased cybersecurity risks due to our reliance on internet technology and the number of our employees working remotely, which may create additional opportunities for cybercriminals to exploit vulnerabilities. Despite our efforts, training and processes to prevent security breaches and incidents, our information technology systems, servers, and those of third parties that we use in our
81
Table of Contents
operations are vulnerable to cybersecurity risks, including cyberattacks such as viruses and worms, phishing attacks and other forms of social engineering, denial-of-service attacks, ransomware attacks, physical or electronic break-ins, third-party or employee theft or misuse, and other negligent actions, errors or malfeasance by employees or other third parties, and similar disruptions from unauthorized tampering with its servers and computer systems or those of third parties that we use in its operations, which could lead to interruptions, delays, loss or corruption of critical data, unauthorized access to or acquisition of health-related and other personal information. In addition, we may be the target of email scams and other social engineering attacks that attempt to acquire personal information or company assets or access to our systems. Despite our efforts to create security barriers to such threats, we may not be able to entirely mitigate these risks. Our third-party service providers face similar risks. Any cyberattack that attempts to obtain our data or assets, including data that we maintain on behalf of its customers, disrupt its service, or otherwise access its systems, or those of third parties we use, or any other security breach or incident, could adversely affect our business, financial condition and operating results, be expensive to remedy, and damage our reputation. We and our third-party service providers may face difficulties or delays in identifying or otherwise responding to any attacks or actual or potential security breaches or security incidents. We may incur significant costs and operational consequences of investigating, remediating, eliminating and putting in place additional tools and devices designed to prevent actual or perceived security breaches and other security incidents, including in response to any actual or perceived incident we may suffer, and substantial costs to comply with any notification or other legal obligations resulting from any security breaches or other security incidents. In addition, any such breaches or incidents, or the perception that they have occurred, may result in negative publicity, and could have an adverse effect on our business, financial condition, and operating results.
Although we maintain insurance coverage that may cover certain liabilities in connection with security breaches and other security incidents, we cannot be certain our insurance coverage will be adequate for liabilities actually incurred, that insurance will continue to be available to us on commercially reasonable terms (if at all) or that any insurer will not deny coverage as to any future claim.
Governmental trade controls, including export and import controls, sanctions, customs requirements and related regimes, could subject us to liability or loss of contracting privileges or limit our ability to compete in certain markets.
Our programs and technologies are subject to U.S. and non-U.S. export controls. Export authorizations may be required for biotechnology products, technologies, or services to be exported outside of the United States, to a foreign person, or outside of a foreign jurisdiction. Our current or future programs or technologies are, and may in the future, be subject to the Export Administration Regulations (“EAR”). If a program, technology, or service meets certain criteria for control under the EAR, then that engineered cell, production process, resulting product, technology, or service would be exportable outside the United States or to a foreign person or from one foreign jurisdiction to another foreign jurisdiction only if we obtain the applicable export license or other applicable authorization including qualifying for a license exception, if required. Compliance with the U.S. and foreign export laws and regulations and other applicable regulatory requirements regarding the sales, shipment and use of our engineering cells, bioprocesses and other technology may affect our ability to work with foreign partners, affect the speed at which we can introduce new products into non-U.S. markets, or limit our ability to sell programs or services or license technologies into some countries.
Additionally, certain materials that we use in our programs are subject to U.S. import controls. We currently have, and may in the course of business need to procure, certain import authorizations, for example, related to plant pests, chemicals, biological agents and other controlled materials, including from the USDA, EPA and CDC. Compliance with applicable regulatory requirements regarding the import of such materials may limit our access to materials critical to our development activities or affect the speed at which we can advance new programs.
Our activities are also subject to the economic sanctions laws and regulations of the United States and other jurisdictions. Such controls prohibit certain transactions, potentially including financial transactions and the transfer of products, technologies and services, to sanctioned countries, governments and persons, without a license or other appropriate authorization. U.S. sanctions policy changes could affect our or our customers’ ability to interact, directly and indirectly, with targeted companies or companies in sanctioned countries.
While we take precautions to comply with U.S. and non-U.S. export control, import control and economic sanctions laws and regulations, we cannot guarantee that such precautions will prevent violations of such laws, including transfers to unauthorized persons or destinations, and including inadvertent violations as a result of a misclassification of a product, technology or service under export control laws. Violations could result in our business being subject to government investigations, denial of export or import privileges, significant fines or penalties, denial of government contracts and reputational harm. Any limitation on our ability to export our engineered cells, production processes, resulting products,
82
Table of Contents
technology, or services, or import materials critical to our programs would likely adversely affect our business and financial condition.
Changes in U.S. and foreign tax laws could have a material adverse effect on our business, cash flow, results of operations or financial conditions.
We are subject to tax laws, regulations and policies of the U.S. federal, state and local governments. Changes in tax laws, as well as other factors, could cause us to experience fluctuations in our tax obligations and otherwise adversely affect our tax positions and/or our tax liabilities. For example, the results of the 2020 presidential and congressional elections in the United States could result in significant changes in tax law that could adversely impact our effective tax rate. In addition, the Organisation for Economic Co-operation and Development has published proposals covering various international tax-related issues, including country-by-country reporting, permanent establishment rules, transfer pricing and tax treaties. Future tax reform resulting from these developments may result in changes that could adversely affect our effective tax rate or result in higher cash tax liabilities. There can be no assurance that our tax payments, tax credits, or incentives will not be adversely affected by these or other initiatives.
We may become subject to lawsuits or indemnity claims in the ordinary course of business, which could materially and adversely affect our business and results of operations.
From time to time, we may in the ordinary course of business be named as a defendant in lawsuits, indemnity claims and other legal proceedings. These actions may seek, among other things, compensation for alleged product liability, personal injury, employment discrimination, breach of contract, property damage and other losses or injunctive or declaratory relief.
The marketing, sale and use of our services engineered cells, production processes and resulting products could lead to the filing of product liability claims were someone to allege that our services, engineered cells, production processes or resulting products failed to perform as designed or intended or caused injury or other harms. A product liability claim could result in substantial damages and be costly and time-consuming for us to defend.
Regardless of merit or eventual outcome, product liability claims may result in:
In the event that such actions, claims or proceedings are ultimately resolved unfavorably to us at amounts exceeding our accrued liability, or at material amounts, the outcome could materially and adversely affect our business and results of operations. In addition, payments of significant amounts, even if reserved, could adversely affect our liquidity position. We maintain product liability insurance, but this insurance may not fully protect us from the financial impact of defending against product liability claims. Any product liability claim brought against us, with or without merit, could increase our insurance rates or prevent us from securing insurance coverage in the future. Additionally, any product liability lawsuit could cause current collaborators to terminate existing agreements or potential collaborators to seek other companies, any of which could impact our business and results of operations.
Our business could be adversely affected by legal challenges to our telehealth partner’s business model.
Certain of our COVID-19 biosecurity offerings rely significantly on healthcare provider orders for testing that are placed on the basis of telemedicine encounters. The ability to conduct telehealth services in a particular state is directly dependent upon the applicable laws governing remote healthcare, the practice of medicine and healthcare delivery in general in such location which are subject to changing political, regulatory and other influences. With respect to telehealth services, state medical boards continue to implement new rules or interpret existing rules in a manner that may limit or restrict the ability of the centers to conduct their business as it has been conducted in the past. Additionally, during the COVID-19 public health emergency, many states enacted waivers and adopted other temporary measures that lifted certain restrictions on out-of-state providers and relaxed licensure requirements to allow greater access to telehealth services during the public health emergency
83
Table of Contents
period. At this time, we cannot predict whether these waivers or temporary measures will remain in place after the end of the public health emergency period. Accordingly, we must monitor compliance with laws in every jurisdiction in which we operate, and we cannot provide assurance that government authorities may nonetheless challenge our activities and arrangements with our telehealth partner and consider them non-compliant. Additionally, it is possible that the laws and rules governing the practice of medicine, including remote healthcare, in one or more jurisdictions may change in a manner deleterious to our business. If a successful legal challenge or an adverse change in the relevant laws were to occur, and we are unable to adapt our business model accordingly, our operations as well as the operations of our telehealth partner in the affected jurisdictions would be disrupted, which could have a material adverse effect on our business, financial condition and results of operations.
Risks Related to our Common Stock, Organizational Structure and Governance
We are not, and do not intend to become, regulated as an “investment company” under the Investment Company Act, and if we were deemed an “investment company” under the Investment Company Act, applicable restrictions could make it impractical for us to continue our business as contemplated and could have a material adverse effect on our business.
An entity generally will be deemed to be an “investment company” for purposes of the Investment Company Act if:
We believe that we are engaged primarily in the business of providing cell engineering services to customers from across a variety of industries and not in the business of investing, reinvesting or trading in securities. We hold ourselves out as a synthetic biology company and do not propose to engage primarily in the business of investing, reinvesting or trading in securities. Accordingly, we do not believe that we are an “orthodox” investment company as defined in Section 3(a)(1)(A) of the Investment Company Act of 1940, as amended (the “Investment Company Act”) and described in the first bullet point above. Furthermore, we believe that less than 40% of our total assets (exclusive of U.S. government securities and cash items) on an unconsolidated basis will be composed of assets that could be considered investment securities. Accordingly, we do not believe that we are an inadvertent investment company by virtue of the 40% tests in Section 3(a)(1)(C) of the Investment Company Act as described in the second bullet point above. In addition, we believe that we are not an investment company under Section 3(b)(1) of the Investment Company Act because we are primarily engaged in a non-investment company business.
The Investment Company Act and the rules thereunder contain detailed parameters for the organization and operation of investment companies. Among other things, the Investment Company Act and the rules thereunder limit or prohibit transactions with affiliates, impose limitations on the issuance of debt and equity securities, generally prohibit the issuance of options and impose certain governance requirements. We intend to conduct our operations so that we will not be deemed to be an investment company under the Investment Company Act or otherwise conduct our business in a manner that does not subject us to the registration and other requirements of the Investment Company Act. In order to ensure that we are not deemed to be an investment company, we may be limited in the assets that we may continue to own and, further, may need to dispose of or acquire certain assets at such times or on such terms as may be less favorable to us than in the absence of such requirement. If anything were to happen which would cause us to be deemed to be an investment company under the Investment Company Act (such as significant changes in the value of our programs or a change in circumstance that results in a reclassification of our interests in our programs for purposes of the Investment Company Act), the requirements imposed by the Investment Company Act could make it impractical for us to continue our business as currently conducted, which would materially adversely affect our business, financial condition and results of operations. In addition, if we were to become inadvertently subject to the Investment Company Act, any violation of the Investment Company Act could subject us to material adverse consequences, including potentially significant regulatory penalties and the possibility that certain of our contracts could be deemed unenforceable.
84
Table of Contents
Only our employees and directors are entitled to hold shares of Class B common stock (including shares of Class B common stock granted or otherwise issued to our employees and directors in the future), which shares have ten votes per share. This limits or precludes other stockholders’ ability to influence the outcome of matters submitted to stockholders for approval, including the election of directors, the approval of certain employee compensation plans, the adoption of certain amendments to our organizational documents and the approval of any merger, consolidation, sale of all or substantially all of our assets, or other major corporate transaction requiring stockholder approval.
Shares of our Class B common stock have ten votes per share, whereas shares of our Class A common stock have one vote per share and shares of our Class C common stock have no voting rights (except as otherwise expressly provided in our amended and restated certificate of incorporation (the “Charter”) or required by applicable law). As of March 17, 2022, our directors and executive officers hold in the aggregate approximately 49.7% of the total voting power of our outstanding capital stock, and our directors, Founders and executive officers hold in the aggregate approximately 19.7% of the total voting power of our outstanding capital stock. Accordingly, holders of shares of Class B common stock are able to significantly influence the outcome of matters submitted to our stockholders for approval, including the election of directors, the approval of certain employee compensation plans, the adoption of amendments to our organizational documents and the approval of any merger, consolidation, sale of all or substantially all of our assets or other major corporate transaction requiring stockholder approval. This concentrated voting power limits or precludes other stockholders’ ability to influence the outcome of these matters. Holders of Class B common stock may have interests that differ from holders of Class A common stock and may vote in a way with which holders of Class A common stock disagree and which may be adverse to the interests of holders of Class A common stock. This concentrated voting power is likely to have the effect of limiting the likelihood of an unsolicited merger proposal, unsolicited tender offer or proxy contest for the removal of directors. As a result, our governance structure and Charter may have the effect of depriving our stockholders of an opportunity to sell their shares at a premium over prevailing market prices and make it more difficult to replace our directors and management. Furthermore, this concentrated voting power could discourage a potential investor from acquiring Class A common stock due to the limited voting power of such stock relative to Class B common stock, which could also adversely affect the trading price of Class A common stock.
Our multi-class stock structure is intended to preserve our existing founder-led governance structure, to promote employee retention and engagement, to facilitate continued innovation and the risk-taking that it requires, to permit us to continue to prioritize our long-term goals rather than short-term results, to enhance the likelihood of continued stability in the composition of our board of directors and its policies, and to discourage certain types of transactions that may involve an actual or threatened acquisition of the company, all of which we believe are essential to the long-term success of our company and to long-term stockholder value. We expect to maintain this concentrated voting power among our founders and employees for the foreseeable future, including by issuing additional shares of Class B common stock to our employees pursuant to our equity compensation plans.
Future transfers of shares of Class B common stock to persons other than Ginkgo directors and employees, or trusts or legal entities through which the right to vote the shares of Class B common stock held thereby is exercised exclusively by one or more of Ginkgo’s directors or employees (any such director, employee, trust or legal entity, an “Eligible Holder”), or the holder of shares of Class B common stock ceasing to be an Eligible Holder, will generally result in those shares converting to shares of Class A common stock on a one-to-one basis, subject to certain exceptions and unless a majority of the independent directors of the our Board determine that such transfer or event will not result in such automatic conversion. Each share of Class B common stock is also convertible at any time at the option of the holder into one share of Class A common stock. The conversion of Class B common stock to Class A common stock over time will have the effect of increasing the relative voting power of those holders of Class B common stock who retain their shares of Class B common stock in the long term. As a result, the relative voting power of holders of Class A common stock is expected to remain limited for a significant period of time, and it is possible that one or more of the persons or entities holding Class B common stock could gain significant voting control as other holders of Class B common stock sell or otherwise convert their shares into Class A common stock. In addition, the conversion of Class B common stock to Class A common stock would dilute holders of Class A common stock in terms of voting power within the Class A common stock. Because holders of Class C common stock have no voting rights (except as otherwise expressly provided in the Charter or required by applicable law), if we issue Class C common stock in the future, the holders of Class B common stock may be able to significantly influence the outcome of matters submitted to our stockholders for approval for a longer period of time than would be the case if we issued Class A common stock rather than Class C common stock in such transactions. A description of the Company’s securities is filed herewith as Exhibit 4.2.
85
Table of Contents
Our share price may change significantly over time, and you may not be able to resell our common stock at or above the price you paid or at all, and you could lose all or part of your investment as a result.
The trading price of our Class A common stock has been in the past and is likely to continue to be volatile. Such volatility may be, in part, attributable to:
These factors among others may materially adversely affect the market price of our Class A common stock, regardless of our actual operating performance. In addition, price volatility may be greater if the public float and trading volume of our common stock are low.
In the past, following periods of market volatility, stockholders have instituted securities class action litigation. If we were involved in securities litigation, it could have a substantial cost and divert resources and the attention of executive management from our business regardless of the outcome of such litigation.
Future sales, or the perception of future sales, by us or our stockholders in the public market could cause the market price for our securities to decline.
The sale of our securities in the public market, or the perception that such sales could occur, could harm the prevailing market price of our securities. These sales, or the possibility that these sales may occur, also might make it more difficult for us to sell equity securities in the future at a time and at a price that we deem appropriate.
As of the consummation of the Business Combination, we had a total of approximately 1,959 million shares of common stock outstanding on a fully-diluted basis, consisting of approximately 1,333 million shares of Class A common stock and approximately 626 million shares of Class B common stock. All shares issued in the merger are freely tradable without registration under the Securities Act, and without restriction by persons other than our “affiliates” (as defined under Rule 144 of the Securities Act, “Rule 144”), including our directors, executive officers and other affiliates. Of these shares, approximately 631 million shares of common stock outstanding on a fully-diluted basis are subject to a one-year lock-up, which is scheduled to expire on September 16, 2022. In addition to the above, there are up to approximately 206 million shares of common stock that may be earned if the trading price is greater than or equal to the earnout price threshold in the table below for any point in a trading day during 20 trading days in a 30 consecutive trading day period, of which
86
Table of Contents
approximately 51.5 million shares were earned as of December 31, 2021. The vast majority of the shares that are part of the earnout will not be subject to lock-up once the earnout conditions are met.
Earnout Price Threshold |
Number of Shares Earned |
$12.50 (earnout conditions have been met) |
Approximately 51.5 million |
$15.00 |
Approximately 51.5 million |
$17.50 |
Approximately 51.5 million |
$20.00 |
Approximately 51.5 million |
In addition, the shares of our common stock reserved for future issuance under our equity incentive plans will become eligible for sale in the public market once those shares are issued, subject to provisions relating to various vesting agreements and, in some cases, limitations on volume and manner of sale applicable to affiliates under Rule 144, as applicable. Our compensation committee of our board of directors may determine the exact number of shares to be reserved for future issuance under our equity incentive plans at its discretion. We are expected to file one or more registration statements on Form S-8 under the Securities Act to register shares of Class A common stock or securities convertible into or exchangeable for shares of Class A common stock issued pursuant to our equity incentive plans. Any such Form S-8 registration statements will automatically become effective upon filing. Accordingly, shares registered under such registration statements will be available for sale in the open market.
Short sellers may engage in manipulative activity intended to drive down the market price of our Class A common stock, which could also result in related regulatory and governmental scrutiny, among other effects.
Short selling is the practice of selling securities that the seller does not own but rather has borrowed or intends to borrow from a third party with the intention of later buying lower priced identical securities to return to the lender. Accordingly, it is in the interest of a short seller of our Class A common stock for the price to decline. At any time, short sellers may publish, or arrange for the publication of, opinions or characterizations that are intended to create negative market momentum. Issuers, like us, whose securities have historically had limited trading history or volumes and/or have been susceptible to relatively high volatility levels can be vulnerable to such short seller attacks. Short selling reports can cause increased volatility in an issuer’s stock price, and result in regulatory and governmental inquiries. On October 6, 2021, such a report was published about us. Shortly after, we received a preliminary and informal inquiry from the U.S. Department of Justice related to such report. Any related inquiry or formal investigation from a governmental organization or other regulatory body, including any inquiry from the SEC, could result in a material diversion of our management’s time and could have a material adverse effect on our business and results of operations.
Our Charter authorizes a large number of shares of Class B common stock for issuance in the future. The future issuance of shares of Class B common stock may have the effect of further concentrating voting power with our employees and other Class B stockholders, and could have an adverse effect on the trading price of Class A common stock.
Under our Charter, we are authorized to issue 4,500,000,000 shares of Class B common stock, which are entitled to ten votes per share. We currently intend to issue additional shares of Class B common stock in the future to existing and newly hired employees pursuant to our equity compensation plans. Our authorized but unissued shares of Class B common stock are available for issuance to Eligible Holders with the approval of our board of directors without stockholder approval, except as may be required by the Listing Rules of the NYSE. In addition, our authorized but unissued shares of Class B common stock are available for issuance to persons other than Eligible Holders only with the approval of majority of our Class B Directors. If we issue additional shares of Class B common stock in the future, holders of shares of Class A common stock, which are entitled to one vote per share, will experience disproportionate voting power dilution relative to economic dilution, and the holders of Class B common stock may be able to significantly influence the outcome of matters submitted to our stockholders for approval for a longer period of time than would be the case if we issued shares of Class A common stock.
See “Risk Factors—Risks Relating to our Organizational Structure and Governance—Only our employees and directors are entitled to hold shares of Class B common stock (including shares of Class B common stock granted or otherwise issued to our employees and directors in the future), which shares have ten votes per share. This limits or precludes other stockholders’ ability to influence the outcome of matters submitted to stockholders for approval, including the election of directors, the approval of certain employee compensation plans, the adoption of amendments to our organizational documents and the approval of any merger, consolidation, sale of all or substantially all of our assets or other major corporate transaction requiring stockholder approval.”
87
Table of Contents
Under our Charter, we are authorized to issue 800,000,000 shares of Class C common stock, which have no voting rights (except as otherwise expressly provided in the Charter or required by applicable law). Any future issuance of Class C common stock may have the effect of extending voting power in Class B common stock, and may discourage potential acquisitions of our business and could have an adverse effect on the trading price of Class A common stock.
Under our Charter, we are authorized to issue 800,000,000 shares of Class C common stock, which have no voting rights (except as required by law). We may in the future issue shares of Class C common stock for a variety of corporate purposes, including financings, acquisitions and investments. Our authorized but unissued shares of Class C common stock are available for issuance with the approval of our board of directors without stockholder approval, except as may be required by the Listing Rules of the NYSE. Because the Class C common stock carries no voting rights (except as otherwise expressly provided in the Charter or required by applicable law), is not convertible into any other capital stock, and is not listed for trading on an exchange or registered for sale with the SEC, shares of Class C common stock may be less liquid and less attractive to any future recipients of these shares than shares of Class A common stock, although we may seek to list the Class C common stock for trading and register shares of Class C common stock for sale in the future. In addition, because our Class C common stock has no voting rights (except as otherwise expressly provided in the Charter or required by applicable law), if we issue Class C common stock in the future, the holders of Class B common stock may be able to significantly influence the outcome of matters submitted to our stockholders for approval for a longer period of time than would be the case if we issued Class A common stock rather than Class C common stock in such transactions. In addition, if we issue Class C common stock in the future, such issuances would have a dilutive effect on the economic interests of Class A common stock and Class B common stock. Any such issuance of Class C common stock could also cause the trading price of Class A common stock to decline.
We cannot predict the effect the multi-class structure of our common stock may have on the trading price of our Class A common stock.
The holding of low-voting stock, such as Class A common stock, may not be permitted by the investment policies of certain institutional investors or may be less attractive to the portfolio managers of certain institutional investors. In addition, certain index providers have announced restrictions on including companies with multiple-class share structures in certain of their indices. In July 2017, FTSE Russell and S&P Dow Jones announced that they would cease to allow most newly public companies with dual- or multi-class capital structures to be included in their indices. Affected indices include the Russell 2000 and the S&P 500, S&P MidCap 400 and S&P SmallCap 600, which together make up the S&P Composite 1500. Under the announced policies, our multi-class capital structure would make our Class A common stock ineligible for inclusion in certain indices, and as a result, mutual funds, exchange-traded funds and other investment vehicles that attempt to passively track those indices would not invest in our common stock. These policies may depress our valuation compared to those of other similar companies that are included. Because of our multi-class stock structure, our Class A common stock will likely continue to be excluded from certain of these indices, and we cannot assure you that other stock indices will not take similar actions. Given the sustained flow of investment funds into passive strategies that seek to track certain indices, exclusion from stock indices would likely preclude investment by many of these funds in our Class A common stock and could make shares of our Class A common stock less attractive to other investors. As a result, the trading price of shares of our Class A common stock could be adversely affected.
88
Table of Contents
Our focus on the long-term best interests of our company and our consideration of all of our stakeholders, including our stockholders, workforce, customers, suppliers, academic researchers, governments, communities and other stakeholders that we may identify from time to time, may conflict with short-term or medium-term financial interests and business performance, which may adversely impact the value of our common stock.
We believe that focusing on the long-term best interests of our company and our consideration of all of our stakeholders, including our stockholders, workforce, customers, suppliers, academic researchers, governments, communities and other stakeholders we may identify from time to time, is essential to the long-term success of our company and to long-term stockholder value. Therefore, we have made decisions, and may in the future make decisions, that we believe are in the long-term best interests of our company and our stockholders, even if such decisions may negatively impact the short- or medium-term performance of our business, results of operations, and financial condition or the short- or medium-term performance of our Class A common stock. Our commitment to pursuing long-term value for the company and its stockholders, potentially at the expense of short- or medium-term performance, may materially adversely affect the trading price of our Class A common stock, including by making owning our Class A common stock less appealing to investors who are focused on returns over a shorter time horizon. Our decisions and actions in pursuit of long-term success and long-term stockholder value, which may include our multi-class stock structure, making investments in R&D and our employees, and investing in and introducing new products and services, may not result in the long-term benefits that we expect, in which case our business, results of operations and financial condition, as well as the trading price of our Class A common stock, could be materially adversely affected.
89
Table of Contents
Item 1B. Unresolved Staff Comments.
None.
Item 2. Properties.
Ginkgo’s headquarters are located in the Seaport district of Boston, Massachusetts and comprise a set of non-cancellable operating leases within a facility totaling approximately 266,000 square feet of office and laboratory space. These lease agreements expire on dates ranging from 2030 to 2036 and each contain one option to extend the lease for a five-year period at then-market rates. We also lease a total of approximately 100,000 square feet of office and lab space in Cambridge, Massachusetts, Emeryville, California, and Utrecht, Netherlands.
In anticipation of expanding facility needs to support future growth, in April 2021, we entered into a lease, as amended, consisting of approximately 260,000 rentable square feet of new office and laboratory space being developed in Boston, Massachusetts near our headquarters. The lease commencement date is estimated to be June 1, 2024, subject to certain extensions, and expires on the fifteenth anniversary of the lease commencement date. The lease includes one option to extend the lease for ten years at then-market rates as well as an expansion option if the owner constructs an additional building on the property.
We currently lease all of our facilities and do not own any real property. We believe our facilities are adequate and suitable for our current needs and that the new lease described above provides significant expansion space. To support future organic growth or merger and acquisition activity, we may enter into new leases, assume lease obligations or acquire property both domestically and internationally and believe that, if needed, suitable or alternative space will be available.
Item 3. Legal Proceedings.
From time to time, we may in the ordinary course of business be named as a defendant in lawsuits, indemnity claims and other legal proceedings. We do not believe any pending litigation to be material, the outcome of which would, in management’s judgment based on information currently available, have a material adverse effect on our results of operations or financial condition. See Note 14 - Commitments and contingencies to the audited consolidated financial statements included elsewhere in this Annual Report on Form 10-K.
Item 4. Mine Safety Disclosures.
Not Applicable.
90
Table of Contents
PART II
Item 5. Market for Registrant’s Common Equity, Related Stockholder Matters and Issuer Purchases of Equity Securities.
Market Information
Our Class A common stock and Public Warrants began trading on the NYSE under the symbols “DNA” and “DNA.WS,” respectively on September 17, 2021. Prior to that date, there was no public trading market for our Class A common stock and Public Warrants.
Holders of Record
As of December 31, 2021, there were approximately 217 stockholders of record of our Class A common stock, 362 stockholders of record of our Class B common stock and 0 stockholders of record of our Class C common stock, which does not include persons whose stock is held in nominee or “street name” accounts through brokers, banks and intermediaries.
Securities Authorized for Issuance Under Equity Compensation Plans
|
|
Number of securities to be issued upon exercise of outstanding options and vesting of outstanding restricted stock units (#) |
|
|
|
|
Weighted-average exercise price of outstanding options ($) |
|
|
Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in the first column) (#) |
|
|
||||
Equity compensation plans approved by security holders (1) |
|
|
193,550,805 |
|
|
(2 |
) |
|
$ |
0.05 |
|
|
|
200,569,979 |
|
(3)(4) |
Equity compensation plans not approved by security holders |
|
|
— |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
Total |
|
|
193,550,805 |
|
|
|
|
$ |
0.05 |
|
|
|
200,569,979 |
|
|
|
Performance Graph
The following graph compares the cumulative total stockholder return on our common stock relative to the cumulative total returns of the S&P 500 Index and the S&P Biotechnology Select Industry Index between September 17, 2021 (the date our common stock began trading on the NYSE after the Business Combination) through December 31, 2021. All values assume a $100 initial investment at market close on September 17, 2021 and data for the S&P 500 and the S&P Biotechnology Select indices assume reinvestment of all dividends.
91
Table of Contents

Recent Sales of Unregistered Securities
During the year ended December 31, 2021, we did not issue or sell any unregistered securities except as previously disclosed in our Current Report on Form 8-K, as originally filed with the SEC on September 20, 2021.
Issuer Purchases of Equity Securities
The following table sets forth information with respect to shares of our Class A and Class B common stock purchased by us during the periods indicated:
|
|
(a) |
|
|
(b) |
|
|
(c) |
|
|
(d) |
|
||||
Period |
|
Total Number of Shares Purchased (1) |
|
|
Average Price Paid per Share |
|
|
Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs |
|
|
Maximum Number (or Approximate Dollar Value) of Shares that May Yet Be Purchased Under the Plans or Programs |
|
||||
October 1, 2021 - October 31, 2021 |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
November 1, 2021 - November 30, 2021 |
|
|
2,520,826 |
|
|
$ |
13.45 |
|
|
|
— |
|
|
|
— |
|
December 1, 2021 - December 31, 2021 |
|
|
23,924 |
|
|
$ |
11.39 |
|
|
|
— |
|
|
|
— |
|
(1) Consists of shares surrendered to us in connection with our payment of the tax withholding obligations upon vesting of restricted stock awards.
Item 6. [Reserved]
92
Table of Contents
Item 7. Management’s Discussion and Analysis of Financial Condition and Results of Operations.
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with the consolidated financial statements and related notes thereto included elsewhere in this Annual Report on Form 10-K. This discussion contains forward-looking statements that reflect our plans, estimates and beliefs that involve risks and uncertainties. As a result of many factors, such as those set forth under “Risk Factors” and “Cautionary Note Regarding Forward Looking Statements” sections elsewhere in the Form 10-K, our actual results may differ materially from those anticipated in these forward-looking statements as a result of many factors, including those discussed in Item 1A “Risk Factors” of this Annual Report on Form 10-K.
Overview
Our mission is to make biology easier to engineer.
Ginkgo is building the industry-standard horizontal platform for cell programming. We use our platform to program cells on behalf of our customers. These “cell programs” are designed to enable biological production of products as diverse as novel therapeutics, key food ingredients, and chemicals currently derived from petroleum. We have worked on cell programs in end markets as diverse as specialty chemicals, agriculture, food, consumer products, and pharmaceuticals. Biology did not evolve by end market. All of these applications run on cells which have a common code—DNA—and a common programming platform can enable all of them. Because of this shared platform, we are able to drive scale and learning efficiencies while maintaining flexibility and diversity in our program areas. Ultimately, customers come to us because they believe we maximize the probability of successfully developing their products.
The foundation of our platform includes two core assets that execute a wide variety of cell programs for customers according to their specifications: our Foundry and our Codebase.
As the platform scales, we have observed a virtuous cycle between our Foundry, our Codebase, and the value we deliver to customers. We believe this virtuous cycle sustains Ginkgo’s growth and differentiated value proposition.
Put simply: we believe that as the platform improves with scale, it drives more scale, which drives further platform improvements, and so on. We believe this positive feedback loop has the potential to drive compounding value creation in the future as every new program we add contributes to both near-term revenues and has the potential to add significant downstream economics.
Our commercial team is organized to both establish new relationships with potential customers (traditional business development) as well as maintain and expand relationships with our existing customers (which we call “alliance management”). We recognize the cross-functional efforts required to sign any new contract and so our business development
93
Table of Contents
teams do not receive a commission or cash bonus based on target sales, but rather a base salary and annual equity grants driven by overall contribution and performance, in line with how we compensate other employees.
Our business development team has both expertise in relevant industries (Consumer & Technology, Industrial & Environment, Agriculture, Food & Nutrition, Pharma & Biotech and Government & Defense) as well as expertise in our Foundry capabilities and synthetic biology. With this background we are able to identify industry or consumer challenges where biology can serve as a solution. Our categories of customers, independent of industry, include potential customers who have research and development (“R&D”) teams with some synthetic biology capabilities where choosing Ginkgo can bring automation, scale and codebase beyond their own; potential customers who are considering but have not yet built lab-scale capabilities where a partnership with Ginkgo allows them to spend their capital on commercialization efforts; and potential customers who are not yet working in synthetic biology whose industries or products stand to be disrupted by biological solutions. Our business development team, with support from our Codebase and Foundry team members, crafts solutions for each of these types of customers through a strategic discussion of customer needs and fit with Ginkgo capabilities.
To grow existing customers, our alliance management team, through close collaboration on our existing programs, seeks technical and business opportunities for our customers that serve as the basis for consideration of future programs. As our programs demonstrate technical success, our existing customers often bring their next strategic R&D needs to our attention.
Our business model mirrors the structure of our platform and we are compensated in two primary ways. First, we charge usage fees for Foundry services, in much the same way that cloud computing companies charge usage fees for utilization of computing capacity or contract research organizations (CROs) charge for services. Additionally, we negotiate a value share with our customers (typically in the form of royalties, milestones, and/or equity interests) in order to align our economics with the success of the programs enabled by our platform. As we add new programs, our portfolio of programs with this “downstream” value potential grows.
With a mission to make biology easier to engineer, we have always recognized the imperative to invest in biosecurity as a key component of our platform. We care how our platform is used, and biosecurity is a necessary complement to synthetic biology that helps us ensure our cell programming work is conducted and deployed responsibly. The near-term growth of the biosecurity sector is highly dependent on domestic and international government initiatives and investment and Ginkgo has been supporting and engaging with domestic and international organizations and governments to help shape the understanding of a robust biosecurity program.
In the second quarter of 2020, in response to the COVID-19 pandemic, we launched our commercial offering of COVID-19 testing products and services for businesses, academic institutions, and other organizations in which we generate product and service revenue. Beginning in the first quarter of 2021, we launched our pooled testing initiative which focuses on providing end-to-end COVID-19 testing and reporting services to public health authorities. We are currently offering pooled testing and reporting services for K-12 schools across the United States, at airports through our partnership with XpresCheck and the CDC, as well as through other congregate settings such as our partnership with Eurofins. In the future, we believe that testing services may have a value proposition internationally and in other use cases including wastewater monitoring and air monitoring.
Generating Economic Value Through Cell Programs
Our cell programming platform is a key enabling technology and source of intellectual property for our customers’ products. We earn Foundry revenue for our research and development (“R&D”) services as well as through a share of the value of products created using our platform.
We structure Foundry revenue to include some combination of the following:
plus,
94
Table of Contents
or,
Downstream value share arrangements which involve equity interests generally fall into two categories: Platform Ventures and Structured Partnerships.
Platform Ventures
Platform Ventures allow leading multinationals to partner with Ginkgo and financial investors to form new ventures in identified market segments with potential to benefit from synthetic biology. In exchange for an equity position in the venture, we contribute license rights to our proprietary cell programming technology and intellectual property, while our partners contribute relevant industry expertise, other resources and venture funding. We also provide R&D services for which we receive cash payments for our costs incurred, plus a margin. Platform Ventures include:
Joyn Bio, LLC
Founded in 2017, Joyn Bio, LLC (“Joyn”) was formed to focus on engineered microbes for use in agricultural applications. Along with certain of our investors, we formed Cooksonia, LLC (“Cooksonia”) which holds a 50% equity interest in Joyn. Bayer CropScience LP contributed cash of $80 million plus intellectual property and holds the remaining 50% equity interest in Joyn. We provided license rights to our intellectual property and platform at inception in return for our equity interest in Joyn, which was recorded at an initial fair value of $97.9 million. The carrying value of our equity method investment in Joyn was $11.7 million as of December 31, 2021. Ginkgo also entered into a Foundry Services Agreement (“Joyn FSA”) with Joyn under which we provide R&D services. Joyn paid us a non-refundable $20.0 million prepayment for services to be provided under the Joyn FSA and made an additional $15.0 million prepayment for services during the year ended December 31, 2019.
Motif FoodWorks, Inc.
Founded in 2018, Motif FoodWorks, Inc. (“Motif”) was formed to focus on the application of synthetic biology to reduce the reliance on animal products in the food industry. We entered into an intellectual property contribution agreement that granted Motif rights to our intellectual property, subject to mutually agreed upon technical development plans. In return for our contribution of intellectual property and access to our platform, we received shares of common stock in Motif. The initial fair value of our common stock investment in Motif was $65.1 million which has subsequently been reduced to a carrying value of zero as a result of the allocation of losses under our accounting for equity method investments. Motif was capitalized through Series A preferred stock financings that raised approximately $119 million in gross proceeds from an investor group which included certain of our investors, Louis Dreyfus Company and Fonterra Co-operative Group Limited. In June 2021, Motif raised an additional $226 million through a Series B preferred stock financing. Ginkgo also entered into a Technical Development Agreement with Motif under which we provide R&D services in return for cash consideration on a cost-plus fixed margin basis. Motif launched its first product, HEMAMI, in 2021.
Allonnia, LLC
Founded in 2019, Allonnia, LLC (“Allonnia”) was formed to focus on the application of synthetic biology in the waste bioremediation and biorecovery industries. We entered into an intellectual property contribution agreement that granted Allonnia rights to our intellectual property, subject to mutually agreed upon technical development plans. In return for our contribution of intellectual property and access to our platform, we received common units in Allonnia with a right to additional units subject to additional closings of Allonnia’s Series A preferred units. The initial fair value of our common units received in Allonnia was $24.5 million, subsequently increased by $12.7 million in 2021, all of which has been reduced
95
Table of Contents
to a carrying value of zero as a result of the allocation of losses under our accounting for equity method investments. Allonnia was capitalized through Series A preferred unit financings that raised approximately $52 million in gross proceeds from an investor group which included certain of our investors and Battelle Memorial Institute. Ginkgo also entered into a Technical Development Agreement with Allonnia under which we provide R&D services in return for cash consideration on a cost-plus fixed margin basis.
Arcaea, LLC (FKA Kalo Ingredients, LLC)
Founded in 2021, Arcaea, LLC (“Arcaea”) was formed to focus on the application of synthetic biology in the beauty and personal care products industry. In March 2021, we entered into an intellectual property contribution agreement that granted Arcaea rights to our intellectual property, subject to mutually agreed upon technical development plans. In return for our contribution of intellectual property and access to our platform, we received common units in Arcaea with a right to additional units subject to additional closings of Arcaea’s Series A preferred units. The initial fair value of our common units received in Arcaea was $11.9 million which has subsequently been reduced to a carrying value of zero as a result of the allocation of losses under our accounting for equity method investments. Arcaea was capitalized through a Series A preferred unit financing that raised approximately $77 million in gross proceeds from an investor group which included certain of our investors, CHANEL and Givaudan. Upon the closing of the Series A preferred unit financing in July 2021, we received an additional 5,229,900 common units in Arcaea. The fair value of our Arcaea common units received in July 2021 of $35.5 million has subsequently been reduced to a carrying value of zero as a result of the allocation of losses under our accounting for equity method investments. Ginkgo also entered into a Technical Development Agreement with Arcaea under which we provide R&D services in return for cash consideration on a cost-plus fixed margin basis.
Ayana Bio, LLC
Founded in September 2021, Ayana Bio, LLC (“Ayana”) was formed to identify and design new bioactive compounds for use as complementary medicine to support human health and wellness. Ayana was capitalized through a Series A funding that raised $30 million in gross proceeds from an investor group comprising certain of our investors. We hold an interest in 9,000,000 common units (representing 100% of common units at inception) of Ayana and have also provided Ayana with certain licenses to our intellectual property for use in the development or production of products that we have agreed to research and develop under technical development plans. We concluded that we hold a variable interest in and are the primary beneficiary of Ayana, and as a result, we have consolidated the financial statements of Ayana into our consolidated financial statements.
Verb Biotics, LLC
Founded in September 2021, Verb Biotics, LLC (“Verb”) was formed to identify and design new strains of probiotic bacteria with advanced properties for human nutrition, health, and wellness. Verb was capitalized through a Series A funding that raised $30 million in gross proceeds from an investor group comprising certain of our investors. We hold an interest in 9,000,000 common units (representing 100% of common units at inception) of Verb and have also provided Verb with certain licenses to our intellectual property for use in the development or production of products that we have agreed to research and develop under technical development plans. We concluded that we hold a variable interest in and are the primary beneficiary of Verb, and as a result, we have consolidated the financial statements of Verb into our consolidated financial statements.
Structured Partnerships
Structured Partnerships allow Ginkgo to: i) partner with early stage synthetic biology product companies to adopt our Foundry as their cell programming R&D platform, in which we offer flexible commercial terms on the Foundry usage fees including the ability to pay a portion or all of such upfront fees in the form of equity, in addition to downstream value share consideration (“Startup Structured Partnership”); and ii) partner with existing entities with complementary assets for high potential synthetic biology applications in a large-scale, multi-program collaboration (“Legacy Structured Partnership”). In the second half of 2021, we entered into our initial five Startup Structured Partnerships in which a component of the Foundry usage fees included upfront payments in the form of equity consideration. In aggregate, the upfront consideration received in
96
Table of Contents
the form of equity was $16.5 million, which will be recognized as revenue over our period of performance. Prior to 2021, we had entered into two Legacy Structured Partnerships:
Genomatica, Inc.
Genomatica, Inc. (“Genomatica”) is a biotechnology company specializing in the development and manufacturing of intermediate and specialty chemicals from both sugar and alternative feedstocks. In 2016 and 2018, we entered into separate preferred stock purchase agreements in which we offered cash and R&D services to Genomatica in exchange for its preferred shares. The initial cost of the investment in Genomatica’s preferred stock was $55.0 million, which is the carrying value of the investment at December 31, 2021 as we account for the investment at historical cost.
Synlogic, Inc.
Synlogic, Inc. (“Synlogic”) is a publicly traded clinical-stage biopharmaceutical company focused on advancing drug discovery and development for synthetic biology-derived medicines. In 2019, we entered into several agreements with Synlogic whereby we purchased Synlogic common stock and warrants to purchase Synlogic common stock and agreed to provide R&D services to Synlogic. At inception, the fair value of Synlogic common stock and warrants was recorded at $35.8 million and $14.4 million, respectively. As of December 31, 2021, the fair value of Synlogic common stock and warrants was $15.3 million and $6.2 million, respectively.
See Notes 10 and 20 of our audited consolidated financial statements included elsewhere in this Annual Report on Form 10-K for further details of our investments in and the material terms of our agreements with our Platform Ventures and Structured Partnerships.
Key Business Metrics
A cell program (or “program”) is the work we do for our customers to enable their product(s) of interest. Programs are defined by a technical development plan. We generally exclude proof-of-concept projects and other exploratory work undertaken on a customer’s behalf from the program count. In the near-term, programs deliver multi-year revenue from platform usage fees. Over the long-term, program growth drives a physical infrastructure scale economic through our Foundry, a data and learning scale economic through our Codebase and accumulation of downstream value share. Our key business metrics comprise New Programs, Current Active Programs, and Cumulative Programs.
|
|
Years Ended December 31, |
|
|||
|
|
2021 |
|
2020 |
|
|
New Programs |
|
31 |
|
|
18 |
|
Current Active Programs |
|
71 |
|
|
49 |
|
Cumulative Programs |
|
105 |
|
|
74 |
|
New Programs
New Programs represent the number of unique programs commenced within the reporting period. As new programs have multi-year durations, we view this metric as an indication of future Foundry revenue growth.
Current Active Programs
Current Active Programs represent the number of unique programs for which we performed R&D services in the reporting period. We view this metric as an indication of current period and future Foundry revenue.
Cumulative Programs
Cumulative Programs represent the cumulative number of unique programs Ginkgo has commenced. We view this metric as an indication of our competitive advantage and as a leading indicator of the mid- to long-term potential economic value derived from downstream value share arrangements. The cumulative number of programs also contributes to Codebase, which accumulates with each additional program we conduct over time and drives better experimental direction and improves the odds of technical success in current and future programs.
97
Table of Contents
We believe the preceding metrics are important to understand our current business. These metrics may change or be substituted for additional or different metrics as our business develops. For example, as our program mix changes, our data gathering abilities expand or our understanding of key business drivers develops, we anticipate updating these metrics or their definitions to reflect such changes.
Business Combination
We entered into the Merger Agreement with Soaring Eagle Acquisition Corp. (“SRNG”) on May 11, 2021. On September 14, 2021, the SRNG shareholders approved and adopted the Merger Agreement and the other proposals described in SRNG’s definitive proxy statement/prospectus included in SRNG’s registration statement on Form S-4 (File No. 333-256121), which was declared effective by the SEC on August 11, 2021. Upon the consummation of the Business Combination on September 16, 2021, SEAC Merger Sub Inc., a wholly owned subsidiary of SRNG (“Merger Sub”), merged with and into Ginkgo, the separate corporate existence of Merger Sub ceased, and Ginkgo survived the merger as a wholly owned subsidiary of SRNG, which was renamed “Ginkgo Bioworks Holdings, Inc.”
The Business Combination was accounted for as a reverse recapitalization in accordance with accounting principles generally accepted in the United States of America (“U.S. GAAP”). Under the guidance in ASC 805, Business Combinations (“ASC 805”), SRNG was treated as the “acquired” company for accounting and financial reporting purposes. We were deemed the accounting predecessor of the combined business, and as the parent company of the combined business, are the successor SEC registrant, meaning that our financial statements for previous periods will be disclosed in future periodic reports filed with the SEC. The most significant change in our reported financial position and results of operations as a result of the Business Combination was a net increase in cash (as compared to our audited consolidated balance sheet as of December 31, 2020) of $1,509.6 million, including $760.0 million in proceeds from the PIPE Investment (as defined below) that was consummated substantially simultaneously with the closing of the Business Combination. The transaction costs for the Business Combination totaled $108.1 million.
As the successor to an SEC-registered and publicly-listed company, we will need to hire additional personnel and implement procedures and processes to address public company regulatory requirements and customary practices. We expect to incur additional expenses as a public company for, among other things, directors’ and officers’ liability insurance, director fees, and additional internal and external accounting, legal and administrative resources.
Modification of Equity Awards in Connection with Business Combination
Prior to the Business Combination, our restricted stock units (“RSUs”) were granted with both a service-based vesting condition and a performance-based vesting condition. We have historically not recognized any stock-based compensation expense associated with these awards as the achievement of the performance condition required a change in control or an initial public offering (both as defined in the underlying award agreement) that was not deemed probable of occurring. The Business Combination did not meet the performance condition required for vesting of our RSUs.
On November 17, 2021 our board of directors modified the vesting terms of RSUs to allow 10% of the RSUs that met the service condition as of the closing of the Business Combination (the “10% RSUs”) to vest with respect to the performance condition, effective as of November 19, 2021, the date on which the Form S-8 registration statement covering such shares became effective. The remaining RSUs vested in full with respect to the performance condition on or before March 15, 2022. The change to the vesting terms was accounted for as a modification and resulted in approximately $1,492.2 million of incremental stock-based compensation expense in the fourth quarter of 2021. The 10% RSUs representing approximately 5.7 million shares were settled in cash for a total cash payment of $76.5 million equal to the fair value of the stock on the Form S-8 effective date. Stock-based compensation expense also increased by $173.5 million related to RSU earnout shares which were also subject to the same performance condition as the underlying RSUs. Additionally, the earnout shares vest upon achieving certain target stock price thresholds. The first target stock price of $12.50 per share was achieved on November 15, 2021.
Acquisition of Dutch DNA Biotech B.V.
On July 1, 2021, we acquired 100% of the equity of Dutch DNA Biotech B.V. (“Dutch DNA”), a company based in the Netherlands with a proprietary platform technology focused on the development of fungal strains and fermentation processes for the production of proteins and organic acids. Dutch DNA's significant expertise and fungal strain assets for the large-scale production of proteins is expected to add a valuable set of tools to Ginkgo's Codebase and broader platform for cell programming. The total acquisition date fair value of the consideration transferred for Dutch DNA was $35.3 million. Dutch
98
Table of Contents
DNA's results of operations have been included in our condensed consolidated statements of operations and comprehensive loss since the date of acquisition.
Components of Results of Operations
Revenue
Foundry Revenue
We generate Foundry revenue through the execution of license and collaboration agreements whereby customers obtain license rights to our proprietary technology and intellectual property for use in the development and commercialization of engineered organisms and derived products. Under these agreements, we typically provide R&D services for cell programming with the goal of producing an engineered cell that meets a mutually agreed specification. Our customers obtain license rights to the output of our services, which are primarily the optimized strains or cell lines, in order to manufacture and commercialize products derived from that licensed strain or cell line. Generally, the terms of these agreements provide that we receive some combination of: (1) Foundry usage fees in the form of (i) upfront payments upon consummation of the agreement or other fixed payments, (ii) reimbursement for costs incurred for R&D services and (iii) milestone payments upon the achievement of specified technical criteria, plus (2) downstream value share payments in the form of (i) milestone payments upon the achievement of specified commercial criteria, (ii) royalties on sales of products from or comprising engineered organisms arising from the collaboration or licensing agreement and (iii) royalties related to cost of goods sold reductions realized by our customers. For the years ended December 31, 2021 and 2020, royalties did not comprise a material amount of our revenue.
Foundry revenue includes transactions with Platform Ventures as well as Structured Partnerships where, as part of these transactions, we received an equity interest in such entities. Specifically related to the Platform Ventures, in these transactions, we received upfront non-cash consideration in the form of common equity interests in these entities, while the Platform Ventures each received cash equity investments from strategic partners and financial investors. We view the upfront non-cash consideration as prepayments for licenses which will be granted in the future as we complete mutually agreed upon technical development plans. In these instances, we also receive cash payments for our costs incurred for the R&D services performed by us, plus a margin. We are not compensated through additional milestone or royalty payments under these arrangements. Our transactions with Genomatica and Synlogic included the purchase of equity securities and the provision of R&D services. As we perform R&D services under the mutually agreed upon development plans, we recognize a reduction in the prefunded obligation based on costs incurred, plus margin basis. Because of our equity holdings in these entities, each is considered as a related party. Revenue from Startup Structured Partnerships was not material in 2021. These arrangements are further described in Notes 10, 11, 20 and 24 of our audited consolidated financial statements included elsewhere in this Annual Report on Form 10-K.
Downstream value share in the form of equity interest appreciation is not recognized as revenue but is expected to contribute to future cash flows upon liquidation, the amount and timing of which is inherently unpredictable. Equity investees are accounted for as equity method investments, cost method investments or carried at fair value.
Biosecurity Revenue
In the second quarter of 2020, in response to the COVID-19 pandemic, we launched our commercial offering of COVID-19 testing products and services for businesses, academic institutions, and other organizations in which we generate product and service revenue. We generate product revenue through the sale of lateral flow assay (“LFA”) diagnostic test kits, polymerase chain reaction (“PCR”) sample collection kits and pooled test kits, all of which we sell to our customers on a standalone basis. We generate service revenue primarily through the sale of our end-to-end COVID-19 testing services which consist of multiple promised goods and services including sample collection kits, physician authorizations, onsite test administration, outsourced laboratory PCR analysis, and access to results reported through a web-based portal.
Generally, the terms of these agreements provide that we are entitled to compensation: (i) upon delivery of diagnostic test kits when no service is provided and (ii) when services are included, upon the reporting of results to the customer.
Beginning in the first quarter of 2021, we launched our pooled testing initiative which focuses on providing end-to-end COVID-19 testing and reporting services to public health authorities. We are currently offering pooled testing and reporting services for K-12 schools across the United States, at airports through our partnership with XpresCheck and the CDC, as well as through other congregate settings such as our partnership with Eurofins and Quest Laboratories in the state of Texas. In the future, we believe that testing services may have a value proposition internationally and in other use cases including
99
Table of Contents
wastewater monitoring and air monitoring. The amount and components of Biosecurity revenue are dependent on the demand for COVID-19 testing products and services which is uncertain in 2022 and beyond.
Costs and Operating Expenses
Cost of Biosecurity Product Revenue
Cost of Biosecurity product revenue consists of costs associated with the sale of diagnostic and sample collection test kits which includes costs incurred to purchase test kits from third parties.
Cost of Biosecurity Service Revenue
Cost of Biosecurity service revenue consists of costs associated with the provision of our end-to-end COVID-19 testing services, which includes costs incurred to provide sample collection kits, physician authorizations, onsite test administration, outsourced laboratory PCR analysis, access to results reported through our proprietary web-based portal and reporting of results to public health authorities.
Research and Development Expenses
The nature of our business, and primary focus of our activities, generates a significant amount of R&D expenses. R&D expenses represent costs incurred by us for the following:
The activities above incur the following expenses:
We expense R&D costs as incurred. As we grow our active programs and customer base and invest in our Foundry and Codebase, we anticipate that our R&D expenses will continue to increase. The nature, timing, and estimated costs required to support our growth will be dependent on advances in technology, our ability to attract new customers and the rate of market penetration within our existing customer industries.
In the fourth quarter of 2021, R&D expenses included a significant adjustment for stock-based compensation expense as a result of the modification of vesting terms of RSUs and the vesting of certain earnout shares (as further described above in “Modification of Equity Awards in Connection with Business Combination”).
General and Administrative Expenses
General and administrative (“G&A”) expenses consist primarily of costs for personnel in executive, business development, finance, human resources, legal and other corporate administrative functions. G&A expenses also include legal fees incurred relating to corporate, intellectual property and patent matters, professional fees incurred for accounting, auditing, tax and administrative consulting services, insurance costs, and facility-related costs not otherwise included in R&D expenses.
We expect our G&A expenses will continue to increase as we pursue organic and inorganic growth initiatives. The increases will likely relate to additional personnel, system costs and increased costs related to business development, finance and legal matters, along with increased expenses related to operating as a publicly traded company, such as fees related to audit, legal and tax services, regulatory compliance programs and investor relations.
In the fourth quarter of 2021, G&A expenses included a significant adjustment for stock-based compensation expense as a result of the modification of vesting terms of RSUs and the vesting of certain earnout shares (as further described above in “Modification of Equity Awards in Connection with Business Combination”).
100
Table of Contents
Interest Income
Interest income consists primarily of interest earned on our cash and cash equivalents.
Interest Expense
Interest expense consists primarily of interest related to our lease financing obligation.
Loss on Equity Method Investments
Loss on equity method investments includes our share of losses from certain of our equity method investments under the Hypothetical Liquidation at Book Value (“HLBV”) method.
Loss on Investments
Loss on investments includes the change in fair value of Synlogic common stock, warrants to purchase Synlogic common stock and change in fair value of Cronos Group Inc. ("Cronos") common stock.
Change in Fair Value of Warrant Liabilities
Change in fair value of warrant liabilities includes the change in fair value of private placement warrants (“Private Placement Warrants”) and publicly-traded warrants (“Public Warrants”), which are classified as liabilities and were assumed as part of the Business Combination.
Gain on Settlement of Partnership Agreement
Gain on settlement of partnership agreement includes payments made by Amyris, Inc. (“Amyris”) under a settlement agreement.
Other (Expense) Income, Net
Other (expense) income, net primarily consists of change in fair value of our convertible notes with Access Bio, Inc. (“Access Bio”) and promissory note with Glycosyn which we have elected to account for under the fair value option and income generated from achieving milestones under our agreement with the National Institutes of Health (“NIH”).
Provision for Income Taxes
Income taxes are recorded in accordance with ASC 740, Income Taxes (“ASC 740”), which provides for deferred taxes using an asset and liability approach. We recognize deferred tax assets and liabilities for the expected future tax consequences of events that have been included in our audited consolidated financial statements or tax returns. Deferred tax assets and liabilities are determined based on the difference between the financial statement and tax bases of assets and liabilities using enacted tax rates in effect for the year in which the differences are expected to reverse. A valuation allowance against deferred tax assets is recorded if, based on the weight of the available evidence, it is more likely than not that some or all the deferred tax assets will not be realized. For all periods presented, we have recorded a valuation allowance against the deferred tax assets that are not expected to be realized.
We account for uncertain tax positions using a more-likely-than-not threshold for recognizing and resolving uncertain tax positions. The evaluation of uncertain tax positions is based on factors, including, but not limited to, changes in the law, the measurement of tax positions taken or expected to be taken in tax returns, the effective settlement of matters subject to audit, new audit activity and changes in facts or circumstances related to a tax position.
As of December 31, 2021, we had federal net operating loss carryforwards of approximately $665.2 million, of which $139.2 million begin to expire in 2029. We have approximately $526.0 million of federal net operating losses as of December 31, 2021 that can be carried forward indefinitely. As of December 31, 2021, we had state net operating loss carryforwards of approximately $529.3 million, of which $485.9 million begin to expire in 2029. We have approximately $43.4 million of state net operating losses as of December 31, 2021 that can be carried forward indefinitely.
101
Table of Contents
Income taxes are determined at the applicable tax rates adjusted for non-deductible expenses, R&D tax credits and other permanent differences. Our income tax provision may be significantly affected by changes to our estimates.
Results of Operations
Comparison of the Years Ended December 31, 2021 and 2020
The following table summarizes our consolidated statements of operations for each period presented:
|
|
Year Ended December 31, |
|
|
|
|
||||||||
(in thousands) |
|
2021 |
|
|
|
2020 |
|
|
Change |
|
||||
Foundry revenue (related party revenue of $47,161 and $42,535 for the years ended 2021 and 2020, respectively) |
|
$ |
112,989 |
|
|
|
|
$ |
59,221 |
|
|
$ |
53,768 |
|
Biosecurity revenue: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Product |
|
|
23,040 |
|
|
|
|
|
8,707 |
|
|
|
14,333 |
|
Service |
|
|
177,808 |
|
|
|
|
|
8,729 |
|
|
|
169,079 |
|
Total revenue |
|
|
313,837 |
|
|
|
|
|
76,657 |
|
|
|
237,180 |
|
Costs and operating expenses: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Cost of Biosecurity product revenue |
|
|
20,017 |
|
|
|
|
|
6,705 |
|
|
|
13,312 |
|
Cost of Biosecurity service revenue |
|
|
109,673 |
|
|
|
|
|
8,906 |
|
|
|
100,767 |
|
Research and development (1) |
|
|
1,149,662 |
|
|
|
|
|
159,767 |
|
|
|
989,895 |
|
General and administrative (1) |
|
|
862,952 |
|
|
|
|
|
38,306 |
|
|
|
824,646 |
|
Total operating expenses |
|
|
2,142,304 |
|
|
|
|
|
213,684 |
|
|
|
1,928,620 |
|
Loss from operations |
|
|
(1,828,467 |
) |
|
|
|
|
(137,027 |
) |
|
|
(1,691,440 |
) |
Other (expense) income: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Interest income |
|
|
837 |
|
|
|
|
|
2,582 |
|
|
|
(1,745 |
) |
Interest expense |
|
|
(2,373 |
) |
|
|
|
|
(2,385 |
) |
|
|
12 |
|
Loss on equity method investments |
|
|
(77,284 |
) |
|
|
|
|
(396 |
) |
|
|
(76,888 |
) |
Loss on investments |
|
|
(11,543 |
) |
|
|
|
|
(3,733 |
) |
|
|
(7,810 |
) |
Change in fair value of warrant liabilities |
|
|
58,615 |
|
|
|
|
|
— |
|
|
|
58,615 |
|
Gain on settlement of partnership agreement |
|
|
23,826 |
|
|
|
|
|
8,286 |
|
|
|
15,540 |
|
Other (expense) income, net |
|
|
(1,733 |
) |
|
|
|
|
7,839 |
|
|
|
(9,572 |
) |
Total other (expense) income, net |
|
|
(9,655 |
) |
|
|
|
|
12,193 |
|
|
|
(21,848 |
) |
Loss before income taxes |
|
|
(1,838,122 |
) |
|
|
|
|
(124,834 |
) |
|
|
(1,713,288 |
) |
Income tax (benefit) provision |
|
|
(1,480 |
) |
|
|
|
|
1,889 |
|
|
|
(3,369 |
) |
Net loss |
|
|
(1,836,642 |
) |
|
|
|
|
(126,723 |
) |
|
|
(1,709,919 |
) |
Net loss attributable to non-controlling interest |
|
|
(6,595 |
) |
|
|
|
|
(114 |
) |
|
|
(6,481 |
) |
Net loss attributable to Ginkgo Bioworks Holdings, Inc. stockholders |
|
$ |
(1,830,047 |
) |
|
|
|
$ |
(126,609 |
) |
|
$ |
(1,703,438 |
) |
(1) In the fourth quarter of 2021, R&D and G&A expenses included a significant adjustment for stock-based compensation expense as a result of the modification of the vesting terms of RSUs and all related earnout shares (as further described above in “Modification of Equity Awards in Connection with Business Combination”). Total stock-based compensation expense inclusive of employer payroll taxes was allocated as follows (in thousands):
|
|
Year Ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Research and development |
|
$ |
930,360 |
|
|
$ |
79 |
|
|
$ |
64 |
|
General and administrative |
|
|
757,247 |
|
|
|
397 |
|
|
|
707 |
|
Total |
|
$ |
1,687,607 |
|
|
$ |
476 |
|
|
$ |
771 |
|
Foundry Revenue
Foundry revenue was $113.0 million for the year ended December 31, 2021 and $59.2 million for the year ended December 31, 2020. The increase of $53.8 million in Foundry revenue was primarily attributable to the progress of programs with existing and new customers, including downstream value share revenue related to the achievement of commercial milestones.
102
Table of Contents
The total number of Current Active Programs increased from 49 in 2020 to 71 in 2021. In 2021, 31 New Programs were commenced. Cumulative Programs increased from 74 in 2020 to 105 in 2021. The number of customers increased from 22 in 2020 to 33 in 2021.
While the majority of Foundry revenue today is made up of Foundry usage fees, as we increase Cumulative Programs and to the extent our customers successfully commercialize products built on our platform, downstream value share is expected to comprise a larger proportion of Foundry revenue. Downstream value share in the form of equity interest appreciation is not recognized as revenue but is expected to contribute to future cash flows upon liquidation, the amount and timing of which is inherently unpredictable.
Biosecurity Revenue
Biosecurity revenue was $200.8 million for the year ended December 31, 2021 and $17.4 million for the year ended December 31, 2020. The increase of $183.4 million in Biosecurity revenue was comprised of an increase in product revenue of $14.3 million and an increase in service revenue of $169.1 million.
The amount and components of Biosecurity revenue are dependent on the demand for COVID-19 testing products and services which is uncertain in 2022 and beyond.
Cost of Biosecurity Product and Service Revenue
Cost of Biosecurity product and service revenue was $129.7 million for the year ended December 31, 2021 and $15.6 million for the year ended December 31, 2020. The increase of $114.1 million was driven by the increase in the demand for our COVID-19 testing products and services.
Research and Development Expenses
Research and development expenses were $1,149.7 million for the year ended December 31, 2021 and $159.8 million for the year ended December 31, 2020. The increase of $989.9 million was primarily attributable to increases in stock-based compensation expense of $930.3 million (inclusive of employer payroll taxes) as a result of the modification of the vesting terms of RSUs and certain earnout shares in the fourth quarter of 2021 (as further described above in “Modification of Equity Awards in Connection with Business Combination”), personnel-related compensation and benefits expense of $22.6 million, rent and facilities expense of $11.8 million, and professional fees of $6.0 million. The remaining increase was attributable to depreciation and amortization expense of $14.9 million and other direct and allocated overhead expenses. Increases in research and development expenses not attributable to stock-based compensation expense supported the Foundry operations, enhancements of Foundry and Codebase, and development of our Biosecurity offering.
General and Administrative Expenses
General and administrative expenses were $863.0 million for the year ended December 31, 2021 and $38.3 million for the year ended December 31, 2020. The increase of $824.6 million was primarily attributable to increases in stock-based compensation expense of $756.9 million (inclusive of employer payroll taxes) as a result of the modification of the vesting terms of RSUs and certain earnout shares in the fourth quarter of 2021 (as further described above in “Modification of Equity Awards in Connection with Business Combination”), personnel-related compensation and benefits expense of $23.5 million, professional fees of $19.9 million, software and technology expense of $7.2 million, rent and facilities expense of $4.2 million, insurance expense of $3.5 million and marketing expense of $2.7 million. The remaining increase was attributable to business taxes and other direct and allocated overhead expenses. Increases in general and administrative expenses not attributable to stock-based compensation expense supported the growth of Foundry and Biosecurity revenue and activities related to public company readiness.
Interest Income
Interest income was $0.8 million and $2.6 million for the years ended December 31, 2021 and 2020, respectively. The decrease of $1.8 million was primarily due to a decrease in interest rates on our cash held in money market accounts.
103
Table of Contents
Interest Expense
Interest expense was $2.4 million for each of the years ended December 31, 2021 and 2020 and was related to our lease financing obligation.
Loss on Equity Method Investments
Loss on equity method investments was $77.3 million and $0.4 million for the years ended December 31, 2021 and 2020, respectively. The increase of $76.9 million was attributable to our equity method investments in Joyn, Allonnia and Arcaea. The fair value of the initial equity we received in Arcaea of $47.4 million during the year ended December 31, 2021 was reduced to zero during the period as a result of the application of the HLBV method. The fair value of the additional equity we received in Allonnia of $12.7 million during the year ended December 31, 2021 was also reduced to zero during the period as a result of the application of the HLBV method. Under the HLBV method, we absorb losses as a common unit holder prior to preferred unit holders due to a substantive profit-sharing agreement where the preferred unit holders receive preferential distribution rights. Because we have no commitment to fund the losses of Arcaea or Allonnia, no further losses on these equity method investments were recognized during the year ended December 31, 2021. The loss on our equity method investment in Joyn increased from $0.4 million to $17.2 million during the years ended December 31, 2020 and 2021, respectively, representing our share of the investee’s losses under the HLBV method.
Loss on Investments
Loss on investments was $11.5 million and $3.7 million for the years ended December 31, 2021 and 2020, respectively. The change of $7.8 million was attributable to fluctuations in the stock price of our shares of Synlogic and Cronos common stock and warrants to purchase Synlogic common stock.
Change in Fair Value of Warrant Liabilities
The change in fair value of warrant liabilities of $58.6 million for the year ended December 31, 2021 was due to a decrease in the estimated fair value of the Private Placement Warrants and a decrease in the quoted price of the Public Warrants.
Gain on Settlement of Partnership Agreement
Gain on settlement of partnership agreement was $23.8 million and $8.3 million for the years ended December 31, 2021 and 2020, respectively. The increase of $15.5 million was due to Amyris making a prepayment in 2021 in full settlement of all amounts due under the partnership agreement.
Other (Expense) Income, Net
Other (expense) income, net was $(1.7) million and $7.8 million for the years ended December 31, 2021 and 2020, respectively. The decrease of $9.5 million was primarily attributable to a $6.6 million decrease in payments received from the NIH, a $4.5 million change in fair value of the Access Bio convertible notes and Glycosyn promissory note, partially offset by net increase in other miscellaneous income.
Non-GAAP Information
In addition to our results determined in accordance with U.S. GAAP, we believe that EBITDA and Adjusted EBITDA, each non-GAAP financial measures, are useful in evaluating our operational performance. We use this non-GAAP financial information to evaluate our ongoing operations and for internal planning and forecasting purposes. We believe that this non-GAAP financial information, when taken collectively, may be helpful to investors in assessing our operating performance.
We define EBITDA as net loss attributable to Ginkgo Bioworks Holdings, Inc. stockholders before the impact of interest income, interest expense, provision for income taxes and depreciation and amortization.
We define Adjusted EBITDA as EBITDA adjusted for stock-based compensation expense, gain or loss on equity method investments, gain or loss on investments, change in fair value of warrant liabilities and other income and expenses. We believe that the use of EBITDA and Adjusted EBITDA provides an additional tool for investors to use in evaluating ongoing operating results and trends because it eliminates the effect of financing activities, investing activities, and certain non-cash charges and other items. Adjusted EBITDA includes non-cash adjustments such as stock-based compensation, gain or loss on equity method investments, and gain or loss on changes in fair value of our investments, warrant liabilities and loans
104
Table of Contents
receivable. Adjusted EBITDA also considers cash components which are not part of our ongoing operating results, such as gains related to settlement payments from Amyris and certain funding received from the NIH to invest in our Biosecurity development related to COVID-19. We believe Adjusted EBITDA, although not a replacement for financial performance measures reported under U.S. GAAP, provides investors with a means to compare our financial measures with those of comparable companies, which may present similar non-GAAP financial measures to investors. However, you should be aware that when evaluating EBITDA and Adjusted EBITDA we may generate future income or incur future expenses similar to those excluded when calculating these measures. In addition, our presentation of these measures should not be construed as an inference that our future results will be unaffected by future income or future expenses similar to those excluded when calculating these measures. Our computation of these measures, especially Adjusted EBITDA, may not be comparable to other similarly titled measures computed by other companies because not all companies calculate these measures in the same way.
Because of these limitations, EBITDA and Adjusted EBITDA should not be considered in isolation or as a substitute for performance measures calculated in accordance with U.S. GAAP. We compensate for these limitations by primarily relying on our U.S. GAAP results supplemented by EBITDA and Adjusted EBITDA. You should review the reconciliation of net loss attributable to Ginkgo Bioworks Holdings, Inc. stockholders to EBITDA and Adjusted EBITDA below and not rely on any single financial measure to evaluate our business.
The following table reconciles net loss attributable to Ginkgo Bioworks Holdings, Inc. stockholders to EBITDA and Adjusted EBITDA for the years ended December 31, 2021 and 2020, respectively:
|
|
Year Ended December 31, |
|
|||||
(in thousands) |
|
2021 |
|
|
2020 |
|
||
Net loss attributable to Ginkgo Bioworks Holdings, Inc. stockholders |
|
$ |
(1,830,047 |
) |
|
$ |
(126,609 |
) |
Interest income |
|
|
(837 |
) |
|
|
(2,582 |
) |
Interest expense |
|
|
2,373 |
|
|
|
2,385 |
|
Income tax (benefit) provision |
|
|
(1,480 |
) |
|
|
1,889 |
|
Depreciation and amortization |
|
|
29,076 |
|
|
|
13,864 |
|
EBITDA |
|
|
(1,800,915 |
) |
|
|
(111,053 |
) |
Stock-based compensation (1) |
|
|
1,687,607 |
|
|
|
476 |
|
Loss on equity method investments (2) |
|
|
74,445 |
|
|
|
282 |
|
Loss on investments (3) |
|
|
11,543 |
|
|
|
3,733 |
|
Change in fair value of warrant liabilities |
|
|
(58,615 |
) |
|
|
— |
|
Gain on settlement of partnership agreement |
|
|
(23,826 |
) |
|
|
(8,286 |
) |
Other (4) |
|
|
3,712 |
|
|
|
(6,574 |
) |
Adjusted EBITDA |
|
$ |
(106,049 |
) |
|
$ |
(121,422 |
) |
Liquidity and Capital Resources
Sources of Liquidity
Prior to the Business Combination, our sources of liquidity have been predominantly from proceeds from equity offerings, convertible notes offerings, payments received for R&D services under license and collaboration arrangements including those received on an upfront basis and upon accomplishment of milestones, payments received from Biosecurity product sales and services, and government grants. Upon the closing of the Business Combination in September 2021, we received net proceeds totaling approximately $1,509.6 million, inclusive of $760.0 million from the PIPE Investment. As of December
105
Table of Contents
31, 2021, we had cash and cash equivalents of $1,550.0 million which we believe will be sufficient to enable us to fund our projected operations through at least the next 12 months from the date of filing of this Annual Report on Form 10-K.
Material Cash Requirements
We anticipate that our expenditures will increase significantly in connection with our ongoing activities, as we:
Leases
We have various noncancelable operating leases for office and laboratory space that begin to expire on dates ranging from 2030 through 2036. As of December 31, 2021, we have minimum rental commitments under noncancelable operating leases of $20.6 million in 2022 and $664.8 million thereafter. See Note 14 to the consolidated financial statements in Part II, Item 8 of this Annual Report on Form 10-K for more information.
Purchase Obligations
We have a noncancelable purchase obligation with a supplier under which we are obligated to pay at least $109.0 million, and up to $150.0 million, over a seven year term ending in 2026 for minimum purchases of certain equipment, associated consumables and other goods and services. The purchase obligation includes variable license fees and milestone payments of up to $11.5 million upon achievement of development and regulatory milestones. As of December 31, 2021, we had approximately $86.5 million remaining under the purchase obligation, with approximately $13 million payable within 12 months. Additionally, we have minimum purchase volume commitments with certain suppliers. See Note 14 to the consolidated financial statements in Part II, Item 8 of this Annual Report on Form 10-K for more information.
Capital Expenditures
We anticipate our cumulative spending on capital expenditures to be in the range of $115 million over the next twelve months, subject to management’s ongoing reassessment, to support our commercial plan as we strategically invest in capacity and technology to deliver new cell programs.
Cash Flows
The following table provides information regarding our cash flows for each period presented:
|
|
Year Ended December 31, |
|
|||||
(in thousands) |
|
2021 |
|
|
2020 |
|
||
Net cash provided by (used in): |
|
|
|
|
|
|
||
Operating activities |
|
$ |
(253,818 |
) |
|
$ |
(135,830 |
) |
Investing activities |
|
|
(73,257 |
) |
|
|
(67,121 |
) |
Financing activities |
|
|
1,534,145 |
|
|
|
90,318 |
|
Effect of exchange rate changes |
|
|
(19 |
) |
|
|
— |
|
Net increase (decrease) in cash, cash equivalents and restricted cash |
|
$ |
1,207,051 |
|
|
$ |
(112,633 |
) |
Operating Activities
Net cash used in operating activities for the year ended December 31, 2021 consisted of net loss of $1,836.6 million, adjusted for net change in operating assets and liabilities of $61.5 million and non-cash charges of $1,644.4 million. The net change in operating assets and liabilities was primarily due to an increase in accounts receivable of $114.1 million driven by an increase in Biosecurity revenue and a decrease in deferred revenue of $10.5 million, partially offset by an increase in accrued expenses and other current liabilities of $44.8 million primarily due to Biosecurity revenue accruals, an increase in deferred
106
Table of Contents
rent of $6.0 million as a result of entering into new leases and expanding the terms of existing leases and an increase in other non-current liabilities of $18.6 million primarily due to a $20.0 million customer deposit liability. Non-cash adjustments primarily consisted of depreciation and amortization of $29.1 million, stock-based compensation expense of $1,606.0 million, loss on equity method investments of $77.3 million and loss on investments of $11.5 million, partially offset by gain on change in fair value of warrant liabilities of $58.6 million and non-cash equity consideration of $24.2 million from milestones associated with a customer collaboration arrangement.
Net cash used in operating activities for the year ended December 31, 2020 consisted of net loss of $126.7 million and a net change in operating assets and liabilities of $26.5 million, offset by non-cash adjustments of $17.4 million. The net change in operating assets and liabilities was primarily due to a decrease in current and non-current deferred revenue of $19.4 million, an increase in accounts receivable and accounts receivable from related parties of $14.2 million and an increase in prepaid expenses and other current assets of $11.4 million, partially offset by an increase in accounts payable of $7.0 million and an increase in accrued expenses and other current liabilities of $8.7 million. Non-cash adjustments primarily consisted of depreciation and amortization of $13.9 million, loss on equity method investments of $0.4 million and loss on investments of $3.7 million, partially offset by changes in the fair value of loans receivable of $1.1 million.
Investing Activities
Net cash used in investing activities for the year ended December 31, 2021 primarily consisted of purchases of property and equipment of $56.5 million associated with Foundry capacity and capability investments, purchase of non-marketable equity securities of $5.0 million and acquisition of Dutch DNA for $12.0 million.
Net cash used in investing activities for the year ended December 31, 2020 primarily consisted of purchases of property and equipment of $57.8 million, including costs associated with Foundry capacity and capability investments and purchase of Access Bio’s convertible notes of $10.0 million.
Financing Activities
Net cash provided by financing activities for the year ended December 31, 2021 primarily consisted of net proceeds received from the Business Combination of $1,509.6 million, non-controlling interest contributions of $59.9 million related to our consolidated variable interest entities (“VIEs”), Ayana and Verb, partially offset by repurchases of common stock from our founders of $25.0 million and tax withholding payments related to net share settlement of equity awards of $9.5 million.
Net cash provided by financing activities for the year ended December 31, 2020 primarily consisted of the net proceeds from the issuance of our Series E convertible preferred stock.
Critical Accounting Estimates
Our management’s discussion and analysis of our financial condition and results of operations is based on our consolidated financial statements, which have been prepared in accordance with U.S. GAAP. The preparation of these consolidated financial statements requires us to make judgments and estimates that affect the reported amounts of assets, liabilities, revenues and expenses and the disclosure of contingent assets and liabilities in our consolidated financial statements. We base our estimates on historical experience, known trends and events and various other factors that we believe to be reasonable under the circumstances, the results of which form the basis for making judgments about the carrying values of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these estimates under different assumptions or conditions. On an ongoing basis, we evaluate our judgments and estimates in light of changes in circumstances, facts and experience. The effects of material revisions in estimates, if any, are reflected in our consolidated financial statements prospectively from the date of change in estimates.
While our significant accounting policies are described in more detail in Note 2 to our audited consolidated financial statements appearing elsewhere in this Annual Report on Form 10-K, we believe the following accounting policies used in the preparation of our consolidated financial statements require the most significant judgments and estimates.
Revenue Recognition
We account for revenue in accordance with ASC 606, Revenue from Contracts with Customers (“ASC 606”). Under ASC 606, we recognize revenue when the customer obtains control of the promised goods or services, at an amount that reflects the consideration we expect to receive in exchange for those goods or services. To determine revenue recognition for arrangements that are within the scope of ASC 606, we perform the following five steps: (i) identify the contract(s) with a
107
Table of Contents
customer, (ii) identify the promises and distinct performance obligations in the contract, (iii) determine the transaction price, (iv) allocate the transaction price to the performance obligations in the contract, and (v) recognize revenue when (or as) we satisfy the performance obligations.
Foundry Revenue
We generate license and service revenue through the execution of license and collaboration agreements whereby customers obtain license rights to our proprietary technology and intellectual property for use in the research, development and commercialization of engineered organisms and derived products. Under these agreements, we typically provide R&D services, which includes the provision of a license to our intellectual property. Additionally, the customer obtains license rights to the output of our services in order to commercialize the resulting output of such services. Generally, the terms of these agreements provide that we receive some combination of: (1) Foundry usage fees in the form of (i) upfront payments upon consummation of the agreement or other fixed payments, (ii) reimbursement for costs incurred for R&D services and (iii) milestone payments upon the achievement of specified technical criteria, plus (2) downstream value share payments in the form of (i) milestone payments upon the achievement of specified commercial criteria, (ii) royalties on sales of products from or comprising engineered organisms arising from the collaboration or licensing agreement and (iii) royalties related to cost of goods sold reductions realized by our customers.
Our collaboration and licensing agreements often contain multiple promises, including (i) licenses and assignments of intellectual property and materials and (ii) R&D services, and we determine whether each of the promises is a distinct performance obligation based on the nature of each agreement. As we are generally performing R&D services that are highly integrated and interrelated to the licenses and assignments of intellectual property and materials, the promises are generally inseparable. As such, we typically combine the R&D services, licenses, and assignments into a single performance obligation. However, for certain agreements, we only grant licenses or effects such transfers and assignments upon the successful completion of the R&D services or delivery of a developed product. For these agreements, we typically consider (i) the R&D services and (ii) the licenses, transfers, and assignments as distinct performance obligations, as each is transferred separately and has a separately identifiable benefit. Options to acquire additional goods and services are evaluated to determine if such options provide a material right to the counterparty that it would not have received without entering into the contract. If so, the option is accounted for as a separate performance obligation. If not, the option is considered a marketing offer which is accounted for as a separate contract upon the counterparty’s election.
At contract inception, we determine the transaction price, including fixed consideration and any estimated amounts of variable consideration. Any upfront cash payment received upon consummation of the agreement is fixed and generally nonrefundable. Variable consideration is subject to a constraint, and amounts are included in the transaction price to the extent that it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is subsequently resolved. Variable consideration may include reimbursement for costs incurred for our R&D efforts, milestone payments upon the achievement of certain technical and commercial criteria, and royalties on sales of products from or comprising engineered organisms arising from the agreement. With respect to the R&D reimbursements and milestone payments, we use the most likely amount method to estimate variable consideration. With respect to agreements that include royalties on sales or other contingent payments based on sales, we apply the royalty recognition constraint which requires a constraint until the royalty or value-sharing transaction occurs. Certain agreements contain payment in the form of equity or other non-cash consideration. Any non-cash consideration is measured at the fair value of the non-cash consideration at contract inception.
For agreements with promises that are combined into a single performance obligation, the entire transaction price is allocated to the single performance obligation. For agreements with multiple performance obligations, the transaction price is allocated to the performance obligations using the relative standalone selling price methodology. For agreements featuring variable consideration, we allocate variable consideration to one or more, but not all, performance obligations if certain conditions are met. Specifically, we assess whether the variable consideration relates solely to our efforts to satisfy the performance obligation and whether allocating such variable consideration entirely to the performance obligation is consistent with the overall allocation objective. If these conditions are not met, we allocate the variable consideration based on the relative standalone selling price methodology. The key assumptions utilized in determining the standalone selling price for each performance obligation include development timelines, estimated R&D costs, commercial markets, likelihood of exercise (in the case of options considered to be material rights), and probabilities of success.
For agreements where the licenses or assignments are considered separate performance obligations or represent the only performance obligation, we recognize revenue at the point in time that we effectively grant the license as the licenses or assignments represent functional intellectual property. For agreements where the licenses and the R&D services represent a
108
Table of Contents
combined performance obligation, we recognize revenue over the period of performance using a measure of progress based on costs incurred to date as compared to total estimated costs.
We evaluate our measure of progress to recognize revenue at each reporting period and, as necessary, adjust the measure of progress and related revenue recognition. Our measure of progress and revenue recognition involves significant judgment and assumptions, including, but not limited to, estimated costs and timelines to complete our performance obligations. We evaluate contract modifications and amendments to determine whether any changes should be accounted for prospectively or on a cumulative catch-up basis. We utilize the right to invoice practical expedient when we have a right to consideration in an amount that corresponds directly with the value of our performance to date.
Royalties are recognized as revenue when sales have occurred as we apply the sales or usage-based royalties recognition constraint. We have determined the application of this exception is appropriate because the license granted in the agreement is the predominant item to which the royalties relate.
As we receive upfront payments for technical services under certain of our arrangements, we evaluate whether any significant financing components exist given the term over which the fees will be earned may exceed one year. Based on the nature of our agreements, there are no significant financing components as the purpose of the upfront payment is not to provide financing, but rather to secure technical services, exclusivity rights, and Foundry capacity, or the timing of transfer of those goods or services is at the discretion of the customer.
Deferred revenue represents consideration received by us in excess of revenue recognized and primarily results from transactions where we receive upfront payments and non-cash equity consideration. In instances where we have received consideration in advance for an undefined number of technical development plans (“TDPs”) under our customer agreements, we record the advance payments as deferred revenue, net of current portion on our consolidated balance sheets. Upon the execution of a specific TDP, we reclassify the estimated consideration to be earned under that TDP within the next twelve months as current deferred revenue. We also classify unexercised material rights related to future TDPs as deferred revenue, net of current portion on our consolidated balance sheets. When a TDP is executed, and the material right is exercised, the amount allocated to the material right, which will be earned within the next twelve months, is reclassified to current deferred revenue. All other deferred revenue is classified as current or non-current based on the timing of when we expect to earn the underlying revenue based upon the projected progress of activities under the TDP.
Variable Interest Entities
We evaluate our variable interests in VIEs and consolidate VIEs when we are the primary beneficiary. We determine whether we are the primary beneficiary of each VIE based on our assessment of whether we possess both (i) the power to direct the activities that most significantly affect the VIE’s economic performance and (ii) the obligation to absorb losses that could be significant to the VIE or the right to receive benefits that could be significant to the VIE. We reevaluate the accounting for our VIEs upon the occurrence of events that could change the primary beneficiary conclusion. With respect to our investments in Motif, Allonnia, Genomatica and Arcaea, we have concluded these entities represent variable interest entities. However, although we hold board representation and are involved in the ongoing development activities of the entities via participation on joint steering committees, we have concluded that we are not the primary beneficiary of these entities. We have reached this conclusion due to the fact that: (i) we do not control the board of directors of Motif, Allonnia, Genomatica or Arcaea and no voting or consent agreements exist between ourselves and other members of each respective board of directors or other investors, (ii) the holders of preferred security interests in Motif, Allonnia, Genomatica and Arcaea hold certain rights that require their consent prior to the taking of certain actions, which include certain significant operating and financing decisions and (iii) our representation on the joint steering committee of each respective entity does not give us control over the development activities of either Motif, Allonnia, Genomatica or Arcaea as all votes must pass by consensus and there are no agreements in place that would require either entity to vote in alignment with ourselves. As our involvement in Motif, Allonnia, Genomatica and Arcaea does not give us the power to control the decisions with respect to the development or other activities, which are the most significant activities of Motif, Allonnia, Genomatica or Arcaea, we have accordingly concluded that we are not the primary beneficiary. Additionally, with respect to Cooksonia’s investment in Joyn, as Cooksonia does not control Joyn’s board of directors, it does not have the power to control the decisions related to the development activities of Joyn, which are the most significant activities of Joyn. Accordingly, Cooksonia is not the primary beneficiary of Joyn.
With respect to Cooksonia, we have concluded that we hold a variable interest in this entity through our equity interest and we are the primary beneficiary of Cooksonia as we control the most significant activities of Cooksonia, we control 100% of the board of directors of Cooksonia and we hold a controlling financial interest in Cooksonia. With respect to Ayana and
109
Table of Contents
Verb, two Platform Ventures formed in September 2021 by us and certain of our investors, we concluded that we hold a variable interest in these entities through our equity interests and are the primary beneficiary of Ayana and Verb as we control the most significant activities of these entities. These conclusions were reached because, as of the primary beneficiary assessment date, for both Verb and Ayana: (i) we have substantive control of the board of directors; (ii) all capital contributions were made by our related parties; and (iii) we or our related parties comprise the entirety of the Joint Steering Committee, the governing body which holds significant oversight with respect to the entities' research and development programs. As a result, we have consolidated the financial statements of Cooksonia, Ayana and Verb in accordance with ASC 810, Consolidation (“ASC 810”), into our consolidated financial statements and have recognized a non-controlling interest associated with the minority equity interest held by the other investors of these VIEs.
Impairment of Long-Lived Assets
We review our long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying value of an asset may not be recoverable. Recoverability is measured by comparing the book values of the assets to the expected future net undiscounted cash flows that the assets are expected to generate. If such assets are considered to be impaired, the impairment to be recognized is measured by the amount by which the book values of the assets exceed their fair value.
Determination of Fair Value of Non-cash Consideration in Platform Ventures
The fair value of non-cash consideration received in relation to our Platform Ventures is in return for the license rights conveyed to the counterparty. We value the non-cash consideration, which is generally common stock or common units, at inception of the agreements using an option pricing method (“OPM”). The OPM used a back-solve methodology to infer the total equity value based on the pricing of the preferred financing round associated with the formation of the respective Platform Ventures, which was contemporaneous with the intellectual property agreements that conveyed our license rights to such Platform Ventures.
Determination of Fair Value of Loans Receivable
We have elected the fair value option under ASC 825, Financial Instruments (“ASC 825”), to account for our loans receivable. We use various valuation techniques to fair value our loans receivable, which are dependent on the terms of the underlying agreements, and record the gains or loss arising from the change in fair value as a component of other (expense) income, net in our consolidated statements of operations and comprehensive loss. As of December 31, 2021 and December 31, 2020, our loans receivable balance primarily consisted of our revolving promissory note with Glycosyn and a series of convertible notes with Access Bio. We used a probability-weighted discounted cash flow valuation approach to value our revolving promissory note with Glycosyn. Under this approach, the present value of the expected cash flows was calculated under various settlement scenarios and then weighted based on the probability of each scenario. Significant inputs used in valuing the Glycosyn promissory note included the probability and timing of each scenario, debt recovery rate and discount rate. We used a binomial lattice model and a Monte-Carlo simulation model to determine the value of our convertible notes with Access Bio as of December 31, 2021 and 2020, respectively. Significant inputs used under the binomial lattice model included equity volatility, time to maturity, risk-free rate, risk-adjusted rate and dividend yield. Under the Monte-Carlo simulation model, the future stock price of Access Bio was simulated over the term of the notes to assess the value of the settlement features which included conversion into stock at a discount determined under a reset provision tied to the stock price of Access Bio and redemption at maturity. Significant inputs used under the Monte Carlo simulation model were the discount rate and equity volatility. Refer to Note 5 of our consolidated financial statements appearing elsewhere in this Annual Report on Form 10-K for additional details.
Business Combinations
We account for business combinations using the acquisition method of accounting. We recognize the identifiable assets acquired and liabilities assumed at their acquisition-date fair values and recognize any excess of the total consideration paid over the fair value of the identifiable net assets as goodwill. Significant management judgments and assumptions are required in determining the fair value of acquired assets and liabilities assumed, particularly intangible assets and their estimated useful lives and the value of contingent consideration payable. As a result, we obtain the assistance of third-party valuation specialists in estimating fair value of intangible assets and contingent consideration payable. Significant estimates in valuing intangible assets include, but are not limited to, the expected costs to reproduce an asset, useful lives and discount rates. Significant estimates in valuing earnout provisions related to contingent consideration payable include, among other factors, future revenue projection of the acquired business, probabilities of achieving certain technical and commercial milestones and appropriate discount rates used in computing present values. While management believes those expectations and
110
Table of Contents
assumptions are reasonable, they are inherently uncertain. Unanticipated market or macroeconomic events and circumstances may occur, which could affect the estimates and assumptions.
Stock-Based Compensation
Stock-based compensation expense is measured based on grant-date fair values and is recognized over the requisite service period. For awards that vest solely based on a service condition, we recognize stock-based compensation expense on a straight-line basis over the requisite service period. For awards that vest based on performance and market conditions, we recognize compensation expense using the accelerated attribution method on a tranche-by-tranche basis. We estimate the grant date fair value of stock options using the Black-Scholes option-pricing model. The Black-Scholes option-pricing model requires the input of certain assumptions, including fair value of common stock (for options granted prior to the Business Combination), expected term, expected volatility, risk-free interest rate and expected dividend yield. For awards with market conditions, we determine the grant date fair value using a Monte Carlo simulation model, which incorporates various assumptions including expected stock price volatility, risk-free interest rate, expected term, and expected dividend yield.
Following the Business Combination, the fair value of our common stock is based on the quoted market price on the NYSE. Prior to the Business Combination, the fair value of our common stock underlying our stock-based awards was determined considering our most recently available third-party valuations of common stock and our assessment of additional objective and subjective factors. These judgments and estimates included: (i) a discount for lack of marketability; (ii) external market data; (iii) historical activity by us in selling equity to outside investors; (iv) our stage of development; (v) rights and preferences of our equity securities that rank senior to common stock; and (vi) the likelihood of the various scenarios, among others. Changes to these assumptions could result in different fair values of common stock.
The assumptions underlying these valuations represent management’s best estimates, which involve inherent uncertainties and the application of management judgment. As a result, if factors or expected outcomes change and we use significantly different assumptions or estimates, the fair value of our stock-based awards could be materially different.
JOBS Act and Emerging Growth Company Status
In April 2012, the JOBS Act was enacted. As an emerging growth company (“EGC”) under the JOBS Act, we may delay the adoption of certain accounting standards until such time as those standards apply to private companies. Other exemptions and reduced reporting requirements under the JOBS Act for EGCs include an exemption from the requirement to provide an auditor’s report on internal controls over financial reporting pursuant to Section 404(b) of the Sarbanes-Oxley Act of 2002, an exemption from any requirement that may be adopted by the Public Company Accounting Oversight Board regarding mandatory audit firm rotation and less extensive disclosure about our executive compensation arrangements. Additionally, the JOBS Act provides that an EGC can take advantage of an extended transition period for complying with new or revised accounting standards. This allows an EGC to delay the adoption of certain accounting standards until those standards would otherwise apply to private companies. We have elected not to opt out of the extended transition period and, therefore, while we are an EGC we will not be subject to new or revised accounting standards while they become applicable to other public companies that are not EGCs, unless we choose to early adopt a new or revised accounting standard.
We will remain classified as an EGC until the earlier of: (i) the last day of our first fiscal year in which we have total annual gross revenues of $1.07 billion or more, (ii) the last day of the fiscal year following the fifth anniversary of completion of the IPO of SRNG, (iii) the date on which we have issued more than $1.0 billion of non-convertible debt instruments during the previous three fiscal years or (iv) the date on which we are deemed a “large accelerated filer” under the rules of the SEC.
Recently Issued Accounting Pronouncements
See Note 2, “Summary of significant accounting policies,” of our consolidated financial statements contained in Part II, Item 8 of this Annual Report on Form 10-K for a discussion of recently issued accounting pronouncements.
111
Table of Contents
Item 7A. Quantitative and Qualitative Disclosures About Market Risk.
Interest Rate Fluctuation Risk
We are exposed to market risk related to changes in interest rates. Our primary exposure to market risk is interest rate sensitivity, which is affected by changes in the general level of U.S. interest rates, particularly because our cash equivalents are invested in short-term U.S. Treasury obligations. However, because of the short-term nature of the instruments in our portfolio, an immediate change in market interest rates of 100 basis points would not have a material impact on the fair market value of our cash and cash equivalents or on our financial position or results of operations.
Foreign Currency Fluctuation Risk
We are subject to foreign currency exchange risk from the translation of the financial statements of our foreign subsidiary, whose financial condition and results of operations are reported in Euros and then translated into U.S. dollars at the applicable currency exchange rate for inclusion in our consolidated financial statements. Foreign currency translation adjustments were $1.7 million for the year ended December 31, 2021. Additionally, we have contracted with and may continue to contract with foreign vendors.
Inflation Fluctuation Risk
Inflation generally affects us by increasing our cost of labor. We do not believe that inflation had a material effect on our business, financial condition or results of operations during the years ended December 31, 2021, 2020 and 2019.
Item 8. Financial Statements and Supplementary Data.
Our consolidated financial statements, together with the report of our independent registered public accounting firm, appear beginning on page F-1 in Part IV, Item 15, “Exhibits, Financial Statement Schedules” and are incorporated herein by reference.
Item 9. Changes in and Disagreements With Accountants on Accounting and Financial Disclosure.
None.
Item 9A. Controls and Procedures.
Evaluation of Disclosure Controls and Procedures
Our disclosure controls and procedures are designed to ensure that information required to be disclosed in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported within the time periods specified in the rules and forms of the SEC, and that such information is accumulated and communicated to our management, including our Chief Executive Officer and Chief Financial Officer, as appropriate, to allow timely decisions regarding required disclosure. Our management, with the participation of our Chief Executive Officer and Chief Financial Officer, evaluated the effectiveness of our disclosure controls and procedures (as defined in Exchange Act Rule 13a–15(e) and 15d-15(e)) as of December 31, 2021. Based on that evaluation, the Chief Executive Officer and Chief Financial Officer concluded that the Company’s disclosure controls and procedures were effective as of December 31, 2021.
Management’s Annual Report on Internal Control Over Financial Reporting
This annual report does not include a report of management's assessment regarding internal control over financial reporting or an attestation report of the company's registered public accounting firm due to a transition period established by rules of the Securities and Exchange Commission for newly public companies.
112
Table of Contents
Changes in Internal Control over Financial Reporting
There were no changes in our internal control over financial reporting (as such term is defined in Exchange Act Rule 13a–15(f) and 15d-15(f)) during the most recent fiscal quarter that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
Item 9B. Other Information.
None.
Item 9C. Disclosure Regarding Foreign Jurisdictions that Prevent Inspections.
Not applicable.
113
Table of Contents
PART III
Item 10. Directors, Executive Officers and Corporate Governance.
Board of Directors and Management
The following is a list of Ginkgo’s directors and executive officers and their ages as of March 17, 2022.
Name |
|
Age |
|
Position |
Jason Kelly |
|
41 |
|
Chief Executive Officer and Founder; Director |
Reshma Shetty |
|
41 |
|
President, Chief Operating Officer and Founder; Director |
Mark Dmytruk |
|
50 |
|
Chief Financial Officer |
Arie Belldegrun |
|
72 |
|
Director |
Marijn Dekkers |
|
64 |
|
Director |
Christian Henry |
|
54 |
|
Director |
Reshma Kewalramani |
|
49 |
|
Director |
Shyam Sankar |
|
40 |
|
Director |
Harry E. Sloan |
|
71 |
|
Director |
Jason Kelly, one of our Founders, is the Chief Executive Officer and a member of Ginkgo’s board of directors. Dr. Kelly has served as a director of CM Life Sciences II Inc. (Nasdaq: CMII), a SPAC with a focus on the life sciences sector, since its IPO in February 2021. Dr. Kelly has a Ph.D. in Biological Engineering and a B.S. in Chemical Engineering and Biology from the Massachusetts Institute of Technology. We believe that Dr. Kelly is qualified to serve on our board of directors as a Founder and due to his knowledge of our company and our business.
Reshma Shetty, one of our Founders, is the President and Chief Operating Officer and a member of Ginkgo’s board of directors. Dr. Shetty currently serves on the Bio Advisory Group at the non-profit Nuclear Threat Initiative. Dr. Shetty has a Ph.D. in Biological Engineering from the Massachusetts Institute of Technology and a B.S. in Computer Science from the University of Utah. We believe that Dr. Shetty is qualified to serve on our board of directors as a Founder and due to her knowledge of our company and our business.
Mark Dmytruk has served as our Chief Financial Officer since joining Ginkgo in 2020. From 2017 to 2020, Mr. Dmytruk served as Executive Vice President, Corporate Strategy and Development, for Syneos Health, a global Contract Research Organization and Contract Commercial Organization serving the biopharmaceutical industry. Syneos Health was formed through the merger of inVentiv Health and INC Research in 2017, and prior to the merger Mr. Dmytruk served at inVentiv Health as Chief of Staff from 2014 to 2017 and President, Patient Outcomes Division, from 2011 to 2014. Prior to inVentiv Health, Mr. Dmytruk served in a variety of roles at Thermo Fisher Scientific (and its predecessor, Fisher Scientific) from 2001 to 2011. As Vice President of Corporate Development, Mr. Dmytruk led the company’s M&A function, contributing to its industry-changing strategy and transformational growth. He also served as Vice President of Finance for the Athena Diagnostics business unit of Thermo Fisher Scientific prior to its sale to Quest Diagnostics. Mr. Dmytruk began his career at Ernst & Young in Canada. Mr. Dmytruk has an M.B.A. from the Sloan School of Management at the Massachusetts Institute of Technology and a Bachelor of Commerce from the University of Alberta.
Arie Belldegrun, M.D., is a member of Ginkgo’s board of directors. Dr. Belldegrun is a leader in the field of cell and gene therapy. Dr. Belldegrun is a co-founder of Allogene Therapeutics, a public biopharmaceutical company, and has served as Executive Chairman of its board of directors since November 2017. From March 2014 until October 2017, Dr. Belldegrun served as the President and Chief Executive Officer of Kite Pharma, Inc. and as a member of its board of directors from June 2009 until October 2017. Dr. Belldegrun currently serves as Chairman of Bellco Capital LLC, a position he has held since 2004, Chairman of Kronos Bio, a position he has held since November 2017, Chairman of UroGen Pharma, Ltd., a position he has held since December 2012, as Chairman and Partner of Two River Group, a position he has held since June 2009, as co-Chairman of Breakthrough Properties LLC and Breakthrough Services, L.L.C., a position he has held since April 2019, as a director of ByHeart, Inc., a position he has held since October 2019, and as a director of IconOVir Bio, Inc., a position he has held since June 2020. Dr. Belldegrun has also served as Senior Managing Director of Vida Ventures, LLC since
November 2017. He is certified by the American Board of Urology and the American Association of Genitourinary Surgeons. Dr. Belldegrun is Research Professor, holds the Roy and Carol Doumani Chair in Urologic Oncology, and Director of the Institute of Urologic Oncology at the David Geffen School of Medicine at the University of California, Los Angeles. Prior to joining UCLA in October of 1988, he was a research fellow at NCI/NIH in surgical oncology and immunotherapy from July 1985 to August 1988 under Dr. Steven Rosenberg. Dr. Belldegrun received his M.D. from the Hebrew University
114
Table of Contents
Hadassah Medical School in Jerusalem before completing his post graduate studies in Immunology at the Weizmann Institute of Science and his residency in Urologic Surgery at Harvard Medical School. We believe that Dr. Belldegrun is qualified to serve on our board of directors due to his extensive knowledge as a leader in the field of cell and gene therapy.
Marijn Dekkers is chairman of Ginkgo’s board of directors. Dr. Dekkers is the Founder and Chairman of Novalis LifeSciences LLC, an investment and advisory firm for the Life Science industry that he founded in 2017. Before that, from 2010 to 2016, Dr. Dekkers served as Chief Executive Officer of Bayer AG. Prior to his time at Bayer, from 2002 to 2009, he served as Chief Executive Officer of Thermo Fisher Scientific. Dr. Dekkers currently serves as a director on the boards of Quantum-SI, Inc. and Cerevel Therapeutics Holdings, Inc., as well as Georgetown University and the Foundation for the National Institutes of Health. He is a former director of Unilever, General Electric and Biogen. Dr. Dekkers began his career in 1985 as a research scientist at General Electric’s Corporate R&D Center. Dr. Dekkers received his PhD and M.S. in chemical engineering from the University of Eindhoven and his B.S in chemistry from the Radboud University. We believe that Dr. Dekkers is qualified to serve on our board of directors due to his extensive knowledge of the life sciences industry, his familiarity with our company and his prior director service.
Christian Henry is a member of Ginkgo’s board of directors. Mr. Henry has served as President and Chief Executive Officer of Pacific Biosciences of California, Inc., a leading sequencing company, since September 2020. From 2005 to January 2017, Mr. Henry was a member of the executive team of Illumina, Inc., a global leader in sequencing. During this tenure at Illumina, he served in a number of roles, including Executive Vice President & Chief Commercial Officer, Senior Vice President of Genomic Solutions, Senior Vice President and General Manager of Life Sciences and Senior Vice President and Chief Financial Officer. Prior to joining Illumina in 2005, Mr. Henry served as the Chief Financial Officer of Tickets.com, Inc. from 2003 to 2005. From 1999 to 2003, Mr. Henry served as Vice President, Finance and Corporate Controller of Affymetrix, Inc. (acquired by Thermo Fisher Scientific in 2016). In 1997, Mr. Henry joined Nektar Therapeutics (formerly Inhale Therapeutic Systems, Inc.) as Corporate Controller, and later as its Chief Accounting Officer from 1997 to 1999. In 1996, Mr. Henry served as General Accounting Manager of Sugen, Inc. Mr. Henry began his career in 1992 at Ernst & Young LLP, where he was a Senior Accountant through 1996. Mr. Henry currently serves as a director and Chairman of the board of WAVE Life Sciences Ltd., and as a director of CM Life Sciences Holdings III LLC. Mr. Henry previously served as Chairman of the board of Pacific Biosciences from August 2018 to September 2020. Mr. Henry holds a B.A. in biochemistry and cell biology from the University of California, San Diego and an M.B.A., with a concentration in finance, from the University of California, Irvine. We believe that Mr. Henry is qualified to serve on our board of directors due to his over 20 years of experience in growing companies in the life sciences industry.
Reshma Kewalramani is a member of Ginkgo’s board of directors. Dr. Kewalramani has been the Chief Executive Officer and President of Vertex Pharmaceuticals Inc. since April 2020 and a member of Vertex’s Board of Directors since February 2020. Dr. Kewalramani was Vertex’s Executive Vice President and Chief Medical Officer from April 2018 through April 2020. She was Vertex’s Senior Vice President, Late Development from February 2017 until March 2018. From August 2004 to January 2017, she served in roles of increasing responsibility at Amgen Inc., most recently as Vice President, Global Clinical Development, Nephrology & Metabolic Therapeutic Area and as Vice President, U.S. Medical Organization. From 2014 through 2019, Dr. Kewalramani was the industry representative to the FDA’s Endocrine and Metabolic Drug Advisory Committee. She completed her internship and residency in Internal Medicine at the Massachusetts General Hospital and her fellowship in Nephrology at the Massachusetts General Hospital and Brigham and Women’s Hospital combined program. Dr. Kewalramani holds a B.A. from Boston University and an M.D. from Boston University School of Medicine. Dr. Kewalramani also completed the General Management Program at Harvard Business School and is an alumnus of the school. We believe that Dr. Kewalramani is qualified to serve on our board of directors due to her extensive experience serving in senior roles at various pharmaceutical companies.
Shyam Sankar is a member of Ginkgo’s board of directors. Mr. Sankar is the Chief Operating Officer and Executive Vice President at Palantir Technologies Inc., where he has worked in various positions since 2006. Prior to his time at Palantir, Mr. Sankar served as the Vice President of Network Management and Director of Business Development for Xoom Corporation. Mr. Sankar has a deep operational background overseeing the development of complex technology from near inception to massive scale. Mr. Sankar received his M.S. in management science and engineering from Stanford University and his B.S. in electrical and computer engineering from Cornell University. We believe that Mr. Sankar is qualified to serve on our board of directors due to his business acumen, leadership experience, and operational background, having overseen the development and expansion of a software company from its near inception through its public listing.
Harry E. Sloan is a founder, public company CEO and a leading investor in the media, entertainment and technology industries. Mr. Sloan is the Chairman and CEO of Eagle Equity Partners II, LLC. Under Mr. Sloan’s leadership, the company has acquired and taken public, through SPACs, several digital media companies including, during 2020, DraftKings and mobile gaming company Skillz. Mr. Sloan has been at the forefront and evolution of the video gaming industry as one of the
115
Table of Contents
founding investors and a Board Member of Zenimax/Bethesda Game Studios, the awarding winning studio acquired by Microsoft in March 2021. After delivering two of the most successful SPAC business combinations to date, Mr. Sloan co-founded Soaring Eagle Acquisition Corp. (Nasdaq: SRNGU), which raised $1.725 billion in its IPO in February 2021 and three months later announced a merger with Boston-based Ginkgo Bioworks Inc. in a deal valued at $17.5 billion. In January 2022, Mr. Sloan and his partners launched Screaming Eagle Acquisition Corp. With a closing of its initial public offering of 75,000,000 units, at a price of $10 per unit, Screaming Eagle is the largest IPO of a public acquisition vehicle since March 2021. Mr. Sloan has served as a director of Skillz, Inc. since December 2020 and DraftKings Inc. since April 2020. He was a director of Soaring Eagle Acquisition Corp. from October 2020 until September 2021, Flying Eagle Acquisition Corp. from March 2020 until December 2020, Diamond Eagle Acquisition Corp. from May 2019 until April 2020, and Videocon d2h Limited from May 2016 until April 2018. We believe that Mr. Sloan is qualified to serve on our board of directors due to his public company experience, business leadership, and operational experience.
Corporate Governance
Ginkgo structures its corporate governance in a manner that it believes closely aligns its interests with those of its stockholders. Notable features of this corporate governance include:
Role of Board in Risk Oversight
The Ginkgo Board has extensive involvement in the oversight of risk management related to Ginkgo and its business and will accomplish this oversight through the regular reporting to the board of directors by the audit committee. The audit committee will represent the Ginkgo Board by periodically reviewing Ginkgo’s accounting, reporting and financial practices, including the integrity of its financial statements, the surveillance of administrative and financial controls and its compliance with legal and regulatory requirements. Through its regular meetings with management, including the finance, legal, internal audit and information technology functions, the audit committee will review and discuss all significant areas of Ginkgo’s business and summarize for the Ginkgo Board all areas of risk and the appropriate mitigating factors. In addition, the Ginkgo Board will receive periodic detailed operating performance reviews from management.
Composition of the Ginkgo Board
Ginkgo’s business and affairs are managed under the direction of its board of directors. Under the Charter, the Ginkgo Board is declassified and all of Ginkgo’s directors are elected each year for one-year terms.
Board Committees
The standing committees of the Ginkgo Board consist of an audit committee, a compensation committee and a nominating and corporate governance committee. The Ginkgo Board may from time to time establish other committees. Ginkgo’s chief executive officer, its president and chief operating officer, and other executive officers will regularly report to the non-executive directors and the audit, the compensation and the nominating and corporate governance committees to ensure effective and efficient oversight of our activities and to assist in proper risk management and the ongoing evaluation of management controls.
Audit Committee
Ginkgo has an audit committee, consisting of Christian Henry, who serves as the chairperson, Harry Sloan, and Marijn Dekkers until February 19, 2022, after which Shyam Shankar joined the committee. Each of Messrs. Henry, Shankar and Sloan qualify as an independent director under the NYSE corporate governance standards and the independence requirements of Rule 10A-3 under the Securities Exchange Act of 1934, as amended (the “Exchange Act”). The Ginkgo Board has determined that Mr. Henry qualifies as an “audit committee financial expert” as such term is defined in Item 407(d)(5) of Regulation S-K and possesses financial sophistication, as defined under the rules of NYSE.
116
Table of Contents
The purpose of the audit committee is to prepare the audit committee report required by the SEC to be included in Ginkgo’s proxy statement and to assist the board of directors in overseeing and monitoring (1) the quality and integrity of the financial statements, (2) compliance with legal and regulatory requirements, (3) Ginkgo’s independent registered public accounting firm’s qualifications and independence, (4) the performance of Ginkgo’s internal audit function and (5) the performance of Ginkgo’s independent registered public accounting firm.
The Ginkgo Board has adopted a written charter for the audit committee, which is available on Ginkgo’s website.
Compensation Committee
Ginkgo has a compensation committee, consisting of Shyam Sankar, who serves as the chairperson, Arie Belldegrun and Christian Henry.
The purpose of the compensation committee is to assist the board of directors in discharging its responsibilities relating to (1) setting Ginkgo’s compensation program and compensation of its executive officers and directors, (2) monitoring Ginkgo’s incentive and equity-based compensation plans and (3) preparing the compensation committee report required to be included in Ginkgo’s proxy statement under the rules and regulations of the SEC.
The Ginkgo Board has adopted a written charter for the compensation committee, which is available on Ginkgo’s website.
Nominating and Corporate Governance Committee
Ginkgo has a nominating and corporate governance committee, consisting of Reshma Kewalramani and Shyam Sankar. Dr. Dekkers served as the chairperson until March 25, 2022, after which Dr. Kewalramani serves as chairperson. The purpose of the nominating and corporate governance committee is to assist the board of directors in discharging its responsibilities relating to (1) identifying individuals qualified to become new board of directors members, consistent with criteria approved by the board of directors, (2) reviewing the qualifications of incumbent directors to determine whether to recommend them for reelection and selecting, or recommending that the board of directors select, the director nominees for the next annual meeting of shareholders, (3) identifying board of directors members qualified to fill vacancies on any board of directors committee and recommending that the board of directors appoint the identified member or members to the applicable committee, (4) reviewing and recommending to the board of directors corporate governance principles applicable to Ginkgo, (5) overseeing the evaluation of the board of directors and management and (6) handling such other matters that are specifically delegated to the committee by the board of directors from time to time.
The Ginkgo Board has adopted a written charter for the nominating and corporate governance committee, which is available on Ginkgo’s website.
Code of Business Conduct
Ginkgo has adopted a code of business conduct and ethics that applies to all of its directors, officers and employees, including its principal executive officer, principal financial officer and principal accounting officer, which is available on Ginkgo’s website. Ginkgo’s code of business conduct and ethics is a “code of ethics”, as defined in Item 406(b) of Regulation S-K. Please note that Ginkgo’s website address is provided as an inactive textual reference only. Ginkgo will make any legally required disclosures regarding amendments to, or waivers of, provisions of its code of ethics on its website.
Delinquent Section 16(a) Reports
Section 16(a) of the Exchange Act requires our directors and executive officers (and persons who own more than 10% of our equity securities) to file reports of holdings and transactions in our common stock with the SEC. To facilitate compliance, we have undertaken the responsibility to prepare and file these reports on behalf of our officers and directors. Based on a review of the reports furnished to us or filed with the SEC and upon information furnished by these parties, the Company believes that during the fiscal year 2021, its executive officers, directors and 10% stockholders timely complied with all Section 16(a) filing requirements, except for a Form 3 amendment that was filed for Dr. Belldegrun on March 28, 2022 to reflect holdings that were inadvertently not reported on his initial Form 3, due to an administrative error.
117
Table of Contents
Item 11. Executive Compensation.
Compensation Committee Interlocks and Insider Participation
No member of the compensation committee was at any time during fiscal year 2021, or at any other time, one of our officers or employees. None of our executive officers has served as a director or member of a compensation committee (or other committee serving an equivalent function) of any entity, one of whose executive officers served as a director of our board of directors or member of our compensation committee.
Independence of the Board of Directors
NYSE rules generally require that independent directors must comprise a majority of a listed company’s board of directors. Based upon information requested from and provided by each director concerning his or her background, employment and affiliations, including family relationships, we have determined that each of Arie Belldegrun, Christian Henry, Reshma Kewalramani, Shyam Sankar and Harry Sloan, representing a majority of Ginkgo’s directors, are “independent” as that term is defined under the applicable rules and regulations of the SEC and the listing requirements and rules of the NYSE.
Ginkgo’s Executive and Director Compensation
This section discusses the material components of the executive compensation program for Ginkgo’s executive officers who are named in the “2021 Summary Compensation Table” below. As an EGC, Ginkgo complies with the executive compensation disclosure rules applicable to “smaller reporting companies,” as such term is defined in the rules promulgated under the Securities Act, which require compensation disclosure for Ginkgo’s principal executive officer and Ginkgo’s two most highly compensated executive officers other than its principal executive officer. These three officers are referred to as Ginkgo’s named executive officers.
In 2021, Ginkgo’s “named executive officers” and their positions were as follows:
This discussion may contain forward-looking statements that are based on Ginkgo’s current plans, considerations, expectations and determinations regarding future compensation programs. The actual compensation programs that Ginkgo adopts may differ materially from the currently planned programs summarized in this discussion.
2021 Summary Compensation Table
The following table sets forth information concerning the compensation of Ginkgo’s named executive officers for the years ended December 31, 2021 and December 31, 2020.
|
|
|
|
|
|
|
|
|
Stock |
|
|
All Other |
|
|
|
|
|||||
|
|
|
|
|
|
|
|
|
Awards |
|
|
Compensation |
|
|
|
|
|||||
Name and Principal Position |
Year |
|
Salary ($)(1) |
|
|
Bonus ($) |
|
|
($)(2) |
|
|
($)(3) |
|
|
Total ($) |
|
|||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
Jason Kelly |
2021 |
|
$ |
250,000 |
|
|
$ |
- |
|
|
$ |
363,924,473 |
|
(4) |
$ |
12,500 |
|
|
$ |
364,186,973 |
|
Chief Executive Officer |
2020 |
|
$ |
250,000 |
|
|
$ |
414,841 |
|
|
$ |
9,854,097 |
|
|
$ |
14,250 |
|
|
$ |
10,533,188 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
Reshma Shetty |
2021 |
|
$ |
250,000 |
|
|
$ |
- |
|
|
$ |
363,924,473 |
|
(4) |
$ |
12,500 |
|
|
$ |
364,186,973 |
|
President and Chief Operating Officer |
2020 |
|
$ |
250,000 |
|
|
$ |
415,386 |
|
|
$ |
9,854,097 |
|
|
$ |
14,250 |
|
|
$ |
10,533,733 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
Mark Dmytruk |
2021 |
|
$ |
425,000 |
|
|
$ |
- |
|
|
$ |
39,629,178 |
|
(4) |
$ |
14,500 |
|
|
$ |
40,068,678 |
|
Chief Financial Officer |
2020 |
|
$ |
63,750 |
|
|
$ |
- |
|
|
$ |
- |
|
|
$ |
2,861 |
|
|
$ |
66,611 |
|
118
Table of Contents
Narrative To Summary Compensation Table
2021 Salaries
The named executive officers receive a base salary to provide a fixed component of compensation reflecting the executive’s skill set, experience, role and responsibilities. No changes were made to the annual base salaries of the named executive officers during 2021.
The 2021 annual base salaries for Ginkgo’s named executives officers were:
Name |
|
2021 Annual Base Salary ($) |
|
|
Jason Kelly |
|
$ |
250,000 |
|
Reshma Shetty |
|
$ |
250,000 |
|
Mark Dmytruk |
|
$ |
425,000 |
|
During 2021, our employees were given the opportunity to participate in a salary exchange program under which they could exchange a portion of their annual base salaries for restricted stock units. The salary to restricted stock units exchange took place on a one-to-four basis such that for each $1 of salary contributed to the program, the employee would receive a number of restricted stock units valued at $4. Mr. Dmytruk was our only named executive officer to participate in this program during 2021.
Equity Compensation
During 2021, Mr. Dmytruk was granted restricted stock units under our 2014 Incentive Plan (the “2014 Plan”). These restricted stock units vest upon the satisfaction of both an “event condition” and a “service condition” on or before the seventh anniversary of the applicable grant date. At the time of grant, the event condition would be satisfied on the first to occur of (i) a change in control or (ii) the earlier of (x) the date six months after the closing of a public offering or (y) March 15 of the calendar year following the effective date of a public offering. See the “Outstanding Equity Awards at Fiscal Year-End” table below for details regarding the service-based vesting schedule applicable to these awards.
In connection with the Business Combination in September 2021, Dr. Kelly and Dr. Shetty were each granted 21,458,317 restricted stock units and agreed to extend the vesting on their restricted stock units granted in 2020. These restricted stock units will vest upon the satisfaction of both an “event condition” (as described above) and a “service condition” on or before the seventh anniversary of the grant date. The service condition will be satisfied on October 1, 2022, subject to continued service with Ginkgo through such date. The 2021 grant of restricted stock units was deemed to satisfy all rights that Dr. Kelly and Dr. Shetty may have had under their Founder Equity Grant Agreements with Ginkgo that were entered into in January 2020.
In connection with the Business Combination, the restricted stock units held by the named executive officers were converted into restricted stock units in Ginkgo and, with respect to the earn-out portion of the consideration paid in the Business Combination, restricted stock. This restricted stock is subject to the service-based vesting schedule of the related restricted stock units and, in addition, only vest based on the attainment of certain stock price thresholds. See the “Outstanding Equity Awards at Fiscal Year-End” table below for additional information. In November 2021, the Ginkgo Board authorized the amendment of all outstanding restricted stock units and shares of restricted stock, including those held by the named executive officers, such that the event condition was deemed satisfied no later than March 15, 2022.
In connection with the Business Combination, we adopted the 2021 Incentive Award Plan (the “2021 Plan”) in order to facilitate the grant of cash and equity incentives to directors, employees (including our named executive officers) and consultants of Ginkgo. We ceased granting awards under the 2014 Plan when the 2021 Plan became effective.
119
Table of Contents
Other Elements of Compensation
Retirement Plan
Ginkgo maintains a 401(k) retirement savings plan for its employees, including Ginkgo’s named executive officers, who satisfy certain eligibility requirements. Ginkgo’s named executive officers are eligible to participate in the 401(k) plan on the same terms as other full-time employees. Under this plan, Ginkgo provides a non-elective safe harbor contribution to all eligible participants equal to up to 5% of eligible compensation, which fully vests once such eligible participant has completed two years of continuous service. Ginkgo believes that providing a vehicle for tax-deferred retirement savings though Ginkgo’s 401(k) plan adds to the overall desirability of Ginkgo’s executive compensation package and further incentivizes Ginkgo’s employees, including Ginkgo’s named executive officers, in accordance with Ginkgo’s compensation policies.
Employee Benefits and Perquisites
Health/Welfare Plans. During their employment, Ginkgo’s named executive officers are eligible to participate in Ginkgo’s employee benefit plans and programs, including medical, dental, vision, life, and disability benefits, to the same extent as Ginkgo’s other full-time employees, subject to the terms and eligibility requirements of those plans.
Outstanding Equity Awards at Fiscal Year-End
The following table summarizes the number of shares of Ginkgo common stock underlying outstanding equity incentive plan awards for each named executive officer as of December 31, 2021.
|
|
Stock Awards |
|
|||||||||||||||||||||
Name |
|
Grant Date |
|
Number of Shares or Units of Stock That Have Not Vested (#) |
|
|
|
|
Market Value of Shares or Units of Stock That Have Not Vested ($)(7) |
|
|
Equity Incentive Plan Awards: Unearned Shares, Units or Other Rights That Have Not Vested (#) |
|
|
|
|
Equity Incentive Plan Awards: Market or Payout Value of Unearned Shares, Units or Other Rights That Have Not Vested ($)(7) |
|
||||||
Jason Kelly |
|
1/1/2020 |
|
|
4,324,037 |
|
|
(1 |
) |
|
$ |
35,932,747 |
|
|
|
389,151 |
|
|
(6 |
) |
|
$ |
3,233,845 |
|
|
|
1/1/2020 |
|
|
129,719 |
|
|
(2 |
) |
|
$ |
1,077,965 |
|
|
|
— |
|
|
|
|
$ |
— |
|
|
|
|
8/18/2021 |
|
|
21,458,317 |
|
|
(1 |
) |
|
$ |
178,318,614 |
|
|
|
1,931,193 |
|
|
(6 |
) |
|
$ |
16,048,214 |
|
|
|
8/18/2021 |
|
|
643,734 |
|
|
(2 |
) |
|
$ |
5,349,430 |
|
|
|
— |
|
|
|
|
$ |
— |
|
|
Reshma Shetty |
|
1/1/2020 |
|
|
4,324,037 |
|
|
(1 |
) |
|
$ |
35,932,747 |
|
|
|
389,151 |
|
|
(6 |
) |
|
$ |
3,233,845 |
|
|
|
1/1/2020 |
|
|
129,719 |
|
|
(2 |
) |
|
$ |
1,077,965 |
|
|
|
— |
|
|
|
|
$ |
— |
|
|
|
|
8/18/2021 |
|
|
21,458,317 |
|
|
(1 |
) |
|
$ |
178,318,614 |
|
|
|
1,931,193 |
|
|
(6 |
) |
|
$ |
16,048,214 |
|
|
|
8/18/2021 |
|
|
643,734 |
|
|
(2 |
) |
|
$ |
5,349,430 |
|
|
|
— |
|
|
|
|
$ |
— |
|
|
Mark Dmytruk |
|
3/2/2021 |
|
|
1,564,440 |
|
|
(3 |
) |
|
$ |
13,000,496 |
|
|
|
198,768 |
|
|
(6 |
) |
|
$ |
1,651,762 |
|
|
|
3/2/2021 |
|
|
48,315 |
|
|
(2 |
) |
|
$ |
401,498 |
|
|
|
— |
|
|
|
|
$ |
— |
|
|
|
|
4/4/2021 |
|
|
47,670 |
|
|
(4 |
) |
|
$ |
396,138 |
|
|
|
5,883 |
|
|
(6 |
) |
|
$ |
48,888 |
|
|
|
4/4/2021 |
|
|
1,472 |
|
|
(2 |
) |
|
$ |
12,232 |
|
|
|
— |
|
|
|
|
$ |
— |
|
|
|
|
8/2/2021 |
|
|
300,619 |
|
|
(5 |
) |
|
$ |
2,498,144 |
|
|
|
30,918 |
|
|
(6 |
) |
|
$ |
256,929 |
|
|
|
8/2/2021 |
|
|
9,236 |
|
|
(2 |
) |
|
$ |
76,751 |
|
|
|
— |
|
|
|
|
$ |
— |
|
|
120
Table of Contents
Executive Compensation Arrangements
Jason Kelly and Reshma Shetty. Neither Dr. Kelly nor Dr. Shetty have entered into employment agreements, offer letters or severance agreements with Ginkgo.
Mark Dmytruk. We have entered into an offer letter with Mr. Dmytruk pursuant to which he will be entitled to receive the following payments and benefits in the event his employment is terminated by Ginkgo without “cause” (as defined in the offer letter), (i) 12 months’ severance pay based on his base salary rate on the date of such termination, to be paid in installments over the 12-month period following the termination date and (ii) up to 12 months’ company-paid health benefits continuation pursuant to COBRA, in each case subject to Mr. Dmytruk’s execution of a general release of claims in favor of Ginkgo.
Mr. Dmytruk also entered into a separate agreement pursuant to which he is subject to employee and customer non-solicitation covenants during the term of his employment or other service with Ginkgo and for one year thereafter. The agreement also includes standard invention assignment and confidential information covenants.
Director Compensation
In connection with the Business Combination, the Ginkgo Board implemented an annual compensation program for its non-employee directors which became effective on September 16, 2021, pursuant to which the non-employee directors are entitled to cash and equity compensation in such amounts necessary to attract and retain non-employee directors that have the talent and skills to foster long-term value creation and enhance the sustainable development of the company. The compensation payable under the program is intended to be competitive in relation to both the market in which the company operates and the nature, complexity and size of Ginkgo’s business.
Ginkgo’s non-employee directors receive the following amounts for their services on the Ginkgo Board under the non-employee director compensation program:
Cash Compensation
121
Table of Contents
Director fees are payable in arrears in four equal quarterly installments, provided that the amount of each payment will be prorated for any portion of a calendar quarter that a non-employee director is not serving on the Ginkgo Board. The Ginkgo Board may permit non-employee directors to elect to receive equity compensation in lieu of cash compensation.
Equity Compensation
Stock options granted under the program have an exercise price equal to the fair market value of Class A common stock on the date of grant and expire not later than ten years after the date of grant. Each Initial Option granted to a non-employee director will vest and become exercisable in substantially equal installments on each of the first three anniversaries of the date of grant. Each Additional Initial Option and the Additional Initial RSUs granted to a non-employee director will vest and become exercisable, as applicable, in a single installment on the day before the next annual meeting of stockholders occurring after the date of the director’s initial election or appointment to the Ginkgo Board. Each Subsequent Option and the Subsequent RSUs will vest and become exercisable, as applicable, in a single installment on the earlier of the first anniversary of the date of grant or the day before the next annual meeting of stockholders occurring after the date of grant. Vesting of the options and restricted stock units granted under the program is subject to the non-employee director’s continued service through each applicable vesting date. In the event of a change in control of Ginkgo, the options and restricted stock units granted under the program will vest in full.
In connection with the Business Combination, Dr. Kewalramani was granted an Initial Option (51,782 shares), an Additional Initial Option (17,649 shares) and an Additional Initial RSU (9,818 restricted stock units) under the program.
The following table sets forth information concerning the compensation of Ginkgo’s non-employee directors for their service on the board of directors for the year ended December 31, 2021.
Name |
|
Fees Earned or Paid in Cash ($)(1) |
|
|
Stock Awards ($)(2) |
|
|
Option Awards ($)(2) |
|
|
Total ($) |
|
||||
Arie Belldegrun |
|
|
16,563 |
|
|
|
— |
|
|
|
— |
|
|
|
16,563 |
|
Marijn Dekkers |
|
|
30,533 |
|
|
|
30,430,090 |
|
|
|
— |
|
|
|
30,460,623 |
|
Christian Henry |
|
|
22,323 |
|
|
|
20,000,194 |
|
|
|
— |
|
|
|
20,022,517 |
|
Reshma Kewalramani |
|
|
15,842 |
|
|
|
132,052 |
|
|
|
532,053 |
|
|
|
679,947 |
|
Evan Lodes |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Shyam Sankar |
|
|
20,163 |
|
|
|
20,625,832 |
|
|
|
— |
|
|
|
20,645,995 |
|
Harry Sloan |
|
|
17,283 |
|
|
|
— |
|
|
|
— |
|
|
|
17,283 |
|
122
Table of Contents
The table below shows the aggregate numbers of options (exercisable and unexercisable), unvested restricted stock and restricted stock units held as of December 31, 2021 by each non-employee director who was serving as of December 31, 2021.
Name |
|
Options Outstanding at Fiscal Year End (#) |
|
|
Restricted Stock Outstanding at Fiscal Year End (#) |
|
|
Restricted Stock Units Outstanding at Fiscal Year End (#) |
|
|||
Arie Belldegrun |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Marijn Dekkers |
|
|
— |
|
|
|
281,217 |
|
|
|
3,124,756 |
|
Christian Henry |
|
|
— |
|
|
|
175,911 |
|
|
|
1,954,629 |
|
Reshma Kewalramani |
|
|
69,431 |
|
|
|
— |
|
|
|
9,818 |
|
Shyam Sankar |
|
|
— |
|
|
|
175,911 |
|
|
|
1,954,629 |
|
Harry Sloan |
|
|
— |
|
|
|
— |
|
|
|
— |
|
Item 12. Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.
Beneficial Ownership of Securities
The following table sets forth information known to the Company regarding the beneficial ownership of Ginkgo common stock by:
Beneficial ownership is determined according to the rules of the SEC, which generally provide that a person has beneficial ownership of a security if he, she or it possesses sole or shared voting or investment power over that security, including options and warrants that are currently exercisable or exercisable within 60 days. Unless otherwise indicated, Ginkgo believes that all persons named in the table below have sole voting and investment power with respect to the voting securities beneficially owned by them.
The beneficial ownership of Ginkgo common stock is based on 1,080,456,818 shares of Ginkgo Class A common stock and 391,147,458 shares of Ginkgo Class B common stock issued and outstanding as of March 17, 2022, unless otherwise specified.
123
Table of Contents
|
|
Class A common stock |
|
|
Class B common stock |
|
|
% of Total Voting Power** |
|
|||||||||||
Name of Beneficial Owner |
|
Shares |
|
|
% |
|
|
Shares |
|
|
% |
|
|
% |
|
|||||
Directors and Executive Officers of Ginkgo |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
Jason Kelly(1) |
|
|
— |
|
|
|
— |
|
|
|
82,431,106 |
|
|
|
21.1 |
% |
|
|
16.5 |
% |
Reshma Shetty(2) |
|
|
— |
|
|
|
— |
|
|
|
165,841,730 |
|
|
|
42.4 |
% |
|
|
33.2 |
% |
Mark Dmytruk (3) |
|
|
— |
|
|
|
— |
|
|
|
674,494 |
|
|
* |
|
|
* |
|
||
Arie Belldegrun (4) |
|
|
522,479 |
|
|
* |
|
|
|
— |
|
|
|
— |
|
|
* |
|
||
Marijn Dekkers(5) |
|
|
7,902,030 |
|
|
* |
|
|
|
— |
|
|
|
— |
|
|
* |
|
||
Christian Henry (6) |
|
|
1,305,943 |
|
|
* |
|
|
|
— |
|
|
|
— |
|
|
* |
|
||
Reshma Kewalramani |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Shyam Sankar (7) |
|
|
1,260,953 |
|
|
* |
|
|
|
— |
|
|
|
— |
|
|
* |
|
||
Harry E. Sloan |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
All Directors and Executive Officers of Ginkgo as a Group (9 individuals) |
|
|
10,991,405 |
|
|
|
1.0 |
% |
|
|
248,947,330 |
|
|
|
63.5 |
% |
|
|
49.7 |
% |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
5% Beneficial Owners of Ginkgo |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||
Entities affiliated with Anchorage Capital Group(8) |
|
|
69,208,354 |
|
|
|
6.4 |
% |
|
|
— |
|
|
|
— |
|
|
|
1.4 |
% |
Bartholomew Canton(9) |
|
— |
|
|
— |
|
|
|
165,841,730 |
|
|
|
42.4 |
% |
|
|
33.2 |
% |
||
Austin Che(10) |
|
— |
|
|
— |
|
|
|
82,920,866 |
|
|
|
21.2 |
% |
|
|
16.6 |
% |
||
Entities affiliated with Baillie Gifford & Co.(11) |
|
|
118,787,590 |
|
|
|
11.0 |
% |
|
|
— |
|
|
|
— |
|
|
|
2.4 |
% |
Cascade Investment, L.L.C. (12) |
|
|
151,865,481 |
|
|
|
14.1 |
% |
|
|
— |
|
|
|
— |
|
|
|
3.0 |
% |
General Atlantic (GK), L.P.(13) |
|
|
111,566,297 |
|
|
|
10.3 |
% |
|
|
— |
|
|
|
— |
|
|
|
2.2 |
% |
Thomas Knight(14) |
|
|
66,110,869 |
|
|
|
6.1 |
% |
|
|
8,972,183 |
|
|
|
2.3 |
% |
|
|
3.1 |
% |
Senator Global Opportunity Master Fund LP(15) |
|
|
76,595,199 |
|
|
|
7.1 |
% |
|
|
— |
|
|
|
— |
|
|
|
1.5 |
% |
* Less than one percent.
** Percentage of total voting power represents voting power with respect to all shares of Ginkgo Class A common stock and Ginkgo Class B common stock, as a single class. Each share of Ginkgo Class B common stock is entitled to 10 votes per share and each share of Ginkgo Class A common stock is entitled to one vote per share. For more information about the voting rights of Ginkgo common stock, see the description of the Company’s securities filed herewith as Exhibit 4.1.
124
Table of Contents
125
Table of Contents
Securities Authorized for Issuance Under Equity Compensation Plans
The following table sets forth information as of December 31, 2021 regarding shares of common stock that may be issued under our equity compensation plans.
|
|
Number of securities to be issued upon exercise of outstanding options and vesting of outstanding restricted stock units (#) |
|
|
|
|
Weighted-average exercise price of outstanding options ($) |
|
|
Number of securities remaining available for future issuance under equity compensation plans (excluding securities reflected in the first column) (#) |
|
|
||||
Equity compensation plans approved by security holders (1) |
|
|
193,550,805 |
|
|
(2 |
) |
|
$ |
0.05 |
|
|
|
200,569,979 |
|
(3)(4) |
Equity compensation plans not approved by security holders |
|
|
— |
|
|
|
|
|
— |
|
|
|
— |
|
|
|
Total |
|
|
193,550,805 |
|
|
|
|
$ |
0.05 |
|
|
|
200,569,979 |
|
|
|
126
Table of Contents
Item 13. Certain Relationships and Related Transactions, and Director Independence.
CERTAIN RELATIONSHIPS AND RELATED PARTY TRANSACTIONS
SRNG
On October 28, 2020, SRNG’s Sponsor purchased an aggregate of 43,125,000 founder shares in exchange for a capital contribution of $25,000, or approximately $0.0006 per share. SRNG’s Sponsor purchased an aggregate of 19,250,000 Private Placement Warrants in connection with SRNG’s IPO, at a price of $1.50 per warrant, or $28,875,000 in the aggregate. See a description of the Company’s securities is filed herewith as Exhibit 4.1.
SRNG currently sub-leases its executive offices at 955 Fifth Avenue, New York, NY, 10075 from Global Eagle Acquisition LLC, an affiliate of our Sponsor. From the time of SRNG’s IPO until the Closing, SRNG reimbursed Global Eagle Acquisition LLC for office space, secretarial and administrative services provided to members of its management team in an amount that did not exceed $15,000 per month.
Prior to the Closing, SRNG’s officers and directors were entitled to reimbursement for any out-of-pocket expenses incurred in connection with activities on SRNG’s behalf such as identifying potential target businesses and performing due diligence on suitable business combinations. SRNG’s audit committee reviewed on a quarterly basis all payments that were made to SRNG’s Sponsor, SRNG’s officers, directors or its or their affiliates.
On May 10, 2021, an affiliate of SRNG’s Sponsor entered into a Subscription Agreement with the Company, pursuant to which it agreed to purchase an aggregate of 7,500,000 shares of New SRNG Class A common stock at $10.00 per share, for an aggregate purchase price of $75,000,000, from the Company in a private placement to close immediately prior to Closing, but after the Domestication.
Ginkgo
The share numbers included in this “Certain Relationships and Related Party Transactions—Ginkgo” section reflect the historical pre-Business Combination shares issued in connection with the relevant transaction, and have not been revised to reflect the accounting of the Business Combination as a reverse recapitalization.
Series E Preferred Stock Financing
From September 2019 through July 2021, Old Ginkgo sold an aggregate of 1,942,610 shares of its Series E preferred stock to the related persons listed below at a purchase price of $150.19 per share, except as described below with respect to the conversion of convertible promissory notes. The following table summarizes purchases of Series E preferred stock from Old Ginkgo by such related persons:
Name |
|
Shares of Series E Preferred Stock |
|
|
Total Purchase Price (Rounded) |
|
||
Entities affiliated with Anchorage Capital Group(1) |
|
|
105,500 |
|
|
$ |
15,053,014 |
|
Entities affiliated with Baillie Gifford & Co.(2) |
|
|
200,479 |
|
|
$ |
29,104,581 |
|
Cascade Investment, L.L.C.(3) |
|
|
268,376 |
|
|
$ |
38,719,465 |
|
General Atlantic (GK), L.P.(4) |
|
|
513,449 |
|
|
$ |
76,109,274 |
|
Novalis Life Sciences Investments I, LP(5) |
|
|
52,755 |
|
|
$ |
7,527,123 |
|
Senator Global Opportunity Master Fund LP(6) |
|
|
70,489 |
|
|
$ |
10,057,534 |
|
Viking Global Opportunities Illiquid Investments Sub-Master LP(7) |
|
|
731,562 |
|
|
$ |
106,379,679 |
|
127
Table of Contents
Convertible Note Financing
In certain cases, the payment of the Total Purchase Price above consisted or, or included, the conversion of convertible promissory notes held by the related persons. From June 2019 through July 2019, Old Ginkgo sold an aggregate of $160,500,000 in principal amount of convertible promissory notes to the related persons listed below. Interest on the principal amount of the convertible promissory notes accrued at the rate of 3.0% per year. The outstanding principal and accrued interest of such convertible promissory notes converted into shares of Series E preferred stock at a discounted purchase price of $142.68 per share and are reflected in the above table. The following table summarizes the convertible promissory notes issued by Old Ginkgo to such related persons:
Name |
|
Principal |
|
|
Entities affiliated with Anchorage Capital Group(1) |
|
$ |
15,000,000 |
|
Entities affiliated with Baillie Gifford & Co.(2) |
|
$ |
19,000,000 |
|
Cascade Investment, L.L.C.(3) |
|
$ |
30,000,000 |
|
General Atlantic (GK), L.P.(4) |
|
$ |
19,000,000 |
|
Novalis Life Sciences Investments I, LP(5) |
|
$ |
7,500,000 |
|
Senator Global Opportunity Master Fund LP(6) |
|
$ |
10,000,000 |
|
Viking Global Opportunities Illiquid Investments Sub-Master LP(7) |
|
$ |
60,000,000 |
|
Series D Preferred Stock Financing
In June 2018, Old Ginkgo sold 52,400 shares of its Series D preferred stock at a purchase price of $47.71 per share for an aggregate purchase price of $2,500,004 to Novalis LifeSciences. Marijn Dekkers, a member of Ginkgo’s board of directors, is an affiliate of Novalis LifeSciences.
Founder Equity Grant Agreements
In January 2020, Old Ginkgo entered into Founder Equity Grant Agreements with each of Jason Kelly, Reshma Shetty, Austin Che and Bartholomew Canton. Dr. Kelly and Dr. Shetty are each a director, officer and holder of more than 5% of Ginkgo’s outstanding capital stock. Dr. Canton and Dr. Che are each a holder of more than 5% of Ginkgo’s outstanding capital stock. Dr. Canton is also a family member of Dr. Shetty. Also in January 2020, each of Dr. Kelly, Dr. Shetty, Dr. Canton and Dr. Che received a restricted stock unit award under the 2014 Plan. The terms of the Founder Equity Grants Agreements and the foregoing restricted stock unit awards are described in the section titled “Ginkgo’s Executive and Director Compensation” in this Annual Report.
Founder Equity Repurchases
In July 2018, Old Ginkgo repurchased 90,017 shares of common stock from each of Jason Kelly, Reshma Shetty, Austin Che, Bartholomew Canton and Thomas Knight at a price of $47.71 per share, which was the then most-recent price per share at which Old Ginkgo had sold convertible preferred stock to investors, for a total purchase price for each of $4,294,711. In September 2021, Old Ginkgo repurchased 11,032 shares of common stock from each of Jason Kelly, Reshma Shetty, Austin Che, Bartholomew Canton and Thomas Knight at a price of $453.20 per share, for a total purchase price for each of $4,999,702. Dr. Kelly and Dr. Shetty are each a director, officer and holder of more than 5% of Ginkgo’s outstanding capital
128
Table of Contents
stock. Dr. Che, Dr. Canton and Dr. Knight are each a holder of more than 5% of Ginkgo’s outstanding capital stock. Dr. Canton is also a family member of Dr. Shetty.
Agreements in Connection with Platform Ventures
In September 2019, Old Ginkgo entered into an agreement with Cascade Investment L.L.C., an affiliated entity of GA GK, and Viking Global Opportunities Illiquid Investments Sub-Master LP, each of which hold more than 5% of Ginkgo’s outstanding capital stock, pursuant to which such related persons were provided with the first right to invest up to an aggregate of $350.0 million for the financial investment portion of new companies launched by Old Ginkgo as part of its Platform Ventures (such as Allonnia). The agreement was terminated in May 2021. Initial investments in new companies launched by Old Ginkgo in connection with the agreement were approximately $12.9 million from Cascade Investment L.L.C., $19.5 million from entities affiliated with GA GK., and $57.8 million from entities affiliated with Viking Global Opportunities Illiquid Investments Sub-Master LP.
In September 2021, Old Ginkgo formed 2 new Platform Ventures (Ayana Bio, LLC and Verb Biotics, LLC) and contributed intellectual property rights to these entities. Simultaneously with the arrangements between Old Ginkgo and each of Ayana Bio, LLC and Verb Biotics, LLC, Cascade Investment L.L.C. and entities affiliated with Viking Global Opportunities Illiquid Investments Sub-Master LP, each of which hold more than 5% of Ginkgo’s outstanding capital stock, invested approximately $7.5 million and $22.5 million, respectively, in each company.
Exchange Agreement
In October 2021 and March 2022, Ginkgo entered into a first and second amended and restated stockholders agreement, respectively, with Viking Global Opportunities Illiquid Investments Sub-Master LP, a holder of more than 5% of Ginkgo’s outstanding capital stock at the time of each agreement, pursuant to which Ginkgo agreed, subject to approval of the Ginkgo Board, to permit such stockholder to exchange a portion of its shares of Ginkgo Class A common stock for shares of Ginkgo Class C common stock on a 1-for-1 basis. The Ginkgo Board approved the exchange and Viking Global Opportunities Illiquid Investments Sub-Master LP effected the exchange in March 2022.
Investors’ Rights Agreement
Old Ginkgo was party to the Third Amended and Restated Investors’ Rights Agreement, dated as of September 9, 2019, which granted registration rights and information rights, among other things, to certain holders of its capital stock, including (i) entities affiliated with Anchorage, entities affiliated with Baillie Gifford & Co., Cascade Investment, L.L.C, GA GK, L.P., and Viking Global Opportunities Illiquid Investments Sub-Master LP, each of which currently holds more than 5% of Ginkgo’s capital stock; (ii) Novalis LifeSciences, which is affiliated with Marijn Dekkers, a Ginkgo director; and (iii) Senator Global Opportunity Master Fund LP, which currently holds more than 5% of Ginkgo’s capital stock and is affiliated with Evan Lodes, a former Old Ginkgo director. This agreement was terminated upon the Closing.
Right of First Refusal and Co-Sale Agreement
Old Ginkgo was party to the Third Amended and Restated Right of First Refusal and Co-Sale Agreement, dated as of September 9, 2019, which granted the right to purchase shares of Old Ginkgo capital stock which certain other stockholders propose to sell to other parties to, among others (i) entities affiliated with Anchorage, entities affiliated with Baillie Gifford & Co., Cascade Investment, L.L.C, GA GK, and Viking Global Opportunities Illiquid Investments Sub-Master LP, each of which currently hold more than 5% of Ginkgo’s capital stock; (ii) Novalis LifeSciences, which is affiliated with Marijn Dekkers, a Ginkgo director; and (iii) Senator Global Opportunity Master Fund LP, which currently holds more than 5% of Ginkgo’s capital stock and is affiliated with Evan Lodes, a former Old Ginkgo director. This agreement was terminated upon the Closing.
Voting Agreement
Old Ginkgo was party to the Third Amended and Restated Voting Agreement, dated as of September 9, 2019, pursuant to which certain holders of its capital stock, including (i) entities affiliated with Anchorage, entities affiliated with Baillie Gifford & Co., Cascade Investment, L.L.C, GA GK, and Viking Global Opportunities Illiquid Investments Sub-Master LP, each of which currently hold more than 5% of Ginkgo’s capital stock; (ii) Novalis LifeSciences, which is affiliated with Marijn Dekkers, a Ginkgo director; and (iii) Senator Global Opportunity Master Fund LP, which currently holds more than 5% of Ginkgo’s capital stock and is affiliated with Evan Lodes, a former Old Ginkgo director, agreed to vote their shares in the manner directed by the agreement. This agreement was terminated upon the Closing.
129
Table of Contents
Director and Officer Indemnification
Ginkgo’s Charter authorizes indemnification and advancement of expenses for its directors and officers to the fullest extent permitted by the DGCL.
PIPE Investment
In May 2021, certain Old Ginkgo related persons entered into Subscription Agreements with SRNG in connection with the Private Placement. The following table summarizes purchases in the Private Placement by such related persons:
Name |
|
SRNG Class A |
|
|
Total Purchase |
|
||
Entities affiliated with Baillie Gifford & Co.(1) |
|
|
10,300,000 |
|
|
$ |
103,000,000 |
|
Cascade Investment, L.L.C.(2) |
|
|
3,000,000 |
|
|
$ |
30,000,000 |
|
General Atlantic (GK), L.P.(3) |
|
|
250,000 |
|
|
$ |
2,500,000 |
|
Senator Global Opportunity Master Fund LP(4) |
|
|
700,000 |
|
|
$ |
7,000,000 |
|
Viking Global Opportunities Illiquid Investments Sub-Master LP(5) |
|
|
2,000,000 |
|
|
$ |
20,000,000 |
|
Policies and Procedures for Related Person Transactions
Our written related person transaction policy sets forth the following policies and procedures for the review and approval or ratification of related person transactions.
A “Related Person Transaction” is a material transaction, arrangement or relationship (or any series of similar transactions, arrangements or relationships) in which the Company (including any of its subsidiaries) was, is or will be a participant and in which any related person had, has or will have a direct or indirect material interest. A transaction involving an amount exceeding $120,000 is presumed to be a “material transaction.” A “Related Person” means:
We have policies and procedures designed to minimize potential conflicts of interest arising from any dealings we may have with our affiliates and to provide appropriate procedures for the disclosure of any real or potential conflicts of interest that may exist from time to time. Specifically, pursuant to its charter, the audit committee has the responsibility to review related person transactions.
130
Table of Contents
Independence of the Board of Directors
NYSE rules generally require that independent directors must comprise a majority of a listed company’s board of directors. Based upon information requested from and provided by each director concerning his or her background, employment and affiliations, including family relationships, we have determined that each of Arie Belldegrun, Christian Henry, Reshma Kewalramani, Shyam Sankar and Harry Sloan, representing a majority of Ginkgo’s directors, are “independent” as that term is defined under the applicable rules and regulations of the SEC and the listing requirements and rules of the NYSE.
131
Table of Contents
Item 14. Principal Accounting Fees and Services.
Our independent public accounting firm is Ernst & Young LLP, Boston, Massachusetts (PCAOB Auditor ID: 42).
Withum Smith+Brown, P.C. (“Withum”) served as the independent registered public accounting firm for Soaring Eagle Acquisition Corp. with respect to the audit of the 2020 financial statements of Soaring Eagle Acquisition Corp. Withum was replaced by Ernst & Young LLP following the consummation of the business combination between SEAC Merger Sub Inc., a wholly owned subsidiary of Soaring Eagle Acquisition Corp., and us on September 16, 2021.
The following table provides a summary of the aggregate fees incurred for Ernst & Young LLP’s services for 2021 and for Withum's services for the period from October 22, 2020 (inception) through December 31, 2020:
|
Fiscal year ended December 31, |
|
|||||
|
2021 |
|
|
2020 |
|
||
Audit fees (a) |
$ |
4,017,109 |
|
|
$ |
40,000 |
|
Audit-related fees (b) |
|
— |
|
|
|
— |
|
Tax fees (c) |
|
321,794 |
|
|
|
— |
|
All other fees (d) |
|
— |
|
|
|
— |
|
Total fees |
$ |
4,338,903 |
|
|
$ |
40,000 |
|
Audit committee pre-approval policy and procedures
All of the fees described above were pre-approved by the audit committee. The audit committee is responsible for the appointment, compensation and oversight of the work of the independent auditor. The audit committee has adopted a pre-approval policy under which the audit committee pre-approves the audit and non-audit services performed by the independent auditor. Proposed services may either be pre-approved within categories presented to the audit committee that include a detailed description of the specific services within such categories along with the budgeted fees or on a case-by-case basis for specific services not contemplated in the original pre-approved categories. The audit committee will, at least annually, review and pre-approve the categories of services (if any).
Finally, in accordance with the pre-approval policy, the audit committee may delegate pre-approval authority to each of its members. Any member to whom this authority is delegated must report any pre-approval decisions to the audit committee at its next meeting.
132
Table of Contents
PART IV
Item 15. Exhibits, Financial Statement Schedules.
Exhibit Number |
Description |
2.1 |
|
2.2 |
|
3.1 |
|
3.2 |
|
3.3 |
|
4.1 |
|
4.2* |
|
4.3 |
|
4.4* |
|
10.1+ |
|
10.2+ |
|
10.3 |
133
Table of Contents
10.4 |
|
10.5 |
|
10.6 |
|
10.7 |
|
10.8 |
|
10.9 |
|
10.10 |
|
10.11 |
|
10.12 |
|
10.13 |
|
10.14†‡ |
|
10.15† |
|
10.16† |
|
10.17 |
134
Table of Contents
10.18† |
|
10.19† |
|
10.20 |
|
10.21† |
|
10.22† |
|
10.23† |
|
10.24† |
|
10.25† |
|
10.26† |
|
10.27† |
|
10.28†‡ |
|
10.29† |
|
10.30†‡ |
|
10.31† |
135
Table of Contents
10.32‡ |
|
10.33 |
|
10.34 |
|
10.35 |
|
10.36 |
|
10.37 |
|
16.1 |
|
21.1 |
|
23.1* |
Consent of Ernst & Young LLP, Independent Registered Public Accounting Firm |
31.1* |
|
31.2* |
|
32.1* |
|
32.2* |
|
101.INS |
Inline XBRL Instance Document – the instance document does not appear in the Interactive Data File because XBRL tags are embedded within the Inline XBRL document. |
101.SCH |
Inline XBRL Taxonomy Extension Schema Document. |
101.CAL |
Inline XBRL Taxonomy Extension Calculation Linkbase Document. |
101.DEF |
Inline XBRL Taxonomy Extension Definition Linkbase Document. |
101.LAB |
Inline XBRL Taxonomy Extension Label Linkbase Document. |
101.PRE |
Inline XBRL Taxonomy Extension Presentation Linkbase Document. |
104 |
Cover Page Interactive Data File (embedded within the Inline XBRL document). |
136
Table of Contents
* Filed herewith.
† The annexes, schedules, and certain exhibits to this Exhibit have been omitted pursuant to Item 601(b)(2) of Regulation S-K. The Registrant hereby agrees to furnish supplementally a copy of any omitted annex, schedule or exhibit to the SEC upon request.
‡ Certain confidential information contained in this Exhibit has been omitted because it is (i) not material and (ii) of the type that the registrant treats as private or confidential.
+ Indicates a management contract of compensatory plan.
Item 16. Form 10-K Summary
None.
137
Table of Contents
SIGNATURES
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended, the Registrant has duly caused this Report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
GINKGO BIOWORKS HOLDINGS, INC. |
|
|
|
|
|
Date: March 28, 2022 |
|
By: |
/s/ Jason Kelly |
|
|
|
Jason Kelly |
|
|
|
Chief Executive Officer |
Pursuant to the requirements of the Securities Exchange Act of 1934, as amended, this Report has been signed below by the following persons on behalf of the Registrant in the capacities and on the dates indicated.
Name |
|
Title |
|
Date |
|
|
|
|
|
/s/ Jason Kelly |
|
Chief Executive Officer and Director |
|
March 28, 2022 |
Jason Kelly |
|
(Principal Executive Officer) |
|
|
|
|
|
|
|
/s/ Mark Dmytruk |
|
Chief Financial Officer |
|
March 28, 2022 |
Mark Dmytruk |
|
(Principal Financial Officer) |
|
|
|
|
|
|
|
/s/ Marie Fallon |
|
Chief Accounting Officer |
|
March 28, 2022 |
Marie Fallon |
|
(Principal Accounting Officer) |
|
|
|
|
|
|
|
/s/ Marijn Dekkers |
|
Director, Chair of the Board |
|
March 28, 2022 |
Marijn Dekkers |
|
|
|
|
|
|
|
|
|
/s/ Arie Belldegrun |
|
Director |
|
March 28, 2022 |
Arie Belldegrun |
|
|
|
|
|
|
|
|
|
/s/ Christian Henry |
|
Director |
|
March 28, 2022 |
Christian Henry |
|
|
|
|
|
|
|
|
|
/s/ Reshma Kewalramani |
|
Director |
|
March 28, 2022 |
Reshma Kewalramani |
|
|
|
|
|
|
|
|
|
/s/ Shyam Sankar |
|
Director |
|
March 28, 2022 |
Shyam Sankar |
|
|
|
|
|
|
|
|
|
/s/ Harry E. Sloan |
|
Director |
|
March 28, 2022 |
Harry E. Sloan |
|
|
|
|
|
|
|
|
|
/s/ Reshma Shetty |
|
President, Chief Operating Officer and Director |
|
March 28, 2022 |
Reshma Shetty |
|
|
|
|
138
Table of Contents
Ginkgo Bioworks HOLDINGS, Inc. and Subsidiaries
Index to Consolidated Financial Statements as of December 31, 2021 and 2020 and for the Years Ended December 31, 2021, 2020 and 2019
|
Page
|
Report of Independent Registered Public Accounting Firm (PCAOB ID: 42) |
F-2 |
F-3 |
|
Consolidated Statements of Operations and Comprehensive Loss |
F-4 |
F-5 |
|
F-7 |
|
F-9 |
F-1
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Stockholders and the Board of Directors of Ginkgo Bioworks Holdings, Inc.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Ginkgo Bioworks Holdings, Inc. and subsidiaries (the Company) as of December 31, 2021 and 2020, the related consolidated statements of operations and comprehensive loss, stockholders’ equity and cash flows for each of the three years in the period ended December 31, 2021, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2021 and 2020, and the results of its operations and its cash flows for each of the three years in the period ended December 31, 2021, in conformity with U.S. generally accepted accounting principles.
Basis for Opinion
These financial statements are the responsibility of the Company's management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ Ernst & Young LLP
We have served as the Company’s auditor since 2018.
Boston, Massachusetts
March 28, 2022
F-2
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Consolidated Balance Sheets
(in thousands, except share and per share data)
|
|
As of December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Assets |
|
|
|
|
|
|
||
Current assets: |
|
|
|
|
|
|
||
Cash and cash equivalents |
|
$ |
1,550,004 |
|
|
$ |
380,801 |
|
Accounts receivable, net |
|
|
131,544 |
|
|
|
16,694 |
|
Accounts receivable - related parties |
|
|
4,598 |
|
|
|
5,212 |
|
Inventory, net |
|
|
3,362 |
|
|
|
2,736 |
|
Prepaid expenses and other current assets |
|
|
33,537 |
|
|
|
21,099 |
|
Total current assets |
|
|
1,723,045 |
|
|
|
426,542 |
|
Property and equipment, net |
|
|
145,770 |
|
|
|
121,435 |
|
Investments |
|
|
102,037 |
|
|
|
74,200 |
|
Equity method investments |
|
|
13,194 |
|
|
|
28,924 |
|
Intangible assets, net |
|
|
21,642 |
|
|
|
3,294 |
|
Goodwill |
|
|
21,312 |
|
|
|
1,857 |
|
Loans receivable, net of current portion |
|
|
— |
|
|
|
13,298 |
|
Other non-current assets |
|
|
43,990 |
|
|
|
5,603 |
|
Total assets |
|
$ |
2,070,990 |
|
|
$ |
675,153 |
|
Liabilities and Stockholders’ Equity |
|
|
|
|
|
|
|
|
Current liabilities: |
|
|
|
|
|
|
|
|
Accounts payable |
|
$ |
8,189 |
|
|
$ |
13,893 |
|
Deferred revenue (includes $12,502 and $22,101 from related parties) |
|
|
33,240 |
|
|
|
28,823 |
|
Accrued expenses and other current liabilities |
|
|
93,332 |
|
|
|
30,505 |
|
Total current liabilities |
|
|
134,761 |
|
|
|
73,221 |
|
Non-current liabilities: |
|
|
|
|
|
|
|
|
Deferred rent, net of current portion |
|
|
18,746 |
|
|
|
12,678 |
|
Deferred revenue, net of current portion (includes $148,319 and $97,977 from related parties) |
|
|
155,991 |
|
|
|
99,652 |
|
Lease financing obligation |
|
|
22,283 |
|
|
|
16,518 |
|
Warrant liabilities |
|
|
135,838 |
|
|
|
— |
|
Other non-current liabilities |
|
|
35,992 |
|
|
|
3,032 |
|
Total liabilities |
|
|
503,611 |
|
|
|
205,101 |
|
|
|
|
|
|
|
|
||
Stockholders’ equity (1): |
|
|
|
|
|
|
|
|
Preferred stock, $0.0001 par value; 200,000,000 shares authorized; none issued |
|
|
— |
|
|
|
— |
|
Class , Class B and Class C common stock $0.0001 par value; 15,800,000,000 shares authorized (Class A 10,500,000,000, Class B 4,500,000,000, Class C 800,000,000); 1,690,990,815 (Class A 1,326,146,808, Class B 364,844,007, Class C 0) and 1,289,014,925 (Class A 974,224,443, Class B 314,790,482, Class C 0) shares issued as of December 31, 2021 and 2020, respectively; 1,611,392,152 (Class A 1,273,976,963, Class B 337,415,189, Class C 0) and 1,288,595,876 (Class A 974,166,577, Class B 314,429,299, Class C 0) shares outstanding as of December 31, 2021 and 2020, respectively |
|
|
161 |
|
|
|
129 |
|
Additional paid-in capital |
|
|
3,804,844 |
|
|
|
929,125 |
|
Accumulated deficit |
|
|
(2,297,925 |
) |
|
|
(467,878 |
) |
Accumulated other comprehensive loss |
|
|
(1,715 |
) |
|
|
— |
|
Total Ginkgo Bioworks Holdings, Inc. stockholders’ equity |
|
|
1,505,365 |
|
|
|
461,376 |
|
Non-controlling interest |
|
|
62,014 |
|
|
|
8,676 |
|
Total stockholders’ equity |
|
|
1,567,379 |
|
|
|
470,052 |
|
Total liabilities and stockholders’ equity |
|
$ |
2,070,990 |
|
|
$ |
675,153 |
|
(1) |
Balances as of December 31, 2020 have been retroactively restated for the reverse recapitalization as described in Note 2. |
The accompanying notes are an integral part of these consolidated financial statements.
F-3
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Consolidated Statements of Operations and Comprehensive Loss
(in thousands, except share and per share data)
|
|
Year Ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Foundry revenue (related party revenue of $47,161, $42,535 and $35,268 for the years ended 2021, 2020 and 2019, respectively) |
|
$ |
112,989 |
|
|
$ |
59,221 |
|
|
$ |
54,184 |
|
Biosecurity revenue: |
|
|
|
|
|
|
|
|
|
|
||
Product |
|
|
23,040 |
|
|
|
8,707 |
|
|
|
— |
|
Service |
|
|
177,808 |
|
|
|
8,729 |
|
|
|
— |
|
Total revenue |
|
|
313,837 |
|
|
|
76,657 |
|
|
|
54,184 |
|
Costs and operating expenses: |
|
|
|
|
|
|
|
|
|
|
||
Cost of Biosecurity product revenue |
|
|
20,017 |
|
|
|
6,705 |
|
|
|
— |
|
Cost of Biosecurity service revenue |
|
|
109,673 |
|
|
|
8,906 |
|
|
|
— |
|
Research and development |
|
|
1,149,662 |
|
|
|
159,767 |
|
|
|
96,299 |
|
General and administrative |
|
|
862,952 |
|
|
|
38,306 |
|
|
|
29,483 |
|
Total operating expenses |
|
|
2,142,304 |
|
|
|
213,684 |
|
|
|
125,782 |
|
Loss from operations |
|
|
(1,828,467 |
) |
|
|
(137,027 |
) |
|
|
(71,598 |
) |
Other (expense) income: |
|
|
|
|
|
|
|
|
|
|
||
Interest income |
|
|
837 |
|
|
|
2,582 |
|
|
|
5,756 |
|
Interest expense |
|
|
(2,373 |
) |
|
|
(2,385 |
) |
|
|
(2,421 |
) |
Loss on equity method investments |
|
|
(77,284 |
) |
|
|
(396 |
) |
|
|
(27,533 |
) |
Loss on investments |
|
|
(11,543 |
) |
|
|
(3,733 |
) |
|
|
(27,200 |
) |
Change in fair value of warrant liabilities |
|
|
58,615 |
|
|
|
— |
|
|
|
— |
|
Gain on settlement of partnership agreement |
|
|
23,826 |
|
|
|
8,286 |
|
|
|
1,587 |
|
Other (expense) income, net |
|
|
(1,733 |
) |
|
|
7,839 |
|
|
|
1,574 |
|
Total other (expense) income, net |
|
|
(9,655 |
) |
|
|
12,193 |
|
|
|
(48,237 |
) |
Loss before income taxes |
|
|
(1,838,122 |
) |
|
|
(124,834 |
) |
|
|
(119,835 |
) |
Income tax (benefit) provision |
|
|
(1,480 |
) |
|
|
1,889 |
|
|
|
22 |
|
Net loss |
|
|
(1,836,642 |
) |
|
|
(126,723 |
) |
|
|
(119,857 |
) |
Net loss attributable to non-controlling interest |
|
|
(6,595 |
) |
|
|
(114 |
) |
|
|
(530 |
) |
Net loss attributable to Ginkgo Bioworks Holdings, Inc. stockholders |
|
$ |
(1,830,047 |
) |
|
$ |
(126,609 |
) |
|
$ |
(119,327 |
) |
Net loss per share attributable to Ginkgo Bioworks Holdings, Inc. (1) common stockholders: |
|
|
|
|
|
|
|
|
|
|||
Basic |
|
$ |
(1.35 |
) |
|
$ |
(0.10 |
) |
|
$ |
(0.10 |
) |
Diluted |
|
$ |
(1.39 |
) |
|
$ |
(0.10 |
) |
|
$ |
(0.10 |
) |
Weighted average common shares outstanding (1) |
|
|
|
|
|
|
|
|
|
|||
Basic |
|
|
1,359,848,803 |
|
|
|
1,274,766,915 |
|
|
|
1,149,000,417 |
|
Diluted |
|
|
1,360,373,343 |
|
|
|
1,274,766,915 |
|
|
|
1,149,000,417 |
|
Comprehensive loss: |
|
|
|
|
|
|
|
|
|
|||
Net loss |
|
$ |
(1,836,642 |
) |
|
$ |
(126,723 |
) |
|
$ |
(119,857 |
) |
Other comprehensive loss: |
|
|
|
|
|
|
|
|
|
|||
Foreign currency translation adjustment |
|
|
(1,715 |
) |
|
|
|
|
|
|
||
Total other comprehensive loss |
|
|
(1,715 |
) |
|
|
|
|
|
|
||
Comprehensive loss |
|
$ |
(1,838,357 |
) |
|
$ |
(126,723 |
) |
|
$ |
(119,857 |
) |
(1) |
Amounts for the year ended December 31, 2020 and 2019 have been retroactively restated for the reverse recapitalization as described in Note 2. |
The accompanying notes are an integral part of these consolidated financial statements.
F-4
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Consolidated Statements of Stockholders’ Equity
(in thousands, except share data)
|
|
Series B, C, D, E Convertible |
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||
|
|
Preferred Stock |
|
|
Old Ginkgo Common Stock |
|
|
Common Stock |
|
|||||||||||||||
|
|
Shares |
|
|
Amount |
|
|
Shares |
|
|
Amount |
|
|
Shares |
|
|
Amount |
|
||||||
Balance as of December 31, 2018 (as previously reported) |
|
|
14,943,599 |
|
|
$ |
149 |
|
|
|
8,555,710 |
|
|
$ |
86 |
|
|
|
— |
|
|
$ |
— |
|
Retroactive application of the reverse recapitalization |
|
|
(14,943,599 |
) |
|
|
(149 |
) |
|
|
(8,555,710 |
) |
|
|
(86 |
) |
|
|
1,153,356,703 |
|
|
|
116 |
|
Balance as of December 31, 2018 |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,153,356,703 |
|
|
|
116 |
|
Exercise of stock options |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
500,621 |
|
|
|
— |
|
Issuance of Series E convertible preferred stock, net of issuance costs of $4,830 |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
69,812,427 |
|
|
|
7 |
|
Beneficial conversion feature of convertible promissory notes |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Conversion of convertible promissory notes into Series E convertible preferred stock |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
69,151,117 |
|
|
|
7 |
|
Vesting of restricted stock awards |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
367,858 |
|
|
|
— |
|
Repurchase of common stock |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Retirement of treasury stock |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(37,626,694 |
) |
|
|
(4 |
) |
Issuance of warrants to purchase convertible preferred stock |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Net loss and comprehensive loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Balance as of December 31, 2019 |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,255,562,032 |
|
|
|
126 |
|
Exercise of stock options |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,921,941 |
|
|
|
— |
|
Issuance of Series E convertible preferred stock, net of issuance costs of $0 |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
30,855,065 |
|
|
|
3 |
|
Vesting of restricted stock awards |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
256,838 |
|
|
|
— |
|
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Net loss and comprehensive loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Balance as of December 31, 2020 |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,288,595,876 |
|
|
|
129 |
|
Issuance of common stock upon exercise or vesting of equity awards |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
91,080,290 |
|
|
|
9 |
|
Vesting of restricted stock - earnouts |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
38,798,801 |
|
|
|
4 |
|
Tax withholdings related to net share settlement of equity awards |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(797,313 |
) |
|
|
— |
|
Founder shares repurchase |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
(2,707,280 |
) |
|
|
— |
|
Issuance of warrants to purchase Series D convertible preferred stock |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Issuance of Series D and B convertible preferred stock upon exercise of warrants |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,013,708 |
|
|
|
— |
|
Issuance of Series E convertible preferred stock in exchange for warrants |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
408,497 |
|
|
|
— |
|
Issuance of common stock for a business acquisition |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
1,633,937 |
|
|
|
— |
|
Issuance of common stock upon reverse recapitalization, net of offering costs (Note 3) |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
193,365,636 |
|
|
|
19 |
|
Assumption of Public and Private Placement Warrants |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Contributions from non-controlling interests |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Foreign currency translation |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
— |
|
Balance as of December 31, 2021 |
|
|
— |
|
|
$ |
— |
|
|
|
— |
|
|
$ |
— |
|
|
|
1,611,392,152 |
|
|
$ |
161 |
|
The accompanying notes are an integral part of these consolidated financial statements.
F-5
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Consolidated Statements of Stockholders’ Equity
(in thousands, except share data)
|
|
Treasury Stock |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||||
|
|
Shares |
|
|
Amount |
|
|
Additional |
|
|
|
Accumulated |
|
|
|
Accumulated |
|
|
|
Non- |
|
|
|
Total |
|
|||||||||||
Balance as of December 31, 2018 (as previously reported) |
|
|
(756,633 |
) |
|
$ |
(24,449 |
) |
|
$ |
450,268 |
|
|
|
$ |
(221,942 |
) |
|
|
$ |
— |
|
|
|
$ |
9,320 |
|
|
|
$ |
213,432 |
|
||||
Retroactive application of the reverse recapitalization |
|
|
(36,379,256 |
) |
|
|
(4 |
) |
|
|
123 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
||||
Balance as of December 31, 2018 |
|
|
(37,135,889 |
) |
|
|
(24,453 |
) |
|
|
450,391 |
|
|
|
|
(221,942 |
) |
|
|
|
— |
|
|
|
|
9,320 |
|
|
|
|
213,432 |
|
||||
Exercise of stock options |
|
|
— |
|
|
|
— |
|
|
|
7 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
7 |
|
||||
Issuance of Series E convertible preferred stock, net of issuance costs of $4,830 |
|
|
— |
|
|
|
— |
|
|
|
208,794 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
208,801 |
|
||||
Beneficial conversion feature of convertible promissory notes |
|
|
— |
|
|
|
— |
|
|
|
(12,651 |
) |
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
(12,651 |
) |
||||
Conversion of convertible promissory notes into Series E convertible preferred stock |
|
|
— |
|
|
|
— |
|
|
|
211,601 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
211,608 |
|
||||
Vesting of restricted stock awards |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
||||
Repurchase of common stock |
|
|
(490,805 |
) |
|
|
(408 |
) |
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
(408 |
) |
||||
Retirement of treasury stock |
|
|
37,626,694 |
|
|
|
24,861 |
|
|
|
(24,857 |
) |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
— |
|
|||||||
Issuance of warrants to purchase convertible preferred stock |
|
|
— |
|
|
|
— |
|
|
|
150 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
150 |
|
||||
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
771 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
771 |
|
||||
Net loss and comprehensive loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
(119,327 |
) |
|
|
|
— |
|
|
|
|
(530 |
) |
|
|
|
(119,857 |
) |
||||
Balance as of December 31, 2019 |
|
|
— |
|
|
|
— |
|
|
|
834,206 |
|
|
|
|
(341,269 |
) |
|
|
|
— |
|
|
|
|
8,790 |
|
|
|
|
501,853 |
|
||||
Exercise of stock options |
|
|
— |
|
|
|
— |
|
|
|
26 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
26 |
|
||||
Issuance of Series E convertible preferred stock, net of issuance costs of $0 |
|
|
— |
|
|
|
— |
|
|
|
94,417 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
94,420 |
|
||||
Vesting of restricted stock awards |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
||||
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
476 |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
— |
|
|
|
|
476 |
|
||||
Net loss and comprehensive loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
(126,609 |
) |
|
|
|
— |
|
|
|
|
(114 |
) |
|
|
|
(126,723 |
) |
||||
Balance as of December 31, 2020 |
|
|
— |
|
|
|
— |
|
|
|
929,125 |
|
|
|
|
(467,878 |
) |
|
|
|
|
|
|
|
8,676 |
|
|
|
|
470,052 |
|
|||||
Issuance of common stock upon exercise or vesting of equity awards |
|
|
— |
|
|
|
— |
|
|
|
167 |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
176 |
|
Vesting of restricted stock - earnouts |
|
|
— |
|
|
|
— |
|
|
|
(4 |
) |
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
Tax withholdings related to net share settlement of equity awards |
|
|
— |
|
|
|
— |
|
|
|
(9,463 |
) |
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
(9,463 |
) |
Founder shares repurchase |
|
|
— |
|
|
|
— |
|
|
|
(24,998 |
) |
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
(24,998 |
) |
Issuance of warrants to purchase Series D convertible preferred stock |
|
|
— |
|
|
|
— |
|
|
|
300 |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
300 |
|
Issuance of Series D and B convertible preferred stock upon exercise of warrants |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
Issuance of Series E convertible preferred stock in exchange for warrants |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
Issuance of common stock for a business acquisition |
|
|
— |
|
|
|
— |
|
|
|
15,160 |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
15,160 |
|
Issuance of common stock upon reverse recapitalization, net of offering costs (Note 3) |
|
|
— |
|
|
|
— |
|
|
|
1,509,610 |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
1,509,629 |
|
Assumption of Public and Private Placement Warrants |
|
|
— |
|
|
|
— |
|
|
|
(194,453 |
) |
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
(194,453 |
) |
Contributions from non-controlling interests |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
59,933 |
|
|
|
|
|
59,933 |
|
Stock-based compensation expense |
|
|
— |
|
|
|
— |
|
|
|
1,579,400 |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
1,579,400 |
|
Foreign currency translation |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
— |
|
|
|
|
|
(1,715 |
) |
|
|
|
|
|
|
|
|
|
(1,715 |
) |
|
Net loss |
|
|
— |
|
|
|
— |
|
|
|
— |
|
|
|
|
|
(1,830,047 |
) |
|
|
|
|
— |
|
|
|
|
|
(6,595 |
) |
|
|
|
|
(1,836,642 |
) |
Balance as of December 31, 2021 |
|
|
— |
|
|
$ |
— |
|
|
$ |
3,804,844 |
|
|
|
|
$ |
(2,297,925 |
) |
|
|
|
$ |
(1,715 |
) |
|
|
|
$ |
62,014 |
|
|
|
|
$ |
1,567,379 |
|
The accompanying notes are an integral part of these consolidated financial statements.
F-6
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Consolidated Statements of Cash Flows
(in thousands)
|
|
Year Ended December 31, |
|
|||||||||||||
|
|
2021 |
|
|
|
2020 |
|
|
|
2019 |
|
|||||
Cash flows from operating activities: |
|
|
|
|
|
|
|
|
|
|
|
|||||
Net loss |
|
$ |
(1,836,642 |
) |
|
|
|
$ |
(126,723 |
) |
|
|
|
$ |
(119,857 |
) |
Adjustments to reconcile net loss to net cash used in operating activities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||
Depreciation and amortization |
|
|
29,076 |
|
|
|
|
|
13,864 |
|
|
|
|
|
10,755 |
|
Stock-based compensation |
|
|
1,606,020 |
|
|
|
|
|
476 |
|
|
|
|
|
771 |
|
Non-cash equity consideration |
|
|
(24,185 |
) |
|
|
|
|
— |
|
|
|
|
|
— |
|
Loss on equity method investments |
|
|
77,284 |
|
|
|
|
|
396 |
|
|
|
|
|
27,533 |
|
Loss on investments |
|
|
11,543 |
|
|
|
|
|
3,733 |
|
|
|
|
|
27,200 |
|
Change in fair value of loans receivable |
|
|
3,508 |
|
|
|
|
|
(1,061 |
) |
|
|
|
|
(914 |
) |
Change in fair value of warrant liabilities |
|
|
(58,615 |
) |
|
|
|
|
— |
|
|
|
|
|
— |
|
Other non-cash activity |
|
|
(270 |
) |
|
|
|
— |
|
|
|
|
(728 |
) |
||
Changes in operating assets and liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Accounts receivable ($614, ($995) and ($2,221) from related parties) |
|
|
(114,094 |
) |
|
|
|
|
(14,228 |
) |
|
|
|
|
(1,843 |
) |
Prepaid expenses and other current assets |
|
|
(2,981 |
) |
|
|
|
|
(11,352 |
) |
|
|
|
|
(4,031 |
) |
Inventory |
|
|
(626 |
) |
|
|
|
|
(2,736 |
) |
|
|
|
|
|
|
Other non-current assets |
|
|
(539 |
) |
|
|
|
|
1,834 |
|
|
|
|
|
(2,361 |
) |
Accounts payable |
|
|
(2,247 |
) |
|
|
|
|
7,019 |
|
|
|
|
|
664 |
|
Accrued expenses and other current liabilities |
|
|
44,796 |
|
|
|
|
|
8,665 |
|
|
|
|
|
4,170 |
|
Deferred revenue, current and non-current ($40,743, ($22,253) and $3,112 from related parties) |
|
|
(10,498 |
) |
|
|
|
|
(19,423 |
) |
|
|
|
|
4,883 |
|
Deferred rent, non-current |
|
|
6,032 |
|
|
|
|
|
1,045 |
|
|
|
|
|
9,095 |
|
Other non-current liabilities |
|
|
18,620 |
|
|
|
|
|
2,661 |
|
|
|
|
|
|
|
Net cash used in operating activities |
|
|
(253,818 |
) |
|
|
|
(135,830 |
) |
|
|
|
(44,663 |
) |
||
Cash flows from investing activities: |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Purchases of property and equipment |
|
|
(56,521 |
) |
|
|
|
|
(57,821 |
) |
|
|
|
|
(22,219 |
) |
Purchases and issuances of loan receivable |
|
|
|
|
|
|
(10,100 |
) |
|
|
|
(2,250 |
) |
|||
Proceeds from loans receivable |
|
|
304 |
|
|
|
|
|
800 |
|
|
|
|
|
|
|
Purchase of investments |
|
|
(5,000 |
) |
|
|
|
|
|
|
|
|
|
(50,133 |
) |
|
Business acquisition, net of cash acquired |
|
|
(12,040 |
) |
|
|
|
|
|
|
|
|
|
|
||
Net cash used in investing activities |
|
|
(73,257 |
) |
|
|
|
(67,121 |
) |
|
|
|
(74,602 |
) |
||
Cash flows from financing activities: |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Proceeds from reverse recapitalization, net of redemptions of $867,253 and offering costs of $108,118 (Note 3) |
|
|
1,509,629 |
|
|
|
|
|
|
|
|
|
|
|
||
Proceeds from exercise of stock options |
|
|
167 |
|
|
|
|
|
26 |
|
|
|
|
|
7 |
|
Repurchases of common stock |
|
|
(24,998 |
) |
|
|
|
|
|
|
|
|
|
(408 |
) |
|
Taxes paid related to net share settlement of equity awards |
|
|
(9,463 |
) |
|
|
|
|
|
|
|
|
||||
Principal payments on capital leases and lease financing obligation |
|
|
(1,123 |
) |
|
|
|
|
(748 |
) |
|
|
|
|
(828 |
) |
Proceeds from lease financing obligation |
|
|
— |
|
|
|
|
— |
|
|
|
|
476 |
|
||
Contributions from non-controlling interests |
|
|
59,933 |
|
|
|
|
|
— |
|
|
|
|
|
— |
|
Proceeds from issuance of convertible promissory notes, net of issuance costs |
|
|
— |
|
|
|
|
— |
|
|
|
|
198,957 |
|
||
Proceeds from issuance of Series E convertible preferred stock, net of issuance costs |
|
|
— |
|
|
|
|
|
91,040 |
|
|
|
|
|
212,181 |
|
Net cash provided by financing activities |
|
|
1,534,145 |
|
|
|
|
90,318 |
|
|
|
|
410,385 |
|
||
Effect of foreign exchange rates on cash and cash equivalents |
|
|
(19 |
) |
|
|
|
|
|
|
|
|
|
|
||
Net increase (decrease) in cash, cash equivalents and restricted cash |
|
|
1,207,051 |
|
|
|
|
(112,633 |
) |
|
|
|
291,120 |
|
||
Cash, cash equivalents and restricted cash, beginning of period |
|
|
385,877 |
|
|
|
|
498,510 |
|
|
|
|
207,390 |
|
||
Cash, cash equivalents and restricted cash, end of period |
|
$ |
1,592,928 |
|
|
|
$ |
385,877 |
|
|
|
$ |
498,510 |
|
||
The accompanying notes are an integral part of these consolidated financial statements.
F-7
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Consolidated Statements of Cash Flows
(in thousands)
Supplemental disclosure of cash flow information: |
|
|
|
|
|
|
|
|
|
|
|
|||||
Cash paid for interest |
|
$ |
2,370 |
|
|
|
$ |
2,572 |
|
|
|
$ |
2,348 |
|
||
Cash paid for income taxes |
|
$ |
61 |
|
|
|
$ |
|
|
|
$ |
31 |
|
|||
Supplemental disclosure of non-cash investing and financing activities: |
|
|
|
|
|
|
|
|
|
|
|
|||||
Purchases of equipment through capital leases |
|
$ |
1,981 |
|
|
|
|
$ |
|
|
|
|
$ |
406 |
|
|
Purchases of property and equipment included in accounts payable and accrued expenses |
|
$ |
1,815 |
|
|
|
|
$ |
14,458 |
|
|
|
|
$ |
605 |
|
Equity received in related parties |
|
$ |
61,554 |
|
|
|
|
$ |
|
|
|
|
$ |
24,480 |
|
|
Purchase of non-marketable equity securities |
|
$ |
10,000 |
|
|
|
|
$ |
|
|
|
|
$ |
|
||
Issuance of common stock for a business acquisition |
|
$ |
15,087 |
|
|
|
|
$ |
|
|
|
|
$ |
|
||
Acquisition date fair value of contingent consideration |
|
$ |
8,760 |
|
|
|
$ |
|
|
|
$ |
|
||||
Purchases and issuances of loans receivable |
|
$ |
|
|
|
|
$ |
375 |
|
|
|
|
$ |
2,744 |
|
|
Initial fair value of warrant liabilities |
|
$ |
194,453 |
|
|
|
|
$ |
|
|
|
|
$ |
|
||
Conversion of convertible promissory notes to preferred stock |
|
$ |
195 |
|
|
|
$ |
|
|
|
$ |
211,608 |
|
|||
Series E convertible preferred stock issuance costs included in accrued expenses |
|
$ |
|
|
|
$ |
|
|
|
$ |
3,380 |
|
||||
Lease financing obligation for build-to-suit lease |
|
$ |
6,120 |
|
|
|
$ |
|
|
|
$ |
|
||||
The following table provides a reconciliation of the cash, cash equivalents and restricted cash balances as of each of the periods shown above:
|
|
As of December 31, |
|
|||||||||||||
|
|
2021 |
|
|
|
2020 |
|
|
|
2019 |
|
|||||
Cash and cash equivalents |
|
$ |
1,550,004 |
|
|
|
|
$ |
380,801 |
|
|
|
|
$ |
495,287 |
|
Restricted cash |
|
|
42,924 |
|
|
|
|
|
5,076 |
|
|
|
|
|
3,223 |
|
Total cash, cash equivalents and restricted cash |
|
$ |
1,592,928 |
|
|
|
$ |
385,877 |
|
|
|
$ |
498,510 |
|
||
The accompanying notes are an integral part of these consolidated financial statements.
F-8
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
1. Organization and Basis of Presentation
Business
The mission of Ginkgo Bioworks Holdings, Inc. (“New Ginkgo”, or the “Company”) is to make biology easier to engineer. The Company designs custom cells for customers across multiple markets. Since inception, the Company has devoted its efforts to improving its platform for programming cells to enable customers to leverage biology to create impactful products across a range of industries. The Company’s platform comprises (i) equipment, robotic automation, software, data pipelines and tools, and standard operating procedures for high throughput genetic engineering, fermentation, and analytics (referred to collectively as the “Foundry”), (ii) a library of proprietary genetic assets and associated performance data (referred to collectively as “Codebase”), and (iii) the Company’s team of expert users, developers and operators of the Foundry and Codebase.
On September 16, 2021, Soaring Eagle Acquisition Corp. (“SRNG”) consummated the merger transaction contemplated by the agreement and plan of merger, dated as of May 11, 2021, and amended on May 14, 2021 (the “Merger Agreement”), by and among SRNG, SEAC Merger Sub Inc., a wholly owned subsidiary of SRNG (“Merger Sub”), and Ginkgo Bioworks, Inc. (“Old Ginkgo”), whereby Merger Sub merged with and into Old Ginkgo, the separate corporate existence of Merger Sub ceased and Old Ginkgo survived the merger as a wholly owned subsidiary of SRNG (the “Business Combination”). In connection with the consummation of the Business Combination, SRNG changed its name to “Ginkgo Bioworks Holdings, Inc.” and, among other transactions contemplated by the Merger Agreement, the existing equity holders of Old Ginkgo exchanged their equity interests of Old Ginkgo for equity interests of New Ginkgo.
As a result of the Business Combination, the shares and corresponding capital amounts and loss per share related to Old Ginkgo’s outstanding convertible preferred stock and common stock prior to the Business Combination have been retroactively restated to reflect the Exchange Ratio established in the Merger Agreement. See Note 3 for additional information on the Business Combination.
Risks and Uncertainties
The Company is subject to a number of risks including rapid technological change, regulatory change, technical feasibility, commercial viability, public perception of genetically modified organisms, uncertain market acceptance of products derived from engineered organisms, alternative means of production, government funding of biosecurity initiatives, data and cybersecurity breaches, and dependence on key vendors and personnel.
Impact of the COVID-19 Pandemic
In March 2020, the World Health Organization declared the novel strain of coronavirus (“COVID-19”) outbreak a global pandemic. Since then, extraordinary actions have been taken by authorities to contain and manage the outbreak and spread of COVID-19 around the world. Consistent with the actions taken by governmental authorities, the Company has taken steps to protect its workforce and support the community efforts. From approximately March 2020 to June 2020, the Company operated at a reduced capacity. The Company also restricted non-essential travel and allowed most of its non-laboratory workforce to work remotely. In June 2020, the Company resumed modified on-site operations for its lab workers following the Center for Disease Control and Prevention guidance on mitigating and preventing the spread of COVID-19 in the workplace. The COVID-19 pandemic caused some disruption in the Company’s operations and the Company experienced partial suspensions and delays in servicing certain customer contracts. However, the Company believes that the COVID-19 pandemic did not have a material adverse impact on its financial condition or results of operations. The Company continues to monitor and assess the effects of COVID-19 on its financial condition, results of operations and cash flows.
2. Summary of Significant Accounting Policies
Basis of Presentation
The accompanying consolidated financial statements have been prepared in conformity with the rules and regulations of the Securities and Exchange Commission ("SEC") and generally accepted accounting principles in the United States of America (“U.S. GAAP”). Any reference in these notes to applicable guidance is meant to refer to the authoritative U.S. GAAP as found in the Accounting Standards Codification (“ASC”) and Accounting Standards Updates (“ASU”) of the Financial Accounting Standards Board (“FASB”).
F-9
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
The Business Combination was accounted for as a reverse recapitalization, in accordance with U.S. GAAP (the “Reverse Recapitalization”). Under this method of accounting, SRNG was treated as the “acquired” company for financial reporting purposes. Accordingly, for accounting purposes, the Reverse Recapitalization was treated as the equivalent of Old Ginkgo issuing stock for the net assets of SRNG, accompanied by a recapitalization. The net assets of SRNG are stated at historical cost, with no goodwill or other intangible assets recorded. The determination of Old Ginkgo as the accounting acquirer was primarily based on the fact that Old Ginkgo’s former shareholders currently have the largest voting interest in New Ginkgo, all of the management of New Ginkgo is comprised of Old Ginkgo’s former executive management, Old Ginkgo's former directors and individuals designated by, or representing, Old Ginkgo shareholders constitute a majority of the initial New Ginkgo Board, and the operations of Old Ginkgo comprise all of the ongoing operations of New Ginkgo.
The consolidated assets, liabilities and results of operations prior to the Reverse Recapitalization are those of Old Ginkgo. The shares and corresponding capital amounts and loss per share prior to the Reverse Recapitalization have been retroactively restated to reflect the Exchange Ratio established in the Merger Agreement.
Principles of Consolidation
The accompanying consolidated financial statements include the accounts of the Company, its wholly owned subsidiaries, majority owned subsidiaries and variable interest entities if the Company is the primary beneficiary. All intercompany accounts and transactions have been eliminated.
Reclassifications
Certain prior year amounts have been reclassified for consistency with the current year presentation. These reclassifications had no effect on the reported results of operations.
Variable Interest Entities
The Company evaluates its variable interests in variable interest entities (“VIE”) and consolidates VIEs when the Company is the primary beneficiary. The Company determines whether it is the primary beneficiary of each VIE based on its assessment of whether the Company possesses both (i) the power to direct the activities that most significantly affect the VIE’s economic performance and (ii) the obligation to absorb losses that could be significant to the VIE or the right to receive benefits that could be significant to the VIE. The Company reevaluates the accounting for its VIEs upon the occurrence of events that could change the primary beneficiary conclusion. As of December 31, 2021 and 2020, the maximum risk of loss related to the Company’s VIEs was limited to the carrying value of its investment in such entities.
Use of Estimates
The preparation of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, the disclosure of contingent assets and liabilities at the date of the consolidated financial statements, and the reported amounts of revenues and expenses during the reporting period. Significant estimates and assumptions used in preparation of these consolidated financial statements include, among others, those related to the fair value of equity instruments and equity awards, revenue recognition, the fair value of loans receivable, the fair value of certain investments, including equity method investments, the fair value of warrant liabilities, accrued expenses, and income taxes.
The Company bases its estimates on historical experience and other market-specific or relevant assumptions that it believes to be reasonable under the circumstances. Reported amounts and disclosures reflect the overall economic conditions that management believes are most likely to occur, and the anticipated measures management intends to take. Actual results could differ materially from those estimates. All revisions to accounting estimates are recognized in the period in which the estimates are revised.
Segment Information
The Company and the Chief Operating Decision Maker (“CODM”), which is comprised of the Chief Executive Officer and the Chief Operating Officer, view the Company’s operations and manage the business as a single operating segment. Strategic decisions are managed centrally, and consistent with this decision-making process, the CODM uses consolidated
F-10
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
financial information for purposes of evaluating performance, allocating resources, as well as forecasting future period financial results. The majority of the Company’s long-lived assets are held in the United States.
For the year ended December 31, 2021, two customers accounted for 17.0% and 10.5% of the Company’s total revenue. For the year ended December 31, 2020, two customers that are related parties accounted for 27.1% and 12.3% of the Company’s total revenue. For the year ended December 31, 2019, three customers that are related parties accounted for 35.0%, 17.3% and 11.5% of the Company’s total revenue and one customer that was not a related party accounted for 13.5% of the Company’s total revenue. No other customers exceeded more than 10% of the Company’s total revenue during the years ended December 31, 2021, 2020 and 2019.
Concentrations of Credit Risk
Financial instruments that potentially subject the Company to concentrations of credit risk primarily consist of cash and cash equivalents, restricted cash, accounts receivable, and loans receivable. The Company’s cash and cash equivalents and restricted cash are maintained in bank deposit accounts and money market funds that regularly exceed federally insured limits. The Company believes that it is not exposed to significant credit risk as its deposits are held in financial institutions that management believes to be of high credit quality. The Company’s accounts receivable primarily consists of amounts due under its Biosecurity contracts; however, concentrations of credit risk associated with these contracts are limited because the customer base is largely made up of state government agencies. The Company has not experienced any material write-offs related to its accounts receivable since inception.
Cash and Cash Equivalents
The Company considers all highly liquid investments with original maturities of three months or less at the date of purchase to be cash equivalents. Cash and cash equivalents include cash held in banks and amounts held in money market accounts. The carrying value of the Company’s cash and cash equivalents approximate fair value due to their short-term maturities.
Restricted Cash
Restricted cash primarily includes cash balances collateralizing letters of credit associated with the Company’s facility leases and a customer prepayment requiring segregation and restrictions in its use in accordance with the customer agreement. Restricted cash is included in other non-current assets on the Consolidated Balance Sheets.
Accounts Receivable, net
Accounts receivable consists of credit extended to customers in the normal course of business and is reported at the estimated net realizable value. Accounts receivable includes unbilled amounts that have been recognized in revenue but have not yet been invoiced based on timing differences and the terms of the underlying arrangements.
The Company maintains an allowance for doubtful accounts to provide for the estimated amounts of receivables that will not be collected. The allowance is based upon an assessment of customer creditworthiness, historical payment experience, the age of outstanding receivables and collateral to the extent applicable. The Company re-evaluates such allowance on a regular basis and adjusts the allowance as needed. Once a receivable is deemed to be uncollectible, such balance is charged against the allowance.
Inventory, net
Inventory mainly consists of diagnostic testing kits purchased from suppliers, testing program supplies and the costs of assembling sample collection kits. Finished goods inventory for lateral flow assay (“LFA”) and polymerase chain reaction (“PCR”) tests are valued at the lower of cost or net realizable value using the first-in first-out method. Raw materials, work in process and finished goods inventory for pooled tests are valued at the lower of cost or net realizable value using the average cost method. Inventory has been reduced by an allowance for excess and obsolete inventory based on the specific identification method.
F-11
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Loans Receivable
The Company has elected the fair value option under ASC 825, Financial Instruments (“ASC 825”) to account for its loans receivable. The Company classifies the current portion of the loans receivable balance as a component of prepaid expenses and other current assets on the Consolidated Balance Sheets based on the principal balance of the loan that matures within one year from the balance sheet date. The Company records loans receivable at fair value and recognizes changes in fair value as a component of other (expense) income, net in the Consolidated Statements of Operations and Comprehensive Loss.
Property and Equipment, net
Property and equipment are stated at cost less accumulated depreciation and amortization. Depreciation and amortization are computed using the straight-line method over the estimated useful lives of the assets or the remaining lease term with respect to leasehold improvement assets. Estimated lives of property and equipment are as follows:
|
Estimated Useful Life |
Computer equipment and software |
2 to 5 years |
Furniture and fixtures |
7 years |
Lab equipment |
1 to 5 years |
Facilities |
15 to 30 years |
Vehicles |
5 years |
Leasehold improvements |
Shorter of useful life or remaining lease term |
Expenditures for maintenance and repairs are expensed as incurred. When assets are retired or otherwise disposed of, the related cost and accumulated depreciation or amortization is removed from the accounts and any resulting gain or loss is reflected in other (expense) income, net in the Consolidated Statements of Operations and Comprehensive Loss.
Construction in progress relates to assets which have not been placed in service as of period end. Facilities relate to assets acquired under a build-to-suit lease arrangement described in Note 14.
Equity Method Investments
The Company utilizes the equity method to account for its investments in common stock, or in-substance common stock, when it possesses the ability to exercise significant influence, but not control, over the operating and financial policies of the investee. The Company uses judgment when determining the level of influence over the operating and financial policies of the investee considering key factors including, among others, the Company’s ownership interest, representation on the board of directors, participation in policy-making decisions and material contractual arrangements and obligations. Income and losses are allocated based upon relative ownership interest unless there is a substantive profit-sharing agreement in place.
For investments with a substantive profit-sharing agreement, the Company utilizes the Hypothetical Liquidation at Book Value (“HLBV”) method to allocate income and losses from the equity method investment. Under the HLBV method, the Company utilizes the capital account at the end of the period assuming the book value of the entity was liquidated or sold minus the same calculation at the beginning of the period. The difference is the share of earnings or losses attributable to the equity method investment.
Under the equity method, if there is a commitment for the Company to fund the losses of its equity method investees, the Company would continue to record its share of losses resulting in a negative equity method investment, which would be presented as a liability on the Consolidated Balance Sheets. Commitments may be explicit and may include formal guarantees, legal obligations, or arrangements by contract. Implicit commitments may arise from reputational expectations, intercompany relationships, statements by the Company of its intention to provide support, a history of providing financial support or other facts and circumstances. When the Company has no commitment to fund the losses of its equity method investees, the carrying value of its equity method investments will not be reduced below zero. The Company had no commitment to fund additional losses of its equity method investments during the years ended December 31, 2021, 2020 and 2019.
The Company evaluates its equity method investments for impairment whenever events or circumstances indicate that the carrying value of the investment may not be recoverable. The Company considers the investee’s financial position, forecasts and economic outlook, and the estimated duration and extent of losses to determine whether a recovery is anticipated. An
F-12
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
impairment that is other-than-temporary is recognized in the period identified. The Company has not recognized an impairment loss related to its equity method investments for the years ended December 31, 2021, 2020 and 2019.
The Company may elect the fair value option for its equity method investments on an investment-by-investment basis. For all equity method investments accounted for under the fair value option, the Company carries the equity method investment at fair value and records all subsequent changes in fair value as a component of loss on equity method investments in the Consolidated Statements of Operations and Comprehensive Loss.
Investments
Investments include warrants and marketable equity securities in publicly-traded companies and non-marketable equity securities in privately-held companies, in each case, in which the Company does not possess the ability to exercise significant influence over the investee.
Investments in warrants and marketable equity securities of publicly-traded companies are measured at fair value with subsequent changes in fair value recorded in loss on investments in the Consolidated Statements of Operations and Comprehensive Loss.
Investments in non-marketable equity securities of privately-held companies that do not have readily determinable fair values are carried at cost, less any impairments, plus or minus changes resulting from observable price changes in orderly transactions for the identical or a similar investment of the same issuer. Each period the Company assesses relevant transactions to identify observable price changes, and the Company regularly monitors these investments to evaluate whether there is an indication of impairment. The Company evaluates whether an investment’s fair value is less than its carrying value using an estimate of fair value, if such an estimate is available. For periods in which there is no estimate of fair value, the Company evaluates whether an event or change in circumstances has occurred that may have a significant adverse effect on the value of the investment. The Company has not recognized an impairment loss, nor any upward or downward adjustments resulting from observable price changes in identical or similar investments, for the years ended December 31, 2021, 2020 and 2019.
Fair Value Measurements
The Company categorizes its assets and liabilities measured at fair value in accordance with the authoritative accounting guidance that establishes a consistent framework for measuring fair value and requires disclosures for each major asset and liability category measured at fair value on either a recurring or nonrecurring basis.
ASC 820, Fair Value Measurement (“ASC 820”), establishes a fair value hierarchy for instruments measured at fair value that distinguishes between assumptions based on market data (observable inputs) and the Company’s own assumptions (unobservable inputs). Observable inputs are inputs that market participants would use in pricing the asset or liability based on market data obtained from sources independent of the Company. Unobservable inputs are inputs that reflect the Company’s assumptions about the inputs that market participants would use in pricing the asset or liability and are developed based on the best information available in the circumstances.
ASC 820 identifies fair value as the exchange price, or exit price, representing the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants. As a basis for considering market participant assumptions in fair value measurements, ASC 820 establishes a three-tier fair value hierarchy that distinguishes among the following:
To the extent that the valuation is based on models or inputs that are either less observable or unobservable in the market, the determination of fair value requires more judgment. Accordingly, the degree of judgment exercised by the Company in
F-13
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
determining fair value is greatest for instruments categorized in Level 3. A financial instrument’s level within the fair value hierarchy is based on the lowest level of any input that is significant to the fair value measurement.
The Company valued its money market fund holdings, loans receivable, marketable equity securities, warrant liabilities and contingent consideration at fair value on a recurring basis. The carrying amounts of the Company’s other financial instruments, which include accounts receivable, certain prepaid expenses and other current assets, accounts payable and accrued expenses and other current liabilities, approximate their fair values due to their short-term nature.
Impairment of Long-Lived Assets
The Company reviews its long-lived assets for impairment whenever events or changes in circumstances indicate that the carrying value of an asset may not be recoverable. Recoverability is measured by comparing the book values of the assets to the expected future net undiscounted cash flows that the assets are expected to generate. If such assets are considered to be impaired, the impairment to be recognized is measured by the amount by which the book values of the assets exceed their fair value. The Company has not recognized an impairment loss for the years ended December 31, 2021, 2020 and 2019.
Business Combinations
The Company accounts for business combinations using the acquisition method of accounting. The Company recognizes the identifiable assets acquired and liabilities assumed at their acquisition-date fair values and recognizes any excess of the total consideration paid over the fair value of the identifiable net assets as goodwill. Any purchase price that is considered contingent consideration is measured at its estimated fair value at the acquisition date and remeasured at each reporting period, with changes in estimated fair value recorded in general and administrative expenses on the Consolidated Statements of Operations and Comprehensive Loss. Acquisition transaction costs are expensed when incurred. The operating results of an acquisition are included in the Company’s consolidated financial statements as of the acquisition date.
Intangible Assets, net
Intangible assets, net consist of certain definite-lived assets including patents, processes and know-how related to technology acquired through business combinations. The Company amortizes such intangible assets on a straight-line basis over their estimated useful life.
The Company reviews intangible assets for impairment whenever events or changes in circumstances have occurred which could indicate that the carrying value of the assets are not recoverable. Recoverability is measured by comparing the carrying value of the intangible assets to the future undiscounted cash flows expected to be generated by the asset. In determining the expected future cash flows, the Company uses assumptions believed to be reasonable, but which are unpredictable and inherently uncertain. Actual future cash flows may differ from the estimates used in impairment testing. The Company recognizes an impairment loss when and to the extent that the estimated fair value of an intangible asset is less than its carrying value. The Company has not recognized an impairment loss for the years ended December 31, 2021, 2020 and 2019.
Goodwill
Goodwill represents the excess of acquisition cost over the fair market value of the net assets acquired. Goodwill is tested for impairment on an annual basis during the fourth quarter or whenever events or changes in circumstances indicate the carrying amount may not be recoverable. The Company considers various qualitative factors that could indicate impairment such as macroeconomic conditions, industry and market environment, technological obsolescence, overall financial performance of the Company, cash flow from operating activities and market capitalization. If the qualitative assessment indicates that it is more likely than not that the fair value of the reporting unit is less than its carrying amount, the Company performs a quantitative assessment to compare the fair value of the reporting unit to its carrying value, including goodwill. If the carrying value of the reporting unit exceeds the fair value, an impairment loss is recognized. A combination of the income approach and the market approach may be used to determine fair value of the reporting unit. The Company has not recognized an impairment loss for the years ended December 31, 2021, 2020 and 2019.
F-14
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Deferred Rent
Deferred rent consists of the difference between cash paid and rent expense recognized on a straight-line basis for the facilities that the Company occupies under operating leases. The Company classifies the current portion of the deferred rent balance as a component of accrued expenses and other current liabilities on the Consolidated Balance Sheets.
Revenue Recognition
The Company accounts for revenue in accordance with ASC 606, Revenue from Contracts with Customers (“ASC 606”). Under ASC 606, the Company recognizes revenue when the customer obtains control of the promised goods or services at an amount that reflects the consideration the Company expects to receive in exchange for those goods or services. To determine revenue recognition for arrangements that are within the scope of ASC 606, the Company performs the following five steps: (i) identify the contract(s) with a customer, (ii) identify the promises and distinct performance obligations in the contract, (iii) determine the transaction price, (iv) allocate the transaction price to the performance obligations in the contract, and (v) recognize revenue when (or as) the Company satisfies the performance obligations.
Foundry Revenue
The Company generates license and service revenue through the execution of license and collaboration agreements whereby customers obtain license rights to the Company’s proprietary technology and intellectual property for use in the research, development and commercialization of engineered organisms, and derived products. Under these agreements, the Company typically provides research and development services, which includes the provision of a license to the Company’s intellectual property. Additionally, the customer obtains license rights to the output of the Company’s services in order to commercialize the resulting output of such services. Generally, the terms of these agreements provide that the Company receives some combination of: (1) Foundry usage fees in the form of (i) upfront payments upon consummation of the agreement or other fixed payments, (ii) reimbursement for costs incurred for research and development services and (iii) milestone payments upon the achievement of specified technical criteria, plus (2) downstream value share payments in the form of (i) milestone payments upon the achievement of specified commercial criteria, (ii) royalties on sales of products from or comprising engineered organisms arising from the collaboration or licensing agreement and (iii) royalties related to cost of goods sold reductions realized by customers.
The Company’s collaboration and licensing agreements often contain multiple promises, including (i) licenses and assignments of intellectual property and materials and (ii) research and development services, and the Company determines whether each of the promises is a distinct performance obligation based on the nature of each agreement. As the Company is generally performing research and development services that are highly integrated and interrelated to the licenses and assignments of intellectual property and materials, the promises are generally inseparable. As such, the Company typically combines the research and development services, licenses, and assignments into a single performance obligation. However, for certain agreements, the Company only grants licenses or effects such transfers and assignments upon the successful completion of the research and development services or delivery of a developed product. For these agreements, the Company typically considers (i) the research and development services and (ii) the licenses, transfers, and assignments as distinct performance obligations, as each is transferred separately and has a separately identifiable benefit.
Options to acquire additional goods and services are evaluated to determine if such options provide a material right to the counterparty that it would not have received without entering into the contract. If so, the option is accounted for as a separate performance obligation. If not, the option is considered a marketing offer which is accounted for as a separate contract upon the counterparty’s election.
At contract inception, the Company determines the transaction price, including fixed consideration and any estimated amounts of variable consideration. Any upfront cash payment received upon consummation of the agreement is fixed and generally non-refundable. Variable consideration is subject to a constraint, and amounts are included in the transaction price to the extent that it is probable that a significant reversal in the amount of cumulative revenue recognized will not occur when the uncertainty associated with the variable consideration is subsequently resolved. Variable consideration may include reimbursement for costs incurred for the Company’s research and development efforts, milestone payments upon the achievement of certain technical and commercial criteria, and royalties on sales of products from or comprising engineered organisms arising from the agreement. With respect to the research and development reimbursements and milestone payments, the Company uses the most likely amount method to estimate variable consideration. With respect to agreements that include royalties on sales or other contingent payments based on sales, the Company applies the royalty recognition constraint which requires a constraint until the royalty or value-sharing transaction occurs. Certain agreements contain
F-15
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
payment in the form of equity or other non-cash consideration. Any non-cash consideration is measured at the fair value of the non-cash consideration at contract inception.
For agreements with promises that are combined into a single performance obligation, the entire transaction price is allocated to the single performance obligation. For agreements with multiple performance obligations, the transaction price is allocated to the performance obligations using the relative standalone selling price methodology. For agreements featuring variable consideration, the Company allocates variable consideration to one or more, but not all, performance obligations if certain conditions are met. Specifically, the Company assesses whether the variable consideration relates solely to its efforts to satisfy the performance obligation and whether allocating such variable consideration entirely to the performance obligation is consistent with the overall allocation objective. If these conditions are not met, the Company allocates the variable consideration based on the relative standalone selling price methodology. The key assumptions utilized in determining the standalone selling price for each performance obligation include development timelines, estimated research and development costs, commercial markets, likelihood of exercise (in the case of options considered to be material rights), and probabilities of success.
For agreements where the licenses or assignments are considered separate performance obligations or represent the only performance obligation, the Company recognizes revenue at the point in time that the Company effectively grants the license as the licenses or assignments represent functional intellectual property. For agreements where the licenses and the research and development services represent a combined performance obligation, the Company recognizes revenue over the period of performance using a measure of progress based on costs incurred to date as compared to total estimated costs.
The Company evaluates its measure of progress to recognize revenue at each reporting period and, as necessary, adjusts the measure of progress and related revenue recognition. The Company’s measure of progress and revenue recognition involves significant judgment and assumptions, including, but not limited to, estimated costs and timelines to complete its performance obligations. The Company evaluates contract modifications and amendments to determine whether any changes should be accounted for prospectively or on a cumulative catch-up basis. The Company utilizes the right to invoice practical expedient when it has a right to consideration in an amount that corresponds directly with the value of the Company’s performance to date.
Royalties are recognized as revenue when sales have occurred as the Company applies the sales or usage-based royalties recognition constraint. The Company has determined the application of this exception is appropriate because the license granted in the agreement is the predominant item to which the royalties relate.
As the Company receives upfront payments for technical services under certain of its arrangements, the Company evaluates whether any significant financing components exist given the term over which the fees will be earned may exceed one year. Based on the nature of the Company’s agreements, there are no significant financing components as the purpose of the upfront payment is not to provide financing, but rather to secure technical services, exclusivity rights, and Foundry capacity, or the timing of transfer of those goods or services is at the discretion of the customer.
Deferred revenue represents consideration received by the Company in excess of revenue recognized and primarily results from transactions where the Company receives upfront payments and non-cash equity consideration. In instances where the Company has received consideration in advance for an undefined number of technical development plans (“TDPs”) under its customer agreements, the Company records the advance payments as deferred revenue, net of current portion on the Consolidated Balance Sheets. Upon the execution of a specific TDP, the Company reclassifies the estimated consideration to be earned under that TDP within the next twelve months as current deferred revenue. The Company also classifies unexercised material rights related to future TDPs as deferred revenue, net of current portion on the Consolidated Balance Sheets. When a TDP is executed, and the material right is exercised, the amount allocated to the material right, which will be earned within the next twelve months, is reclassified to current deferred revenue. All other deferred revenue is classified as current or non-current based on the timing of when the Company expects to earn the underlying revenue based upon the projected progress of activities under the TDP.
Collaboration Arrangements
For arrangements that do not represent contracts with a customer, the Company analyzes its collaboration transactions to assess whether they are within the scope of ASC 808, Collaborative Arrangements (“ASC 808”), to determine whether such arrangements involve joint operating activities performed by parties that are both active participants in the activities and exposed to significant risks and rewards that are dependent on the commercial success of such activities. To the extent the
F-16
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
arrangement is within the scope of ASC 808, the Company assesses whether aspects of the arrangement between the Company and its collaboration partner are within the scope of other accounting literature. If the Company concludes that some or all aspects of the arrangement represent a transaction with a customer, the Company accounts for those aspects of the arrangement within the scope of ASC 606.
Biosecurity Revenue
In 2020, the Company launched its commercial offering of COVID-19 testing products and services for businesses, academic institutions, and other organizations in which the Company generates product and service revenue. Beginning in the first quarter of 2021, the Company launched its pooled testing initiative which focuses on providing end-to-end COVID-19 testing services to public health authorities. The Company currently offers pooled testing and reporting services for K-12 schools across the United States, at airports through its partnership with XpresCheck and the CDC, as well as through other congregate settings such as its partnership with Eurofins. The Company sells COVID-19 test kits on a standalone basis or as part of an end-to-end testing service. The Company records product revenue from sales of LFA, PCR, and pooled test kits. The Company records service revenue from sales of its end-to-end COVID-19 testing services, which consist of multiple promised goods and services including sample collection kits, physician authorizations, onsite test administration, outsourced laboratory PCR analysis, and access to results reported through the Company’s proprietary web-based portal. The Company recognizes its product and service revenue using the five-step model under ASC 606, Revenue from Contracts with Customers (“ASC 606”).
Product revenue is recognized when the test kits are shipped and risk of loss is transferred to the carrier. The Company’s test kits are generally not subject to a customer right of return except for product recalls under the rules and regulations of the U.S. Food and Drug Administration (“FDA”). The Company has elected to include shipping and handling fees billed to customers as a component of Biosecurity revenue.
Service revenue from the Company’s end-to-end COVID-19 testing services is recognized upon completion of the tests and release of the test results on the web-based portal. The Company has identified one performance obligation in its testing services contracts that represents a series of distinct goods or services that are substantially the same and that have the same pattern of transfer to the customer, with each test as a distinct service within the series. As the price for the testing services is fixed under each customer contract, the Company has elected the practical expedient to recognize revenue at the amount to which it has the right to invoice for services performed. The Company’s testing services contracts are generally one year or less in length and contain fixed unit pricing. Under typical payment terms for testing services, amounts are billed monthly in arrears for services performed or in advance based on contractual billing terms.
Cost of Biosecurity Revenue
Cost of Biosecurity product revenue consists of costs associated with the sale of diagnostic and sample collection test kits which includes costs paid to purchase test kits from third parties. Cost of Biosecurity service revenue consists of costs associated with the provision of the Company’s end-to-end COVID-19 testing services, which includes costs paid to provide sample collection kits, physician authorizations, onsite test administration, outsourced laboratory PCR analysis, access to results reported through a web-based portal and reporting of results to public health authorities.
Research and Development Costs
Research and development costs are expensed as incurred. Research and development costs consist of direct and indirect internal costs related to specific projects and initiatives, acquired intellectual property deemed to be in-process research and development, as well as fees paid to other entities that conduct certain research and development activities on the Company’s behalf.
Patent Costs
The Company expenses all costs as incurred in connection with the filing, prosecution, maintenance, defense, and enforcement of patent applications, including direct application fees and related legal and consulting expenses. Patent costs are included in general and administrative expenses within the Consolidated Statements of Operations and Comprehensive Loss.
F-17
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Stock-Based Compensation
The Company measures and recognizes compensation expense for all stock-based awards based on estimated grant-date fair values recognized over the requisite service period. For awards that vest solely based on a service condition, the Company recognizes compensation expense on a straight-line basis over the requisite service period. For awards that vest based on multiple conditions, the Company recognizes compensation expense using the accelerated attribution method on a tranche-by-tranche basis over the requisite service period such that the amount of compensation expense recognized at each reporting period is at least equal to the vested tranches at that date. For awards with a performance-based vesting condition, the Company recognizes stock-based compensation when achievement of the performance condition is deemed probable, and upon achieving a performance condition that was not previously considered as probable, records a cumulative catch-up adjustment to reflect the portion of the grantee’s requisite service that has been provided to date. For awards with market conditions, the compensation expense recognized over the requisite service period is not reversed if the market condition is not satisfied. The Company recognizes forfeitures as they occur.
The Company estimates the grant date fair value of stock options using the Black-Scholes option-pricing model. The Black-Scholes option-pricing model requires the input of subjective assumptions, including fair value of common stock (for options granted prior to the Business Combination), expected term, expected volatility, risk-free interest rate and expected dividend yield. The expected term was generally determined using the "simplified" method for standard options. The Company determined expected volatility using the historical volatility of the stock prices of similar publicly traded peer companies. The risk-free interest rate was based on the yield available on U.S. Treasury zero-coupon issues similar in duration to the expected term of the stock options. The Company has not paid, and does not expect to pay, dividends in the foreseeable future.
For awards with market conditions, the Company determines the grant date fair value using a Monte Carlo simulation model, which incorporates various assumptions including expected stock price volatility, risk-free interest rates, expected term, and expected dividend yield. The Company determines expected volatility using the historical volatility of the stock prices of similar publicly traded peer companies. The risk-free interest rate is based on the yield available on U.S. Treasury zero-coupon issues similar in duration to the expected term of the awards. The expected term is equal to the contractual term and a dividend yield of zero is assumed.
For awards granted prior to the Business Combination, the Company utilized the hybrid method to estimate the grant date fair value of its common stock underlying its stock-based awards. The hybrid method is a probability-weighted expected return method (“PWERM”) where the equity value in at least one scenario is allocated using an option pricing method (“OPM”). Under the PWERM, the value of the common stock is estimated based on the probability-weighted present value of expected future investment returns considering various liquidity events and the rights and privileges of each class of equity. Under the OPM, each class of stock is treated as a call option on the Company’s equity value, with exercise prices based on the liquidation preferences of the convertible preferred stock. The Black-Scholes model is used to price the call options which includes assumptions for the time to liquidity and volatility of equity value. A discount for lack of marketability is then applied to the common stock value. There are significant judgments and estimates inherent in determining the fair value of the common stock. These judgments and estimates include factors, both subjective and objective, including: (i) a discount for lack of marketability; (ii) external market data; (iii) historical activity by the Company in selling equity to outside investors; (iv) the Company’s stage of development; (v) rights and preferences of the Company’s equity securities that rank senior to common stock; and (vi) the likelihood of various liquidity events, among others. Changes to these assumptions could result in different fair values of common stock.
Income Taxes
The Company accounts for income taxes using the asset and liability method, which requires the recognition of deferred tax assets and liabilities for the expected future tax consequences of events that have been recognized in the consolidated financial statements or in the Company’s tax returns. Under this method, deferred tax assets and liabilities are determined based on differences between the financial statement carrying amounts and the tax bases of the assets and liabilities using the enacted tax rates in effect in the years in which the differences are expected to reverse. A valuation allowance against deferred tax assets is recorded if, based on the weight of the available evidence, it is more likely than not that some or all of the deferred tax assets will not be realized. Potential for recovery of deferred tax assets is evaluated by considering several factors, including estimating the future taxable profits expected, estimating future reversals of existing taxable temporary differences, considering taxable profits in carryback periods, and considering prudent and feasible tax planning strategies.
The Company accounts for uncertain tax positions using a more-likely-than-not threshold for recognizing and resolving uncertain tax positions. The evaluation of uncertain tax positions is based on factors including, but not limited to, changes in
F-18
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
the law, the measurement of tax positions taken or expected to be taken in tax returns, the effective settlement of matters subject to audit, new audit activity, and changes in facts or circumstances related to a tax position. The Company evaluates uncertain tax positions on an annual basis and adjusts the level of the liability to reflect any subsequent changes in the relevant facts surrounding the uncertain positions. The Company accounts for interest and penalties related to uncertain tax positions as part of its provision for income taxes. As of December 31, 2021 and 2020, the Company did not have any uncertain tax positions and no accrued interest or penalties related to uncertain tax positions. The Company does not expect a material change in unrecognized tax benefits in the next twelve months.
Warrant Liabilities
The Company classifies Private Placement Warrants and Public Warrants (both defined and discussed in Note 15) as liabilities. At the end of each reporting period, changes in fair value during the period are recognized as change in fair value of warrant liabilities on the Consolidated Statements of Operations and Comprehensive Loss. The Company will continue to adjust the warrant liability for changes in the fair value until the earlier of (a) the exercise or expiration of the warrants or (b) the redemption of the warrants, at which time the warrants will be reclassified to additional paid-in capital.
Foreign Currency Translation
The Company's reporting currency is the U.S. dollar while the functional currency of the Company's non-U.S. subsidiary, Ginkgo Bioworks Netherlands, BV, is the Euro. The financial statements of the non-U.S. subsidiary are translated into U.S. dollars in accordance with ASC 830, Foreign Currency Matters, using period-end exchange rates for assets and liabilities, average exchange rates in the period for revenues and expenses and historical exchange rates for equity. Foreign currency translation adjustments are recorded as a component of other comprehensive income (loss) on the Consolidated Statements of Operations and Comprehensive Loss and accumulated in other comprehensive income (loss) in stockholders' equity.
Comprehensive Loss
Comprehensive loss is defined as the change in equity during a period from transactions and other events and circumstances from non-owner sources. Other comprehensive loss consists of foreign currency translation adjustments.
Net Loss per Share
The Company follows the two-class method when computing net loss per share attributable to Ginkgo Bioworks Holdings, Inc. common stockholders as the Company has issued shares that meet the definition of participating securities. The two-class method determines net loss per share for each class of common and participating securities according to dividends declared or accumulated and participation rights in undistributed earnings. The two-class method requires earnings for the period to be allocated between common and participating securities based upon their respective rights to share in the earnings as if all earnings for the period had been distributed. During periods of loss, there is no allocation required under the two-class method since the participating securities do not have a contractual obligation to fund the losses of the Company.
Basic net loss per share is computed by dividing the net loss attributable to Ginkgo Bioworks Holdings, Inc. common stockholders by the weighted average number of common shares outstanding for the period. Diluted net loss per share is equal to the net loss attributable to Ginkgo Bioworks Holdings, Inc. common stockholders less the gain (if any) on the change in fair value of warrant liabilities, divided by the weighted average number of common shares outstanding for the period, including the effect of potentially dilutive common shares. For purposes of this calculation, outstanding options to purchase shares of common stock, unvested RSAs, unvested RSUs, warrants to purchase shares of common stock and contingently issued earnout shares are considered potentially dilutive common shares.
Recent Accounting Pronouncements
The Company is an emerging growth company as defined in the Jumpstart Our Business Startups Act of 2012, under which it may take advantage of an extended transition period for complying with new or revised accounting standards until such time as those standards apply to private companies. The Company has elected not to opt out of this extended transition period and, as a result, these consolidated financial statements may not be comparable to companies that comply with new or revised accounting pronouncements as of public company effective dates.
F-19
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Recently Adopted Accounting Pronouncements
In October 2018, the FASB issued ASU 2018-17, Consolidation (Topic 810): Targeted Improvements to Related Party Guidance for Variable Interest Entities (“ASU 2018-17”). The provisions of ASU 2018-17 modify the guidance under ASC 810 related to the evaluation of indirect interests held through related parties under common control when determining whether fees paid to decision makers and service providers are variable interests. Indirect interests held through related parties that are under common control are no longer considered to be the equivalent of direct interests in their entirety and instead should be considered on a proportional basis. This guidance more closely aligns with accounting of how indirect interests held through related parties under common control are considered for determining whether a reporting entity must consolidate a variable interest entity. The Company adopted ASU 2018-17 on January 1, 2021 and the adoption did not have a material impact on the Company’s consolidated financial statements and related disclosures.
Recently Issued Accounting Pronouncements
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842): Amendments to the FASB Accounting Standards Codification (“ASC 842”), which has been clarified and amended by various subsequent updates. ASC 842 requires lessees to record a right-of-use ("ROU") asset and a lease liability on the balance sheet for all leases with a lease term of more than 12 months. ASC 842 also requires additional disclosures about the amount, timing and uncertainty of cash flows arising from leases. The Company is required to adopt ASC 842 as of January 1, 2022. In connection with the adoption of ASC 842, the Company will apply the modified retrospective approach and recognize a cumulative-effect adjustment to the opening balance of accumulated deficit in the period of adoption. The Company has elected to apply the package of practical expedients that allows for not reassessing (i) whether any expired or existing contracts are or contain leases, (ii) the lease classification of any expired or existing leases, and (iii) the accounting for initial direct costs for any existing leases. The Company has also elected, by class of underlying asset, not to apply the recognition requirements of ASC 842 to short-term leases. While the Company continues to assess the various impacts of adoption, the most significant effects will primarily relate to (1) the recognition of an ROU asset and lease liability on the balance sheet for the Company’s existing operating leases; and (2) providing significant new disclosures about leasing activities. The Company does not anticipate that the adoption of ASC 842 will have a material impact on its results of operations and cash flows.
In June 2016, the FASB issued ASU 2016-13, Financial Instruments-Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments, and subsequently issued multiple amendments to the standard (collectively, “ASU 2016-13”). The provisions of ASU 2016-13 modify the impairment model to utilize an expected loss methodology in place of the currently used incurred loss methodology and require a consideration of a broader range of reasonable and supportable information to inform credit loss estimates. The guidance is effective for the Company on January 1, 2023, with early adoption permitted. The Company is currently evaluating the impact that the implementation of this standard will have on its consolidated financial statements and related disclosures.
In December 2019, the FASB issued ASU 2019-12, Simplifying the Accounting for Income Taxes (“ASU 2019-12”). The provisions of ASU 2019-12 eliminate certain exceptions related to the approach for intraperiod tax allocation and deferred tax liabilities for outside basis differences and clarify when a step-up in the tax basis of goodwill should be considered part of a business combination or a separate transaction. It also clarifies and simplifies other aspects of the accounting for income taxes. The guidance is effective for the Company on January 1, 2022. The adoption of ASU 2019-12 is not expected to have a material impact on the Company's consolidated financial statements and related disclosures.
In January 2020, the FASB issued ASU 2020-01, Investments—Equity Securities (Topic 321), Investments—Equity Method and Joint Ventures (Topic 323), and Derivatives and Hedging (Topic 815)—Clarifying the Interactions between Topic 321, Topic 323, and Topic 815 (a consensus of the FASB Emerging Issues Task Force) (“ASU 2020-01”). ASU 2020-01 addresses accounting for the transition into and out of the equity method and provides clarification of the interaction of rules for equity securities, the equity method of accounting, and forward contracts and purchase options on certain types of securities. The guidance is effective for the Company on January 1, 2022. The adoption of ASU 2020-01 is not expected to have a material impact on the Company's consolidated financial statements and related disclosures.
In August 2020, the FASB issued ASU 2020-06, Debt—Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging- Contracts in Entity’s Own Equity (Subtopic 815-40) (“ASU 2020-06”) which simplifies the accounting for convertible instruments by reducing the number of accounting models and the number of embedded conversion features that could be recognized separately from the host contract. Additionally, ASU 2020-06 removes certain settlement conditions that are required for contracts in an entity's own equity to qualify for the derivatives scope exception. The guidance also modifies diluted earnings per share calculations by requiring entities to use the if-converted method for
F-20
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
convertible instruments and to assume share settlement when an instrument can be settled in cash or shares. The guidance is effective for the Company on January 1, 2024 with early adoption permitted. The Company is currently evaluating the impact that the implementation of this standard will have on its consolidated financial statements and related disclosures.
In November 2021, the FASB issued ASU 2021-10, Government Assistance (Topic 832): Disclosures by Business Entities about Government Assistance ("ASU 2021-10"). This update requires annual disclosures about transactions with a government that are accounted for by applying a grant or contribution accounting model by analogy including: (1) the types of transactions; (2) the accounting for the transactions; and (3) the effect of the transactions on a business entity’s financial statements. ASU 2021-10 is effective for the Company on January 1, 2022. The adoption of ASU 2021-10 is not expected to have a material impact on the Company's consolidated financial statements and related disclosures.
3. Business Combination
On September 16, 2021 (the “Closing Date”), the Company and SRNG completed the merger transaction contemplated by the Merger Agreement (the “Closing"), with Old Ginkgo surviving the merger as a wholly owned subsidiary of SRNG.
Pursuant to the Merger Agreement, SRNG acquired all of the outstanding equity interests of Old Ginkgo for approximately $15.8 billion in aggregate consideration in the form of common stock of New Ginkgo valued at $10 per share (the “Base Equity Consideration”). The Base Equity Consideration was allocated among Old Ginkgo equity holders based on an exchange ratio of 49.080452 ("Exchange Ratio"). Accordingly, upon the closing of the Business Combination, all shares of Old Ginkgo Class A common stock and Old Ginkgo Class B common stock issued and outstanding immediately prior to the Business Combination converted into New Ginkgo Class A common stock and New Ginkgo Class B common stock, respectively, each with a par value of $0.0001 per share, based on the Exchange Ratio. All equity awards under Old Ginkgo's stock incentive plans were assumed by the Company and converted into comparable equity awards that are settled or exercisable for shares of the Company’s common stock. As a result, (i) each outstanding stock option to acquire Old Ginkgo common stock was converted into an option to purchase approximately 49.080452 shares of New Ginkgo common stock, (ii) each outstanding share of restricted common stock was converted into approximately 49.080452 shares of restricted common stock of New Ginkgo and (iii) each outstanding award of restricted stock units was assumed and converted into a restricted stock unit having the same terms and conditions as applied to the Old Ginkgo restricted stock unit so converted but relating to approximately 49.080452 shares of common stock of New Ginkgo.
In addition to the Base Equity Consideration, the equity holders of Old Ginkgo received approximately 188.7 million shares of New Ginkgo common stock (the “Earnout Consideration”), which are subject to forfeiture to the extent that the vesting conditions described below are not satisfied on or before the fifth anniversary of the Closing (the "Earnout Period"). If at any point during the trading hours of a trading day, for any 20 trading days within any period of 30 consecutive trading days during the Earnout Period, the trading price per share of the Company's Class A common stock is greater than or equal to:
The Company evaluated the earnout shares and concluded that they qualify for the scope exception from derivative accounting in ASC 815-10-15-74 and meet the criteria for equity classification under ASC 815-40. The Company determined that the earnout shares underlying rollover equity awards (i.e., restricted stock awards, restricted stock units and options) granted under the Company's stock incentive plans (together the "Rollover Equity Awards") that are unvested as of the Closing Date are within the scope of ASC 718 (see Note 18). The remaining earnout shares issued to holders of Old Ginkgo common stock and those earnout shares underlying vested Rollover Equity Awards were initially measured at fair value at Closing and recorded within additional paid-in-capital ("APIC") and had no net impact on APIC. Since those earnout shares are equity-classified, there is no remeasurement unless reclassification is required. Upon meeting an earnout target, the earnout shares delivered to the equity holders are recorded in equity as shares outstanding with the appropriate allocation to par value of common stock and APIC. The first earnout target of $12.50 was met on November 15, 2021 and, as a result, approximately 38.8 million earnout shares became vested and outstanding.
In connection with the entry into the Merger Agreement, Eagle Equity Partners III, LLC, a Delaware limited liability company (the “Sponsor”), forfeited 11,534,052 of its shares of New Ginkgo Class A common stock and an additional 16,737,183 of its shares of New Ginkgo Class A common stock (the "Sponsor Earnout Shares") became subject to vesting
F-21
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
and forfeiture conditions identical to those applicable to the Earnout Consideration issued to Old Ginkgo equity holders. Similar to the Earnout Consideration, the Sponsor Earnout Shares were accounted for as equity classified instruments and were included as merger consideration and recorded in additional paid-in capital. The Sponsor Earnout Shares are considered legally issued and outstanding shares of common stock subject to restrictions on transfer and do not participate in the earnings or losses of the Company prior to vesting.
The Business Combination is accounted for as a reverse recapitalization, in accordance with U.S. GAAP. Under this method of accounting, SRNG was treated as the “acquired” company for financial reporting purposes. Accordingly, the Business Combination was treated as the equivalent of Old Ginkgo issuing stock for the net assets of SRNG, accompanied by a recapitalization. The net assets of SRNG are stated at historical cost, with no goodwill or other intangible assets recorded.
PIPE Investment
On May 11, 2021, concurrently with the execution of the Merger Agreement, SRNG entered into subscription agreements with certain accredited investors (the “PIPE Investors”). In connection with the consummation of the Business Combination on September 16, 2021, the PIPE Investors collectively consummated investments for 76,000,000 shares of the Company's Class A common stock at a price of $10.00 per share (the "PIPE Shares") for an aggregate amount of $760.0 million (the “PIPE Investment”).
Summary of Net Proceeds
The following table summarizes the elements of the net proceeds from the Business Combination as of December 31, 2021 (in thousands):
Cash - SRNG Trust and cash (net of redemptions) |
|
$ |
857,747 |
|
Cash - PIPE Investment |
|
|
760,000 |
|
Less: Payment of underwriter fees and other offering costs |
|
|
(108,118 |
) |
Net proceeds from the Business Combination |
|
$ |
1,509,629 |
|
Summary of Shares Issued
The following table summarizes the number of shares of common stock outstanding immediately following the consummation of the Business Combination:
SRNG shares outstanding prior to the Business Combination |
|
|
215,625,000 |
|
Less: redemption of SRNG shares prior to the Business Combination |
|
|
(86,725,312 |
) |
Less: SRNG shares forfeited |
|
|
(11,534,052 |
) |
Common stock of SRNG (1) |
|
|
117,365,636 |
|
Shares issued pursuant to the PIPE Investment |
|
|
76,000,000 |
|
Business Combination and PIPE Investment shares |
|
|
193,365,636 |
|
Conversion of Old Ginkgo Series B preferred stock to common stock |
|
|
203,346,152 |
|
Conversion of Old Ginkgo Series C preferred stock to common stock |
|
|
228,641,430 |
|
Conversion of Old Ginkgo Series D preferred stock to common stock |
|
|
302,464,716 |
|
Conversion of Old Ginkgo Series E preferred stock to common stock |
|
|
170,227,108 |
|
Conversion of Old Ginkgo common stock (2) |
|
|
387,016,194 |
|
Total shares of New Ginkgo common stock outstanding immediately following the Business Combination |
|
|
1,485,061,236 |
|
(1)
(2)
F-22
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
4. Acquisition
On July 1, 2021, the Company completed an acquisition of 100% of the equity of Dutch DNA Biotech B.V. (“Dutch DNA”), a company based in the Netherlands with a proprietary platform technology focused on the development of fungal strains and fermentation processes for the production of proteins and organic acids. Dutch DNA's significant expertise and fungal strain assets for the large-scale production of proteins is expected to add a valuable set of tools to the Company's Codebase and broader platform for cell programming.
The following table summarizes the preliminary acquisition date fair value of the consideration transferred for Dutch DNA (in thousands):
Cash |
|
$ |
11,451 |
|
Fair value of Class A common stock |
|
|
15,087 |
|
Contingent consideration |
|
|
8,760 |
|
Total Dutch DNA consideration |
|
$ |
35,298 |
|
The fair value of the Class A common stock issued as part of the consideration paid for Dutch DNA was determined using the then-most recently available third-party valuation of the Company's common stock. The contingent consideration arrangement requires the Company to pay up to a maximum of $20.0 million to the seller upon the achievement of certain technical and commercial milestones by Dutch DNA pursuant to a Technical Development Agreement executed between the Company and Dutch DNA prior to the close of the acquisition. Refer to Note 5 for further discussion of the fair value of the contingent consideration liability.
The acquisition was accounted for in accordance with ASC 805, Business Combinations. Dutch DNA's results of operations have been included in the Consolidated Statements of Operations and Comprehensive Loss since the date of acquisition, which were not material. The Dutch DNA acquisition does not represent a material business combination, and therefore pro forma financial information is not provided. The Company allocated the purchase price to the tangible and identifiable intangible assets acquired and liabilities assumed based on their respective estimated fair values on the acquisition date. The fair value of the intangible assets was determined using the replacement cost method which estimates the cost the Company would incur in rebuilding the technology. The excess purchase price consideration was recorded as goodwill and is made up of the future potential value of the acquired intellectual property and the assembled workforce. The Company incurred $0.6 million of acquisition-related costs which were included in general and administrative expenses in the Consolidated Statements of Operations and Comprehensive Loss.
The following table summarizes the acquisition date fair values of assets acquired and liabilities assumed as of the acquisition date (in thousands):
Cash |
|
$ |
387 |
|
Accounts receivable |
|
|
149 |
|
Prepaid expenses and other current assets |
|
|
170 |
|
Property, plant and equipment |
|
|
234 |
|
Intangibles (1) |
|
|
20,500 |
|
Goodwill (2) |
|
|
15,177 |
|
Accounts payable |
|
|
(194 |
) |
Accrued expenses and other current liabilities |
|
|
(137 |
) |
Other non-current liabilities |
|
|
(988 |
) |
Net assets acquired |
|
$ |
35,298 |
|
(1) Estimated useful life of 15 years.
(2) Non-deductible for tax purposes.
The purchase price allocation presented above has been finalized as of the fourth quarter of 2021 and includes a $5.0 million measurement period adjustment related to deferred income taxes. As a result, goodwill associated with Dutch DNA was $19.5 million as of December 31, 2021, including currency translation adjustments.
5. Fair Value Measurements
F-23
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
No transfers between levels have occurred during the periods presented. The following tables present information about the Company’s financial assets and liabilities measured at fair value on a recurring basis (in thousands):
|
|
As of December 31, 2021 |
|
|||||||||||||||||||
|
|
Total |
|
|
|
Level 1 |
|
|
|
Level 2 |
|
|
|
Level 3 |
|
|||||||
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
Money market funds, included in cash and cash equivalents |
|
$ |
1,482,063 |
|
|
|
|
$ |
1,482,063 |
|
|
|
|
$ |
|
|
|
|
$ |
|
||
Synlogic, Inc. common stock, included in investments |
|
|
15,345 |
|
|
|
|
|
15,345 |
|
|
|
|
|
|
|
|
|
|
|
||
Synlogic, Inc. warrants, included in investments |
|
|
6,166 |
|
|
|
|
|
|
|
|
|
|
6,166 |
|
|
|
|
|
|
||
Cronos Group Inc. common stock, included in investments |
|
|
10,331 |
|
|
|
|
|
|
|
|
|
|
10,331 |
|
|
|
|
|
|
||
Loans receivable, included in prepaid expenses and other current assets |
|
|
11,559 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
11,559 |
|
||
Total assets |
|
$ |
1,525,464 |
|
|
|
|
$ |
1,497,408 |
|
|
|
|
$ |
16,497 |
|
|
|
|
$ |
11,559 |
|
Liabilities: |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||
Public Warrants, included in warrant liabilities |
|
$ |
77,280 |
|
|
|
|
$ |
77,280 |
|
|
|
|
$ |
|
|
|
|
$ |
|
||
Private Placement Warrants, included in warrant liabilities |
|
|
58,558 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
58,558 |
|
||
Contingent consideration, included in other non-current liabilities |
|
|
8,467 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
8,467 |
|
||
Total liabilities |
|
$ |
144,305 |
|
|
|
|
$ |
77,280 |
|
|
|
|
$ |
|
|
|
|
$ |
67,025 |
|
|
|
|
As of December 31, 2020 |
|
|||||||||||||
|
|
Total |
|
|
Level 1 |
|
|
Level 2 |
|
|
Level 3 |
|
||||
Assets: |
|
|
|
|
|
|
|
|
|
|
|
|
||||
Money market funds, included in cash and cash equivalents |
|
$ |
372,537 |
|
|
$ |
372,537 |
|
|
$ |
|
|
$ |
|
||
Synlogic, Inc. common stock, included in investments |
|
|
13,696 |
|
|
|
13,696 |
|
|
|
|
|
|
|
||
Synlogic, Inc. warrants, included in investments |
|
|
5,504 |
|
|
|
|
|
|
5,504 |
|
|
|
|
||
Loans receivable, included in prepaid expenses and other current assets |
|
|
2,268 |
|
|
|
|
|
|
|
|
|
2,268 |
|
||
Loans receivable, net of current portion |
|
|
13,298 |
|
|
|
|
|
|
|
|
|
13,298 |
|
||
Total assets |
|
$ |
407,303 |
|
|
$ |
386,233 |
|
|
$ |
5,504 |
|
|
$ |
15,566 |
|
The fair value of the warrants to purchase Synlogic common stock (Note 10) is calculated as the value of the underlying common stock, less the related unpaid exercise price and represents a Level 2 measurement within the fair value hierarchy.
During the year ended December 31, 2021, the Company received 2,934,980 shares of Cronos Group Inc. ("Cronos") common stock as consideration for the achievement of certain target productivity milestones related to two cultured cannabinoids under the collaboration agreement with Cronos to produce eight cultured cannabinoids. The fair value of the Cronos common stock is calculated as the quoted price of the common stock adjusted for short-term marketability restrictions. Accordingly, these shares are classified as Level 2 financial instruments.
Loans Receivable
As of December 31, 2021 and 2020, loans receivable primarily consisted of a revolving promissory note with Glycosyn, LLC (“Glycosyn”) which is secured by the assets of Glycosyn, including certain intellectual property such as patents and copyrights held by Glycosyn (“Glycosyn Promissory Note”) and a series of convertible notes with Access Bio, Inc. (“Access Bio Convertible Notes”). The fair value of the Glycosyn Promissory Note and Access Bio Convertible Notes were determined based on significant inputs not observable in the market, which represent a Level 3 measurement within the fair value hierarchy. Significant changes in these unobservable inputs in isolation could have resulted in a significantly lower or higher fair value measurement. Refer to Note 6 for additional details on loans receivable.
As of December 31, 2021, the Company estimated the fair value of the Glycosyn Promissory Note using a probability-weighted discounted cash flow model under a dissolution scenario with partial recovery and no recovery as Glycosyn was in default on that date (see Note 6). The significant assumptions used in valuing the Glycosyn Promissory Note were scenario probabilities of 50%, a recovery rate on first lien debt of 63% and a discount rate of 15%. As of December 31, 2020, the Company used a probability-weighted discounted cash flow model under four settlement scenarios: (i) a qualified financing which resulted in a 20% conversion discount, (ii) repayment upon change in control, (iii) a dissolution scenario and (iv)
F-24
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
repayment in accordance with the terms of the note. The significant assumptions used in valuing the Glycosyn Promissory Note included the expected timing and probability of each scenario, ranging from 1 to 2.5 years and from 10% to 40%, respectively, and a discount rate of 15%. The weighted average timing of the scenarios weighted based on the probability of each scenario was 1.2 years as of December 31, 2020.
As of December 31, 2021, the Company estimated the fair value of the Access Bio Convertible Notes using a binomial lattice model using the following key assumptions: 85.5% equity volatility, 0.88 years to maturity, 0.3% risk-free rate, 30.9% risk-adjusted rate and 0% dividend yield. As of December 31, 2020, the Company estimated the fair value of the Access Bio Convertible Notes using a Monte-Carlo simulation model as the conversion price of the notes had not yet reset to the minimum reset price. Under the Monte-Carlo simulation model, the future stock price of Access Bio, Inc. (“Access Bio”) was simulated over the term of the note to assess the value of the settlement features which included conversion into stock at a discount determined under a reset provision tied to the stock price of Access Bio and redemption at maturity. The key inputs into the Monte Carlo simulation model were a discount rate of 32.8% and volatility of 88.5%.
The following table provides a reconciliation of loans receivable measured at fair value using Level 3 significant unobservable inputs (in thousands):
|
|
2021 |
|
|
2020 |
|
||
Balance at January 1 |
|
$ |
15,566 |
|
|
$ |
4,830 |
|
Purchases and issuances |
|
|
|
|
|
10,475 |
|
|
Proceeds from loans receivable |
|
|
(304 |
) |
|
|
(800 |
) |
Conversion of promissory notes |
|
|
(195 |
) |
|
|
|
|
Change in fair value |
|
|
(3,508 |
) |
|
|
1,061 |
|
Balance at December 31 |
|
$ |
11,559 |
|
|
$ |
15,566 |
|
Warrant Liabilities
The fair value of the Private Placement Warrants have been estimated using a Monte Carlo simulation model as of the date the Company assumed the warrants from SRNG and subsequently as of the balance sheet date. The fair value of the Public Warrants have been measured based on the quoted price of such warrants on the NYSE. The estimated fair value of the Private Placement Warrants is determined using Level 3 inputs. Inherent in a Monte Carlo simulation are assumptions related to expected stock-price volatility, expected life, risk-free interest rate and dividend yield. Material increases (or decreases) in any of those inputs may result in a significantly higher (or lower) fair value measurement. The Company estimates the volatility of its Private Placement Warrants based on implied volatility from the Company’s Public Warrants and from historical volatility of select peer company’s common stock that matches the expected remaining life of the warrants. The risk-free interest rate is based on the U.S. Treasury zero-coupon yield curve for a maturity similar to the expected remaining life of the warrants. The expected life of the warrants is assumed to be equivalent to their remaining contractual term. The dividend yield is based on the historical rate, which the Company anticipates remaining at zero. Refer to Note 15 for additional details on the Company’s warrant liabilities.
The following table provides quantitative information regarding Level 3 inputs used in the recurring valuation of the Private Placement Warrants as of their measurement dates:
|
|
September 16, 2021 (Closing) |
|
|
December 31, 2021 |
|
||
Exercise price |
|
$ |
11.50 |
|
|
$ |
11.50 |
|
Stock price |
|
$ |
11.42 |
|
|
$ |
8.31 |
|
Volatility |
|
|
53.1 |
% |
|
|
58.7 |
% |
Term (in years) |
|
|
5.00 |
|
|
4.71 |
|
|
Risk-free interest rate |
|
|
0.84 |
% |
|
|
1.25 |
% |
The following table provides a reconciliation of the Private Placement Warrants measured at fair value using Level 3 significant unobservable inputs (in thousands):
|
|
2021 |
|
|
Balance at January 1 |
|
$ |
— |
|
Additions pursuant to the Business Combination |
|
|
90,263 |
|
Change in fair value |
|
|
(31,705 |
) |
Balance at December 31 |
|
$ |
58,558 |
|
F-25
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Contingent Consideration
In connection with the acquisition of Dutch DNA, the Company recorded contingent consideration liabilities for the estimated fair value of earnout payments up to a maximum of $20.0 million payable to the seller upon the achievement of certain technical and commercial milestones by Dutch DNA pursuant to a Technical Development Agreement executed between the Company and Dutch DNA prior to the close of the acquisition. The contingent consideration liabilities are measured at fair value and are based on significant inputs not observable in the marketplace, which represent a Level 3 measurement. Material increases (or decreases) in any of those inputs may result in a significantly higher (or lower) fair value measurement. The fair value of the earnouts was estimated using a combination of probability weighted present value and discounted cash flow models. The key valuation inputs used as of December 31, 2021 and the acquisition date were management's estimate of the probability of achieving each milestone ranging from 10% to 80% and projections related to Dutch DNA's after-tax revenues for each of the calendar years through 2046. The earnout payments were discounted at rates ranging from 9% to 11.31% as of December 31, 2021 and from 6.93% to 9% as of the acquisition date.
The following table provides a reconciliation of the contingent consideration measured at fair value using Level 3 significant unobservable inputs (in thousands):
|
|
2021 |
|
|
Balance at January 1 |
|
$ |
— |
|
Acquisition |
|
|
8,760 |
|
Change in fair value |
|
|
(293 |
) |
Balance at December 31 |
|
$ |
8,467 |
|
6. Loans Receivable
Glycosyn Promissory Note
In October 2018, the Company provided a revolving promissory note to Glycosyn which has been amended several times since inception. The Glycosyn Promissory Note is convertible at a discount, at the Company’s election, into equity securities of Glycosyn upon Glycosyn’s first issuance of equity securities, other than an underwritten public offering, from which Glycosyn receives gross proceeds of at least $10.0 million. In addition, Glycosyn is obligated to immediately repay the outstanding balance of the loan, plus accrued interest, upon a change in control event. In January 2021, the existing terms of the Glycosyn Promissory Note were amended to add an additional $0.2 million to the principal balance, extend the number of interest-only payments through June 30, 2021 and to increase the interest rate from 7.5% to 12.5% in the event of default. In July 2021, the parties entered into an additional amendment to extend the number of interest-only payments through the end of 2021 and to accelerate the maturity date to December 31, 2021. As of December 31, 2021, the Glycosyn Promissory Note was in default with an unpaid principal balance of $5.4 million and a fair value of $1.8 million, which is recorded in prepaid expenses and other current assets on the Consolidated Balance Sheets. As of December 31, 2020, the Glycosyn Promissory Note had an unpaid principal balance of $5.3 million and a fair value of $4.5 million, of which $2.0 million was recorded in prepaid expenses and other current assets and the remainder in loans receivable, net of current portion on the Consolidated Balance Sheets. For the years ended December 31, 2021, 2020 and 2019, the (loss) gain on the change in fair value of the Glycosyn Promissory Note was $(2.5) million, $0.5 million and $(0.9) million, respectively, which was recorded in other (expense) income, net on the Consolidated Statements of Operations and Comprehensive Loss.
Access Bio Convertible Notes
In November 2020, the Company entered into a convertible note subscription agreement with Access Bio, a supplier of the Company’s diagnostic test kits. The Access Bio Convertible Notes are due in November 2022 in the aggregate principal amount of $10.0 million plus a 2% rate of return compounded annually. The Access Bio Convertible Notes are convertible into a number of shares of common stock of Access Bio, a company listed on the Korea Stock Exchange, of up to $10.0 million based on a fixed foreign currency exchange rate and a conversion price subject to certain adjustments, including reset adjustments each quarter based on the trading price of Access Bio’s stock. The adjusted conversion price cannot be reduced to less than 70% of the initial conversion price and the reset adjustments cannot increase the effective conversion ratio. The Access Bio Convertible Notes are convertible at the Company’s election any time following the first anniversary of the issuance date of the notes and prior to the 30th day before the maturity date. Additionally, subject to certain provisions, the Company has the option to cause Access Bio to repurchase, or Access Bio has the option to repurchase, a portion of the outstanding balance under the notes (up to the entire balance in the case of the Company’s option) at a price to ensure a 2% rate of return compounded annually.
F-26
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
As of December 31, 2021, the fair value of the Access Bio Convertible Notes was $9.8 million, which was recorded in prepaid expenses and other current assets on the Consolidated Balance Sheets. As of December 31, 2020, the fair value of the Access Bio Convertible Notes was $10.7 million, which was recorded in loans receivable, net of current portion on the Consolidated Balance Sheets. The (loss) gain from the change in fair value of the Access Bio Convertible Notes during the years ended December 31, 2021 and 2020 was $(0.9) million and $0.7 million, respectively, and was recorded in other (expense) income, net on the Consolidated Statements of Operations and Comprehensive Loss.
7. Prepaid Expenses and Other Current Assets
Prepaid expenses and other current assets consisted of the following (in thousands):
|
|
As of December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Prepaid expenses |
|
$ |
9,739 |
|
|
$ |
9,727 |
|
Prepaid insurance |
|
|
9,199 |
|
|
|
1,127 |
|
Prepaid inventory |
|
|
144 |
|
|
|
6,536 |
|
Loans receivable |
|
|
11,559 |
|
|
|
2,268 |
|
Other receivables |
|
|
2,198 |
|
|
|
761 |
|
Other current assets |
|
|
698 |
|
|
|
680 |
|
Prepaid expenses and other current assets |
|
$ |
33,537 |
|
|
$ |
21,099 |
|
8. Inventory, net
Inventory, net consisted of the following (in thousands):
|
|
As of December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Finished goods |
|
$ |
3,264 |
|
|
$ |
2,756 |
|
Raw materials |
|
|
64 |
|
|
|
|
|
Work in process |
|
|
50 |
|
|
|
|
|
Less: Inventory reserve |
|
|
(16 |
) |
|
|
(20 |
) |
Inventory, net |
|
$ |
3,362 |
|
|
$ |
2,736 |
|
9. Property and Equipment, net
Property and equipment, net consisted of the following (in thousands):
|
|
As of December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Facilities |
|
$ |
12,762 |
|
|
$ |
12,762 |
|
Furniture and fixtures |
|
|
4,617 |
|
|
|
2,165 |
|
Lab equipment |
|
|
113,963 |
|
|
|
51,072 |
|
Computer equipment and software |
|
|
10,129 |
|
|
|
6,204 |
|
Leasehold improvements |
|
|
55,033 |
|
|
|
40,435 |
|
Construction in progress |
|
|
10,278 |
|
|
|
42,575 |
|
Vehicles |
|
|
40 |
|
|
|
|
|
Total property and equipment |
|
|
206,822 |
|
|
|
155,213 |
|
Less: Accumulated depreciation |
|
|
(61,052 |
) |
|
|
(33,778 |
) |
Property and equipment, net |
|
$ |
145,770 |
|
|
$ |
121,435 |
|
As of December 31, 2021 and 2020, capital leases totaling $4.1 million and $3.3 million, respectively, were included in lab equipment, with related accumulated depreciation of $2.1 million and $2.4 million, respectively.
F-27
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Depreciation expense for the years ended December 31, 2021, 2020 and 2019 totaled $26.9 million, $12.6 million and $9.6 million, respectively, inclusive of $0.7 million, $0.7 million and $0.6 million, respectively, related to capital leases.
10. Investments and Equity Method Investments
The Company partners with other investors to form new ventures, including Joyn, Motif Foodworks, Inc. (“Motif”), Allonnia, LLC (“Allonnia”) and Arcaea LLC (“Arcaea”, FKA Kalo Ingredients LLC) (collectively "Platform Ventures"). The Company also partners with existing entities, including Genomatica, Inc. (“Genomatica”) and Synlogic, Inc. (“Synlogic”) (collectively, "Structured Partnerships"), with complementary assets for high potential synthetic biology applications. The Company or its subsidiaries hold equity interests in these Platform Ventures and Structured Partnerships. The Company's investments in Platform Ventures are accounted for under the equity method. The Company's investments in Synlogic, a publicly traded company, are carried at fair value. As of December 31, 2021 and 2020, the Company held 6,340,771 shares of Synlogic common stock and warrants to purchase an aggregate of 2,548,117 shares of Synlogic common stock. Prior to the third quarter of 2021, the Company's investment in Synlogic common stock was classified as an equity method investment based on the Company’s ownership interest in Synlogic and accounted for under the fair value option. Due to a decrease in the level of ownership interest during the third quarter of 2021, the investment was reclassified from equity method investments to investments on the Consolidated Balance Sheets, and from loss on equity method investments to (loss) gain on investments on the Consolidated Statements of Operations and Comprehensive Loss for all periods presented. However, the Company continues to account for its investments in Synlogic at fair value.
The Company’s non-marketable equity securities are investments in privately held companies without readily determinable fair values. The investment in Genomatica preferred stock and other non-marketable equity securities are initially recorded using the measurement alternative at cost and subsequently adjusted to fair value for any impairment and observable price changes in orderly transactions for identical or similar investments of the same issuer. As of December 31, 2021 and 2020, no impairment or adjustment from observable price changes were recognized related to investments accounted for under the measurement alternative.
Investments and equity method investments consisted of the following (in thousands):
|
|
As of December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Investments: |
|
|
|
|
|
|
||
Genomatica, Inc. preferred stock |
|
$ |
55,000 |
|
|
$ |
55,000 |
|
Synlogic, Inc. common stock |
|
|
15,345 |
|
|
|
13,696 |
|
Synlogic, Inc. warrants |
|
|
6,166 |
|
|
|
5,504 |
|
Cronos Group Inc. common stock |
|
|
10,331 |
|
|
|
— |
|
Non-marketable equity securities |
|
|
15,195 |
|
|
|
— |
|
Total |
|
$ |
102,037 |
|
|
$ |
74,200 |
|
Equity method investments (1): |
|
|
|
|
|
|
||
Joyn Bio, LLC |
|
$ |
11,694 |
|
|
$ |
28,924 |
|
Other |
|
|
1,500 |
|
|
|
— |
|
Total |
|
$ |
13,194 |
|
|
$ |
28,924 |
|
(1)
F-28
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
(Loss) gain on investments and equity method investments consisted of the following (in thousands):
|
|
Year Ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
(Loss) gain on investments: |
|
|
|
|
|
|
|
|
|
|||
Synlogic, Inc. common stock |
|
$ |
1,649 |
|
|
$ |
(2,663 |
) |
|
$ |
(19,403 |
) |
Synlogic, Inc. warrants |
|
|
662 |
|
|
|
(1,070 |
) |
|
|
(7,797 |
) |
Cronos Group Inc. |
|
|
(13,854 |
) |
|
|
|
|
|
|
||
Total |
|
$ |
(11,543 |
) |
|
$ |
(3,733 |
) |
|
$ |
(27,200 |
) |
Loss on equity method investments: |
|
|
|
|
|
|
|
|
|
|||
Joyn Bio, LLC |
|
$ |
(17,230 |
) |
|
$ |
(396 |
) |
|
$ |
(1,730 |
) |
Allonnia, LLC |
|
|
(12,698 |
) |
|
|
|
|
|
(24,480 |
) |
|
Arcaea, LLC |
|
|
(47,356 |
) |
|
|
|
|
|
|
||
Glycosyn, LLC |
|
|
|
|
|
|
|
|
(1,323 |
) |
||
Total |
|
$ |
(77,284 |
) |
|
$ |
(396 |
) |
|
$ |
(27,533 |
) |
11. Variable Interest Entities
Consolidated Variable Interest Entities
As of December 31, 2021, the Company has consolidated three VIEs: Cooksonia, Ayana Bio, LLC (“Ayana”) and Verb Biotics, LLC (“Verb”), as the Company holds variable interests in and was deemed to be the primary beneficiary of the VIEs. The other investors’ equity interests in the consolidated VIEs are presented as non-controlling interests in the accompanying consolidated financial statements.
The Company holds a 70% equity interest in Cooksonia, which was formed by the Company and certain other investors for the purposes of holding the Company’s investment in Joyn. The Company concluded that it holds a variable interest in and is the primary beneficiary of Cooksonia as it controls the most significant activities of Cooksonia by controlling 100% of the board of directors of Cooksonia and holds a controlling financial interest in Cooksonia. As a result, the Company has consolidated the financial statements of Cooksonia in accordance with ASC 810, Consolidation (“ASC 810”) into its consolidated financial statements and has recognized a non-controlling interest associated with the minority equity interest held by other investors of Cooksonia, which together hold the remaining 30% equity interest in Cooksonia.
The Company holds an interest in 9,000,000 common units (representing 100% of common units at inception) in each of Ayana and Verb, two Platform Ventures formed in September 2021 by the Company and certain of its investors. The Company has also provided Ayana and Verb with certain licenses to intellectual property for use in the development or production of products that the parties agree to research and develop under technical development plans. Additionally, in September 2021, Ayana and Verb entered into a Series A Preferred Unit Purchase Agreement under which each entity sold 9,000,000 Series A preferred units to certain of the Company’s investors for aggregate proceeds of approximately $30.0 million each. The Company concluded that it holds a variable interest in and is the primary beneficiary of Ayana and Verb as it controls the most significant activities of these entities. These conclusions were reached because, as of the primary beneficiary assessment date, for both Verb and Ayana: (i) the Company has substantive control of the board of directors; (ii) all capital contributions were made by related parties of New Ginkgo; and (iii) New Ginkgo or its related parties comprise the entirety of the Joint Steering Committee, the governing body which holds significant oversight with respect to the entities' research and development programs.
The following table presents the carrying amounts and classification of the VIEs’ assets and liabilities included in the Consolidated Balance Sheets:
F-29
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
|
|
As of December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Cash and cash equivalents |
|
$ |
58,025 |
|
|
$ |
— |
|
Prepaid expenses and other current assets |
|
|
737 |
|
|
|
— |
|
Equity method investments |
|
|
11,694 |
|
|
|
28,924 |
|
Total assets |
|
$ |
70,456 |
|
|
$ |
28,924 |
|
|
|
|
|
|
|
|
||
Accounts payable |
|
$ |
188 |
|
|
$ |
— |
|
Accrued expenses and other current liabilities |
|
|
440 |
|
|
|
— |
|
Total liabilities |
|
$ |
628 |
|
|
$ |
— |
|
Unconsolidated Variable Interest Entities
With respect to the Company’s investments in Motif, Allonnia, Genomatica and Arcaea, the Company has concluded these entities represent VIEs. However, although the Company holds board representation and is involved in the ongoing development activities of the entities via its participation on joint steering committees, the Company has concluded that it is not the primary beneficiary of these entities. This conclusion is supported by the fact that: (i) it does not control the board of directors of any of Motif, Allonnia, Genomatica or Arcaea, and no voting or consent agreements exist between the Company and other members of each respective board of directors or other investors, (ii) the holders of preferred security interests in Motif, Allonnia, Genomatica and Arcaea hold certain rights that require their consent prior to the taking of certain actions, which include certain significant operating and financing decisions, and (iii) the Company’s representation on the joint steering committee of each respective entity does not give it control over the development activities of any of Motif, Allonnia, Genomatica or Arcaea, as all votes must pass by consensus and there are no agreements in place that would require any of the entities to vote in alignment with the Company. As the Company’s involvement in Motif, Allonnia, Genomatica and Arcaea does not give it the power to control the decisions with respect to their development or other activities, which are their most significant activities, the Company has concluded that it is not the primary beneficiary of Motif, Allonnia, Genomatica or Arcaea.
With respect to Cooksonia’s investment in Joyn, as Cooksonia does not control Joyn’s board of directors, it does not have the power to control the decisions related to the development activities of Joyn, which are its most significant activities. Accordingly, the Company has concluded that Cooksonia is not the primary beneficiary of Joyn.
Additionally, the Company holds equity interests in certain privately-held companies that are not consolidated as the Company is not the primary beneficiary. As of December 31, 2021 and 2020, the maximum risk of loss related to the Company’s unconsolidated VIEs was limited to the carrying value of its investment in such entities.
Refer to Notes 10 and 20 for additional details on the Company’s investments and equity method investments.
12. Goodwill and Intangible Assets, net
Goodwill consisted of the following (in thousands):
|
|
As of December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Beginning balance |
|
$ |
1,857 |
|
|
$ |
1,857 |
|
Goodwill acquired in Dutch DNA acquisition |
|
|
15,177 |
|
|
|
— |
|
Measurement period adjustment (see Note 4) |
|
|
5,000 |
|
|
|
— |
|
Currency translation |
|
|
(722 |
) |
|
|
— |
|
Ending balance |
|
$ |
21,312 |
|
|
$ |
1,857 |
|
F-30
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Intangible assets, net consisted of the following (in thousands):
|
|
Weighted Average |
|
|
Gross |
|
|
|
Accumulated |
|
|
|
Net (1) |
|
||||||
Balances as of December 31, 2021 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Acquired technology |
|
|
13.3 |
|
|
$ |
25,038 |
|
|
|
|
$ |
(3,396 |
) |
|
|
|
$ |
21,642 |
|
Balances as of December 31, 2020 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
||||||
Acquired technology |
|
|
6.0 |
|
|
$ |
5,490 |
|
|
|
$ |
(2,196 |
) |
|
|
$ |
3,294 |
|
||
Amortization expense was $1.2 million, $0.5 million and $0.5 million for the years ended December 31, 2021, 2020 and 2019, respectively. Future amortization expense for each of the next five years will be $1.8 million.
13. Accrued Expenses and Other Current Liabilities
Accrued expenses and other current liabilities consisted of the following (in thousands):
|
|
As of December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Employee compensation and benefits |
|
$ |
3,650 |
|
|
$ |
3,037 |
|
Professional fees |
|
|
14,871 |
|
|
|
6,381 |
|
Property and equipment |
|
|
991 |
|
|
|
10,017 |
|
Product revenue accruals |
|
|
4,565 |
|
|
|
|
|
Biosecurity service revenue accruals |
|
|
28,726 |
|
|
|
1,440 |
|
Inventory related accruals |
|
|
3,538 |
|
|
|
|
|
Lab supplies |
|
|
560 |
|
|
|
4,276 |
|
External research and development expenses |
|
|
11 |
|
|
|
2,594 |
|
Liability classified stock-based compensation (Note 18) |
|
|
26,612 |
|
|
|
|
|
Capital lease obligation |
|
|
747 |
|
|
|
485 |
|
Other current liabilities |
|
|
9,061 |
|
|
|
2,275 |
|
Accrued expenses and other current liabilities |
|
$ |
93,332 |
|
|
$ |
30,505 |
|
14. Commitments and Contingencies
Lease Obligations
The Company has entered into various noncancelable operating leases for office and lab space in Boston and Cambridge, Massachusetts, Emeryville, California and Utrecht, Netherlands. The leases expire on dates ranging from 2030 to 2036 and contain periods of free rent, escalating rent, tenant improvement incentives, renewal periods, and expansion options. The Company recognizes rent expense on a straight-line basis over the term of each lease, inclusive of the free rent periods and reduced by the amortization of the tenant incentives.
The Company’s headquarters and primary operations are located in the Seaport district of Boston, Massachusetts and are comprised of a number of leases across 21, 23, 25 and 27 Drydock Avenue, which represent the Company’s most significant lease arrangements. In April 2021, the Company entered into an operating lease for a new life science campus spanning two buildings that is being constructed at 1 Au Bon Pain Way and 3 Anchor Way, Boston, Massachusetts near the Company's headquarters. The following summarizes the key terms of such leases:
21-23-25 Drydock Avenue
In March 2016, the Company entered into a noncancelable operating lease for approximately 87,000 square feet of office and lab space. The lease is comprised of five suites, the first of which was delivered to the Company in April 2016. The Company currently occupies four suites totaling approximately 70,000 square feet and the Company anticipates occupying the remaining suite in 2022. The lease contains periods of free rent, annual rent increases and tenant improvement incentives totaling $5.3 million. The lease expires in January 2030 and contains one option to extend the lease term for five years at
F-31
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
then-market rates. The lease is secured by a $1.4 million letter of credit recorded in other non-current assets on the Consolidated Balance Sheets.
At the time the Company took possession of the first three suites, the premises were in shell condition and required substantial work prior to occupancy. The Company was deemed the accounting owner during the construction period as the improvements constituted structural elements of the project. Accordingly, the Company capitalized the fair value of the leased space upon delivery from the landlord and recorded a corresponding lease financing obligation. The Company also capitalized the construction costs, leasehold improvements, and interest incurred during the construction period. Construction was complete, and the assets were placed in service, for the first three suites in September 2016, December 2017, and January 2019, respectively. Upon completion of the construction, the Company evaluated the lease and determined it did not meet the criteria for sale-leaseback treatment. Accordingly, the Company depreciates the capitalized assets and recognizes interest expense related to the lease financing obligation using the effective interest rate method over the lease term. For the years ended December 31, 2021, 2020 and 2019, the Company recognized $0.4 million of depreciation expense and $2.3 million of interest expense related to the lease. As of December 31, 2021 and 2020, the aggregate lease financing obligation for the capitalized suites totaled $16.5 million and $16.8 million, respectively.
27 Drydock Avenue
Beginning in December 2011, the Company entered into a series of noncancelable operating leases with the same landlord for an aggregate of approximately 130,000 square feet of office and lab space. In September 2021, the Company executed an amendment expanding the leased premises by approximately 48,000 square feet and extending the lease term by six years from January 2030 to January 2036. The Company has an option to extend the lease term for five years at then-market rates. The lease contains periods of free rent, annual rent increases and aggregate tenant improvement allowances of $16.1 million. As of December 31, 2021 and 2020, the unamortized balance of the tenant improvement allowance was $10.0 million and $8.1 million, respectively. The lease is secured by a letter of credit totaling $2.9 million and $1.6 million as of December 31, 2021 and 2020, respectively, recorded in other non-current assets on the Consolidated Balance Sheets.
1 Au Bon Pain Way and 3 Anchor Way
In April 2021, the Company entered into a noncancelable operating lease consisting of approximately 152,000 square feet of a new life science campus spanning two buildings that is being constructed on the property to be known as The Foundry at Drydock located at 1 Au Bon Pain Way and 3 Anchor Way, Boston, Massachusetts near the Company's headquarters. The lease commencement date is estimated to be June 1, 2024 for a portion of the premises and September 1, 2024 for the remaining premises. The lease expires on the fifteenth anniversary of the lease commencement date and includes one option to extend the lease for ten years at then-market rates. In September 2021, the Company exercised its expansion option to include the entire rentable area of the primary building resulting in approximately 262,000 square feet of leased space in aggregate. Annual base rent for the first lease year will be approximately $21.6 million, subject to annual rent increases. The lease contains periods of free rent, tenant improvement allowances based on a rate per square foot and a $14.7 million letter of credit which is recorded in other non-current assets on the Consolidated Balance Sheets. The Company has concluded that it is the deemed owner (for accounting purposes) of the buildings during the construction period. As a result, the Company has recorded $6.1 million to construction in progress in property and equipment, net and a corresponding $6.1 million lease financing obligation in its Consolidated Balance Sheets as of December 31, 2021 related to project costs incurred to date by the owner-lessor.
The Company subleases a portion of its office and lab space to Joyn and Motif. The sublease with Joyn runs coterminous with the Foundry Services Agreement (Note 20) and the sublease with Motif expires in November 2024. The Company receives approximately $0.2 million and $0.7 million per year under subleases with Joyn and Motif, respectively, and records sublease income in other (expense) income, net on the Consolidated Statements of Operations and Comprehensive Loss. As of December 31, 2021, future minimum sublease rentals under noncancelable subleases totaled $2.1 million.
F-32
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Rent expense under operating leases was $17.7 million, $7.0 million and $6.1 million for the years ended December 31, 2021, 2020 and 2019, respectively. Future minimum lease payments under noncancelable operating lease agreements, inclusive of payments for the lease financing obligations, as of December 31, 2021 are as follows (in thousands):
Years Ending December 31, |
|
Minimum Lease Payments (1) |
|
|
2022 |
|
$ |
20,600 |
|
2023 |
|
|
22,505 |
|
2024 |
|
|
33,469 |
|
2025 |
|
|
47,376 |
|
2026 |
|
|
44,092 |
|
Thereafter |
|
|
517,399 |
|
Total |
|
$ |
685,441 |
|
(1)
The Company enters into certain capital leases for lab equipment used in research and development activities. Lease terms range from to five years, may include bargain purchase options, and have fixed monthly rental payments. Future minimum lease payments under capital leases as of December 31, 2021 are as follows (in thousands):
Years Ending December 31, |
|
Minimum Lease Payments |
|
|
2022 |
|
$ |
816 |
|
2023 |
|
|
681 |
|
2024 |
|
|
438 |
|
2025 |
|
|
163 |
|
2026 |
|
|
— |
|
Thereafter |
|
|
— |
|
Total noncancelable payments |
|
$ |
2,098 |
|
Less: Imputed interest expense |
|
|
(127 |
) |
Present value of future minimum lease payments |
|
$ |
1,971 |
|
Purchase Obligations
In September 2019, the Company executed a noncancelable collaboration agreement with Berkeley Lights, Inc. (“Berkeley Lights”), under which the Company will incorporate Berkeley Lights’ Platform into its Foundry for engineering of biotherapeutics and cell-based products and the parties will jointly develop workflows for the Company's use of the Berkeley Lights’ Platform. Under the collaboration agreement, the Company is obligated to pay Berkeley Lights at least $109.0 million, and up to $150.0 million, over a seven year term for (i) Berkeley Lights’ efforts under the workflow development plans and for (ii) purchases of certain equipment, associated consumables, and other goods and services. The purchase obligation includes variable license fees based on usage of the Berkeley Lights’ Platform and milestone payments of up to $11.5 million upon achievement of development and regulatory milestones. All such license fees and milestone payments are applied against the minimum purchase commitment. The minimum purchase commitment will increase to $150.0 million if Berkeley Lights achieves certain performance targets.
For contract years one and two, which represents an 18-month period, the minimum purchase commitments must be met each year. For contract years three through seven, the minimum purchase commitment is measured on a cumulative basis with any excess applied towards the next years’ minimum. Minimum purchase commitments for the remaining contract years of the collaboration agreement are as follows (in thousands):
Contract Years |
|
Minimum Purchase Commitment |
|
|
October 1, 2020 - March 31, 2022 |
|
|
15,000 |
|
April 1, 2022 - March 31, 2023 |
|
|
14,000 |
|
April 1, 2023 - March 31, 2024 |
|
|
17,500 |
|
April 1, 2024 - March 31, 2025 |
|
|
17,500 |
|
Thereafter |
|
|
35,000 |
|
Total |
|
$ |
99,000 |
|
F-33
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
In March 2018, the Company entered into a noncancelable supply agreement with Twist Bioscience Corporation (“Twist”), a supplier of synthetic DNA. Under the supply agreement, the Company is obligated to purchase specified volumes of synthetic DNA subject to quarterly minimums over a four year term. Products purchased that contribute to the quarterly minimums can vary based on the Company’s discretion and the minimums can be adjusted up or down based on certain scenarios. During the years ended December 31, 2021, 2020 and 2019, the Company incurred $8.9 million, $10.4 million and $8.3 million, respectively, of research and development expenses under its supply agreement with Twist.
Indemnification Agreements
The Company enters into standard indemnification agreements and has agreements with indemnification clauses in the ordinary course of business. Under such arrangements, the Company indemnifies, holds harmless and agrees to reimburse the indemnified party for losses suffered or incurred by the indemnified party, who are generally the Company’s business partners. The terms of these indemnification arrangements are generally perpetual and effective any time after contract execution. The maximum potential liability resulting from these indemnification arrangements may be unlimited. The Company has never incurred costs to defend lawsuits or settle claims as a result of such indemnifications and the Company is not aware of any indemnification arrangements that could have a material effect on its financial position, results of operations or cash flows, and it has not accrued any liabilities related to such obligations as of December 31, 2021.
Legal Proceedings
The Company is not currently party to any material legal proceedings. As of each reporting date, the Company evaluates whether or not a potential loss amount or range of loss amounts is reasonably estimable and probable of being incurred and whether such amounts meet the requirements to be accrued or disclosed pursuant to ASC 450, Contingencies (“ASC 450”). The Company expenses costs related to such legal proceedings as incurred.
Registration Rights
In connection with the closing of the Business Combination, the Company entered into an amended and restated registration rights agreement (the “Registration Rights Agreement”) among the Company, SRNG and certain Old Ginkgo stockholders. Pursuant to the Registration Rights Agreement, the Company will be required to register for resale securities held by the stockholders. The Company will have no obligation to facilitate more than two demands per calendar year for each of the SRNG or the Ginkgo Holders (as defined in the Registration Rights Agreement) that the Company register such stockholders’ securities. In addition, the holders have certain “piggyback” registration rights with respect to registrations initiated by the Company. The Company will bear the expenses incurred in connection with the filing of any registration statements pursuant to the Registration Rights Agreement.
15. Warrant Liabilities
Upon the closing of the Business Combination, the Company assumed 34,499,925 publicly-traded warrants (“Public Warrants”) and 17,325,000 private placement warrants (the "Private Placement Warrants") held by the Sponsor. Both the Public Warrants and the Private Placement Warrants were issued in conjunction with the consummation of SRNG’s initial public offering on February 26, 2021. Each whole warrant entitles the holder to purchase one share of the Company’s Class A common stock at a price of $11.50 per share, subject to adjustments. The warrants will expire five years from the completion of the Business Combination, or earlier upon redemption or liquidation.
No Public Warrants will be exercisable for cash unless the Company has an effective and current registration statement covering the issuance of the shares of common stock issuable upon exercise of the Public Warrants. On November 23, 2021, the Company’s registration statement covering such shares became effective. The Company may redeem the outstanding Public Warrants:
F-34
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
If the Company calls the Public Warrants for redemption, as described above, its management will have the option to require any holder that wishes to exercise the Public Warrants to do so on a “cashless basis,” as described in the warrant agreement. The exercise price and number of ordinary shares issuable upon exercise of the Public Warrants may be adjusted in certain circumstances including in the event of a share dividend, extraordinary dividend or recapitalization, reorganization, merger or consolidation. However, except as described above, the Public Warrants will not be adjusted for issuances of ordinary shares at a price below its exercise price. Additionally, in no event will the Company be required to net cash settle the Public Warrants.
The Private Placement Warrants are identical to the Public Warrants, except that (i) the Private Placement Warrants will be exercisable on a cashless basis and be non-redeemable so long as they are held by the initial purchasers or their permitted transferees and (ii) the Private Placement Warrants and the Class A ordinary shares issuable upon exercise of the Private Placement Warrants were entitled to registration rights, which was satisfied on November 23, 2021 when the Company’s registration statement covering such shares became effective. If the Private Placement Warrants are held by someone other than the initial purchasers or their permitted transferees, the Private Placement Warrants will be redeemable by the Company and exercisable by such holders on the same basis as the Public Warrants.
As of December 31, 2021, the aggregate values of the Public Warrants and the Private Placement Warrants was $77.3 million and $58.6 million, respectively, representing warrants outstanding to purchase 34.5 million shares and 17.3 million shares, respectively, of the Company's Class A common stock. The warrants are accounted for as liabilities in accordance with ASC 815-40 and are presented within warrant liabilities on the Consolidated Balance Sheets. The warrant liabilities are measured at fair value at inception and on a recurring basis, with changes in fair value presented within change in fair value of warrant liabilities on the Consolidated Statement of Operations and Comprehensive Loss. See Note 5 for additional information.
16. Convertible Promissory Notes
In June 2019, the Company entered into a Note Purchase Agreement (“NPA”) with certain existing investors. In connection with the NPA, the Company issued convertible promissory notes (“Convertible Promissory Notes”) and received $199.0 million in cash proceeds, net of issuance costs of $1.0 million. The Convertible Promissory Notes carried interest at the rate of 3% per annum and had a maturity date of June 21, 2021. Pursuant to the NPA, all of the outstanding principal and interest under the Convertible Promissory Notes were to be automatically converted into (i) preferred stock issued in connection with the Company’s next financing that resulted in at least $50.0 million of gross proceeds (“NPA Qualified Financing”) at a 5% discount, (ii) common stock issued in connection with the filing of an effective registration statement pursuant to an initial public offering, or (iii) cash equal to the greater of (x) one and a half times the outstanding principal and interest accrued immediately prior to a sale or change in control event (as defined in the NPA) in which the Company or one of its subsidiaries was a party, or (y) the amount each investor would have received if the outstanding principal and accrued interest had been converted into Series D convertible preferred stock immediately prior to such sale or change in control event. On the maturity date, the Convertible Promissory Notes were to be automatically converted into shares of Series D convertible preferred stock, at a predetermined conversion rate, which was less than the fair value of Series D convertible preferred stock at the date of issuance of the Convertible Promissory Notes. The Company determined that at the Convertible Promissory Notes’ commitment date, this conversion feature was beneficial to the investors and, as such, calculated and recorded a beneficial conversion feature (“BCF”). The intrinsic value of the BCF, which was calculated utilizing the fair value of the underlying Series D convertible preferred stock and effective conversion price on the commitment date, was $199.0 million and was recorded as a debt discount with an offset to additional paid-in capital. The debt discount was amortized to interest expense using the effective interest method through the maturity date of the Convertible Promissory Notes. For the year ended December 31, 2019, the Company recorded interest expense of $0.1 million related to the amortization of the debt discount in the Consolidated Statements of Operations and Comprehensive Loss.
The Company’s Series E convertible preferred stock issuance in September 2019 (Note 17) met the criteria of an NPA Qualified Financing. Accordingly, the Convertible Promissory Notes were converted into Series E convertible preferred stock. In connection with the NPA Qualified Financing and the associated conversion, the Company was required to account for the repurchase of the BCF. The total repurchase price associated with the reacquisition of the BCF in connection with the settlement of the Convertible Promissory Notes was the issuance of 1,408,934 shares of Series E convertible preferred stock valued at $211.6 million. The intrinsic value of the BCF upon the NPA Qualified Financing was measured based on the intrinsic value of the conversion option at the settlement date which was in excess of the repurchase price. Therefore, the
F-35
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
entire $211.6 million was allocated to the reacquisition of the BCF which was recorded as a reduction to additional paid-in capital. As a result of the extinguishment of the Convertible Promissory Notes, the Company recorded a gain of $0.1 million in other (expense) income, net in the Consolidated Statements of Operations and Comprehensive Loss for the year ended December 31, 2019.
17. Stockholders' Equity
The Consolidated Statement of Stockholders’ Equity has been retroactively adjusted for all periods presented to reflect the Business Combination and reverse recapitalization as described in Note 3.
Old Ginkgo Convertible Preferred Stock
In 2019, the Company received $208.8 million in cash proceeds from the issuance of 69,812,427 shares of Series E convertible preferred stock to various investors at $3.06 per share, net of $4.8 million in issuance costs. In conjunction with the issuance of the Series E preferred stock, $201.0 million of principal and accrued interest associated with the Convertible Promissory Notes (Note 16) was converted at a 5% discount into 69,151,117 shares of Series E preferred stock. In May and July of 2020, the Company received $94.4 million in cash proceeds from the issuance of an additional 30,855,065 shares of Series E preferred stock to various investors at $3.06 per share.
Immediately prior to the closing of the Business Combination on September 16, 2021, all outstanding Series B, C, D, and E convertible preferred stock converted into shares of Old Ginkgo common stock on a one-for-one basis. Upon closing of the Business Combination, those shares converted into an aggregate 904.7 million shares of New Ginkgo's Class A common stock pursuant to the Exchange Ratio established in the Merger Agreement. All fractional shares were rounded down.
New Ginkgo Preferred Stock
The Company is authorized to issue 200,000,000 shares of preferred stock with a par value $0.0001 per share. The Company’s board of directors are authorized, without stockholder approval, to issue such shares of preferred stock in one or more series, to establish from time to time the number of shares to be included in each such series, and to fix the designations, powers, voting, and other rights, preferences and privileges of the shares. There were no issued and outstanding shares of preferred stock as of December 31, 2021.
Common Stock
The Company is authorized to issue 15,800,000,000 shares of common stock, including 10,500,000,000 shares of Class A common stock, par value $0.0001 per share, 4,500,000,000 shares of Class B common stock, par value $0.0001 per share, and 800,000,000 shares of Class C common stock, par value $0.0001 per share.
Voting
Holders of Class A common stock are entitled to one vote per share and holders of Class B common stock are entitled to ten votes per share. Holders of Class C common stock are not entitled to vote except as otherwise expressly provided in the certificate of incorporation or required by applicable law.
Dividends
Common stockholders are entitled to receive dividends, as may be declared by the board of directors. Different classes of common stock are legally entitled to equal per share distributions whether through dividends or liquidation. No dividends have been declared to date.
Conversion
Each share of Class B common stock is convertible at any time at the option of the holder into one share of Class A common stock. Generally, shares of Class B common stock will convert automatically into Class A common stock upon the holder ceasing to be an Eligible Holder (i.e., director, employee, trust or legal entity of New Ginkgo), unless otherwise determined by affirmative vote of a majority of independent directors of New Ginkgo.
F-36
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Treasury Stock
During the year ended December 31, 2019, the Company repurchased 490,805 shares of common stock from its employees. The fair value of the common stock on the repurchase date was recorded as a reduction to stockholders’ equity and the excess $0.1 million paid over the fair value was recorded as additional compensation expense. Upon the repurchase, the Company returned all shares of treasury stock to authorized and unissued shares of common stock by deducting the carrying value of treasury stock from common stock and additional paid-in capital. As of December 31, 2021 and 2020, no shares of common stock remained in treasury.
Common Stock Reserved for Future Issuances
The Company had the following common stock reserved for future issuance as of the date indicated:
|
|
December 31, 2021 |
|
|
Stock options issued and outstanding |
|
|
25,228,853 |
|
Restricted stock units outstanding |
|
|
168,321,952 |
|
Shares available for grant under the 2021 Plan |
|
|
200,569,979 |
|
Shares available for grant under the ESPP |
|
|
20,000,000 |
|
Warrants to purchase Class A common stock |
|
|
51,824,925 |
|
Total common stock reserved for future issuances (1) |
|
|
465,945,709 |
|
(1)
18. Stock-Based Compensation
2021 Incentive Award Plan
On September 16, 2021, the 2021 Incentive Award Plan (the “2021 Plan”) became effective. The 2021 Plan provides for the grant of stock options, including incentive stock options (“ISOs”) and nonqualified stock options, stock appreciation rights, restricted stock, dividend equivalents, RSUs and other stock or cash-based awards to employees, consultants and directors of New Ginkgo and its subsidiaries.
The aggregate number of shares of common stock available for issuance under the 2021 Plan, which may be issued as Class A common stock and/or Class B common stock, was initially 200,440,957 shares. As of December 31, 2021, 200,569,979 shares are available for future issuance under the 2021 Plan. The number of shares of common stock reserved for issuance under the 2021 Plan will automatically increase for ten years on January 1 of each year, starting on January 1, 2022, in an amount equal to the lesser of (a) 4.0% of the aggregate number of shares of common stock outstanding on the final day of the immediately preceding calendar year and (b) such smaller number of shares as is determined by the Board. The maximum number of shares of common stock that may be issued pursuant to the exercise of incentive stock options granted under the 2021 Plan is 200 million shares. Shares issued under the 2021 Plan may consist of authorized but unissued shares, shares purchased on the open market or treasury shares.
2021 Employee Stock Purchase Plan
On September 16, 2021, the 2021 Employee Stock Purchase Plan (the “ESPP”) became effective. The ESPP authorizes (i) the grant of options that are intended to qualify for favorable U.S. federal tax treatment under Section 423 of the Internal Revenue Code of 1986 (the “Section 423 Component”) and (ii) the grant of options that are not intended to be tax-qualified (the “Non-Section 423 Component”). All of the Company’s employees are expected to be eligible to participate in the ESPP. However, with respect to the Section 423 Component, an employee may not be granted rights to purchase stock under the ESPP if the employee, immediately after the grant, would own (directly or through attribution) stock possessing 5% or more of the total combined voting power or value of all classes of the Company’s common stock.
The ESPP permits the Company to deliver up to 20 million shares of common stock pursuant to awards issued under the ESPP, which may be Class A common stock and/or Class B common stock. The number of shares of common stock reserved for issuance under the ESPP will automatically increase each January 1, beginning on January 1, 2022, by an amount equal to the lesser of (a) 1% of the aggregate number of shares of common stock outstanding on the final day of the immediately preceding calendar year and (b) such smaller number of shares as is determined by the Board, provided that no more than 100 million shares may be issued under the Section 423 Component. Prior to or in connection with issuing any shares of common
F-37
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
stock under the ESPP, the ESPP administrator may convert awards covering shares of Class B common stock to Class A common stock. As of December 31, 2021, no awards have been granted under the ESPP.
2014 Stock Incentive Plan
The 2014 Stock Incentive Plan (the “2014 Plan”) provided for the Company to grant options, stock appreciation rights, restricted stock, restricted stock units (“RSUs”) and other stock-based awards. From and after the effective date of the 2021 Incentive Award Plan, the Company ceased granting awards under the 2014 Plan. However, the 2014 Plan continues to govern the terms and conditions of the outstanding awards previously granted thereunder. Shares of common stock underlying any awards that are forfeited, cancelled, repurchased, or otherwise terminated by the Company under the 2014 Plan will be added back to the shares available for issuance under the 2021 Incentive Award Plan.
2008 Stock Incentive Plan
The 2008 Stock Incentive Plan (the “2008 Plan”) provided for the Company to grant options and restricted stock awards (“RSAs”). From and after the effective date of the 2014 Stock Incentive Plan, the Company ceased granting awards under the 2008 Plan. However, the 2008 Plan continues to govern the terms and conditions of the outstanding awards previously granted thereunder. Shares of common stock underlying any awards that are forfeited, cancelled, repurchased, or otherwise terminated by the Company under the 2008 Plan will be added back to the shares available for issuance under the 2021 Incentive Award Plan.
Stock Options
Options outstanding under the 2008 Plan and 2014 Plan are fully vested. Options outstanding under the 2021 Plan consists of options granted to a non-employee director and vest in three equal annual installments. All stock options expire no later than ten years after the grant date. The exercise price of each option under the 2021 Plan is equal to the closing price of the Company’s common stock on the date of grant.
A summary of stock option activity for the year ended December 31, 2021 is presented below:
|
|
Number |
|
|
Weighted |
|
|
Weighted |
|
|
|
Aggregate |
|
|||||
|
|
|
|
|
|
|
|
(in years) |
|
|
|
(in thousands) |
|
|||||
Outstanding as of December 31, 2020 |
|
|
33,354,871 |
|
|
$ |
0.02 |
|
|
|
|
|
|
|
|
|||
Granted |
|
|
1,664,251 |
|
|
$ |
0.50 |
|
|
|
|
|
|
|
|
|||
Exercised |
|
|
(10,969,639 |
) |
|
$ |
0.02 |
|
|
|
|
|
|
|
|
|||
Forfeited |
|
|
(1,594,820 |
) |
|
$ |
0.02 |
|
|
|
|
|
|
|
|
|||
Outstanding as of December 31, 2021 |
|
|
22,454,663 |
|
|
$ |
0.05 |
|
|
|
2.32 |
|
|
|
|
$ |
185,620 |
|
Exercisable as of December 31, 2021 |
|
|
22,385,232 |
|
|
$ |
0.02 |
|
|
|
2.30 |
|
|
|
|
$ |
185,620 |
|
(1)
The aggregate intrinsic value of stock options exercised during the years ended December 31, 2021, 2020 and 2019 was $91.0 million, $5.3 million and $1.1 million, respectively. The weighted-average fair value of options granted during the year ended December 31, 2021 was $8.97 per share and was calculated using the following assumptions. No options were granted during 2020 and 2019.
|
|
Year Ended |
|
|
|
|
December 31, 2021 |
|
|
Risk-free interest rate |
|
|
0.11 |
% |
Dividend yield |
|
|
0 |
% |
Expected volatility |
|
|
88.6 |
% |
Expected term (in years) |
|
|
0.96 |
|
Restricted Stock and Restricted Stock Units
RSAs granted under the 2014 Plan are subject to a service-based vesting condition and generally vest in equal monthly installments over four years. RSUs granted under the 2014 Plan are subject to two vesting conditions: (i) a service-based
F-38
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
vesting condition that is generally met over four years with 25% of the shares vesting on the first anniversary of the grant date with monthly vesting thereafter, and (ii) a performance-based vesting condition that is met through a liquidity event in the form of either a change of control or an initial public offering (“the performance condition”). RSUs granted under the 2021 Plan are subject to a service-based vesting condition only that is generally met over four years with 25% of the shares vesting on the first anniversary of the grant date with monthly vesting thereafter.
Prior to the Business Combination, no stock-based compensation expense had been recognized related to RSUs granted under the 2014 Plan as the performance condition was not probable of being met and the Business Combination did not meet the definition of a liquidity event as defined in the 2014 Plan. As a result of the Business Combination, on November 17, 2021 the Board of Directors modified the vesting terms of RSUs granted under the 2014 Plan to allow 10% of the RSUs that met the service condition as of the closing of the Business Combination (the “10% RSUs”) to vest with respect to the performance condition, effective as of November 19, 2021, the date on which the Form S-8 registration statement covering such shares became effective. In addition, on November 17, 2021 the Board of Directors modified the vesting terms of the remaining RSUs granted under the 2014 Plan such that they will vest in full with respect to the performance condition on or before March 15, 2022 (the original service-based vesting condition is still applicable). As a result of these modifications, the performance condition for all RSUs granted under the 2014 Plan became probable of being met during the fourth quarter of 2021. As the performance condition was not probable of being met prior to the modification, the RSU awards were remeasured as of the modification date pursuant ASC 718 and the Company recorded a cumulative-catch up adjustment to reflect the change in the probability assessment. The modification resulted in approximately $1,492.2 million of incremental stock-based compensation expense recognized in the fourth quarter of 2021 based on the modification-date fair value. The Company cash settled the 10% RSUs for a total cash payment of $76.5 million equal to the fair value of the stock on the Form S-8 effective date.
In September 2021, the Board of Directors modified the terms of RSUs granted to non-employee directors by adding a cash settlement feature to the awards which allowed the non-employee directors to elect to settle in cash up to 50% of their RSUs that were vested with respect to the service condition on or prior to December 31, 2021 (the “50% RSUs”). The director RSUs were subject to the same performance condition as all other RSUs granted under the 2014 Plan. In the fourth quarter of 2021, all directors elected to cash settle the 50% RSUs. As a result, the 50% RSUs are classified as liability awards and the liability is measured at fair value at the reporting date. The aggregate fair value of the liability classified awards was $26.6 million as of December 31, 2021 which is included in accrued expenses and other current liabilities on the Consolidated Balance Sheets.
A summary of the RSU and RSA activity for the year ended December 31, 2021 is presented below:
|
|
Restricted Stock Units |
|
|
Restricted Stock Awards |
|
||||||||||
|
|
Number of |
|
|
Weighted |
|
|
Number of |
|
|
Weighted |
|
||||
Nonvested as of December 31, 2020 |
|
|
124,932,207 |
|
|
$ |
1.74 |
|
|
|
419,049 |
|
|
$ |
1.99 |
|
Granted |
|
|
133,307,479 |
|
|
$ |
13.53 |
|
|
|
— |
|
|
|
|
|
Vested |
|
|
(85,829,389 |
) |
|
$ |
13.39 |
|
|
|
(236,427 |
) |
|
$ |
1.99 |
|
Forfeited |
|
|
(4,088,345 |
) |
|
$ |
5.23 |
|
|
|
— |
|
|
|
|
|
Nonvested as of December 31, 2021 |
|
|
168,321,952 |
|
|
$ |
13.58 |
|
|
|
182,622 |
|
|
$ |
1.99 |
|
(1) The weighted average grant date fair value of awards nonvested as of December 31, 2020 and awards forfeited prior to the modification date reflect the original grant date fair value and not the modification-date fair value.
The weighted average grant date fair value of RSUs granted during the year ended December 31, 2021 was $13.53, which represents the weighted average of the modification-date fair value and any post modification grant date fair values. The weighted average grant date fair value of RSUs granted during the years ended December 31, 2020 and 2019 was $2.68 and $1.78 per share, respectively, and is no longer relevant for expense recognition due to the modification in the fourth quarter of 2021. The weighted average grant date fair value of the RSAs granted during the year ended December 31, 2019 was $1.99 per share. No RSAs were granted during 2021 and 2020.
The aggregate fair value of the RSUs that vested during the year ended December 31, 2021 was $1,149.5 million. No RSUs vested during 2020 and 2019 as the performance condition was not probable of being met. The aggregate fair value of the RSAs that vested during the years ended December 31, 2021, 2020 and 2019 was $0.5 million, $0.5 million and $0.7 million, respectively.
F-39
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Earnouts
As described in Note 3, the holders of Rollover Equity Awards outstanding immediately prior to the effective time of the Business Combination received a proportional amount of the Earnout Consideration, which is divided into four equal tranches subject to vesting during the five years after the Closing Date (the “Earnout Period”). The earnout shares in respect of the Rollover Equity Awards are subject to the same terms and conditions as the underlying Rollover Equity Awards (including with respect to vesting and termination-related provisions). Additionally, the earnout shares in respect of the Rollover Equity Awards are subject to a market condition that will be met when the trading price of the Company's common stock is greater than or equal to $12.50, $15.00, $17.50 and $20.00 for any 20 trading days within any period of 30 consecutive trading days during the Earnout Period (collectively, the “Earnout Targets”). To the extent that the Earnout Targets are not achieved during the Earnout Period, the portion of the Earnout Consideration that remains subject to vesting and forfeiture at the end of the Earnout Period will be forfeited to New Ginkgo for no consideration and cancelled.
As described above, the earnout shares related to Old Ginkgo RSUs ("Earnout RSUs") are subject to the same performance condition as the underlying RSUs. As a result of the November 2021 modification to the RSUs described above, the performance condition became probable of being met in the fourth quarter of 2021. The modification resulted in approximately $173.5 million of incremental stock-based compensation expense recognized in the fourth quarter of 2021 related to the Earnout RSUs based on the modification-date fair value. The first earnout target of $12.50 per share was met on November 15, 2021 and the earnout shares related to the first tranche of the Earnout Consideration for which the service condition had also been met became vested and were settled, less shares withheld to cover tax withholding obligations.
The grant date fair value of Earnout RSUs was estimated on the Closing Date and remeasured on the modification date using a Monte Carlo simulation model with the following assumptions:
|
|
Year Ended |
|
|
|
|
December 31, 2021 |
|
|
Risk-free interest rate |
|
0.84% - 1.21% |
|
|
Expected volatility |
|
53.1% - 81% |
|
|
Expected term (in years) |
|
4.83 - 5 |
|
|
Dividend yield |
|
|
— |
|
A summary of activity during the year ended December 31, 2021 for the Earnout RSUs and the earnout shares underlying Old Ginkgo RSAs ("Earnout RSAs") is presented below:
|
|
Number of |
|
|
Weighted |
|
||
Nonvested as of December 31, 2020 |
|
|
— |
|
|
|
|
|
Granted |
|
|
30,539,475 |
|
|
$ |
12.91 |
|
Vested |
|
|
(2,580,570 |
) |
|
$ |
13.33 |
|
Forfeited |
|
|
(95,780 |
) |
|
$ |
12.92 |
|
Nonvested as of December 31, 2021 |
|
|
27,863,125 |
|
|
$ |
12.87 |
|
The aggregate fair value of the Earnout RSUs and Earnout RSAs that vested during the year ended December 31, 2021 was $34.4 million.
Stock-Based Compensation
Stock-based compensation expense was allocated as follows (in thousands):
|
|
Year Ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Research and development |
|
$ |
926,730 |
|
|
$ |
79 |
|
|
$ |
64 |
|
General and administrative |
|
|
755,835 |
|
|
|
397 |
|
|
|
707 |
|
Total |
|
$ |
1,682,565 |
|
|
$ |
476 |
|
|
$ |
771 |
|
As of December 31, 2021, there was $0.5 million of unrecognized compensation expense related to stock options to be recognized over a weighted-average period of 2.4 years, $2,160.8 million of unrecognized compensation expense related to
F-40
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
RSUs and Earnout RSUs to be recognized over a weighted-average period of 1.6 years and $0.4 million of unrecognized compensation expense related to RSAs and Earnout RSAs to be recognized over a weighted-average period of 1.0 year.
19. Revenue Recognition
Disaggregation of Revenue
The following table sets forth the percentage of total Foundry revenue by industry:
|
|
Year Ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Consumer and technology |
|
|
36 |
% |
|
|
12 |
% |
|
|
19 |
% |
Food and nutrition |
|
|
25 |
% |
|
|
35 |
% |
|
|
39 |
% |
Industrial and environment |
|
|
16 |
% |
|
|
29 |
% |
|
|
13 |
% |
Agriculture |
|
|
8 |
% |
|
|
13 |
% |
|
|
18 |
% |
Pharma and Biotech |
|
|
8 |
% |
|
|
2 |
% |
|
|
2 |
% |
Government and Defense |
|
|
7 |
% |
|
|
9 |
% |
|
|
9 |
% |
Total Foundry revenue |
|
|
100 |
% |
|
|
100 |
% |
|
|
100 |
% |
The Company’s revenue is derived from customers located primarily in the United States. For the years ended December 31, 2021, 2020, and 2019, the Company’s revenue from customers within the United States comprised 86%, 88% and 81%, respectively, of total revenue.
Contract Balances
The Company recognizes a contract asset when the Company transfers goods or services to a customer before the customer pays consideration or before payment is due, excluding any amounts presented as accounts receivable. The Company did not have any contract assets as of December 31, 2021 and 2020.
Contract liabilities, or deferred revenue, primarily consist of payments received in advance of performance under the contract or when the Company has an unconditional right to consideration under the terms of the contract before it transfers goods or services to the customer. The Company’s collaborative arrangements with its equity investees and related parties typically include upfront payments consisting of cash or non-cash consideration for future research and development services and non-cash consideration in the form of equity securities for licenses that will be transferred in the future. The Company records the upfront cash payments and fair value of the equity securities as deferred revenue.
The Company also invoices customers based on contractual billing schedules, which results in the recording of deferred revenue to the extent payment is received prior to the Company’s performance of the related services. Contract liabilities are recognized as revenue as (or when) the Company performs under the contract.
Of the Company’s $128.5 million in deferred revenue at December 31, 2020, $28.8 million was recognized as revenue during 2021. Of the Company’s $147.9 million in deferred revenue at December 31, 2019, $25.5 million was recognized as revenue during 2020.
Performance Obligations
The aggregate amount of the transaction price that was allocated to performance obligations that have not yet been satisfied or are partially satisfied as of December 31, 2021 and 2020 was $21.1 million and $20.7 million, respectively. The Company has elected the practical expedient not to provide the remaining performance obligation disclosures related to contracts for which the Company recognizes revenue on a cost-plus basis in the amount to which it has the right to invoice and for contracts with a term of one year or less. As of December 31, 2021, of the performance obligations not yet satisfied or partially satisfied, approximately 90% is expected to be recognized as revenue during the years 2022 to 2026. The remainder cannot be reasonably estimated due to uncertainty about the timing of future events, including development milestones. When a milestone subject to the variable consideration constraint is achieved, the Company updates its estimate of the transaction price to include the milestone payment and records a cumulative catch-up in revenue. During the year ended December 31, 2021, the Company recorded a $6.4 million cumulative catch-up in revenue primarily due to recognition of previously constrained variable consideration related to milestones. The cumulative catch-up adjustment in years 2020 and 2019 was not material.
F-41
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
20. Significant Collaboration Transactions
Arcaea LLC (FKA Kalo Ingredients, LLC)
Summary of Arrangement
Arcaea was formed in March 2021 to focus on the application of synthetic biology in the personal care products industry. In March 2021, the Company entered into (i) an Intellectual Property Contribution Agreement (“Arcaea IP Agreement”) that granted Arcaea a license to certain of the Company’s intellectual property, (ii) a Technical Development Agreement (“Arcaea TDA”) that establishes the terms under which the Company will provide technical research and development services, and (iii) a Common Unit Issuance Agreement (“Arcaea CUIA”) which compensates the Company for its intellectual property contribution. Contemporaneous with these transactions, Arcaea entered into a Series A Preferred Unit Purchase Agreement under which it sold 1,755,000 Series A preferred units to certain of the Company’s investors, for aggregate proceeds of approximately $19.5 million. The Series A Preferred Unit Purchase Agreement provided for the sale and issuance of up to an additional 7,245,000 Series A preferred units subsequent to the initial closing. In subsequent closings during 2021, Arcaea issued an additional 5,139,900 Series A preferred units to existing and third-party investors for aggregate proceeds of approximately $57.1 million and closed its Series A preferred unit financing. As a result, the Company received an additional 5,229,900 common units in Arcaea for total consideration of $35.5 million.
Under the Arcaea IP Agreement, the Company licensed certain intellectual property to Arcaea for use in the development or the production of Arcaea’s products that the parties will subsequently agree to research and develop under technical development plans (“TDPs”). The license rights provide Arcaea with the ability to commercialize the specified products from the corresponding TDP under the Arcaea TDA. In return for the license to the intellectual property, Arcaea has agreed to issue the Company up to 9,000,000 common units in accordance with certain terms and conditions set forth within the agreements. The Company received 1,755,000 common units upon execution of the Arcaea CUIA and an additional 5,229,900 common units upon closing of the Series A preferred unit financing in July 2021 (as discussed above). No additional common units are expected to be issued to the Company.
Under the Arcaea TDA, the parties jointly agree on TDPs, through equal representation on a joint steering committee, under which the Company will perform agreed-upon research and development services in return for consideration on a cost-plus basis for all services provided.
Accounting Analysis
The common unit investment in Arcaea is considered an equity method investment as a result of the Company’s ability to exercise significant influence over Arcaea’s financial and operating policies through its ownership of common units. The initial carrying value of the equity method investment in Arcaea is the fair value of the common units of $11.9 million received in exchange for the Arcaea IP Agreement which, as discussed below, was accounted for as deferred revenue at inception. The fair value of Arcaea’s common units was determined at inception of the agreements using the option pricing method. The option pricing method used a back-solve methodology to infer the total equity value based on the pricing of the Series A preferred unit financing, which was contemporaneous with the Arcaea IP Agreement. Further, the Company determined the rights to up to an additional 7,245,000 common units did not meet the definition of a freestanding financial instrument and are not representative of a derivative. The right to the additional common units is considered variable consideration that is fully constrained at inception and until the contingencies related to the issuance of the additional shares are resolved.
The Series A preferred units issued by Arcaea receive a liquidation preference prior to common units. As such, the Company concluded that this represents a substantive profit-sharing arrangement, and the Company is recognizing earnings and losses on the equity method investment using the HLBV method. The Company recorded a $11.9 million loss on its equity method investment in Arcaea in 2021. The loss allocated to the Company primarily relates to Arcaea’s accounting for the non-cash consideration related to the Arcaea IP Agreement as in-process research and development, which resulted in the full value of the Company’s intellectual property contribution being expensed in 2021. As of December 31, 2021, the carrying value of the equity method investment in Arcaea has been reduced to zero. There is no commitment for the Company to provide further financial support to Arcaea, and therefore the carrying value of the equity method investment will not be reduced below zero.
The relationship with Arcaea is a vendor-customer relationship and is within the scope of ASC 606, as the provision of services and corresponding license rights are considered a part of the Company’s ordinary activities. The common units issued to the Company represent non-cash consideration. While the Arcaea TDA has been executed by the parties and provides the payments terms for future services, the Arcaea TDA does not provide for any transfer of goods or services
F-42
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
between the parties. However, the Company will provide licenses and services upon execution of the contemplated TDPs. Accordingly, the Company concluded that the Arcaea TDA, in combination with the Arcaea CUIA, met the definition of a contract under ASC 606. Each TDP executed under the Arcaea TDA will be accounted for in accordance with ASC 606.
The Company’s performance obligations under the contract consist of ten material rights to future technical research and development services and commercial licenses under individual TDPs that the Company expects to execute under the Arcaea TDA. The material rights represent an advance payment for the license rights, which will be granted upon the execution of future TDPs. As there is no additional payment for these license rights when future TDPs are executed, the Company has determined that there is a material right associated with each of the contemplated additional TDPs under the Arcaea TDA. The Company has allocated approximately $1.2 million of the upfront non-cash consideration to each of the ten material rights based on the estimated standalone selling price of the performance obligations. During the year ended December 31, 2021, the additional non-cash consideration received of $35.5 million, which is representative of previously constrained variable consideration, was allocated to each of the ten performance obligations under the arrangement with Arcaea of $3.6 million each consistent with the initial relative selling price allocation. Unexercised material rights are recorded as non-current deferred revenue until such time as the parties execute a TDP conveying a commercial license.
Upon the execution of a TDP underlying a material right, the Company is obligated to provide technical research and development services under the TDP and a license to applicable patents and other intellectual property designed and developed under the TDP. The technical research and development services and license provided under a TDP are highly interdependent and interrelated with one another. Without the Company’s knowledge, expertise, and platform, there would not be a licensable strain or other commercializable product to transfer to Arcaea. Further, Arcaea has rights to development intellectual property created as part of each TDP, irrespective of the result of the development. Therefore, each executed TDP underlying a material right consists of one combined performance obligation for the technical research and development services and license to be provided by the Company.
For each TDP underlying a material right, the transaction price consists of variable consideration for the most likely amount of estimated consideration to be received under the cost-plus arrangement and non-cash consideration allocated to the material rights. As the services performed by the Company under a TDP create or enhance an asset that Arcaea controls as the asset is created or enhanced, the Company satisfies the performance obligation and recognizes revenue over time. The Company uses an input method that compares total costs incurred relative to total estimated cost to complete to estimate progress under the contract. Any revisions to the estimated total budgeted costs to complete, and the resulting impact to revenue recognition, are reflected in the period of the change through a cumulative catch-up adjustment.
As of December 31, 2021, the Company had a deferred revenue balance of $47.4 million with Arcaea. During the year ended December 31, 2021, the Company recognized revenue of $3.7 million from services provided to Arcaea.
Allonnia, LLC
Summary of Arrangement
In December 2019, the Company entered into (i) an Intellectual Property Contribution Agreement (“Allonnia IP Agreement”) that granted Allonnia a license to certain of the Company’s intellectual property, (ii) a Technical Development Agreement (“Allonnia TDA”) that establishes the terms under which the Company is providing technical development services, and (iii) a Common Unit Issuance Agreement (“CUIA”) which provides for the issuance of common units of Allonnia to the Company in exchange for the license rights granted under the Allonnia IP Agreement. Contemporaneous with these agreements, Allonnia entered into a Series A Preferred Unit Purchase Agreement under which Allonnia sold 2,970,000 Series A Preferred Units to certain of the Company’s investors, as well as a third-party investor, for aggregate proceeds of approximately $33.0 million. Allonnia also agreed to issue an additional 630,000 Series A Preferred Units to a strategic partner as compensation for the delivery of future services to Allonnia. The Series A Preferred Unit Purchase Agreement also provided for the sale and issuance of up to an additional 5,400,000 Series A Preferred Units subsequent to the initial closing. In 2020, Allonnia issued an additional 1,844,911 Series A Preferred Units, 1,664,911 of which were sold for aggregate proceeds of $18.5 million and 180,000 of which were issued in exchange for the rights to certain intellectual property which will vest based on the achievement of milestones associated with the development of the intellectual property received. In 2021, Allonnia issued an additional 22,500 Series A Preferred Units for aggregate proceeds of $0.2 million and closed their Series A Preferred Unit financing.
Under the Allonnia IP Agreement, the Company licensed intellectual property to Allonnia for use in the development or the production of its products that the parties will subsequently agree to develop under TDPs. The license rights provide Allonnia
F-43
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
with the ability to commercialize the specified products from the corresponding strain or enzyme, which can only be developed by the Company under the Allonnia TDA. The Company received 3,600,000 common units as consideration for the license upon execution of the Allonnia IP Agreement and an additional 1,867,411 common units during the year ended December 31, 2021 in connection with the closing of the Series A preferred unit financing.
Under the Allonnia TDA, the parties jointly agree, through equal representation on a joint steering committee, on TDPs for specific strains and enzymes, in which the Company will perform agreed upon development services in return for consideration on a cost-plus basis for all services provided.
Accounting Analysis
The common unit investment in Allonnia is considered an equity method investment as a result of the Company’s ability to exercise significant influence over Allonnia's financial and operating policies through its ownership of common units. The initial carrying value of the equity method investment in Allonnia is the fair value of the common units of $24.5 million received in exchange for the Allonnia IP Agreement which, as discussed below, was accounted for as deferred revenue at inception. The fair value of Allonnia’s common units was determined at inception of the agreements using the option pricing method. The option pricing method used a back-solve methodology to infer the total equity value based on the pricing of the Series A Preferred Unit financing, which was contemporaneous with the Allonnia IP Agreement. Further, the Company determined the rights to up to an additional 5,400,000 common units did not meet the definition of a freestanding financial instrument and are not representative of a derivative. The right to the additional common units is considered variable consideration that is fully constrained at inception and until the contingencies related to the issuance of the additional shares are resolved. This contingency was resolved in 2021 when the Company received an additional 1,867,411 common units in connection with the closing of the Series A preferred unit financing.
The Series A Preferred Units issued by Allonnia receive a liquidation preference prior to common units. As such, the Company concluded that this represents a substantive profit-sharing arrangement and the Company is recognizing earnings and losses on the equity method investment using the HLBV method. The Company recorded a loss on equity method investment of $24.5 million in 2019 and $12.7 million in 2021 as a result of the application of the HLBV method. The loss allocated to the Company primarily relates to Allonnia’s accounting for the non-cash consideration related to the Allonnia IP Agreement as in-process research and development, which resulted in the full value of the Company’s intellectual property contribution being expensed in the year that the shares were issued. As of December 31, 2021, the carrying value of the equity method investment in Allonnia has been reduced to zero. There is no commitment for the Company to provide further financial support to Allonnia and therefore the carrying value of the equity method investment will not be reduced below zero.
The relationship with Allonnia is a vendor-customer relationship and is within the scope of ASC 606 as the provision of services and corresponding license rights are considered a part of the Company’s ordinary activities and the common units represent non-cash consideration. While the Allonnia TDA has been executed by the parties and provides the payments terms for future services, the Allonnia TDA does not provide for any transfer of goods or services between the parties. However, the Company will provide licenses and services upon execution of the contemplated TDPs. Accordingly, the Company concluded that the Allonnia TDA met the definition of a contract under ASC 606 and each TDP executed under the Allonnia TDA will be accounted for in accordance with ASC 606.
The Company’s performance obligations under the contract consist of a ten material rights related to the estimated number of TDPs the parties expect to execute under the Allonnia TDA. The material rights represent an advance payment for the license rights which will be granted upon the execution of each TDP. As there is no additional payment for these license rights upon execution of a TDP, the Company has determined that there is a material right associated with each of the contemplated future TDPs. The Company has allocated $2.5 million of the upfront non-cash consideration to each of the ten performance obligations under the contract based on the estimated standalone selling price of the performance obligations. Unexercised material rights are recorded as non-current deferred revenue until such time as the parties execute a TDP.
Upon the execution of each TDP, the Company is obligated to provide development services under the TDP and a license to applicable patents and other intellectual property to the ingredient developed under the plan. The license and research and development services under a TDP are highly interdependent and interrelated with one another. Without the Company’s knowledge, expertise, and platform, there would not be a licensable strain or other commercializable product to transfer to Allonnia. Further, Allonnia has rights to all development intellectual property created as part of each TDP, irrespective of the
F-44
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
result of the development. Therefore, each executed TDP consists of one combined performance obligation for the license and research and development services to be performed by the Company.
For each TDP, the transaction price consists of variable consideration for the most likely amount of estimated consideration to be received under the cost-plus arrangement and the $2.5 million allocation of the fixed non-cash consideration. As the services performed by the Company create or enhance an asset that Allonnia controls as the asset is created or enhanced, the Company satisfies the performance obligation and recognizes revenue over time. The Company uses an input method that compares total costs incurred relative to total estimated cost to complete to estimate progress under the contract. Any revisions to the estimated total budgeted costs to complete, and the resulting impact to revenue recognition, are reflected in the period of the change through a cumulative catch-up adjustment. In 2021, the additional non-cash consideration of $12.7 million, which represents previously constrained variable consideration, was allocated to all of the performance obligations consistent with the initial relative selling price allocation and a cumulative catch up was recognized for the TDPs in process.
As of December 31, 2021 and 2020, the Company had a deferred revenue balance of $38.0 million and $26.1 million, respectively, with Allonnia. During the years ended December 31, 2021, 2020 and 2019, the Company recognized revenue of $5.1 million, $5.0 million and $0 million, respectively, from services provided to Allonnia.
Motif FoodWorks, Inc.
Summary of Arrangement
In September 2018, the Company entered into (i) an Intellectual Property Contribution Agreement (“Motif IP Agreement”) with Motif that granted Motif a license to certain of the Company’s intellectual property and (ii) a Technical Development Agreement (“Motif TDA”) that establishes the terms under which the Company is providing technical development services.
Under the Motif IP Agreement, the Company licensed intellectual property to Motif for use in strain development to produce ingredients that the parties will subsequently agree to develop under TDPs. The license rights provide Motif with the ability to commercialize the specified ingredients from the corresponding strain, which can only be developed by the Company under the Motif TDA. In return for the license to the intellectual property, Motif granted the Company 9,000,900 shares of common stock. Concurrent with the Motif IP Agreement, Motif also sold 8,100,720 shares of Series A preferred stock to certain of the Company’s investors, as well as third-party investors, for aggregate proceeds of approximately $90.0 million.
The Motif TDA governs the procurement of the Company’s expertise and technical development services to collaborate in the research, development, and commercialization of specified ingredients. Under the Motif TDA, the parties jointly agree on TDPs for specific ingredients, in which the Company will perform agreed upon development services in return for consideration on a cost-plus fixed margin basis for all services provided. At inception, the Company estimated that it would execute ten TDPs with Motif.
Accounting Analysis
The investment in Motif common stock is considered an equity method investment as a result of the Company’s ability to exercise significant influence over the financial and operating policies through its common stock ownership. The initial carrying value of the equity method investment in Motif is the fair value of the common stock received in exchange for the Motif IP Agreement of $65.1 million which, as discussed below, is being accounted for as non-cash consideration under ASC 606. As Motif’s Series A preferred stockholders receive a liquidation preference prior to common stock, the Company concluded that this represents a substantive profit-sharing arrangement. Accordingly, the Company is recognizing earnings and losses on the equity method investment using the HLBV method. The Company recorded a loss on equity method investment of $65.1 million from inception through December 31, 2018 which reduced the carrying value to zero. The loss allocated to the Company primarily relates to Motif’s accounting for the non-cash consideration related to the Motif IP Agreement as in-process research and development, which resulted in the full value of Company’s intellectual property contribution being expensed in the period ended December 31, 2018, at which time the carrying value of the equity method investment in Motif had been reduced to zero. There is no commitment for the Company to provide further financial support to Motif and therefore the carrying value of the equity method investment will not be reduced below zero. As a result, no loss was recognized during the years ended December 31, 2021, 2020 and 2019 on the equity method investment.
The overall arrangement with Motif is a vendor-customer relationship and is within the scope of ASC 606 as the provision of development services and corresponding license rights are considered a part of the Company’s ordinary activities. The licenses contemplated under the Motif IP Agreement are contingent upon a TDP being agreed to by the parties under the
F-45
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Motif TDA and only relate to strains that are developed under a TDP. While the TDPs require approval by the parties, the parties initially estimated that ten TDPs would be negotiated under the arrangement.
The Company’s performance obligations under the Motif IP Agreement consist of ten material rights, related to the initial set of ingredients that the parties desired to develop in the first two years. The material rights represent an advance payment for the license rights which will be granted upon the execution of each TDP. As there is no additional payment for these license rights upon execution of a TDP, the Company has determined that there is a material right associated with each of the contemplated TDPs. The common stock received under the Motif IP Agreement is considered non-cash consideration and has been recognized at fair value. The Company determined the fair value of the common stock was $65.1 million at inception of the agreement with the assistance of a third-party valuation specialist, which was initially recorded as non-current deferred revenue. The option pricing model used a back-solve methodology to determine the total equity value based on the pricing of the Series A financing, which was contemporaneous with the Motif IP Agreement. The Company has allocated $6.5 million to each of the ten material rights. The Company allocated the transaction price based on the estimated standalone selling price of the material rights which is, in turn, based on the intrinsic value of the right and the probability of exercise.
Upon the execution of each TDP, the Company is obligated to provide development services under the TDP and a license to applicable patents and other intellectual property to the ingredient developed under the plan. The license and research and development services under a TDP are highly interdependent and interrelated with one another. Without the Company’s knowledge, expertise and platform, there would not be a licensable strain or other commercializable product to transfer to Motif. Further, Motif has rights to all development intellectual property created as part of each TDP, irrespective of the result of the development. Therefore, each executed TDP consists of one combined performance obligation for the license and research and development services to be performed by the Company.
For each TDP, the transaction price consists of variable consideration for the most likely amount of estimated consideration to be received under the cost-plus arrangement and the $6.5 million which was allocated to the associated material right under the Motif IP Agreement. As the services performed by the Company create or enhance an asset (i.e., the specified ingredient) that Motif controls as the asset is created or enhanced, the Company satisfies the performance obligation and recognizes revenue over time. The Company uses an input method that compares total costs incurred relative to total estimated cost to complete to estimate progress under the contract. Any revisions to the estimated total budgeted costs to complete, and the resulting impact to revenue recognition, are reflected in the period of the change through a cumulative catch-up adjustment.
As of December 31, 2021 and 2020, the Company had a deferred revenue balance of $52.2 million and $54.0 million, respectively, with Motif. During the years ended December 31, 2021, 2020 and 2019, the Company recognized revenue of $20.2 million, $20.8 million and $19.0 million, respectively, from services provided to Motif.
Genomatica, Inc.
2016 Genomatica Agreement
In 2016, the Company purchased Series A preferred stock of Genomatica, Inc. (“Genomatica”), a biotechnology company specializing in the development and manufacturing of intermediate and specialty chemicals from both sugar and alternative feedstocks. The Company also entered into a Collaboration Agreement with Genomatica (“Genomatica Collaboration”) in connection with the financing. The Genomatica Collaboration was entered into to share expertise on biotechnology solutions. Specifically, Genomatica provided the Company with scale-up and process optimization functions, and the Company has provided Genomatica with certain technology development functions generally centered on high throughput strain engineering capabilities. The Genomatica Collaboration’s focus was on obtaining new customers for either party that could benefit from the combined expertise of both parties, and the agreement provides for profit-sharing allocations between Genomatica and the Company depending on the category of the potential product. Each party is responsible for their own costs incurred under an agreed upon TDP.
2018 Genomatica Agreement
In September 2018, the Company entered into a stock purchase agreement with Genomatica under which it received $40.0 million of Series B preferred stock from Genomatica. In lieu of cash consideration, the Company entered into a Foundry Terms of Service Agreement (“Genomatica FSA”) with Genomatica in which the Company would provide up to $40.0 million in services at no charge to Genomatica (“Initial Prepayment”). The Genomatica FSA terminated the Genomatica Collaboration and changed the pricing terms for work performed under TDPs to a cost-plus fixed margin agreement.
F-46
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
Genomatica can apply a portion of the $40.0 million in prepaid services to outstanding invoices under the Genomatica FSA, subject to certain limitations that require cash payment for services over certain monthly thresholds. Further, while the Genomatica FSA replaced the Genomatica Collaboration, any fees that would have been paid to or by the Company under contracts previously governed by the Genomatica Collaboration continued to be shared between the parties. These amounts are either (i) added to, if payable to the Company, or (ii) reduced from, if payable to Genomatica, the balance of the prepaid services over the term of the arrangement, with certain restrictions. As of December 31, 2021 and 2020, the Company has received $8.3 million and $6.9 million, respectively, under the Genomatica FSA. All contracts previously governed by the Genomatica Collaboration have ended as of December 31, 2021, therefore, no additional payments are expected.
Accounting Analysis
The Company concluded the preferred stock investment was not in-substance common stock and therefore did not qualify for accounting as an equity method investment. Rather, the Company concluded the preferred stock investment should be accounted for as an equity security as it represents an ownership interest in Genomatica that is not mandatorily redeemable nor does the Company have the unilateral right to redeem the preferred stock. Genomatica’s preferred stock is not exchange-traded and does not have a readily determinable fair value. Therefore, the Company accounts for the Genomatica preferred stock under the measurement alternative for equity investments that do not have a readily determinable fair value, which in this case is at historical cost. As of December 31, 2021 and 2020, the cost of the investment in Genomatica’s preferred stock was $55.0 million and is included in investments on the Consolidated Balance Sheets. As of December 31, 2021 and 2020, no adjustments have been recognized related to the preferred stock investment as a result of the application of the measurement alternative.
Under the Genomatica Collaboration, the Company was entitled to receive a portion of fees earned from third party customers of Genomatica that were within the scope of the agreement. The Company accounted for the collaboration under ASC 808, however the Company applied ASC 606 by analogy for measurement and recognition purposes. Under the Genomatica Collaboration, the Company’s promises consisted of (i) licenses to the Company’s intellectual property, related to the specified development work, and (ii) research and development services. The Company determined that there was a single, combined performance obligation consisting of research services and licenses to certain intellectual property. The Company recognized the revenue for the combined performance obligation using an over-time input method, as the Company’s performance under the contract created or enhanced the target product or strain as such product or strain was developed. The Company measured progress based on the cost incurred relative to total forecasted cost.
The Genomatica FSA represents a modification to the Genomatica Collaboration that resulted in a change in transaction price from milestones to a cost-plus fixed margin structure. The Genomatica FSA did not result in the addition of any distinct promised goods or services, and the Company’s remaining obligation post-modification was to finish the partially satisfied development work that had commenced under the Genomatica Collaboration. This performance obligation was satisfied during the year ended December 31, 2019 and the parties have entered into subsquent TDPs under the Genomatica FSA.
As of December 31, 2021 and 2020, the Company had a deferred revenue balance of $17.1 million and $30.1 million, respectively, with Genomatica. During the years ended December 31, 2021, 2020 and 2019, the Company recognized revenue of $12.9 million, $9.4 million and $6.2 million, respectively, from services provided to Genomatica.
Joyn Bio, LLC
Summary of Arrangement
In September 2017, the Company and certain other investors formed Cooksonia for the purposes of holding the Company’s investment in Joyn. Concurrently, Cooksonia entered into a commitment agreement with Bayer CropScience LP (“Bayer”) to form Joyn. Joyn is focused on research, development, discovery, and commercialization of engineered microbes for use in agriculture. The initial program uses advanced techniques in biology to study and engineer naturally occurring soil microbes and their nitrogen-fixing genes to enable crops to produce their own fixed nitrogen and reduce the nitrogen fertilizer required.
The Company contributed $5.0 million in cash and certain intellectual property to Cooksonia in exchange for a 70% equity interest in Cooksonia (“Class A Units”). Cooksonia received $20.0 million in cash from another investor, who is a related party of the Company, for a 20% equity interest in Cooksonia (“Class B Units”). Cooksonia also received certain intellectual property from Genomatica and issued Genomatica a 10% equity interest in Cooksonia (“Cooksonia Class C Units”) and paid Genomatica $5.0 million in cash. Subsequently, Cooksonia contributed $20.0 million and all intellectual property received from the Company and Genomatica in exchange for a 50% equity interest in Joyn. Bayer contributed $20.0 million in cash
F-47
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
funding plus specified intellectual property. In addition, Bayer committed to contribute up to an additional $60.0 million to be paid subject to certain funding procedures. In return, Bayer obtained a 50% equity interest in Joyn. The agreements may be terminated by mutual agreement, following a change in control, and for breach.
Joyn is governed by a Board of Managers (“Joyn Board”) comprised of equal representation of the Company and Bayer. The Joyn Board has all the rights, powers, obligations, and authority to manage the business and affairs of Joyn.
The Company also entered into a Foundry Services Agreement (“Joyn FSA”) with Joyn under which the Company will provide Joyn with technical services and preferred access to the Company’s facilities. Joyn paid the Company a non-refundable $20.0 million prepayment for services to be provided under the Joyn FSA (“Joyn Prepaid Services”). The Joyn Prepaid Services can be utilized for technical services performed by the Company, its subcontractors, and third parties involved in the performance of the overall technical services. Amounts due to the Company are applied to the balance of Joyn Prepaid Services as earned. During the year ended December 31, 2019, Joyn made an additional $15.0 million prepayment for services (“Joyn Additional Prepaid Services”). Under certain Joyn termination scenarios, any amount of unused Joyn Additional Prepaid Services shall be repaid by the Company to Joyn.
Accounting Analysis
From inception, the Company’s investment in Cooksonia has represented a controlling financial interest, resulting in consolidation of Cooksonia within the Company’s consolidated financial statements (see Note 11). The initial cash and in-kind contributions the Company made to Cooksonia have been recorded at carrying value as the transaction was with entities under common control. All assets of Cooksonia after the initial investments, net of the amounts paid to Genomatica, were contributed to Joyn for a 50% equity interest in Joyn. The initial carrying value of the Company’s equity interest in Cooksonia was $13.1 million, comprised of the initial $5.0 million cash investment and an $8.1 million adjustment for Cooksonia’s claim on net assets in accordance with ASC 810, Consolidation (“ASC 810”) recognized to reflect a certain investor’s liquidation preference in a termination event that represents a substantive profit-sharing agreement. The initial carrying value of the non-controlling interest was comprised of cash and intellectual property contributions from the other investors of $29.7 million, less the $8.1 million adjustment for the non-controlling interest holders’ claim on the net assets of Cooksonia.
Cooksonia accounts for its 50% equity interest in Joyn as an equity method investment based on the size of its equity interest and its influence on the board of directors. The equity method investment in Joyn was recorded at an initial carrying value of $97.9 million, which is the fair value of Cooksonia’s interest in Joyn. The fair value was determined by management with the assistance of a third-party valuation specialist. The option pricing model used a back-solve methodology to determine the total equity value based on the pricing of the Class B Units which were exchanged for cash. The license of intellectual property to Joyn has been accounted for under ASC 606 as described below. Upon liquidation, the net assets of Joyn are not distributed in accordance with each party’s respective ownership interest. Depending on the circumstances or type of liquidation event, Bayer or Cooksonia may receive certain preference payments or priority in the assets that are distributed. These preferences represent a substantive profit-sharing arrangement and, accordingly, Cooksonia recognizes earnings and losses on its equity method investment using the HLBV method. Refer to Note 11 for additional details on Cooksonia's investment in Joyn.
The Company accounts separately under ASC 606 for Cooksonia’s contribution of its intellectual property and the services performed by the Company under technical project plans governed by the Joyn FSA. The Company accounts for the intellectual property sale and the technical services separately as the two agreements were not negotiated with a single commercial objective, the consideration under each agreement is not interdependent, and the intellectual property contribution from Cooksonia is separate and distinct from the research and development services performed under the Joyn FSA.
The Company considers the granting of licenses to the Company’s intellectual property as part of its ordinary business activities and, therefore, Cooksonia’s contribution of intellectual property to Joyn represents a contract with a customer. The intellectual property contains multiple licenses for which control transfers at inception and all revenue associated with the licenses was recognized during the year ended December 31, 2017.
The Joyn FSA functions as a master services agreement that provides the framework for the ongoing research and development services relationship between the Company and Joyn. The Joyn FSA does not create a contract under ASC 606 as it does not identify goods or services to be performed nor does it define consideration under the contract. Upon the execution of a technical project plan under the Joyn FSA, the arrangement qualifies as a contract under ASC 606.
F-48
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
The Company accounts for each technical project separately. Each technical project plan provides for distinct services in the context of the contract, has been separately negotiated with Joyn, focuses on different specified strains with separate scopes of work, and has its own budget. The sole performance obligation under each individual technical project plan consists of the research and development services as the requisite licenses were transferred prior to the execution of the technical project plans. The transaction price for each technical project plan is determined at plan inception based on the consideration that the Company negotiated in exchange for the services to be provided. The Company’s performance under each technical project plan creates or enhances assets under Joyn’s control. Joyn receives the benefits of the output of the research and development services which allow Joyn to make strategic business decisions on the direction of each product candidate. Therefore, the Company satisfies the respective performance obligations and recognizes revenue over time.
As of December 31, 2021 and 2020, the Company had a deferred revenue balance of $4.6 million and $9.9 million, respectively, with Joyn, representing the remaining balance of the prepaid services. The entire deferred revenue balance remains refundable under certain termination scenarios. During the years ended December 31, 2021, 2020 and 2019, the Company recognized revenue of $5.3 million, $7.3 million and $9.3 million, respectively, from services provided to Joyn for which the balance was applied against deferred revenue.
Amyris, Inc.
During 2017, the Company terminated its collaborative relationship with Amyris, Inc. (“Amyris”) as provided in the Amyris Collaboration Agreement and executed a settlement arrangement (“Partnership Agreement”) under which the Company is entitled to receive (i) value share payments owed to the Company under the Amyris Collaboration Agreement, (ii) payments of $0.8 million each quarter commencing on December 31, 2018 through the quarter ended September 30, 2022, and (iii) payments due under an interest bearing $12.0 million promissory note.
The parties amended the agreements during the year ended December 31, 2020 to defer certain payments and provide Amyris waivers for noncompliance with certain covenants. As of December 31, 2020, the Company was owed (i) the $12.0 million principal balance on the promissory note which matures on October 19, 2022 and (ii) payments under the Partnership Agreement, as amended, which includes quarterly payments of $0.2 million to $0.3 million through September 2022 and an end of term payment of $9.8 million on October 19, 2022.
The Company concluded that all amounts due are a settlement for accounting purposes as the payments are being made without any obligation from the Company to Amyris. The balance due on the promissory note and right to payments due under the Partnership Agreement are not recognized in the Company’s financial statements until the gain is realized. The Company recognizes any payments made under the Partnership Agreement and promissory note, including interest, when the cash is received as a component of other (expense) income. On November 15, 2021, the Company received a $22.8 million payment from Amyris in full settlement of all amounts due under the Partnership Agreement including (i) the $12.0 million principal balance on the promissory note and all interest due, (ii) all quarterly payments due under the Partnership Agreement through September 2022 and (iii) an end of term payment of $9.8 million. Payments received from Amyris are recorded as gain on settlement of partnership agreement in the Consolidated Statements of Operations and Comprehensive Loss.
Synlogic, Inc.
Summary of Arrangement
In June 2019, the Company entered into several agreements with Synlogic, a publicly traded clinical-stage biopharmaceutical company focused on advancing drug discovery and development for synthetic biology-derived medicines. The Company entered into a Subscription Agreement with Synlogic whereby it purchased 6,340,771 shares of common stock at $9.00 per share for a total purchase price of $57.1 million, which represented a 19.9% equity interest in Synlogic. The Company also entered into a Warrant Agreement whereby it received the right to purchase 2,548,117 shares of common stock of Synlogic at an exercise price of $9.00 per share. The Company made a non-refundable prepayment related to the exercise price of the warrant equal to $8.99 per share for a total payment of $22.9 million. The warrant is only exercisable to the extent the Company’s interest in Synlogic does not exceed 19.99%. The Company also entered into a Foundry Services Agreement (“Synlogic FSA”) whereby Synlogic provided $30.0 million in cash as a non-refundable prepayment for Foundry services. The prepaid Foundry services can be utilized for development of collaboration strains. Services performed under the services agreement will be applied to the prepaid amount based on the contractual rates included in the contract, based on costs incurred plus a fixed margin. Work will be performed under the Synlogic FSA pursuant to TDPs. Each TDP will pursue the development of a specific collaboration strain and/or production protocol. The Synlogic FSA will terminate upon the earlier
F-49
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
of the exhaustion of the prepayment amount in full or the fifth anniversary of the effective date of the agreement and may be extended in certain circumstances.
Accounting Analysis
The overall arrangement with Synlogic includes the Subscription Agreement whereby the Company purchased shares of Synlogic common stock, the Warrant Agreement whereby the Company prepaid a significant portion of the exercise price of the warrant to purchase Synlogic common stock, which is non-refundable, and the Synlogic FSA whereby the Company will perform services for Synlogic. The Company concluded that these agreements should be considered one arrangement for accounting purposes as they were entered into at the same time and negotiated as a package with a single commercial objective.
At inception, the common stock investment in Synlogic was considered an equity method investment as the Company did not have a controlling financial interest in Synlogic but did have the ability to influence the financial and operating policies through its ownership of common stock. The Company elected to apply the fair value option to account for the equity method investment as the fair value of Synlogic’s common stock is objectively determinable based on quoted market prices in an active market for the identical securities. At inception, the fair value of the equity method investment in Synlogic was recorded at $35.8 million as a component of equity method investments on the Consolidated Balance Sheet. Beginning with the third quarter of 2021, due to a decrease in the level of ownership, the investment no longer qualifies for the equity method and was reclassified from equity method investments to investments on the Condensed Consolidated Balance Sheets, and from loss on equity method investments to (loss) gain on investments on the Condensed Consolidated Statements of Operations and Comprehensive Loss for all periods presented. However, the Company continues to apply the fair value option to account for its investments in Synlogic. The Company has also elected to apply the fair value option to account for the warrant to purchase Synlogic common stock, which at inception was recorded at $14.4 million as a component of investments on the Consolidated Balance Sheet. See Note 5 for additional information related to the fair value measurements of Synlogic common stock and the Synlogic warrants and Note 10 for additional information related to the net gains and losses recognized during the periods presented related to these securities.
The Company concluded that the TDPs represent contracts with a customer and will be accounted for under ASC 606. At inception, Synlogic prepaid $30.0 million for services under the Synlogic FSA. The prepaid services were reduced by $29.8 million, which represents the excess of the aggregate $80.0 million the Company paid to purchase Synlogic’s common stock and warrant over the respective fair values of those instruments. This resulted in a deferred revenue balance of $0.2 million at inception, which is being recognized over the period in which the Company will provide services to Synlogic. The Company recognized nominal amounts of revenue during each of the years ended December 31, 2021, 2020 and 2019 from services provided to Synlogic. As of December 31, 2021 and 2020, the Company had a deferred revenue balance of less than $0.1 million with Synlogic.
21. Employee Benefit Plan
The Company has a 401(k) retirement plan covering substantially all employees. Under the retirement plan, employees make voluntary contributions and the Company makes a 5% non-elective contribution for all employees based on compensation, subject to IRS contribution limits. For the years ended December 31, 2021, 2020 and 2019, the Company contributed $3.7 million, $2.2 million and $1.6 million, respectively, to the retirement plan.
22. Income Taxes
For the years ended December 31, 2021, 2020 and 2019, the loss before income taxes consisted of the following (in thousands):
|
|
Year Ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Domestic |
|
$ |
(1,837,497 |
) |
|
$ |
(124,834 |
) |
|
$ |
(119,835 |
) |
Foreign |
|
|
(625 |
) |
|
|
— |
|
|
|
— |
|
Total |
|
$ |
(1,838,122 |
) |
|
$ |
(124,834 |
) |
|
$ |
(119,835 |
) |
F-50
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
For the years ended December 31, 2021, 2020 and 2019, the Company incurred the following income tax (benefit) expense (in thousands):
|
|
Year Ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Current state income tax |
|
$ |
1 |
|
|
$ |
26 |
|
|
$ |
22 |
|
Deferred federal income tax |
|
|
(413 |
) |
|
|
581 |
|
|
|
— |
|
Deferred state income tax |
|
|
(912 |
) |
|
|
1,282 |
|
|
|
— |
|
Deferred foreign income tax |
|
|
(156 |
) |
|
|
— |
|
|
|
— |
|
Income tax (benefit) expense |
|
$ |
(1,480 |
) |
|
$ |
1,889 |
|
|
$ |
22 |
|
A reconciliation of income tax (benefit) expense computed at the statutory corporate income tax rate to the effective income tax rate for the years ended December 31, 2021, 2020 and 2019 is as follows:
|
|
Year Ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Federal income tax at statutory rate |
|
|
21.0 |
% |
|
|
21.0 |
% |
|
|
21.0 |
% |
State income tax |
|
|
4.5 |
% |
|
|
4.5 |
% |
|
|
4.2 |
% |
Change in valuation allowance |
|
|
(23.9 |
)% |
|
|
(31.3 |
)% |
|
|
(25.2 |
)% |
Executive compensation |
|
|
(2.0 |
)% |
|
|
— |
|
|
|
— |
|
Equity investments |
|
|
(0.7 |
)% |
|
|
(0.6 |
)% |
|
|
(5.7 |
)% |
Tax credits |
|
|
0.9 |
% |
|
|
4.8 |
% |
|
|
4.4 |
% |
Non-deductible expenses and change in fair value of warrant liability |
|
|
0.9 |
% |
|
|
(0.2 |
)% |
|
|
(0.1 |
)% |
Other expenses |
|
|
(0.6 |
)% |
|
|
0.3 |
% |
|
|
1.4 |
% |
Effective tax rate |
|
|
0.1 |
% |
|
|
(1.5 |
)% |
|
|
— |
|
The Company’s deferred tax assets and liabilities consist of the following (in thousands):
|
|
Year Ended December 31, |
|
|||||
|
|
2021 |
|
|
2020 |
|
||
Deferred tax assets: |
|
|
|
|
|
|
||
Net operating loss carryforwards |
|
$ |
174,127 |
|
|
$ |
91,467 |
|
Tax credit carryforwards |
|
|
37,455 |
|
|
|
20,338 |
|
Accrued expenses |
|
|
2,690 |
|
|
|
1,265 |
|
Deferred revenue |
|
|
45,928 |
|
|
|
28,590 |
|
Stock-based compensation |
|
|
318,049 |
|
|
|
— |
|
Amortizable intangibles |
|
|
3,834 |
|
|
|
4,198 |
|
Tenant allowance |
|
|
2,927 |
|
|
|
2,206 |
|
Deferred tax assets before valuation allowance |
|
|
585,010 |
|
|
|
148,064 |
|
Valuation allowance |
|
|
(583,107 |
) |
|
|
(143,827 |
) |
Deferred tax assets, net of valuation allowance |
|
|
1,903 |
|
|
|
4,237 |
|
Deferred tax liabilities: |
|
|
|
|
|
|
||
Amortizable intangibles |
|
|
(4,722 |
) |
|
|
— |
|
Property and equipment |
|
|
(830 |
) |
|
|
(830 |
) |
Basis differences |
|
|
(1,522 |
) |
|
|
(5,270 |
) |
Deferred tax liabilities |
|
|
(7,074 |
) |
|
|
(6,100 |
) |
Net deferred taxes |
|
$ |
(5,171 |
) |
|
$ |
(1,863 |
) |
Activity in the deferred tax assets valuation allowance is summarized as follows (in thousands):
|
|
Beginning of |
|
|
Additions |
|
|
End of |
|
|||
Deferred tax assets valuation allowance: |
|
|
|
|
|
|
|
|
|
|||
Year ended December 31, 2021 |
|
$ |
143,827 |
|
|
$ |
439,280 |
|
|
$ |
583,107 |
|
Year ended December 31, 2020 |
|
$ |
104,745 |
|
|
$ |
39,082 |
|
|
$ |
143,827 |
|
F-51
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
The Company has evaluated the positive and negative evidence bearing upon its ability to realize the deferred tax assets. The Company considered its history of cumulative net losses incurred since inception and has concluded that it is more likely than not that it will not realize the benefits of the deferred tax assets. Accordingly, a valuation allowance has been established against the deferred tax assets as of December 31, 2021 and 2020 that are not expected to be realized. The Company reevaluates the positive and negative evidence at each reporting period. The valuation allowance increased on a net basis by approximately $439.3 million during the year ended December 31, 2021 primarily due to an increase in the deferred tax asset related to stock-based compensation and the increase in the net operating losses and tax credits carryforwards.
As of December 31, 2021, the Company had federal net operating loss carryforwards of approximately $665.2 million, of which $139.2 million begin to expire in 2029. The Company has approximately $526.0 million of federal net operating losses as of December 31, 2021 that can be carried forward indefinitely. As of December 31, 2021, the Company had state net operating loss carryforwards of approximately $529.3 million, of which $485.9 million begin to expire in 2029. The Company has approximately $43.4 million of state net operating losses as of December 31, 2021 that can be carried forward indefinitely.
As of December 31, 2021, the Company had federal research and development tax credit carryforwards of approximately $23.3 million which begin to expire in 2029. As of December 31, 2021, the Company also had state research and development and investment tax credit carryforwards of approximately $18.0 million which begin to expire in 2030.
Under Sections 382 and 383 of the U.S. Internal Revenue Code, if a corporation undergoes an ownership change, the corporation’s ability to use its pre-change net operating loss carryforwards and other pre-change tax attributes, such as research tax credits, to offset its post-change income and taxes may be limited. In general, an ownership change generally occurs if there is a cumulative change in its ownership by 5% stockholders that exceeds 50 percentage points over a rolling three-year period. Similar rules may apply under U.S. state tax laws. The Company may have experienced an ownership change in the past and may experience ownership changes in the future as a result of future transactions in its share capital, some of which may be outside of the Company’s control. As a result, if the Company earns net taxable income, the Company's ability to use its pre-change net operating loss carryforwards, or other pre-change tax attributes, to offset U.S. federal and state taxable income and taxes may be subject to significant limitations.
We assess the impact of various tax reform proposals and modifications to existing tax treaties in all jurisdictions where we have operations to determine the potential effect on our business and any assumptions we have made about our future taxable income. We cannot predict whether any specific proposals will be enacted, the terms of any such proposals or what effect, if any, such proposals would have on our business if they were to be enacted. Beginning in 2022, the Tax Cuts and Jobs Act of 2017 eliminates the currently available option to deduct research and development expenditures and requires taxpayers to amortize them over five years. The U.S. Congress is considering legislation that would defer the amortization requirement to future periods, however, we have no assurance that the provision will be repealed or otherwise modified.
The Company files tax returns as prescribed by the tax laws of the jurisdictions in which the Company operates. In the normal course of business, the Company is subject to examination by federal and state jurisdictions, where applicable. There are currently no pending tax examinations. As of December 31, 2021, the Company’s tax years are still open under statute from 2018 to the present in the United States and 2016 to the present in the Netherlands. To the extent the Company has tax attribute carryforwards, the tax years in which the attribute was generated may still be adjusted upon examination by the Internal Revenue Service or state taxing authorities to the extent utilized in a future period.
The Company accounts for uncertain tax positions using a more likely than not threshold for recognizing and resolving uncertain tax positions. The evaluation of uncertain tax positions is based on factors that include, but are not limited to, changes in tax law, the measurement of tax positions taken or expected to be taken in tax returns, the effective settlement of matters subject to audit, new audit activity and changes in facts or circumstances related to a tax position. The Company evaluates uncertain tax positions on an annual basis and adjusts the level of the liability to reflect any subsequent changes in the relevant facts surrounding the uncertain positions. The Company accounts for interest and penalties related to uncertain tax positions as part of its provision for income taxes. As of December 31, 2021 and 2020, the Company had no recorded liabilities for uncertain tax positions and had no accrued interest or penalties related to uncertain tax positions. The Company does not expect a material change in unrecognized tax benefits in the next twelve months.
F-52
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
23. Net Loss per Share
As a result of the Business Combination, the Company has retroactively restated the weighted average shares outstanding prior to September 16, 2021 to give effect to the Exchange Ratio.
The Company computes net loss per share of the Class A common stock and Class B common stock using the two-class method required for participating securities. The earnings per share amounts are the same for the different classes of common stock because the holders of each class are legally entitled to equal per share distributions whether through dividends or liquidation. The calculation of basic and diluted earnings per common share are as follows (in thousands, except per share amounts):
|
|
Year ended December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Numerator: |
|
|
|
|
|
|
|
|
|
|||
Net loss attributable to Ginkgo Bioworks Holdings, Inc. stockholders, basic |
|
$ |
(1,830,047 |
) |
|
$ |
(126,609 |
) |
|
$ |
(119,327 |
) |
Change in fair value of warrant liabilities |
|
$ |
58,615 |
|
|
|
— |
|
|
|
— |
|
Net loss attributable to Ginkgo Bioworks Holdings, Inc. stockholders, diluted |
|
$ |
(1,888,662 |
) |
|
$ |
(126,609 |
) |
|
$ |
(119,327 |
) |
Denominator |
|
|
|
|
|
|
|
|
|
|||
Weighted average common shares outstanding, basic |
|
|
1,359,848,803 |
|
|
|
1,274,766,915 |
|
|
|
1,149,000,417 |
|
Weighted average effect of dilutive outstanding warrants |
|
|
524,540 |
|
|
|
— |
|
|
|
— |
|
Weighted average common shares outstanding, diluted |
|
|
1,360,373,343 |
|
|
|
1,274,766,915 |
|
|
|
1,149,000,417 |
|
Basic net loss per share |
|
$ |
(1.35 |
) |
|
$ |
(0.10 |
) |
|
$ |
(0.10 |
) |
Diluted net loss per share |
|
$ |
(1.39 |
) |
|
$ |
(0.10 |
) |
|
$ |
(0.10 |
) |
The following potential common shares, presented based on amounts outstanding at each period end, were excluded from the calculation of diluted net loss per share attributable to Ginkgo Bioworks Holdings, Inc. common stockholders for the periods presented because including them would have been anti-dilutive:
|
|
As of December 31, |
|
|||||||||
|
|
2021 |
|
|
2020 |
|
|
2019 |
|
|||
Warrants to purchase Class A common stock |
|
|
— |
|
|
|
1,020,187 |
|
|
|
1,020,187 |
|
Outstanding stock options |
|
|
25,228,853 |
|
|
|
33,354,871 |
|
|
|
35,276,812 |
|
Unvested RSUs |
|
|
168,321,952 |
|
|
|
124,932,207 |
|
|
|
70,119,944 |
|
Unvested RSAs |
|
|
182,622 |
|
|
|
419,049 |
|
|
|
675,887 |
|
New Ginkgo and Sponsor earnout shares (1) |
|
|
160,995,237 |
|
|
|
— |
|
|
|
— |
|
|
|
|
354,728,664 |
|
|
|
159,726,314 |
|
|
|
107,092,830 |
|
(1) Represents earnout shares for which the vesting conditions have not been satisfied.
F-53
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
24. Related Parties
Related party transactions included in the Consolidated Balance Sheets, excluding the Company’s investments and equity method investments, are summarized below (in thousands):
|
|
As of December 31, |
|
|||||||
|
|
2021 |
|
|
|
2020 |
|
|||
Accounts receivable: |
|
|
|
|
|
|
|
|||
Joyn |
|
$ |
5 |
|
|
|
|
$ |
— |
|
Motif |
|
|
3,020 |
|
|
|
|
|
2,403 |
|
Genomatica |
|
|
— |
|
|
|
|
|
1,500 |
|
Allonnia |
|
|
849 |
|
|
|
|
|
1,309 |
|
Arcaea |
|
|
724 |
|
|
|
|
— |
|
|
|
|
$ |
4,598 |
|
|
|
$ |
5,212 |
|
|
Deferred revenue, current and non-current: |
|
|
|
|
|
|
|
|||
Joyn |
|
$ |
4,608 |
|
|
|
|
$ |
9,862 |
|
Motif |
|
|
52,171 |
|
|
|
|
|
53,952 |
|
Genomatica |
|
|
17,111 |
|
|
|
|
|
30,128 |
|
Allonnia |
|
|
38,016 |
|
|
|
|
|
26,064 |
|
Arcaea |
|
|
47,356 |
|
|
|
|
|
— |
|
Other equity investees |
|
|
1,559 |
|
|
|
|
72 |
|
|
|
|
$ |
160,821 |
|
|
|
|
$ |
120,078 |
|
Related party transactions included in the Consolidated Statements Operations and Comprehensive Loss, excluding the losses on the Company’s investments and equity method investments, are summarized below (in thousands):
|
|
Year Ended December 31, |
|
|||||||||||||
|
|
2021 |
|
|
|
2020 |
|
|
|
2019 |
|
|||||
Foundry revenue: |
|
|
|
|
|
|
|
|
|
|
|
|||||
Joyn |
|
$ |
5,254 |
|
|
|
|
$ |
7,273 |
|
|
|
|
$ |
9,349 |
|
Motif |
|
|
20,224 |
|
|
|
|
|
20,798 |
|
|
|
|
|
18,986 |
|
Genomatica |
|
|
12,868 |
|
|
|
|
|
9,431 |
|
|
|
|
|
6,248 |
|
Allonnia |
|
|
5,126 |
|
|
|
|
|
4,960 |
|
|
|
|
|
|
|
Arcaea |
|
|
3,676 |
|
|
|
|
|
|
|
|
|
|
|
||
Other equity investees |
|
|
13 |
|
|
|
|
73 |
|
|
|
|
685 |
|
||
|
|
$ |
47,161 |
|
|
|
|
$ |
42,535 |
|
|
|
|
$ |
35,268 |
|
Refer to Notes 10 and 20 for additional details on the Company’s investments and equity method investments held in its related parties.
25. Subsequent Events
The Company announced subsequent to year end a change in its reporting structure to formalize Concentric as a separate business unit. The purpose of the reorganization is to strengthen the Company's focus on the Biosecurity market opportunity which the Company believes is broader than its current offering of the Concentric testing program. The Biosecurity business unit will initially consist of the Concentric platform along with additional biosecurity activities as they are developed potentially including wastewater monitoring, supporting rapid vaccine readiness and future biology-based countermeasures for infectious disease. Beginning in the first quarter of 2022, the Company will report its segment results under two reportable segments: Cell Programming / Foundry and Biosecurity.
On March 11, 2022, the Company entered into a definitive agreement to acquire FGen AG (“FGen”), a Swiss company specializing in strain development and optimization. FGen has developed an ultra-high-throughput screening platform built on nanoliter reactor technology which the Company believes will enhance its cell screening capabilities and potentially increase the likelihood of finding enzymes, pathways, and strains or cell lines that perform to diverse product specifications. Under the terms of the agreement, the Company will acquire 100% of the equity of FGen for total consideration of $17.5 million, subject to certain adjustments, payable in New Ginkgo class A common stock, and additional contingent
F-54
Table of Contents
Ginkgo Bioworks Holdings, Inc. and Subsidiaries
Notes to Consolidated Financial Statements
consideration up to $25.0 million related to, among other things, the successful integration and deployment of the FGen technology across the Company's programs. The transaction is expected to close in April 2022.
F-55
Similar companies
See also AMGEN INC - Annual report 2024 (10-K 2024-12-31) Annual report 2025 (10-Q 2025-03-31)See also GILEAD SCIENCES, INC. - Annual report 2022 (10-K 2022-12-31) Annual report 2023 (10-Q 2023-09-30)
See also Moderna, Inc. - Annual report 2022 (10-K 2022-12-31) Annual report 2024 (10-Q 2024-06-30)
See also BioNTech SE
See also BIOGEN INC. - Annual report 2023 (10-K 2023-12-31) Annual report 2025 (10-Q 2025-03-31)
