MetaVia Inc. - Annual Report: 2020 (Form 10-K)
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, DC 20549
Form 10-K
(Mark One)
☒ | ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 |
| For the fiscal year ended December 31, 2020 |
OR
☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF SECURITIES EXCHANGE ACT OF 1934 |
| For the transition period from to |
Commission file number 001-37809
NeuroBo Pharmaceuticals, Inc.
(Exact name of Registrant as specified in its charter)
Delaware |
| 47-2389984 |
(State or other jurisdiction of incorporation or organization) |
| (IRS Employer Identification No.) |
|
|
|
200 Berkeley Street, Office 19th Floor Boston, Massachusetts |
| 02116 |
(Address of principal executive offices) |
| (Zip Code) |
(857) 702-9600
(Registrant’s telephone number, including area code)
Securities registered pursuant to Section 12(b) of the Securities Exchange Act of 1934:
| ||||
Title of Each Class |
| Trading symbol(s) |
| Name of Exchange on Which Registered |
Common stock, $0.001 par value | | NRBO |
| The Nasdaq Stock Market LLC |
Securities registered pursuant to Section 12(g) of the Act: None
Indicate by check mark whether the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act. Yes ☐ No ☒
Indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or Section 15(d) of the Act. Yes ☐ No ☒
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes ☒ No ☐
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes ☒ No ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer ☐ | Accelerated filer ☐ |
Non-accelerated filer ☒ | Smaller reporting company ☒ |
|
|
Emerging growth company ☒ |
|
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☒
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.
Yes ☐ No ☒
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12(b)-2 of the Securities Exchange Act of 1934). Yes ☐ No ☒
The aggregate market value of the registrant’s common stock held by non-affiliates of the registrant was approximately $23.4 million based on the closing price on the Nasdaq Capital Market as of June 28, 2020, the last business day of the registrant’s most recently completed second fiscal quarter.
The number of outstanding shares of the registrant’s common stock, $0.001 par value, as of April 9, 2021 was 22,171,182.
NEUROBO PHARMACEUTICALS, INC.
FORM 10-K
| | |
| | |
5 | ||
49 | ||
86 | ||
86 | ||
86 | ||
86 | ||
| | |
| | |
| | |
86 | ||
87 | ||
Management’s Discussion and Analysis of Financial Condition and Results of Operations | 87 | |
100 | ||
101 | ||
Changes In and Disagreements With Accountants on Accounting and Financial Disclosure | 131 | |
131 | ||
132 | ||
| | |
| | |
| | |
132 | ||
135 | ||
Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters | 140 | |
Certain Relationships and Related Transactions and Director Independence | 142 | |
145 | ||
| | |
| | |
| | |
146 | ||
| | |
151 | ||
2
SPECIAL NOTE REGARDING FORWARD-LOOKING STATEMENTS
This Annual Report on Form 10-K for the fiscal year ended December 31, 2020 contains “forward-looking statements” within the meaning of the Securities Act of 1933, as amended (the “Securities Act”), and the Securities Exchange Act of 1934, as amended (the “Exchange Act”). Forward-looking statements are neither historical facts nor assurances of future performance. Instead, these forward-looking statements contain information about our expectations, beliefs or intentions regarding our product development and commercialization efforts, business, financial condition, results of operations, strategies or prospects, and other similar matters. These forward-looking statements are based on management’s current expectations and assumptions about future events, which are inherently subject to uncertainties, risks and changes in circumstances that are difficult to predict. These statements may be identified by words such as “expects,” “plans,” “projects,” “will,” “may,” “anticipates,” “believes,” “should,” “intends,” “estimates,” and other words of similar meaning.
Actual results could differ materially from those contained in forward-looking statements. Many factors could cause actual results to differ materially from those in forward-looking statements, including those matters discussed below, as well as those listed in Item 1A. Risk Factors.
Other unknown or unpredictable factors that could also adversely affect our business, financial condition and results of operations may arise from time to time. Given these risks and uncertainties, the forward-looking statements discussed in this report may not prove to be accurate. Accordingly, you should not place undue reliance on these forward-looking statements, which only reflect the views of NeuroBo Pharmaceuticals, Inc.’s management as of the date of this report. We undertake no obligation to update or revise forward-looking statements to reflect changed assumptions, the occurrence of unanticipated events or changes to future operating results or expectations, except as required by law.
3
SUMMARY RISK FACTORS
Our business is subject to a number of risks, as fully described in “Item 1A. Risk Factors” in this Annual Report. The principal factors and uncertainties include, among others:
| ● | NeuroBo’s pursuit of potential therapeutic and prophylactic treatments for COVID-19 is at an early stage and subject to many risks. NeuroBo may be unable to receive approval for any of its COVID-19 product candidates a timely manner, if at all, and its COVID-19 product candidate may never be approved. |
| ● | The regulatory pathway for ANA001 is continually evolving, and may result in unexpected or unforeseen challenges. |
| ● | NeuroBo has determined to postpone the initiation of Phase 3 clinical trials of NB-01 under present circumstances and has terminated all of its agreements with contract research organizations related to NB-01. NeuroBo may not be able to successfully develop NB-01 pursuant to other alternatives, including as an orphan drug or as a nutraceutical candidate. |
| ● | NeuroBo has only incurred losses since inception. NeuroBo expects to incur losses for the foreseeable future and may never achieve or maintain profitability. |
| ● | The regulatory review and approval processes of the FDA and comparable foreign regulatory authorities are lengthy, time-consuming and inherently unpredictable, and if NeuroBo is ultimately unable to obtain regulatory approval for its product candidates, including pursuant to the guidelines applicable to NB-01 and NB-02 as botanical drug products, and the guidelines applicable to Gemcabene, its business will be substantially harmed. |
| ● | NeuroBo’s profits from Gemcabene sales will be limited pursuant to our contingent value rights obligations, and NeuroBo, therefore, may, at any time and in its sole and absolute discretion, discontinue any and all further efforts to develop, divest or otherwise monetize Gemcabene, particularly as a treatment for cardiovascular conditions. |
| ● | NeuroBo faces substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than it does. |
| ● | NeuroBo’s commercial success depends upon attaining significant market acceptance of its product candidates, if approved, among hospitals, physicians, patients and healthcare payors. |
| ● | Even if NeuroBo is able to commercialize a future pharmaceutical drug candidate, the profitability of such product candidate will likely depend in significant part on third-party reimbursement practices, which, if unfavorable, would harm its business. |
| ● | Product liability lawsuits against NeuroBo could cause it to incur substantial liabilities and could limit commercialization of any product candidate that it may develop. |
| ● | If NeuroBo is unable to establish sales and marketing capabilities or to selectively enter into agreements with third parties to sell and market its product candidates, it may not be successful in commercializing its product candidates if and when they are approved. |
| ● | NeuroBo has relied and will rely on third-party clinical research organizations (CROs) to conduct its preclinical studies and clinical trials. If these CROs do not successfully carry out their contractual duties or meet expected deadlines, NeuroBo may not be able to obtain regulatory approval for or commercialize its product candidates and its business could be substantially harmed. |
| ● | NeuroBo relies on third parties to manufacture its product candidates and preclinical and clinical drug supplies. |
| ● | If NeuroBo is unable to obtain and maintain sufficient intellectual property rights, its competitive position could be harmed. |
| ● | NeuroBo may not be able to protect or practice its intellectual property rights throughout the world. |
| ● | NeuroBo may become involved in lawsuits to protect or enforce its intellectual property, which could be expensive, time consuming and unsuccessful. |
4
| ● | NeuroBo may be subject to damages resulting from claims that its employees or NeuroBo has wrongfully used or disclosed alleged trade secrets of their former employers. |
| ● | NeuroBo currently has a limited number of employees and our future success depends on its ability to retain our executive officers and to attract, retain and motivate qualified personnel. |
| ● | NeuroBo’s trade secrets are difficult to protect and if NeuroBo is unable to protect the confidentiality of its trade secrets, its business and competitive position would be harmed. |
| ● | NeuroBo relies significantly on information technology and any failure, inadequacy, interruption or security lapse of that technology or loss of data, including any cyber security incidents, could compromise sensitive information related to its business, prevent NeuroBo from accessing critical information or expose NeuroBo to liability which could harm its ability to operate its business effectively and adversely affect its business and reputation. |
| ● | NeuroBo is an "emerging growth company" and a "smaller reporting company" and cannot be certain whether the reduced reporting requirements applicable to such companies could make its common stock less attractive to investors. |
| ● | NeuroBo’s largest shareholder may use its significant interest to take actions not supported by NeuroBo’s other shareholders, including to initiate or support shareholder activism, an unsolicited takeover proposal, or a proxy contest, which could negatively impact NeuroBo’s business. |
Overview
NeuroBo Pharmaceuticals Inc. (the “Company,” “NeuroBo,” “we,” “us” or “our”) is a clinical-stage biotechnology company with four therapeutics programs designed to impact a range of indications in coronavirus, neurodegenerative and cardiometabolic disease:
| ● | ANA001, our lead drug candidate, is a proprietary oral niclosamide formulation and was developed as a treatment for patients with moderate coronavirus disease (COVID-19). Niclosamide is a potential oral antiviral and anti-inflammatory agent with a long history of use and well-understood safety in humans. ANA001 is currently being studied in a 60-subject Phase 2 clinical trial conducted in the United States. We plan to initiate a Phase 3 development program for ANA001 in the third quarter of 2021. |
| ● | NB-01 is primarily focused on the development of a treatment for painful diabetic neuropathy (PDN). We believe NB-01 could also treat a range of neuropathic conditions, including chemotherapy-induced peripheral neuropathy and post-traumatic peripheral neuropathy. |
| ● | NB-02 has the potential to treat the symptoms of cognitive impairment and modify the progression of neurodegenerative diseases associated with the malfunction of a protein called tau, and with amyloid beta plaque deposition. |
| ● | Gemcabene is currently being assessed as an acute indication for COVID-19. Gemcabene was previously being developed for the treatment of dyslipidemia, a serious medical condition that increases the risk of life-threatening cardiovascular disease, and was focused on orphan indications such as homozygous familial hypercholesterolemia (HoFH), as well as severe hypertriglyceridemia (SHTG). |
We were initially established to advance NB-01 and NB-02, which were originally developed by the South Korean pharmaceutical company Dong-A ST Co., Ltd. (“Dong-A ST”). NB-01 has been in-licensed by us from Dong-A ST for
5
exclusive worldwide rights, except for South Korea. We acquired NB-02 from Dong-A ST, and we hold the full worldwide commercial rights for NB-02.
December 2020 Acquisition of ANA Therapeutics, Inc.
On December 31, 2020, we acquired ANA Therapeutics, Inc. (“ANA”), a privately held biotechnology company developing ANA001. The transaction was unanimously approved by each of the board of directors of the Company and ANA.
December 2019 Completion of Reverse Acquisition of Gemphire
On December 30, 2019, the Company completed a business combination (the “2019 Merger”) with Gemphire Therapeutics, Inc. (“Gemphire”) in accordance with the terms of the Agreement and Plan of Merger and Reorganization, dated as of July 24, 2019, as amended on October 29, 2019 (the “2019 Merger Agreement”). Gemphire was a clinical-stage biopharmaceutical company focused on developing and commercializing Gemcabene.
Prior to completion of the 2019 Merger, Gemphire entered into a Contingent Value Rights Agreement (the “Original CVR Agreement”) with Grand Rapids Holders’ Representative, LLC, as representative of Gemphire’s stockholders prior to the 2019 Merger, (the “Holders’ Representative”), and Computershare Inc. and Computershare Trust Company, N.A. as the rights agents (collectively, the “Rights Agent”). Under the Original CVR Agreement, which the Company assumed in connection with the 2019 Merger, the holders of Gemphire shares at the time of the 2019 Merger (the “CVR Holders”) were entitled to receive 80% of the proceeds from the grant, sale, or transfer of rights to Gemcabene.
Amendment of Original CVR Agreement
On March 23, 2021, the Company entered into a First Amendment to Contingent Value Rights Agreement with the Holders’ Representative and the Rights Agent to amend the Original CVR Agreement (as amended, the “Current CVR Agreement”). Pursuant to the Current CVR Amendment, (i) the CVR Holders will continue to receive 80% of the proceeds from the grant, sale, or transfer of rights to Gemcabene as a treatment for cardiovascular conditions and (ii) the CVR Holders will now also receive 10% of the proceeds from the grant, sale, or transfer of rights to Gemcabene as a treatment for any indication outside of treating cardiometabolic diseases, including COVID-19.
Strategy
Our goal is to discover, develop and commercialize novel therapeutics designed to impact a range of indications in neurodegenerative and cardiometabolic disease and nutraceuticals for their respective health areas. The key elements of our business strategy to achieve this goal include:
| ● | | Pursue ANA001 as a treatment for COVID-19. |
| ● | | Explore alternatives for NB-01, including assessing whether to pursue NB-01 as an orphan drug and/or as a nutraceutical product. |
| ● | | Conduct feasibility studies to identify a specified rare disease relevant to NB-01. |
| ● | | Explore alternatives for NB-02, including assessing whether to pursue NB-02 as an orphan drug. |
| ● | | Extend the pipeline of drug indications by leveraging the potential of NB-01 and NB-02 in neurodegenerative diseases such as neuropathic pain in rare diseases and tauopathies. As we continue to build and develop our product portfolio, we may opportunistically pursue strategic partnerships that maximize the value of our pipeline. |
| ● | | Explore additional therapeutic indications for Gemcabene that may strengthen our pipeline of assets to treat viral diseases, including COVID-19. |
| ● | | Continue to hire highly qualified management and personnel in advancing drug development, achieving marketing approval, and implementing our corporate growth strategy. |
6
Product Candidates
ANA001: Treatment of COVID-19 Symptoms
ANA001, NeuroBo’s lead drug candidate, is a proprietary oral niclosamide formulation and was developed as a treatment for patients with moderate COVID-19 (patients not requiring ventilators). Niclosamide is a potential oral antiviral and anti-inflammatory agent with a long history of use and well-understood safety in humans. ANA001 is currently being studied in a 60-subject Phase 2/3 clinical trial conducted at up to 20 clinical sites in the United States. . Niclosamide has demonstrated both antiviral and immunomodulatory activity with possible downstream effects on coagulation abnormalities observed in COVID-19. In preclinical research by an independent academic group published in Antimicrobial Agents and Chemotherapy, niclosamide inhibited viral replication in vitro and was more potent than remdesivir in the same assay.
Specifically, studies have shown that niclosamide prevents replication of SARS-CoV-2 at very low concentrations and that the compound appears to exhibit three distinct mechanisms of action: 1) acting as a potent antiviral to a broad homology of other viruses including influenza; 2) reducing inflammation without suppressing the immune system; and 3) providing bronchodilation, which is a useful pulmonary mechanism for at-risk patients with underlying cardiovascular and/or pulmonary conditions.
As a result, the Company believes ANA001 has the potential to reduce the viral load and inflammation associated with cytokine dysregulation, acute respiratory distress syndrome (ARDS), and coagulation abnormalities and thus improve time to clinical improvement as defined as hospital discharge recorded using the World Health Organization (“WHO”) Ordinal Scale for Clinical Improvement.
Background
ANA001 is a proprietary oral niclosamide formulation and was developed as a treatment for patients with moderate COVID-19. Niclosamide is an oral antiviral and anti-inflammatory agent with a long history of safety in humans. The active pharmaceutical ingredient (API) of ANA001 is niclosamide (Figure 1), a chlorinated salicylanilide with anthelmintic, antiviral, anti-inflammatory and bronchodilator activity. Niclosamide was discovered in 1958 and was approved by the U.S. Food and Drug Administration (the “FDA”) in 1982 under New Drug Application (NDA) 018669 (Bayer Pharmaceuticals) for the treatment of tapeworm infections, although it has since been voluntarily discontinued from marketing in the U.S., and all patents or applications that were originally filed by Bayer AG to cover niclosamide or its use in treating tapeworm have been expired or abandoned. Niclosamide is approved in several other countries for the treatment of tapeworm infections and is on the WHO’s List of Essential Medicines (World Health Organization, 2019) and has been used to safely treat thousands of patients. In the past several years, mounting evidence has accumulated that niclosamide is a multifunctional drug that is able to regulate multiple signaling pathways and biological processes, suggesting that it may be developed as a novel treatment for more than just helminthic infection. Niclosamide is being studied for a variety of clinical indications beyond its use as an anthelmintic, such as cancer, rheumatoid arthritis, diabetic neuropathy, metabolic syndrome and COVID-19.
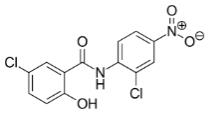
Fig. 1: Chemical structure of niclosamide.
Necessity to Develop a COVID-19 Therapeutic
Coronaviruses (CoVs) are single‐stranded RNA viruses that infect a wide variety of animals and primarily cause respiratory tract infections in humans. Recent outbreaks of novel CoVs, including SARS-CoV and MERS-CoV, have caused significant international concern and mortality. Surpassing both of these in severity and loss of life, the current outbreak of SARS-CoV-2 represents a severe public health emergency. COVID-19, the disease caused by a SARS-CoV-
7
2, continues to spread worldwide. As of March 20, 2021, more than 123 million confirmed cases of COVID-19 have been reported worldwide, with more than 2.7 million deaths from the disease. In the United States alone, the total number of COVID-19 cases as of March 20, 2021 is more than 30 million and the total number of deaths is more than 550,000. In addition to this tremendous toll, the Centers for Disease Control and Prevention (CDC) has reported that patients who have recovered from COVID-19 often exhibit long-term negative impacts such as fatigue, shortness of breath, cough, joint pain and chest pain. These symptoms require extended hospitalizations, resulting in health care costs in excess of $10,000/day. Therefore, it is necessary to develop countermeasures, such as therapeutics, that can improve the outcomes for patients who become infected with COVID-19.
Despite widespread efforts to slow the spread of the virus, transmission is likely to continue even after a vaccine is developed, or until natural infection approaches the herd immunity threshold. On December 11, 2020, the FDA issued the first emergency use authorization (EUA) for the Pfizer-BioNTech COVID-19 vaccine for the prevention of COVID-19 for individuals 16 years of age and older. On December 18, 2020, the FDA issued a second EUA for the Moderna COVID-19 vaccine. On February 27, 2021, the FDA issued a third EUA for the Johnson & Johnson single-shot COVID-19 vaccine. Other companies are expected to submit applications to the FDA for EUA of their COVID-19 vaccines. Distribution of the vaccines has commenced, initially with front-line health-care workers receiving vaccinations to be followed by individuals deemed at high-risk of contracting COVID-19 or for severe illness from COVID-19. During the initial phases of the vaccine distribution, vaccine shortages and delays have been caused by high demand outstripping supply and challenging distribution logistics. With vaccine production ramping up and improvements to distribution logistics, experts estimate that the vaccines will be available to the general public by late spring/early summer of 2021.
A simple model can provide scenarios that help understand the number of infections and deaths that will occur in a naive population exposed to COVID-19. Table 1 uses the population, vaccine effectiveness and percentage of the population as inputs and calculates the exposed population and deaths (assuming a 2% mortality rate). If there is no vaccine and a 2% mortality rate, the model estimates total mortality of 6,560,000. Using the same model with a 90% effective vaccine with 90% vaccination rate and 2% mortality rate results in an exposed population of >60,000,000 and total mortality of 1,246,000. The model has several shortcomings and is unable to represent all of the uncertainty of the COVID-19 pandemic. However, these data underscore that even in a country with widespread vaccination, there is still a significant exposed population and substantial mortality remains.
Table 1: Exposed population and calculated deaths based on exposed population and vaccine effectiveness1
Population | Vaccine Effectiveness (%) | Vaccinated (%) | Exposed Population | Deaths (2% mortality) |
328,000,000 | 0 | 0 | 328,000,000 | 6,560,000 |
328,000,000 | 70 | 70 | 167,280,000 | 3,345,000 |
328,000,000 | 70 | 90 | 121,400,000 | 2,427,000 |
328,000,000 | 90 | 50 | 180,400,000 | 3,608,000 |
328,000,000 | 90 | 70 | 121,400,000 | 2,427,000 |
328,000,000 | 90 | 90 | 62,320,000 | 1,246,000 |
1. Source: http://www.cassbeth.com/covid-19/return-to-life/index.html
Even in the presence of effective vaccines, the rapid spread of COVID-19, as well as the risk of continued infection, underscores the importance of developing treatments for patients diagnosed with COVID-19.
Potential treatments under investigation include drugs that are already approved to treat other conditions and drugs still in development. Several classes of drugs are being investigated for potential treatments, including convalescent plasma, monoclonal antibodies, immune modulators and antivirals. To date, two therapies have been approved by the FDA for the treatment of COVID-19 (remdesivir and Dexamethasone) and three pharmaceutical treatments have received EUA from the FDA through randomized clinical trials: baricitinib + remdesivir, Regeneron’s antibody cocktail and
8
bamlanivimab. However, on November 20, 2020 the WHO recommends against the use of remdesivir in COVID-19 patients.
The critical need for effective treatments and the volume of urgent activity to find them underscore the tremendous potential value of an effective therapeutic. This value predominantly comes from two sources: health-related benefits and economy-related impacts. Health-related benefits include increased quality-adjusted life years (QALYs) that result from reducing mortality, symptom severity and duration. Potential benefits also include savings in healthcare expenditures that result from shorter hospital stays and less intensive use of healthcare resources generally.
It is important to note that each of these treatments, with the exception of dexamethasone, are delivered intravenously. This method of delivery complicates and prevents patients from taking the drug themselves in an outpatient environment.
In addition, with the exception of dexamethasone, none of these therapies have been shown to decrease mortality rates in COVID-19 patients. Further, the WHO has recommended against the use of remdesivir in COVID-19 patients.
A study conducted at the University of Southern California showed that:
| ● | a hypothetical treatment administered outside the hospital that reduces hospitalization risk by 50% results in 285,000 fewer hospitalizations, up to 71,000 fewer deaths, and up to $88 billion in value by the end of 2021; and |
| ● | a hospital-based treatment that reduces mortality and length of stay by 30% saves 51,000 to 85,000 lives, and generates up to $106 billion in value by the end of 2021. |
ANA001 has the potential to both reduce hospitalizations and decrease mortality, increasing the likelihood of therapeutic benefit to patients and significantly reducing healthcare expenditures.
Development Rationale
Niclosamide has broad in vitro antiviral activity (Figure 2) (Fan et al., 2019; Andersen et al., 2019; Jurgeit et al., 2012; Mazzon et al., 2019; Xu et al., 2020; Wu et al., 2004; Wen et al., 2007; Gassen et al., 2019) including activity against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Additionally, niclosamide has in vitro anti-inflammatory properties due to its inhibition of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and signal transducer and activator of transcription 3 (STAT3), two main drivers to induce the expression of proinflammatory cytokines. In addition, a recent in vitro study showed that niclosamide works as a potent bronchodilator that relaxed histamine induced constriction of human bronchial rings (Miner et al., 2019).
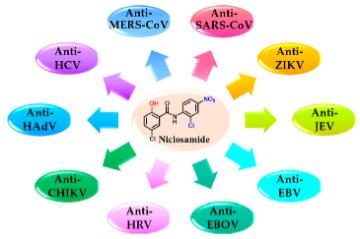
Fig. 2: Broad antiviral activity of niclosamide (modified from Xu et al, 2020). Niclosamide has demonstrated in vitro activity against severe acute respiratory syndrome coronavirus (SARS-CoV), Middle East respiratory syndrome coronavirus (MERS-CoV), Zika virus (ZIKV), Japanese encephalitis virus (JEV), hepatitis C virus (HCV), Ebola virus (EBOV), human rhinoviruses (HRVs), Chikungunya virus (CHIKV), human adenovirus (HAdV), and Epstein−Barr virus (EBV).
Antiviral Activity of Niclosamide
Niclosamide has broad in vitro antiviral activity (Figure 2) (Xu et al., 2020) including severe acute respiratory syndrome coronavirus (SARS-CoV) and Middle East respiratory syndrome coronavirus (MERS-CoV) (Wu et al., 2004, Wen et al., 2007, Gassen et al., 2019). SARS-CoV, MERS-CoV and SARS-CoV-2 share 86% homology (Wilder-Smith, et al., 2020) and it is therefore reasonable to assume that niclosamide has in vitro antiviral efficacy against SARS-CoV-2.
9
Three recent independent studies confirmed potent activity of niclosamide against SARS-CoV-2. The reported IC50 values were 0.15 µM (49 ng/mL) (Figure 3A; Shi (manuscript in preparation)), 0.28 µM (92 ng/mL) (Figure 3B) (Jeon et al., 2020) and 0.17 µM (56 ng/mL) (Figure 3C) (Gassen et al., 2020) resulting in an average IC50 of 0.2 µM (65 ng/mL). For comparison, the IC50 values of remdesivir, chloroquine and lopinavir (other current or former drug candidates as COVID-19 therapeutics) were 11.41 μM (3,732 ng/mL), 7.28 μM (2,381 ng/mL) and 9.12 μM (2,983 ng/mL), respectively (Jeon et al., 2020) making niclosamide 57, 36 and 46 times more effective respectively. As it was shown for MERS-CoV (Gassen et al., 2019), also SARS-CoV-2 replication is reduced by niclosamide via SKP2-inhibition, thus enhancing autophagy (Gassen et al., 2020).Furthermore, it was shown that pretreating cells for 24 h with 5 µM (1,636 ng/mL) niclosamide followed by drug washout and viral infection reduced SARS-CoV-2 replication significantly and as potently as spermidine, a natural enhancer of autophagy (Figure 4) (Gassen et al., 2020). This indicates that niclosamide has potential as a prophylactic COVID-19 treatment and that serum concentrations may not need to continuously exceed in vitro inhibitory levels to be effective. In summary, these results highlight the potency of niclosamide to inhibit replication of coronaviruses, especially SARS-CoV-2.
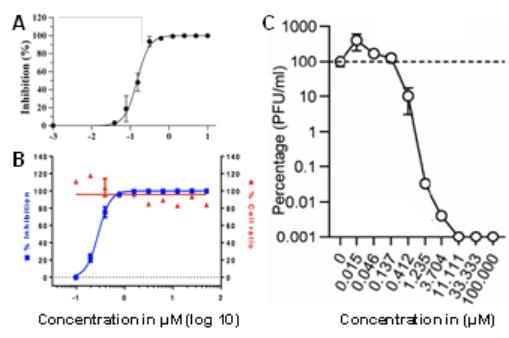
Fig. 3: Antiviral activity of niclosamide. (A) Vero E6 cells were infected with nano luciferase reporter SARS-CoV-2 in the presence of increasing concentrations of niclosamide. At 24 h post-infection, luciferase activity was measured. Shown are mean values from triplicate experiments ± standard deviation. Modified from Shi, 2020 (manuscript in preparation). (B) Vero cells were infected with SARS-CoV-2 in the presence of increasing concentrations of niclosamide. 24 h post infection (p.i.), cells were scored by immunofluorescence for the presence of the viral N protein. Modified from Jeon et al., 2020. (C) VeroFM cells were infected with SARS-CoV-2 and treated with increasing concentrations of niclosamide. SARS-CoV-2 plaque forming units (PFU) were determined 48 h p.i. by plaque assay. Shown are means from triplicate experiments ± standard error of the mean. Modified from Gassen et al., 2020.
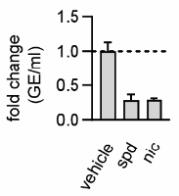
Fig. 4: Niclosamide has prophylactic properties. VeroFM cells were treated with 100 μM spermidine (spd), 5 μM niclosamide (nic) or left untreated (vehicle, veh) 24 h prior to infection with SARS-CoV-2. SARS-CoV-2 genome equivalents per ml (GE) were determined by real-time RT-PCR at 24 h post infection (p.i.). Data are presented as GE/mL (left) or as fold difference (right). Modified from Gass en et al., 2020.
Anti-Inflammatory Activity of Niclosamide
COVID-19 is a cytokine release syndrome (Hirano and Murakami, 2020) that is associated – amongst others – with ARDS, a widespread inflammation in the lungs and increased blood clotting. Two transcription factors - nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and signal transducer and activator of transcription 3 (STAT3) – are key drivers of cytokine release. Niclosamide was shown to inhibit them in vitro thus decreasing the production of proinflammatory cytokines.
10
NF-κB is a transcription factor that induces the expression of pro-inflammatory cytokines. In vitro experiments with U2OS cells demonstrated that niclosamide inhibited NF-kB transcription, its binding to deoxyribonucleic acid (DNA), tumor necrosis factor (TNF)-induced phosphorylation of IκBα, translocation of p65 into the nucleus, and expression of NF-κB-regulated downstream genes. The IC50 of niclosamide to inhibit NF-kB transcription was 0.13 µM (42.5 ng/mL) (Figure 5) (Jin et al., 2010).
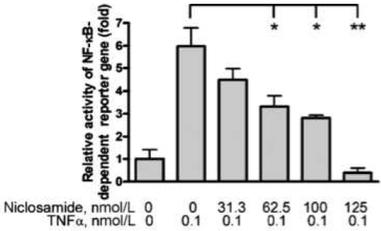
Fig. 5: Niclosamide prevents TNF-induced NF-κB activation. U2OS cells were co-transfected with NF-κB–TATA-Luc and renilla luciferase reporter plasmids. 24 h later, cells were first treated with different concentrations of niclosamide and then with 0.1 nmol/L TNFα, and luciferase intensity was measured. Shown are means of the fold change ± standard error of three independent experiments. * P < 0.05; ** P < 0.01. Modified from Jin et al., 2010.
Signal transducers and activators of transcription (STATs) are a class of transcription factors that regulate cellular and biological processes, including immune responses and angiogenesis, by modulating the expression of specific target genes (Yu et al., 2007). Upon stimulation by cytokines such as interleukin 6 (IL-6), tyrosine residue 705 (Tyr-705) in the STAT3 SH2 domain is phosphorylated, consequently inducing STAT3 to dimerize, translocate into the nucleus, and induce its binding to specific DNA response elements of target genes (Schuringa et al., 2000). Niclosamide has been shown to inhibit activation and transcriptional function of STAT3 in vitro. HeLa cells were transfected with a luciferase reporter driven by a promoter sequence with 7 STAT3 binding sites so that luciferase becomes active upon STAT3 binding. Niclosamide prevented binding and thus the transcriptional function of STAT3 with an IC50 of 0.25 μM (81.8 ng/mL) (Figure 6) (Ren et al., 2010).
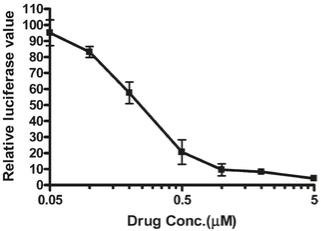
Fig. 6: Niclosamide inhibits STAT3 activity. Luciferase reporter activity was evaluated after 24 h of treatment with different concentrations of niclosamide. Relative luciferase units were the ratio of the absolute activity of firefly luciferase to that of renilla luciferase. Results are representatives of three independent experiments. Modified from Ren et al., 2010.
11
Bronchodilator Activity of Niclosamide
Additionally, niclosamide has been shown to have activity in vitro as a potent bronchodilator by inhibiting protein transmembrane member 16A (TMEM16A), a calcium-activated chloride channel. This ion channel is implicated in controlling both airway smooth muscle cell contraction and epithelial mucin secretion. The EC50 of niclosamide to relax carbachol induced constriction of human bronchial rings was 240 ng/mL (0.7 µM) (Fig. 7) (Miner et al., 2019).
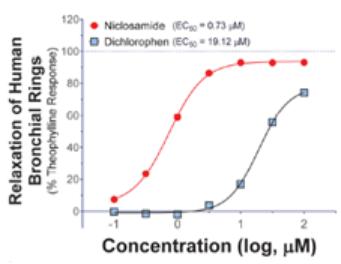
Fig. 7: Niclosamide induces relaxation of pre-contracted human bronchial rings. Human bronchial rings were contracted with carbachol and then treated with increasing concentrations of niclosamide (red curve). Modified from Miner et al., 2019.
12
ANA001/Niclosamide Preclinical Development
Table 2 summarizes pharmacology and safety studies with niclosamide in various animal models.
Table 2: Pharmacology and safety studies with niclosamide
Pharmacology and Toxicity Studies | ||
Type of Study | Model Used | Outcome/Findings |
Single and repeat oral dose toxicity | Range finding in rats ● 1-day ● 24-day ● 30-day ● 14-week ● 1-year ● 381-day | No signs of intoxication |
Single and repeat oral dose toxicity | Range finding in rabbits ● 1-day ● 11-day ● 25-day | No signs of intoxication |
Single and repeat oral dose toxicity | Range finding in dogs ● 1-day, ● 24-day ● 28-day ● 32-day ● 84-96-day ● 366-393-day | No signs of intoxication |
Single and repeat oral dose toxicity | Range finding in cats ● 1-day ● 12-day ● 24-day ● 4-week | No signs of intoxication |
Genotoxicity | In vitro (bacterial and lymphocyte reverse mutation) and in vivo (sister chromatid exchange and chromosomal aberration in mouse model) | No signs of genotoxic potential |
Carcinogenicity | Studies with rats: ● 14-week ● 365-381-day Studies with dogs ● 366-393-day | No signs of carcinogenicity |
Embryo fetal development studies | Range finding and development studies in rats and rabbits | No signs of teratogenicity, embryotoxicity nor toxicity to pregnant dams or their offspring. Niclosamide was approved as a pregnancy class B drug and is thus permitted to be used during pregnancy. |
Safety Pharmacology Studies
Studies to support the initial FDA marketing approval of niclosamide in 1982 were conducted prior to establishment of the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines, therefore the current standard battery of safety pharmacology studies pertaining to the central nervous,
13
cardiovascular and respiratory systems that are recommended in the ICH S7A guideline were not conducted. However, there is a substantial amount of data from human and animal exposures to support the expected safety of the proposed clinical development program. Relevant data provided in the available literature related to safety pharmacology in the nervous system, cardiovascular system, respiratory system and gastrointestinal system are summarized in Table 3 below.
Table 3: Safety pharmacology studies with niclosamide
Safety Pharmacology Studies | |||
Type of Study | Model Used | Outcome/Findings | Reference |
Nervous System | Mouse: 30 mg/kg niclosamide were administered intraperitoneally on 5 days per week for a total duration of 3 weeks | ● Niclosamide selectively diminished the malignant potential of primary human glioblastoma cells (pGBMs) by suppressing the Wnt, Notch, mTOR, and NF-κB signaling pathways. ● No drug-related adverse side effects were observed. | Wieland et al., 2013 |
Nervous System | Mouse: Oxaliplatin and niclosamide were administered intraperitoneally (200 μl injections) according to this weekly schedule: On day 1, either oxaliplatin (10 mg/kg) or vehicle and 6 hours later either niclosamide (10 mg/kg) or vehicle. On days 3 and 5 either niclosamide (10 mg/kg) or vehicle. This was repeated for 4-8 weeks. | ● Niclosamide prevented tactile hypoesthesia and thermal hyperalgesia and abrogated membrane hyperexcitability and also prevented intraepidermal nerve fiber density reduction and demyelination. ● No drug-related adverse side effects were observed | Cerles et al., 2017 |
Nervous System | Rat: Niclosamide was administered by intrathecal (0.05 mg/kg) and intraperitoneal (i.p.) (60 mg/kg) injection as well as oral gavage (18.8, 37.7, 75, 150 and 300 mg/kg). | ● Niclosamide reversed pain-related behavior in a mechanical hyperalgesia model of neuropathic pain. ● Neither treatment caused drug-related adverse effects | Ai et al., 2016 |
Cardiovascular System | Isolated rat mesenteric arteries | ● Niclosamide (0.5 µM; 164 ng/mL) relaxed vasoconstriction induced by phenylephrine (PE). ● Pretreatment with 0.5 µM (164 ng/mL) niclosamide for 20 minutes transiently inhibited PE-induced vasoconstriction. | Li et al., 2017 |
Respiratory System | Isolated human bronchial rings | ● Niclosamide reversed carbachol evoked bronchoconstriction with an EC50 of 240 ng/mL (0.7 µM). | Miner et al., 2019 |
Bioavailability of Niclosamide in Animal Studies
Table 4 summarizes bioavailability (expressed as maximum serum concentration, Cmax) of niclosamide upon oral administration in mice, rats, rabbits and dogs.
Table 4: Pharmacokinetic Data of Niclosamide After Oral Administration
Dose | Cmax | Reference | |
Mouse | 200 mg/kg | 893.7 ng/mL (2.7 μM; first peak), 78 ng/mL (0.2 μM; second peak) | Osada et al., 2011 |
Mouse | 50 mg/kg | 960±45 ng/mL (2.9±0.1 μM) | Lodagekar et al., 2019 |
Rat | 5 mg/kg | 354 ± 152 ng/mL (1.1±0.5 μM) | Chang et al., 2006 |
Rat | 5 mg/kg | 160 ng/mL (0.5 μM; first peak), 45 ng/mL (0.1 μM; second peak) | Lin et al., 2016 |
Rabbit | 100 mg/kg | 1.84 ± 0.3 μg/mL (5.6±0.9 μM) | Rehman et al., 2018 |
Dog | 125 mg/kg | 1.6 µg/mL (4.9 μM) | Andrews et al., 1982 |
ANA001/Niclosamide Clinical Development
Completed and Ongoing Clinical Studies
Between 1971 and 1978, niclosamide was administered to 6,365 patients under a U.S. IND. Doses were up to and including 2,000 mg/day for 7 days. There were 2,385 evaluable patients, of which 13.3% reported side effects, all of
14
which were mild or moderate, with none requiring treatment discontinuation. These included nausea/emesis in 4.1%, abdominal discomfort/loss of appetite in 3.4%, diarrhea in 1.6%, drowsiness/dizziness/headache in 1.4%, and skin rash/pruritis in 0.3% of patients (NDA 018669 Review Documentation).
In addition, according to the Safety Review of Niclosamide, Pyrantel, Triclabendazole and Oxamniquine report of the WHO, there have been 84 reported adverse drug reactions related to niclosamide between 1975 and 2004 in the WHO database. They include 173 reports from 16 countries and the most common reactions are related to skin and appendages (41 reports), GI tract (37 reports), cardiovascular system (28 reports) and anaphylactic reactions (9 reports).
Clinical studies with niclosamide for various indications (including COVID-19) are summarized in Table 5, Table 6, Table 7, Table 8 and Table 9.
Table 5: Human Studies in Healthy Volunteers
Study Title | N | Dose Regimen | Reference |
Phase 1 Clinical Study | 20 | Group 1: placebo Group 2: 2 g once per day for 3 days; this regimen was repeated after 6 days | NDA 018669 |
Unknown | Unknown | Adults: single dose 1,000-2,000 mg | Hecht and Gloxhuber, 1960, translated |
Unknown | Children (6-15 years): single dose 750-1,000 mg |
NCT = National clinical trial
15
Table 6: Human Studies in Patients (Tapeworm)
Study Title | N | Dose Regimen | Reference |
Phase 3 Clinical Study | 2,385 evaluable subjects | 2,000 mg/day for 1-7 days | NDA 018669 |
Targeted Screening for Taenia Solium Tapeworms | 1,811 | single oral dose of 1,000 mg (11-34 kg), 1,500 mg (35-50 kg), 2,000 mg (>50 kg) | NCT01296958 |
Unknown | 3,663 (patients infected with T. saginata) | adults: single oral dose of 2,000 mg, | NDA 018669 |
Unknown | 297 (patients infected with D. latum) | 2,000 mg single oral dose | NDA 018669 |
Unknown | 464 (patients infected with H. nana; world-wide survey) | daily administration for 5-7 days | NDA 018669 |
Unknown | 34 (patients infected with T. saginata) | 2,000 mg oral dose (1,000 mg followed by 1,000 mg after one hour) | Abrams et al., 1963 |
Niclosamide treatment of cestodiasis | 47 | patients <75 lbs: single 1,000 mg oral dose OR single 2,000 mg oral dose on day 1, followed by 500 mg daily for 5-7 days | Perera et al., 1970 |
Niclosamide as treatment for tapeworm infection in man | 86 | >15 years: single oral dose of 2,000 mg for 1 day or 5-7 days <15 years, >34 kg: single oral dose of 1,500 mg OR single oral dose of 1,500 mg on day 1, followed by 1,000 mg daily for 5-7 days <15 years, 11-34 kg: single oral dose of 1,000 mg OR single oral dose of 1,500 mg on day 1, followed by 500 mg daily for 5-7 days | Jones, 1979 |
Unknown | 5 | oral dose of 1,000-2,000 mg on day 1, followed by a 500 mg daily dose for the following 6 days | Ostrosky-Wegman et al., 1986 |
NCT = National clinical trial
16
Table 7: Human Studies in Patients (Oncology)
Study Title | N | Dose Regimen | Reference |
A phase I study of niclosamide in combination with enzalutamide in men with castration-resistant prostate cancer | 5 | oral doses of 500 mg, 1,000 mg or 1,500 mg 3 times daily for 4 weeks | Schweizer et al., 2018 and |
Phase II trial to investigate the safety and efficacy of orally applied niclosamide in patients with metachronous or synchronous metastases of a colorectal cancer progressing after therapy: the NIKOLO trial | 37 (estimated) | 2,000 mg orally until disease progression or toxicity | Burock, et al., 2018, Burock et al., 2020 and |
Abiraterone Acetate, Niclosamide, and Prednisone in Treating Patients with Hormone-Resistant Prostate Cancer | 40 (estimated) | Oral dose twice a day; courses repeat every 4 weeks in the absence of disease progression or unacceptable toxicity | NCT02807805 |
Enzalutamide and Niclosamide in Treating Patients with Recurrent or Metastatic Castration-Resistant Prostate Cancer | 12 (estimated) | Oral dose twice a day in combination with enzalutamide on week 1-4; courses repeat every 4 weeks in the absence of disease progression or unacceptable toxicity | NCT03123978 |
Niclosamide for Familial Adenomatous Polyposis | 72 (estimated) | 650 mg orally once a day for 6 months | NCT04296851 |
A Study of Niclosamide in Patients with Resectable Colon Cancer | 1 | orally daily from day 1-7 prior to surgery | NCT02687009 |
NCT = National clinical trial
Table 8: Human Studies in Patients (Various Indications)
Study Title | N | Dose Regimen | Reference |
Niclosamide With Etanercept in Rheumatoid Arthritis | 110 | 500 mg BID with 50 mg etanercept weekly for 8 weeks | NCT03160001 |
Niclosamide Role in Diabetic Nephropathy | 60 (estimated) | 1,000 mg orally once daily for six months in combination with maximum tolerated dose of ACE inhibitors | NCT04317430 |
A Study of Niclosamide Enemas in Subjects with Active Ulcerative Proctitis or Ulcerative Proctosigmoiditis | 51 (estimated) | 150 mg/60 mL or 450 mg/60 mL enemas given BID for 6 weeks | NCT03521232 |
NCT = National clinical trial
17
Table 9: Human Studies in Patients (COVID-19)
Study Title | N | Dose Regimen | Reference |
Niclosamide for Patients With Mild to Moderate Disease From Novel Coronavirus (COVID-19) | 100 (estimated) | 2,000 mg orally once daily for 7 days | NCT04399356 |
Phase I Study to Evaluate the Safety, Tolerability, and Pharmacodynamics (PD) of DWRX2003 (Niclosamide IM Depot) Injection Following Intramuscular Administration in COVID-19 Patients | 40 (estimated) | Four intramuscular injections of DWRX2003 at pre-defined injection sites (total amount of niclosamide administered per dosing group is 96 mg, 288 mg, 480 mg, 672 mg and 960 mg, respectively) | NCT04541485 (not yet recruiting) |
A Double-blind, Randomized, Placebo-controlled, Single-ascending Dose Phase I Study to Evaluate the Safety, Tolerability and Pharmacokinetic Properties of Niclosamide Injectable (DWRX2003) Following Intramuscular Administration in Healthy Volunteers | 24 (estimated) | Four intramuscular injections of DWRX2003 at pre-defined injection sites (total amount of niclosamide administered per dosing group is 288 mg, 576 mg and 960 mg, respectively) | NCT04592835 (not yet recruiting) |
Phase I Study to Evaluate the Safety, Tolerability, Pharmacodynamics (PD) and Pharmacokinetics (PK) of DWRX2003 (Niclosamide IM Depot) Injection Following Intramuscular Administration in Healthy Volunteers | 32 (estimated) | Four intramuscular injections of DWRX2003 at pre-defined injection sites (total amount of niclosamide administered per dosing group is 144 mg, 432 mg, 960 mg and 1,200 mg, respectively) | NCT04524052 (not yet recruiting) |
A Double-blind, Randomized, Placebo-controlled, Single-ascending Dose Phase I Study to Evaluate the Safety, Tolerability and Pharmacokinetic Properties of Niclosamide Injectable (DWRX2003) Following Intramuscular Administration in Healthy Volunteers | 24 (estimated) | Two intramuscular injections of DWRX2003 at pre-defined injection sites (total amount of niclosamide administered per dosing group is 96 mg, 432 mg and 960 mg, respectively) | NCT04749173 (recruiting) |
A Phase 2/3 Randomized and Placebo-Controlled Study of ANA001 in Moderate and Severe COVID-19 Patients | 436 (estimated) | 1,000 mg orally twice per day for 7 consecutive days | NCT04603924 (recruiting) |
A Randomized, Double-Blind, Single and Multiple Ascending Dose Study to Assess the Safety and Pharmacokinetics of Niclosamide in Healthy Adults | 66 (estimated) | SAD study: single oral dose (1,000 mg, 2,000 mg or 3,000 mg) MAD study: oral dose twice daily or thrice daily for 7 consecutive days (total daily dose will not exceed 2,000 mg) | NCT04705415 (recruiting) |
Efficacy of HYdroxychloroquine and DILtiazem-nIClosamide Combination for the Treatment of Non-severe Forms of SARS-CoV2 Infection in Patients With Co-morbidities: Multicenter, Randomized, Open-labeled Controlled Trial | 480 (estimated) | Niclosamide 500 mg × 4 on day 1 then 500 mg × 2 per day + diltiazem 60 mg × 3 per day for 10 days | NCT04372082 |
A Phase 2 Randomized Double Blind, Placebo-controlled Study on the Safety and Efficacy of Niclosamide in Patients With COVID-19 | 148 (estimated) | Oral dose (400 mg 3 times daily for 14 consecutive days) | NCT04542434 (not yet recruiting) |
Phase 2, Multicentre, Randomized, Double Blind, 2 Arms Placebo-controlled Study in Adults With Moderate COVID-19 With Gastrointestinal Signs and Symptoms | 100 (estimated) | Oral dose (3 times daily for 14 consecutive days, dose unknown) | NCT04436458 (not yet recruiting) |
A Phase III, Randomized, Placebo-controlled, Clinical Trial to Evaluate the Efficacy and Safety of Co-administered Niclosamide in Patients Treated With an Established Regimen for Novel Coronavirus Infectious Disease (COVID-19) | 200 (estimated) | 200 mg/10 mL suspension administered 3 times daily for 5 consecutive days | NCT04558021 (recruiting) |
NCT = National clinical trial
18
Bioavailability of Niclosamide in Human Studies
As ANA001 is orally administered, it can be taken by hospitalized, ambulatory and non-hospitalized individuals and it can be administered in both ambulatory and acute care environments. Although niclosamide has low bioavailability, data from available literature clearly demonstrate that blood levels of niclosamide after oral administration exceed effective in vitro concentrations:
| ▪ | Oral administration of a single dose of 2,000 mg of niclosamide reached maximal systemic serum concentrations (Cmax) in humans of 0.76-18.3 μM (249-5,986 ng/mL) (Andrews et al., 1982). |
| ▪ | A study in prostate cancer patients showed that 149-182 ng/mL (0.46-0.56 µM) become available after a single oral dose of 1,000 mg (Schweizer et al., 2018). |
| ▪ | In a recent study, colorectal cancer patients received 2,000 mg of niclosamide orally once a day until disease progression or toxicity (up to four months). Plasma levels mainly peaked 240 minutes after the first niclosamide administration with a median Cmax of 2.03 µM (665 ng/mL) (Burock et al., 2018). |
The lower bounds of the reported Cmax values fall within the effective in vitro ranges of niclosamide as an antiviral agent (0.15-0.28 µM, 49-92 ng/mL) (Shi, 2020, manuscript in preparation; Jeon et al., 2020; Gassen et al., 2020) and an anti-inflammatory drug that inhibits NF-kB and STAT3 at 0.13 µM (42.5 ng/mL) (Ji et al., 2010) and 0.25 µM (81.8 ng/mL) (Ren et al., 2010), respectively. It is therefore anticipated that the dose regimen foreseen for the ANA clinical development program will provide sufficient systemic and intracellular drug levels for effective antiviral and anti-inflammatory activity.
In summary, the following characteristics support the use of niclosamide as an oral treatment for COVID-19:
| ▪ | Niclosamide significantly inhibits in vitro replication of SARS-CoV-2 with an IC50 of 0.15 µM-0.28 µM (49-92 ng/mL) (Shi, unpublished results; Jeon et al., 2020; Gassen et al., 2020). |
| ▪ | Pretreatment of mammalian cells for 24 h with 5 µM (1,636 ng/mL) niclosamide followed by drug washout and viral infection reduced SARS-CoV-2 replication significantly (Gassen et al., 2020) pointing to the fact that niclosamide could be used as a pre-exposure prophylaxis. |
| ▪ | Niclosamide has anti-inflammatory properties that are expected to be relevant in COVID-19 patients. Niclosamide inhibits NF-kB and STAT3 with an IC50 of 0.13 µM (42.5 ng/mL) (Jin et al., 2010) and 0.25 μM (81.8 ng/mL), respectively (Ren et al., 2010). |
| ▪ | Bronchodilation may be helpful in ARDS patients. Niclosamide is a potential bronchodilator based on in vitro results that showed that niclosamide relaxed carbachol induced constriction of human bronchial rings with an EC50 of 240 ng/mL (0.7 µM) (Miner et al., 2019). |
| ▪ | In humans, a single oral dose of 1,000 mg reached maximal systemic serum concentrations of 149-182 ng/mL (0.46-0.56 µM) (Schweizer et al., 2018). A single oral dose of 2,000 mg reached maximal systemic serum concentrations of 249-5,986 ng/mL (0.76-18.3 μM) (Andrews et al., 1982) and 2.03 µM (665 ng/mL) (Burock et al., 2018), respectively. These concentrations exceed effective in vitro concentrations ~2-4-fold suggesting that a sufficient amount of drug becomes bioavailable through the course of 2,000 mg daily to provide effective antiviral and anti-inflammatory effects. |
| ▪ | Similar to remdesivir, ANA001 has a short serum half-life (0.9 h for remdesivir vs. ~4 h for niclosamide (Schweizer et al., 2018) but in both cases intracellular concentrations are more relevant than serum PK values with regard to therapeutic effects). |
| ▪ | Extensive preclinical studies showed that niclosamide does not cause significant toxicity, nor carcinogenicity, mutagenicity or embryotoxicity. |
| ▪ | Niclosamide is an FDA approved drug that has been used to treat tapeworms in humans (adults, children and pregnant women) with a well-understood safety profile upon oral administration. |
Taken together, these data demonstrate the potential for ANA001 to treat COVID-19 and support its further development.
ANA001 Phase 2/3 Clinical Development
ANA001 is currently being tested in a U.S. Phase 2/3 study titled “A Phase 2/3 Randomized and Placebo-Controlled Study of ANA001 in Moderate COVID-19 Patients” (NCT04603924). Niclosamide has demonstrated both antiviral and immunomodulatory activity with possible downstream effects on coagulation abnormalities observed in COVID-19. These effects support the development of ANA001, an oral formulation of niclosamide, for the treatment of COVID-19. It is
19
therefore anticipated that ANA001 will reduce viral load and inflammation associated with cytokine dysregulation, acute respiratory distress syndrome (ARDS), and coagulation abnormalities and thus improve time to clinical improvement as defined as hospital discharge recorded using the WHO Ordinal Scale for Clinical Improvement.
The study consists of two parts:
| ▪ | Study Part 1: Includes 60 subjects randomized in 1:1 ratio to receive ANA001 or matching placebo to assess the safety and tolerability of ANA001 1,000 mg BID for 7 days. An independent Data Monitoring Committee will review the safety profile of ANA001 1,000 mg PO BID prior to the initiation of Part 2 of the study. |
| ▪ | Study Part 2: Includes 376 subjects randomized in 1:1 ratio to receive ANA001 or matching placebo to demonstrate the statistical superiority of ANA001 1,000 mg PO BID for 7 consecutive days compared to matching placebo in the treatment of subjects with moderate COVID-19 infection. Additionally, the safety profile of ANA001 will be assessed compared to placebo. |
The primary endpoints of Phase 2 are:
| ▪ | Treatment-emergent Adverse Events (AEs), Severe AEs (SAEs), deaths, and discontinuations due to an AE; and |
| ▪ | Vital signs and laboratory (hematology, chemistry, and coagulation) parameters. |
The secondary endpoints of Phase 2 are:
| ▪ | Median time (in hours) to hospital discharge (where discharge is defined as a score of 1 or 2 in the WHO Ordinal Scale for Clinical Improvement); and |
| ▪ | Plasma concentrations will be explored on Days 1, 2, 3, or Day 4. |
The primary endpoints of Phase 3 are:
| ▪ | Time to clinical improvement as measured by median time (in hours) to hospital discharge (where discharge is defined as a score of 1 or 2 in the WHO Ordinal Scale for Clinical Improvement); |
| ▪ | Treatment-emergent AEs, SAEs, deaths, and discontinuations due to an AE; and |
| ▪ | Vital signs and laboratory (hematology, chemistry, and coagulation) parameters. |
The secondary endpoints of Phase 3 are:
| ▪ | Mean change from baseline (BL) in NEWS2 on Day 8 and Day 15; and |
| ▪ | Mean number of days on rescue treatment (COVID-19 therapies that are FDA approved or have emergency-use authorization) within 15 days after enrollment. |
ANA001 Development Plan through NDA
An NDA is the classical vehicle through which the FDA approves a new pharmaceutical for sale and marketing in the US. However, for COVID-19 drugs and biological products (remdesivir, hydroxychloroquine, monoclonal antibodies and convalescent plasma), the FDA has granted EUA to make treatments during this pandemic available without having all safety and efficacy data available. NeuroBo expects data readout of the Phase 2 trial in the second or third quarter of 2021. NeuroBo will request a Type C meeting with the FDA in the third quarter of 2021 to discuss whether data justify an EUA or if data readout from the Phase 3 trial and other additional studies will be necessary for approval of ANA001 as a COVID-19 therapeutic. In case an EUA will not be issued, NeuroBo will pursue an NDA via a 505(b)(2).
20
NB-01
NB-01 addresses a range of mechanisms that contribute to neuropathic pain and nerve degeneration in diabetic and other peripheral neuropathies. These include a decrease in key inflammatory markers, restoration of nerve growth factor (NGF) to normal levels, and reduction of advanced glycation end products (AGEs). Inflammation is a central factor in pain generation and other peripheral neurodegenerative diseases. NB-01 reduces levels of TNF-a and IL-6, both of which are markers of inflammation. NB-01 also reduces AGEs, which are implicated in diabetes-related complications. AGE inhibitors have been clinically tested as potential treatments for these complications. NB-01 also restores the neurotrophin NGF, which is involved in nerve growth, maintenance and repair. NB-01 has been shown in animal models to alleviate symptoms of PDN.
Background
Based on third-party research, the U.S. population with diabetes is estimated at 30.3 million people. At least half of these individuals will develop diabetic neuropathy, and up to 25% of those individuals will develop neuropathic pain. According to the industry intelligence firm GlobalData plc, as of 2018, the global PDN market was responsible for approximately $3.6 billion in annual sales, approximately $2.6 billion of which is concentrated in the U.S. The same source projects that the global PDN market will increase to approximately $7.1 billion in annual sales by 2026 with approximately $4.8 billion of such sales concentrated in the U.S. Products to address PDN make up about 60% of the market, and products to address indications such as chemotherapy-induced and post-traumatic neuropathic pain are estimated to constitute an additional 20% of the market. The market is characterized by a significant unmet need, with more than 50% of patients not adequately responding to first-line therapy and patients experiencing significant side effects with existing approved therapies.
In the U.S., there are currently only three FDA-approved treatments for PDN: pregabalin (Lyrica); duloxetine (Cymbalta) and tapentadol (Nucynta ER). Despite an established treatment protocol for PDN based on these approved therapeutics, the current treatment paradigm for patients suffers from numerous shortcomings as a result of their negative side effects associated with the available FDA-approved drug products. The first line of therapy typically consists of anti-epileptic drugs (AEDs) such as gabapentin and pregabalin, which are insufficient on their own in that they have been shown to exhibit only moderate efficacy accompanied by moderate to severe side effects such as somnolence and dizziness in some patients, and, even after drug treatment, 50% to 70% of patients still experience pain. If pain persists beyond treatment with AEDs, as it often does, the second line of therapy typically consists of prescriptions for anti-depressants (SNRIs and TCAs), which have been shown to reduce pain only by an additional 20% when added to AED treatment. Treatment with anti-depressants is also associated with significant drug-to-drug interactions. If pain persists beyond treatment with AEDs and anti-depressants, the third line of therapy typically consists of opiates, which are only appropriate as a short-term option and have been shown to exhibit potentially harmful addictive and habit-forming side effects. A significant number of mortalities from drug overdose have been caused by opiates. Beyond the potential side effects, the existing approved therapies for PDN are burdened by additional safety and efficacy concerns.
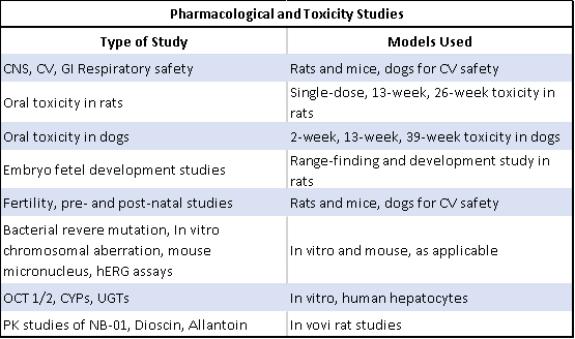
NB-01 Preclinical development
Extensive and comprehensive preclinical pharmacology, safety and toxicology studies have been completed with NB-01, as detailed in the table below. Among the safety and toxicology studies completed are: (i) central nervous system (CNS), cardiovascular (CV), gastrointestinal (GI), and respiratory safety in rats, mice and dogs; (ii) a single-dose 13-week and
21
26-week oral toxicity study in rats; (iii) a single-dose 13-week and 26-week oral toxicity study in dogs; (iv) range-finding embryo fetal development studies in rats; and (v) fertility, pre-and post-natal studies in rats.
In addition, in mechanism of action studies conducted by Dong-A ST, NB-01 induced nerve regeneration in streptozotocin (STZ)-induced and db/db diabetes mouse models with a significant increase in axon diameter and thickness of myelin sheath, returning thickness and diameter to almost the naturally occurring levels. Similar results were achieved in rat models, including the streptozotocin (STC) diabetes model. NGF has been shown to be lowered in diabetes and diabetic neuropathy animal models, and the administration of NB-01 in these models shows elevation of endogenous NGF to near-normal levels. Preclinical studies have demonstrated that NB-01 has a demonstrable impact on reduction of AGEs as well as inflammatory markers (TNF-alpha and interleukin-6) which are implicated in nerve degeneration in diabetes.
Additional studies have been completed on the effect of NB-01 on thermal and mechanical hyperalgesia in mouse models, including the STZ diabetes model and genetic (db/db) diabetes model. The data from these studies have demonstrated that NB-01 alleviates both thermal and mechanical hyperalgesia relative to the control.
With respect to additional neuropathic indications, NB-01 has also been studied for its effects on rat models of chemotherapy-induced neuropathic pain and chronic constriction injury (CCI). In these studies, NB-01 demonstrated an analgesic effect on rats, measured by threshold of paw pressure tolerance, during treatment with paclitaxel and with CCI. In both cases, the paw pressure threshold was significantly elevated following dosing with NB-01.
NB-01 Phase 2 Clinical Development
Completed Phase 2 trial in Korea. A 15-site, 128-subject, double blind, dose ranging, randomized, placebo-controlled Phase 2 trial to assess the efficacy and safety of NB-01 in the treatment of subjects with PDN has been completed in Korea. Three doses of NB-01 were evaluated versus placebo in 128 subjects (32 per dose group), administered daily for an 8-week treatment period. The treatment groups were placebo or one of NB-01 100 mg, 200 mg, or 300 mg, administered three times daily (TID), for a total daily NB-01 dose of 300 mg, 600 mg or 900 mg, respectively. The primary endpoint of the study was reduction in the average daily Pain Numerical Rating Scale (NRS) score from baseline at 8 weeks. Secondary endpoints included percentage reduction in NRS at 8 weeks, Patient Global Impression of Improvement (PGI-I) scale, Clinical Global Impression of Severity, and change from baseline in the NRS based on a daily patient diary.
22
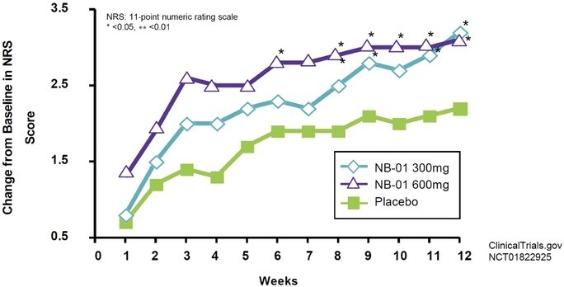
Completed Phase 2 trial in the United States. A 14-site, 128-subject, double blind, dose ranging, randomized, placebo-controlled Phase 2 trial to assess the efficacy and safety of NB-01 in the treatment of subjects with PDN has been completed in the United States. Three doses of NB-01 were evaluated versus placebo in 128 subjects (32 per dose group), administered daily for a 12-week treatment period. The treatment groups were placebo or one of NB-01 100 mg, 200 mg, or 300 mg, administered three times daily (“TID”) for a total daily NB-01 dose of 300 mg, 600 mg or 900 mg, respectively. The primary endpoint of the study was reduction in the clinic visit Pain Numerical Rating Scale (“NRS”) score at 12 weeks. Secondary endpoints included percentage reduction in clinic visit NRS score at 12 weeks, proportion of subjects with at least 30% improvement in the clinic visit pain NRS score, proportion of responders in the Patient Global Impression of Improvement (“PGI-I”) scale, and change from baseline in the NRS based on a daily patient diary.
14 U.S. sites, 128 subjects, 3 doses vs. placebo
(600mg and 300mg doses shown here)
Results of Phase 2 U.S. Clinical Trial for NB-01
Measured as a change from baseline in NRS score over the course of 12 weeks, NB-01 was observed to be generally well tolerated in its Phase 2 study at doses ranging from 300 mg to 900 mg against placebo, as summarized in the table below.
Measured in terms of changes in the mean NRS score at week 12 in the Phase 2 study, patients treated with the 300 mg and 600 mg doses showed statistically significant improvement from baseline in pain scores. As summarized in the table below, patients treated with the 300 mg dose experienced an average 45% change from the baseline NRS score, and patients treated with the 600 mg dose experienced an average 47% change from the baseline NRS score.
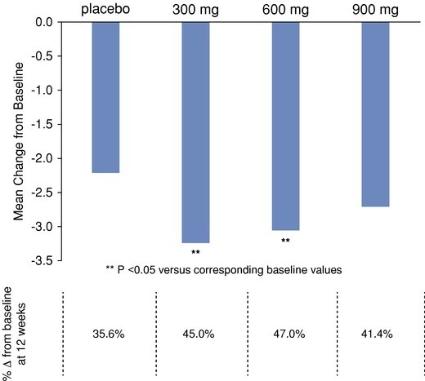
Mean Change in NRS Score at Week 12 Following NB-01 Dosing
23
During the Phase 2 study, patients in each dose group experienced a number of adverse events, including nausea and pruritus, but not at a level higher than those of subjects who received placebo.
Future Development of NB-01
In light of the present business environment including the impact of the COVID-19 disease that emerged in December 2019 as a global threat, we have determined that any attempt to conduct Phase 3 clinical trials for NB-01, as previously announced, would be difficult if not impossible in the short or medium term. To conserve financial resources, in the first quarter of 2020 we directed our contract research organization (CRO) partners and other vendors working on the Phase 3 clinical trials of NB-01 to cease all work and we gave notice of termination of our existing contract arrangements with each of them.
We are currently re-evaluating alternatives to bring the NB-01 asset to the market through a different regulatory pathway. Development of NB-01 as an orphan drug is among the alternatives we are considering, and we may conduct feasibility studies to identify a rare disease relevant to NB-01. Additionally, we are considering marketing the NB-01 product line as nutraceutical (non-pharmaceutical) products. There is no assurance that we will be able to pursue either alternative for NB-01. There is no assurance that we will be able to pursue any of these alternatives for NB-01. See “We have determined to postpone the initiation of Phase 3 clinical trials of NB-01 under present circumstances, and we have terminated all of our agreements with contract research organizations related to NB-01. We may not be able to successfully develop NB-01 pursuant to other alternatives, including as an orphan drug or as a nutraceutical candidate” in Part I, Item 1A, “Risk Factors,” of this Annual Report on Form 10-K.
NB-02
NB-02 is in development for the symptomatic and disease modifying treatment of neurodegenerative diseases, including Alzheimer's disease and tauopathies. In preclinical studies, we have observed the mechanisms of action of NB-02 to include inhibition of tau phosphorylation, acetylcholinesterase (AChE) inhibition, inhibition of Ab toxicity and amyloid plaque formation, and anti-inflammatory effects.
Specifically, in both in vitro and in vivo models, NB-02 has demonstrated inhibition of AChE, as is the case with three of the current drugs on the market to treat the symptoms of Alzheimer's disease. It has also demonstrated inhibition of tau phosphorylation and of amyloid plaque formation, both mechanisms believed to contribute to the progression of neurodegenerative diseases.
NeuroBo acquired NB-02 from Dong-A ST on January 18, 2018. NeuroBo has full worldwide rights to all disease indications for NB-02 from the asset acquisition and does not have further obligations in future payments to Dong-A ST however; if NeuroBo wishes to sell products using NB-02 in the Republic of South Korea, Dong-A ST is entitled to certain notice rights and rights to negotiate with respect to any distribution agreement for the sale of NB-02 in such territory.
Background
Alzheimer’s disease (AD) is a progressive and chronic neurodegenerative disease characterized by memory and cognitive deterioration beyond normal aging that becomes severe enough to interfere with daily tasks. It is the most common form of dementia. AD is characterized by the loss of neurons and synapses in the cerebral cortex and certain subcortical regions. Different mechanisms have been implicated in the underlying cause of the cognitive and functional impairments observed in AD. Degeneration of the cholinergic nervous system has been shown to be closely linked to the impairment of cognitive functions. Also, neurodegeneration caused by the buildup of two structural abnormalities known as beta-amyloid (βA) plaques and hyper-phosphorylated tau protein (pTau) aggregates that leads to neurofibrillary tangle formation is thought to play a major role in the pathogenesis of AD. However, neurodegeneration in AD appears to be a multi-factorial event, in which various genetics as well as environmental risk factors may play a role sequentially and/or in parallel.
Despite the need, there is no cure for AD. Currently available treatments can only temporarily provide symptomatic relief without the ability to control disease progression. As the life expectancy increases, the prevalence of aging-
24
associated diseases such as AD has also dramatically increased and has become a major public health concern. Therefore, there is an urgent need for the development of AD drugs that are capable of more than just relieving the symptoms. The current goal in AD therapeutics research is to search for drugs/interventions that can directly address the underlying disease processes of AD, also known as disease-modifying therapy (DMT), to delay or even prevent disease progression.
Based on the preclinical studies, NB-02 has both symptomatic relief benefits and disease modifying mechanism of action. Specifically, in in vivo studies, NB-02 was shown to up-regulate nerve growth factor (NGF), brain-derived neurotrophic factor (“BNDF”) and cellular antioxidant defense system, which is indicative of neuroprotection and neuronal survival. Decrease in the accumulation of Aβ protein level and tau protein hyper-phosphorylation was also observed, which suggests NB-02 has disease modification efficacy by clearance of the toxic proteins that represent the neuropathological indices of AD. Furthermore, NB-02 was shown to reverse cognition impairment by suppressing AChE activity. The findings from these nonclinical studies collectively suggested that NB-02 could be a treatment candidate for AD via multiple mechanisms of action including cognition enhancement and disease modification.
Development Plan
NB-02 has shown considerable promise as a neuroprotective agent in preclinical studies, demonstrating a multimodal mechanism of action including inhibition of tau phosphorylation, AChE inhibition, inhibition of Ab toxicity and amyloid plaque formation, and anti-inflammatory effects. We intend to further leverage the benefits of tau modulation by NB-02 in conjunction with the other pathway effects to explore treatment of certain dementias, such as tauopathy indications. We believe that leveraging the therapeutic advantages of NB-02 will drive a paradigm shift in the treatment of Alzheimer’s disease and other neurodegenerative diseases. Although NB-02 is almost ready for the submission of an IND application to the FDA, we intend to postpone the first human clinical trials until global macroeconomic conditions improve. Additionally, we are assessing whether to pursue further development of NB-02 as an orphan drug.
Gemcabene
Gemcabene is a novel, once-daily, oral therapy designed to target known lipid metabolic pathways to lower levels of LDL-C, hsCRP and triglycerides. Gemcabene shares many of the attributes of statin therapy, including broad therapeutic applications, convenient route of administration and cost-effective manufacturing process, but does not appear to increase the reporting of myalgia when added to statin therapy. Gemcabene has also shown additive LDL-C lowering in combination with stable low, moderate or high-intensity statin therapy. As described below, we licensed global rights to Gemcabene from Pfizer in April 2011. The license with Pfizer was renegotiated to April 2024.
Gemcabene was being evaluated in a Phase 2 randomized, double-blind, placebo-controlled study to assess its efficacy safety and tolerability in patients with severe hypertriglyceridemia. In January 2016, the Gemcabene Phase 2 clinical study was placed on partial clinical hold as the FDA requested 2-year rat and mouse carcinogenicity studies to be completed and submitted. The study currently remains on partial clinical hold for the treatment of dyslipidemia. NeuroBo is currently assessing the path forward for Gemcabene for the indication for COVID-19 and does not expect to direct additional resources towards Gemcabene as a cardiovascular therapy in the near term.
Background
Gemcabene's mechanism of action is multifaceted. In the liver, Gemcabene acts in two major ways to reduce levels of circulating LDL-C and triglycerides: (i) inhibition of the two metabolic pathways that synthesize precursors (i.e., cholesterol and fatty acids) of VLDL-C, LDL-C and triglycerides and (ii) stimulation of a liver mechanism known as the remnant receptor pathway that removes particles that contain cholesterol and triglycerides from the blood. Gemcabene's stimulation of this remnant receptor pathway involves enhanced removal of an LDL-C precursor known as very low-density lipoprotein remnants. With regard to Gemcabene's anti-inflammatory properties, in human clinical trials and animal studies, to date, Gemcabene has been shown to significantly reduce plasma levels of CRP. Furthermore, in preclinical studies of dyslipidemia as well as NASH, Gemcabene inhibited production of a number of known pro-inflammatory molecules (e.g., CRP, CCR2, CCR5, IL-6, TNF-alpha, MCP-1 and MIP1-beta) as well as pro-fibrotic factors (e.g., TIMP-1, MMP-2). Overall, Gemcabene's multifaceted mechanism of action provides the potential for
25
safely addressing multiple major risk factors in a broad array of cardiometabolic patients who have an elevated risk of cardiovascular or liver disease, even when taking conventional therapies.
Clinical Experience with Gemcabene
Gemcabene has been assessed in 25 Phase 1 and Phase 2 clinical trials. Across these trials, over 1,500 adult subjects have participated, including healthy volunteers and patients with various underlying conditions. Of these subjects, over 1,100 have been exposed to at least one dose of Gemcabene.
Across the Gemphire-sponsored clinical trials, Gemcabene was observed to be well tolerated at single doses up to 1,500 mg and multiple doses up to 900 mg/day. Safety of the subjects in these trials was evaluated by adverse event (“AE”) monitoring, clinical laboratory assessments, electrocardiograms (ECGs), physical examinations, and vital sign assessments. Across all trials, 10 Gemcabene treated healthy volunteers or patients reported a treatment-emergent severe adverse event (“SAE”), none of which were considered by the clinician to be related to Gemcabene. No deaths occurred in any of the trials. AEs reported were generally mild to moderate in intensity with the most common events being headache, weakness, nausea, dizziness, upset stomach, infection and abnormal bowel movements. Gemcabene, when compared with placebo, was not associated with an increased incidence of myalgia or liver enzyme elevations, whether as monotherapy or in combination with statin therapy. Elevated levels of liver enzymes, specifically alanine transaminase (“ALT”) and/or aspartate aminotransferase (“AST”), were observed in three patients (0.27% of Gemcabene treated subjects). These three patients had ALT or AST levels more than three times the upper limit of normal (“ULN”) returning to near baseline after cessation of treatment. Small mean increases in serum creatinine and blood urea nitrogen (“BUN”) have been observed in some trials. The increase in creatinine values was reversible returning to baseline within approximately four weeks of cessation of Gemcabene. No clinically meaningful changes were observed in physical examinations or vital signs, including blood pressure.
In addition, Gemcabene demonstrated promising clinical pharmacology attributes across 15 completed Company-sponsored Phase 1 trials in healthy subjects, such as once-daily dosing, no meaningful drug-drug interactions with high-intensity statins and no observed food effect. Gemcabene can be taken with or without food. Gemcabene was observed to: (i) be rapidly absorbed following oral administration with time of maximum concentration within two hours and (ii) reach maximum plasma concentration (Cmax) and area under the curve over 24 hours (AUC0-24) that were dose proportional following both single-and multiple-dose administration. Steady state concentrations were achieved within six days of repeated dose administration. Average half-life ranged from 32 to 41 hours. Gemcabene's primary route of elimination was renal. No significant drug-drug interactions (“DDIs”) were observed with digoxin, a cardiovascular drug for the treatment of atrial fibrillation or statins (atorvastatin, simvastatin and rosuvastatin) used as background therapy in patients with HoFH, HeFH and many SHTG patients.
Gemcabene has been evaluated in ten Company-sponsored Phase 2 trials across a diverse patient population. These trials explored safety, tolerability and efficacy using multiple doses of Gemcabene as monotherapy and in combination with low-, moderate- and high-intensity statins. In Company-sponsored Phase 2 trials, patients treated with Gemcabene were observed to have significantly lowered LDL-C, hsCRP and triglycerides.
Development Plan
NeuroBo is currently exploring additional therapeutic indications for Gemcabene that may strengthen NeuroBo’s pipeline of assets to treat viral diseases, including COVID-19. NeuroBo intends to evaluate Gemcabene as a treatment for COVID-19 both as a stand-alone treatment and in combination with ANA001. NeuroBo intends to pursue the ANA001/Gemcabene combination as a new chemical entity with the FDA which, if successful, could grant NeuroBo extended patent exclusivity.
The historical development of Gemcabene is as follows. In August 2018, Gemphire announced that it had completed and submitted to the FDA the results from its two year rodent carcinogenicity studies. These studies were submitted as part of a request for the FDA to remove the partial clinical hold that prevents human studies of Gemcabene that are greater than six months in duration. In response to its submission, the FDA did not lift the hold, requested that Gemphire provide additional data, including two preclinical studies, namely, a subchronic (13 week) study of Gemcabene in PPARa knock-out mice and a study of Gemcabene in in vitro PPAR transactivation assays using monkey and canine PPAR isoforms and informed Gemphire that an End-of-Phase 2 (EOP2) meeting to reach agreement on the design of
26
Phase 3 registration and long-term safety exposure trials for its target indications in dyslipidemia would not take place until such time, if ever, as the clinical hold is lifted.
In late 2017 and early 2018, Gemphire announced the initiation of two non-Company investigator-initiated proof-of-concept Phase 2 trials in Pediatric Non-Alcoholic Fatty Liver Disease (NAFLD) and in Familial Partial Lipodystrophy Disease (FPLD).
In August 2018, the Data Safety Monitoring Board (DSMB) halted the Pediatric NAFLD trial early due to “unanticipated problems” in the first three patients. Specifically, ALT was increased in 2 of these 3 subjects beyond baseline levels. In addition, all 3 subjects had an increase in liver fat fraction as measured by MRI PDFF. All 6 subjects treated in this study gained weight and had increased TGs during study treatment. These observations are in contrast to the totality of the evidence from other Gemcabene trials. In addition, there was evidence of non-compliance to the dosing regimen and patient non-adherence to dietary and lifestyle guidelines, as well as inconsistencies in biomarkers. The six pediatric patients that were enrolled in the study were followed for a 12 month safety monitoring period post final dose which is now complete. During this follow-on period there were no drug related adverse events reported. There was one serious non-related adverse event of hospitalization of subacute spinal cord infarction/embolism. No deaths or other SAEs were reported.
In June 2019, Gemphire reported topline data from the FPLD trial. Overall Gemcabene treatment resulted in a median change in serum triglycerides (TGs) of –19.6% for the five patients at twelve weeks (the primary endpoint) with a range of TG responses from +40.4% to –52.9% and three patients showing decreases. Gemcabene was generally well tolerated and safe. Nonsignificant fluctuations in ALT, AST, serum creatinine and eGFR were observed. Four of 5 subjects completed the study; one subject withdrew due to an AE of right quadrant pain considered related to Gemcabene. There was one SAE of benign paroxysmal positional vertigo considered unrelated to treatment.
On July 24, 2019, Gemphire announced that it had entered into the Beijing SL License Agreement pursuant to which Gemphire has granted to Beijing SL an exclusive, royalty-bearing license to develop and commercialize products containing Gemcabene for the treatment of any human disease in mainland China, Taiwan, Hong Kong and Macau.
With respect to the partial clinical hold that prevents human studies of Gemcabene that are greater than six months in duration, Gemphire has completed the in vitro PPAR transactivation studies and the subchronic study of Gemcabene in PPARa knock-out mice. In May 2020, we received written communication from the FDA that the clinical development program for Gemcabene remains on a partial clinical hold.
Licensing Agreements
License Agreement with YourChoice
In connection with the acquisition of ANA, we assumed a license agreement (the “YourChoice Agreement”) between ANA and YourChoice Therapeutics, Inc. (“YourChoice”). Pursuant to the YourChoice Agreement, YourChoice granted to ANA, during the term of the YourChoice Agreement, an exclusive, worldwide, fee-bearing license derived from the licensed intellectual property throughout the world. The fees due under the YourChoice Agreement include certain single-digit royalty payments and milestone payments in the aggregate of $19.5 million. The term of the YourChoice Agreement will expire on the expiration or invalidation of the last of the licensed patents under the YourChoice Agreement.
License Agreement with Dong-A ST for NB-01
On January 18, 2018, we entered into an exclusive license agreement with Dong-A ST, a leading pharmaceutical company specializing in the discovery, development, manufacture and marketing of pharmaceutical products and biosimilars, which agreement was amended on April 18, 2018 and July 24, 2019. Dong-A ST is headquartered in Seoul, South Korea and listed on the Korean stock exchange. Under the terms of the agreement, we obtained an exclusive, royalty-bearing, worldwide (except for the Republic of Korea) license to make, use, offer to sell, sell and import products covered by certain Dong-A ST intellectual property rights in its proprietary compound designated as DA-9801 (NB-01). Our license rights cover any and all applications and markets for the therapeutic, health, nutrition or well-being
27
of humans. We may grant sublicenses to any affiliate or third party. We are responsible for all future patent prosecution costs.
Dong-A ST retained the exclusive right to conduct clinical studies in the Republic of Korea and sell products to end users in Korea. NeuroBo grants Dong-A ST an exclusive, royalty free right and license to use, solely for Dong-A ST's commercialization of products in Korea, any inventions, designs and technology developed by us in its performance of the agreement. If Dong-A ST terminates the agreement due to a breach by us or bankruptcy event, then this technology is licensed exclusively to Dong-A ST at no charge. We will also negotiate in good faith to supply product to Dong-A ST for clinical studies and sale of products to end-users in Korea under a separate supply agreement.
We are obligated to use commercially reasonable efforts to develop products for use in each of the United States, the European Union, Japan and the People's Republic of China. If we terminate, discontinue or suspend, for longer than 12 months, the development of any product listed as a product under development in any development plan provided to Dong-A ST (other than for reasons of force majeure or requirements of applicable law), then we are deemed in breach of this development obligation, and Dong-A ST may terminate for cause after a 60-day cure period. We are obligated to use commercially reasonable efforts to commercialize products worldwide throughout the term of the agreement.
In connection with obtaining the licenses we paid Dong-A ST total consideration of $2 million consisting of a one-time upfront license fee and shares of common stock.
We may be required to pay development milestone payments of up to an aggregate of $98 million related to publication of Phase 3 clinical trial data, the first NDA submission in any country, and NDA approval in the United States, the European Union, Japan and the People's Republic of China. We may also be required to pay sales milestone payments in a specified amount, related to the first time that aggregate net sales of products exceed specified amounts in a calendar year.
We are required to pay Dong-A ST commercial milestone payments of up to an aggregate of $80 million and a royalty between a single digit and a low double digit percentage of net sales of products. The royalty rate increases as annual net sales increase.
The term of the agreement continues on a country-by country and product-by-product basis until the later of the 12th anniversary of the first commercial sale of such product in such country or expiration or termination of the last valid claim within the patent rights covering the product. The royalty rate is then reduced by 30% in any country that prohibits the payment of royalties on a patent license beyond the expiration or invalidation of the last valid claim covering the product.
Either Dong-A ST or we may terminate the agreement if the other party is in material breach of the agreement and has not cured or started to cure the breach within 60 days of notice of such breach, or is subject to a bankruptcy or insolvency event. We may terminate the agreement at any time upon 90 days’ written notice.
We may assign our rights under the agreement in connection with a merger, consolidation, or sale of substantially all of its assets, with prior written notice to Dong-A ST, and if the successor entity agrees in writing to be bound by the agreement.
Pfizer License Agreement
In August 2018, an Amended and Restated License Agreement with Pfizer (the “Pfizer Agreement”) for the research, development, manufacture and commercialization of Gemcabene went into effect. This agreement amended and restated in full the prior license agreement with Pfizer dated April 16, 2011.
The Pfizer Agreement included milestone payments to Pfizer totaling up to $37 million upon the achievement of certain milestones, including the first NDA (or its foreign equivalent) in any country, regulatory approval in each of the United States, Europe and Japan, the first anniversary of the first regulatory approval in any country, and upon achieving certain aggregate sales levels of Gemcabene. Future milestone payments under the Pfizer Agreement, if any, would not be expected to begin for at least several years and extend over a number of subsequent years.
28
Pfizer will also receive tiered royalties on a country-by-country basis based upon the annual amount of net sales as specified in the Pfizer Agreement until the later of: (i) five years after the first commercial sale in such country; (ii) the expiration of all regulatory or data exclusivity for Gemcabene in such country; and (iii) the expiration or abandonment of the last valid claim of the licensed patents, including any patent term extensions or supplemental protection certificates in such country. The royalty rates range from the high single digits to the mid-teens depending on the level of net sales. The royalty rates are subject to reduction during certain periods when therapeutically-equivalent generic products represent a certain market share of prescription volume in the country. Under the Pfizer Agreement, commercially reasonable efforts must be used to develop and commercialize Gemcabene.
The Pfizer Agreement will expire upon expiration of the last royalty term. On expiration (but not earlier termination), we will have a perpetual, exclusive, fully paid-up, royalty-free license under the licensed patent rights and related data to make, use, develop, commercialize, import and otherwise exploit the clinical product candidate Gemcabene. Either party may terminate the Pfizer Agreement for the other party's material breach following a cure period or immediately upon certain insolvency events relating to the other party. Pfizer may immediately terminate the Pfizer Agreement in the event that (i) we or any of our affiliates or sublicenses contests or challenges, or supports or assists any third party to contest or challenge, Pfizer's ownership of or rights in, or the validity, enforceability or scope of any of the patents licensed under the Pfizer Agreement or (ii) we or any of our affiliates or sublicensees fails to achieve the first commercial sale in at least one country by April 16, 2024.
License Agreement with Beijing SL
As of July 23, 2019, Beijing SL has an exclusive royalty-bearing license to research, develop, manufacture and commercialize pharmaceutical products comprising, as an active ingredient, Gemcabene in the territory comprised of mainland China, Hong Kong, Macau and Taiwan. We retain all rights to Gemcabene outside of the territory. The parties have agreed to collaborate with respect to development and commercialization activities under the Beijing SL License Agreement through a joint steering committee composed of an equal number of representatives of Beijing SL and us.
Beijing SL will be responsible, at its expense, for developing and commercializing products containing Gemcabene in the territory, with certain assistance from us. To the extent mutually agreed to in writing, the parties will collaborate on the Phase 3 clinical trial for HoFH or other clinical trials, with us as the sponsor, and designed to enroll patients both inside and outside the territory, but Beijing SL will be responsible, at its expense, for the conduct of any such study to the extent solely in the territory. Beijing SL will be responsible for development activities, including non-clinical and clinical studies directed at obtaining regulatory approval of the licensed product in the territory. Beijing SL has agreed to use commercially reasonable efforts to commercialize the licensed products for each indication that receives regulatory approval in the territory and shall prepare and present a commercialization plan that shall be subject to approval by the joint steering committee.
Pursuant to the Beijing SL License Agreement, Beijing SL made an upfront gross payment of $2.5 million. Additionally, with respect to each licensed product, Beijing SL will pay (i) payments for specified developmental and regulatory milestones (including submission of a NDA to China's National Medical Product Administration, dosing of the first patient in a Phase 3 clinical trial in mainland China and regulatory approval for the first and each additional indication of a Licensed Product in the Territory) totaling up to $6 million in the aggregate and (ii) payments for specified global net sales milestones of up to $20 million in the aggregate multiplied by the ratio of the net sales of a licensed product divided by the global net sales of a licensed product, which net sales milestone payments are payable once, upon the first achievement of such milestone.
Beijing SL will also be obligated to pay tiered royalties ranging from the mid-teens to twenty percent on the net sales of all licensed products in the territory until the latest of (a) the date on which any applicable regulatory exclusivity with respect to such Licensed Product expires in such region, (b) the expiration or abandonment of the last valid patent claim or joint patent claim covering such Licensed Product in each region and (c) the fifth anniversary of the first commercial sale of such Licensed Product in such region. Future milestone payments under the Beijing SL License Agreement, if any, are not expected to begin for at least one year and will extend over a number of subsequent years.
Either party may terminate the Beijing SL License Agreement (x) with written notice for the other party's material breach following a cure period or (y) if the other party becomes subject to certain insolvency proceedings. In addition,
29
we may terminate the Beijing SL License Agreement in its entirety if Beijing SL or its affiliates or sublicensees commence a proceeding challenging the validity, enforceability or scope of any of our patents.
The Beijing SL License Agreement contemplates that parties shall, no later than twelve months prior to the anticipated date of the first commercial sale of a licensed product, if any, negotiate in good faith and execute a commercial supply agreement, pursuant to which Beijing SL shall purchase from us, and we shall use commercially reasonable efforts to supply, Gemcabene or licensed product for clinical or commercial purposes, as applicable, until manufacturing and regulatory transfers are complete.
ANA001 is a small molecule drug candidate that can be synthesized using readily available raw materials and based on conventional chemical processes. Contract manufacturers produce both the drug substance and drug product required for the preclinical studies and clinical trials of ANA001. All of the contract manufacturers have updated GMP certificates and all of the drug products are being manufactured under current good manufacturing practices (GMP), a quality system regulating CMC activities.
ANA001 capsules are manufactured under GMP to support all clinical trials. More specifically, drug substance and drug product manufacturing process and analytical method development have been optimized and updated based on ICH/FDA guidelines. There is solid stability data for both the drug substance and drug product. The current contract manufacturers have been producing, and could produce in the future, bulk drug substance and drug product for use in our preclinical studies and clinical trials on a purchase order basis.
NB-01
NB-01 is derived from two plant species native to China, Dioscorea Rhizome and Dioscoreae Nipponicae Rhizoma. Both species have been previously used in traditional Chinese medicine (TCM) for the treatment of arthritis-related pain, muscular pain and pain related to other conditions such as Kashin-Beck disease. Traditional Chinese medicine (TCM) is a style of traditional medicine built on a foundation of more than 2,500 years of Chinese medical practice that includes various forms of herbal medicine, acupuncture, massage (tui na), exercise (qigong), and dietary therapy.
While the characterization of the full composition of NB-01 and underlying active compounds is underway, certain compounds have been identified for purposes of product screening and quality control. These include allantoin and dioscin, the chemical structures for which are shown in the figure below. Allantoin is a marker of the D. Rhizome extract and dioscin is a marker of the D. Nipponicae Rhizoma extract. Signature high-performance liquid chromatography (HPLC) chemical profile assays are established for both markers. These markers are used to show the drug quality profile during the manufacturing of the drug extract from the plant species and the final drug product formulation used in the human clinical studies.

NB-01 is manufactured in a highly monitored and controlled manner to ensure rigorous batch-to-batch consistency that yields a complex mixture of active compounds. NB-01 is considered a "botanical drug product" by the FDA, which defines this class of products to include plant materials, algae, macroscopic fungi, and combinations thereof. As a result, it has unique features that must be taken into account during the drug development process. Plant species used for the production of our compounds are cultivated on dedicated, Good Agricultural Practices (GAP)-compliant acreage in accordance with established WHO standards for starting materials of plant or herbal origin, as recommended by FDA its guidelines for botanical drug development. Production of the drug substance from the botanical raw material involves modern harvesting and extraction processes incorporating state-of-the-art molecular biology and analytical chemistry methodologies.
30
The manufacturing process and analytical testing methodologies have been validated and the adherence to regulatory requirements of the processes have been audited by two firms, Amarex and FDAMap, well-experienced in the review and audit of botanical drug requirements of the FDA. The drug substance, an ethanol extract of the two plant species, combined in a specific weight ratio, is manufactured in KGC Yebon, in South Korea in a GMP-compliant process, and has been audited by Amarex and FDAMap. The drug substance has completed process validation and analysis method validation, and demonstrated 36-month stability. The drug product is manufactured by Dong-A ST in South Korea in a GMP-compliant process, and is audited by Amarex and FDAMap. The final drug product has completed process validation and analysis method validation, and demonstrated 36-month stability.
NB-02
NB-02 is derived from two plant materials, Morus alba Linne and the peel of Poria cocos Wolf. NB-02 is manufactured in a highly monitored and controlled manner to ensure rigorous batch-to-batch consistency that yields a complex mixture of active compounds. NB-02 is considered a "botanical drug product" by the FDA, which defines this class of products to include plant materials, algae, macroscopic fungi, and combinations thereof. As a result, NB-02 has unique features that must be taken into account during the drug development process. Plant species used for the production of our compounds are cultivated on dedicated, GAP-compliant acreage in accordance with established WHO standards for starting materials of plant or herbal origin, as recommended by FDA its guidelines for botanical drug development. Production of the drug substance from the botanical raw material involves modern harvesting and extraction processes incorporating state-of-the-art molecular biology and analytical chemistry methodologies.
Gemcabene
Gemcabene is a small molecule drug candidate that can be synthesized as a single polymorph crystalline monocalcium salt, using readily available raw materials and based on conventional chemical processes. Contract manufacturers produce both the drug substance and drug product required for the preclinical studies and clinical trials of Gemcabene. All of the contract manufacturers have updated GMP certificates and all of the drug products are being manufactured under current good manufacturing practices (GMP), a quality system regulating CMC activities.
Gemcabene Immediate Release (IR) tablets are manufactured under GMP to support all clinical trials. More specifically, drug substance and drug product manufacturing process and analytical method development have been optimized and updated based on ICH/FDA guidelines. In addition, Gemcabene is successfully manufactured in multiple strengths of tablets under GMP: 150mg, 300mg, and 600mg. There is solid stability data for both the drug substance and drug product. The current contract manufacturers have been producing, and could produce in the future, bulk drug substance and drug product for use in our preclinical studies and clinical trials on a purchase order basis.
Competition
The pharmaceutical and biotechnology industries are characterized by rapidly advancing technologies, intense competition and a strong emphasis on proprietary products. We face potential competition from many different sources, including major pharmaceutical, specialty pharmaceutical and biotechnology companies, academic institutions, governmental agencies and public and private research institutions. Any product candidates that we successfully develop and commercialize will compete with existing therapies and new therapies that may become available in the future.
Some of our competitors may have significantly greater financial resources and expertise in research and development, manufacturing, preclinical testing, conducting clinical trials, obtaining regulatory approvals and marketing approved products than we do. Other firms may also compete with us in recruiting and retaining qualified scientific and management personnel and establishing clinical trial sites and patient enrollment for clinical trials, as well as in acquiring technologies complementary to, or necessary for our programs. Mergers and acquisitions in the pharmaceutical, biotechnology and diagnostic industries may result in even more resources being concentrated among a smaller number of our competitors. Smaller or early-stage companies may also prove to be significant competitors with us, particularly through collaborative arrangements with large and established companies.
Our commercial opportunity could be reduced or eliminated if our competitors develop and commercialize therapeutics that are safer, more effective, have fewer or less severe side effects, are more convenient or are less expensive than any
31
products that we may develop. Our competitors also may obtain marketing approvals for their products more rapidly than we may obtain approval for our products, which could result in our competitors establishing a strong market position before we are able to enter the market. In addition, our ability to compete may be affected because in some cases insurers or other third-party payors, including government programs, seek to encourage the use of generic products. This may have the effect of making branded products less attractive, from a cost perspective, to buyers.
ANA001 and Gemcabene—COVID-19
We expect that, if approved, ANA001 and/or Gemcabene will compete with a number of drugs that are being studied for the treatment of symptoms of COVID-19. Two therapies are currently approved by the FDA for the treatment of symptoms of COVID-19 (remdesivir and Dexamethasone), and three have received EUA from the FDA (baricitinib + remdesivir, Regeneron’s antibody cocktail and bamlanivimab). We are aware of other therapies currently being studied in clinical trials for the treatment of COVID-19, including favipiravir, convalescent plasma, oleandrin, ivermectin and molnupiravir.
With respect to ANA001, in addition to the marketed therapies, we are aware of several companies currently developing and commercializing niclosamide for the treatment of COVID-19 symptoms, including Daewoong, Union Therapeutics, TFF and FirstWave.
NB-01—Painful Diabetic Neuropathy
We expect that, if approved, NB-01 will compete with currently approved drug therapies for painful diabetic neuropathy, including pregabalin, duloxetine, and tapentadol HCl. We are also aware of a number of therapies that are approved to treat other types of neuropathic pain, and that various therapies are used off-label to treat neuropathic pain. In addition to the marketed therapies, we are aware of several companies currently developing therapies for neuropathic pain, including Biogen Inc., Cara Therapeutics, Inc., Daiichi Sankyo Company, Eliem Therapeutics Inc, Immune Pharmaceuticals Inc., Novartis AG, and Xenoport Inc.
NB-02—Cognitive disease and Tauopathies
We expect that, if approved, NB-02 will compete with the currently approved therapies for management of cognitive disease including Alzheimer's disease. In Alzheimer's disease, four drugs are currently approved by the FDA for the treatment of symptoms of Alzheimer's disease, based on AChE inhibition (three drugs) and NMDA receptor antagonism (one drug). In addition to the marketed therapies, we are aware of several companies currently developing therapies for Alzheimer's disease, including Eisai Co., Ltd., Hoffman-LaRoche, Otsuka Pharmaceuticals, Inc., Novartis AG, and Avanir Pharmaceuticals, and Biohaven Pharmaceuticals.
Gemcabene —Dyslipidemia
We expect that, assuming we re-engage our effort to develop Gemcabene as a cardiovascular therapy, if approved, Gemcabene will compete with large pharmaceutical and biopharmaceutical companies, specialty pharmaceutical and generic drug companies, academic institutions, government agencies and research institutions. We are aware of other therapies approved to lower LDL, both statin and non-statin based therapies, as well as medications written off label to treat the disease. In addition, we are aware of other therapies to lower triglycerides. Lipid-lowering therapies currently on the market that would compete with Gemcabene, if approved, include the following:
| ● | | statins, such as Crestor marketed by AstraZeneca, Livalo marketed by Kowa Pharmaceuticals America, Inc. (Kowa), Zocor marketed by Merck & Co., Inc. (Merck), Lipitor marketed by Pfizer, and their generic versions; |
| ● | | cholesterol absorption inhibitors, such as Zetia, marketed by Merck; |
| ● | | apoB antisense Kynamro marketed by Genzyme Corporation, a Sanofi company, and MTTP inhibitor Juxtapid marketed by Aegerion Pharmaceuticals, Inc.; |
| ● | | combination therapies, such as Vytorin and Liptruzet, both marketed by Merck; |
32
| ● | | other lipid-lowering monotherapies and fixed dose combinations, including: fibrates, such as TriCor and Trilipix, both marketed by AbbVie Inc. (AbbVie), and Lipofen marketed by Kowa; niacin, such as Niaspan marketed by AbbVie; bile acid sequestrants, such as Welchol, marketed by Daiichi Sankyo Inc.; combination therapies, such as Advicor and Simcor, both of which are marketed by AbbVie; Pemafibrate (PPARalpha agonist) being marketed by Kowa; and the generic versions of these drugs; Nexletol and Nexlizet marketed by Esperion; |
| ● | | triglyceride lowering therapies including: prescription fish oils, such as Lovaza marketed by GlaxoSmithKline, Epanova marketed by AstraZeneca and Vascepa marketed by Amarin Corporation plc; Waylivra marketed by Akcea; |
| ● | | PCSK9 inhibitors, such as Praluent, developed by Sanofi-Aventis U.S. LLC, and Regeneron Pharmaceuticals, Inc. and Repatha marketed by Amgen Inc; and |
| ● | | anti-inflammatory agents such as canakinumab, developed by Novartis. |
Several other pharmaceutical companies have other lipid-lowering therapies in development that may be approved for marketing in the United States or outside of the United States. Based on publicly available information, we believe the current therapies in development that would compete with Gemcabene include, but are not limited to, the following:
| ● | | for HoFH, RGEN-1500 being developed by Regeneron Pharmaceuticals, Inc. MGL-3196 developed by Madrigal Pharmaceuticals (Madrigal) for HoFH, and ALN-PCSsc being developed by The Medicines Company and Alnylam Pharmaceuticals, Inc.; |
| ● | | for HeFH and ASCVD, drugs include: oral cholesteryl ester transfer protein inhibitors, such as anacetrapib being developed by Merck and TA-8995 being developed by Amgen/Dezima; ATP citrate lyase inhibitor, ETC-1002 developed by current Esperion; PCSK9 inhibitors, such as ALN-PCSsc (inclisiran) being developed by Novartis; apoA antisense agent AKCEA-APO(a)-LRx being developed by Akcea and Novartis; apabetalone (RVX-208) being developed by Resvelogix; and MGL-3196 developed by Madrigal (HeFH only); and |
| ● | | for SHTG, AKCEA-APOCIII-LRx and AKCEA-ANGPTL3-LRx both being developed by Akcea Pharmaceuticals, Inc.; Evinacumab ANGPTL-3 in development by Regeneron; CaPre (long-chain omega-3 phospholipid) being developed by Acasti; and pemafibrate being developed by KOWA. |
Intellectual Property
The term of individual patents depends upon the legal term of the patents in the countries in which they are obtained. In most countries, including the United States, the patent term is 20 years from the earliest filing date of a non-provisional patent application or a Patent Cooperation Treaty (PCT) application to which a U.S. application claims priority. In the United States, a patent's term may be lengthened by patent term adjustment, which compensates a patentee for administrative delays by the U.S. Patent and Trademark Office, or the USPTO, in examining and granting a patent, or may be shortened if a patent is terminally disclaimed over an earlier filed patent. The term of a U.S. patent that covers a drug or biological product may also be eligible for patent term extension when approval from the FDA is granted, provided statutory and regulatory requirements are met. In the future, our product candidates receive approval from the FDA or foreign regulatory authorities, we expect to apply for patent term extensions on issued patents covering those products, depending upon the length of the clinical trials for each drug and/or other factors. There can be no assurance that any of our pending patent applications will issue or that we will benefit from any patent term extension or other favorable adjustment to the term of any of its patents.
As with other biotechnology and pharmaceutical companies, our ability to maintain and solidify our proprietary and intellectual property position for our product candidates, including NB-01 and NB-02, its preclinical compounds, and its core technologies will depend on our success in obtaining effective patent claims and enforcing those claims if granted. However, patent applications that we may file or license from third parties may not result in the issuance of patents. We also cannot predict the breadth of claims that may be allowed or enforced in our patents. Any issued patents that we may receive in the future may be challenged, invalidated or circumvented. For example, prior to March 16, 2013, in the United States, patent applications were subject to a “first to invent” rule of law. Applications effectively filed on or after March 16, 2013, are subject to a “first to file” rule of law.
33
Discoveries reported in the scientific literature often lag the actual discoveries, and patent applications in the United States and other jurisdictions are typically not published until 18 months after filing, or in some cases not at all. We cannot be certain that any existing application will be subject to the “first to file” or “first to invent” rule of law, that we or our licensor were the first to make the inventions claimed in our existing patent portfolio subject to the prior laws, or that we or our licensor were the first to file for patent protection of such inventions subject to the new laws. If third parties prepare and file patent applications in the United States that also claim technology we have claimed in our patents or patent applications, we may have to participate in interference or derivation proceedings and/or invalidation proceedings in the USPTO, which could result in substantial costs to us, even if the eventual outcome is favorable. In addition, because of the extensive time required for clinical development and regulatory review of a product candidate we may develop, it is possible that, before any of our product candidates can be commercialized, any related patent may expire or remain in force for only a short period following commercialization, thereby reducing any advantage of any such patent.
In addition to patents, we rely upon unpatented trade secrets, know-how, and continuing technological innovation to develop and maintain its competitive position. We seek to protect our proprietary information, in part, by using confidentiality agreements with our collaborators, scientific advisors, employees and consultants, and invention assignment agreements with its employees. We also have agreements requiring assignment of inventions with selected consultants, scientific advisors and collaborators. The confidentiality agreements are designed to protect our proprietary information and, in the case of agreements or clauses requiring invention assignment, to grant us ownership of technologies that are developed under those agreements.
Our ability to commercialize product candidates depends in large part on our ability to obtain and maintain intellectual property protection for our product candidates. Our policy is to seek to protect our intellectual property position by, among other methods, filing U.S. and foreign patent applications related to the technology, inventions and improvements that are important to the development and implementation of our business strategy. We also rely on trade secrets, know-how and continuing technological innovation to develop and maintain our proprietary position.
ANA001
As of December 31, 2020, our intellectual property portfolio for ANA001 included four U.S. provisional applications directed to niclosamide formulation. A PCT application and/or a non-provisional U.S. application in the U.S. to which claim priority to the U.S. provisional applications may be filed in 2021. Patent applications may be issued in the U.S. and any countries in which the Company files national phase applications of the PCT application. The patents issued from the national phase applications are estimated to expire 2041.
As described in more detail above under “Licensing Agreements – License Agreement with YourChoice,” pursuant to the YourChoice Agreement, the Company has licensed several patent applications relating to ANA from YourChoice. A PCT application to which claims priority to the U.S. provisional applications was filed in 2021. Patent applications may be issued in any countries in which the Company files national phase applications of the PCT application. The patents issued from the national phase applications are estimated to expire 2041.
NB-01 and NB-02
As of December 31, 2020, our intellectual property portfolio for NB-01 included four issued U.S. patents, comprised of one patent directed to composition of matter and three patents directed to use, and two pending U.S. non-provisional patent applications, comprised of one directed to composition of matter and another directed to use, and 65 granted foreign patents and one pending application, these patents are related to its NB-01 clinical programs in peripheral neuropathy and neurological conditions. The issued patents have expiration dates ranging from October 27, 2026 to June 22, 2033. The patent issuing from the application, if any, is expected to expire December 2031. The jurisdictions for the foreign patents and application include: Brazil, Canada, China, the European Patent Convention (including Austria, Belgium, Finland, France, Germany, Greece, Hungary, Italy, Netherlands, Poland, Portugal, Romania, Spain, Switzerland, Turkey, and the United Kingdom), India, Japan, Mexico, the Republic of Korea, and Russia. One patent family including some of the above patents for NB-01 is assigned to University-Industry Cooperation Group of Kyung Hee University, and is exclusively licensed from Kyung Hee University to Dong-A ST and then from Dong-A ST to us pursuant to the terms of the corresponding agreements. The other two patent families including the other above patents and patent applications for NB-01 are assigned to Dong-A ST and exclusively licensed to us.
34
As of December 31, 2020, our intellectual property portfolio for NB-02 included two issued U.S. patents, two pending U.S. non-provisional patent applications, 24 foreign granted patents, and 9 foreign patent applications. Patents issuing from these applications, if any, are expected to expire around 2035. The issued patents have an expiration date of December 3, 2035 and December 19, 2035. The jurisdictions for the foreign patents and applications include: Brazil, Canada, China, the European Patent Convention (including Austria, Belgium, Finland, France, Germany, Greece, Hungary, Italy, Netherlands, Poland, Portugal, Romania, Spain, Switzerland, Turkey, and the United Kingdom), India, Japan, Mexico, the Republic of Korea, and Russia. All of the above patents and patent applications for NB-02 were assigned to us.
Gemcabene
As of March 22, 2021, our intellectual property portfolio relating to Gemcabene included eight issued U.S. patents, seven pending U.S. patent applications, 36 foreign-granted patents and 46 foreign patent applications directed to formulations, compositions, methods of use and methods of manufacturing. The Gemcabene intellectual property includes both owned and Pfizer-licensed issued and pending patents in the United States and foreign jurisdictions. The issued patents in the United States and foreign countries have expiration dates between July 2021 and November 2036. The patents in the United States and foreign countries that may be issued from pending applications, if any, are expected to expire between December 2031 and October 2039. The jurisdictions for the foreign countries include Argentina, Australia, Brazil, Canada, China, Europe, Hong Kong, India, Israel, Japan, Mexico, New Zealand, Philippines, Korea, Russia, Singapore, South Africa, Taiwan and Thailand.
Government Regulation
Government authorities at the federal, state and local level in the United States and in other countries extensively regulate, among other things, the research, development, testing, manufacture (including any manufacturing changes), packaging, storage, recordkeeping, labeling, advertising, promotion, distribution, marketing, post-approval monitoring and reporting, import and export of pharmaceutical products, such as those we are developing.
United States — FDA Regulation
In the United States, pharmaceutical products are subject to extensive regulation by the FDA. The Federal Food, Drug, and Cosmetic Act, or the FDCA, and other federal and state statutes and regulations, govern, among other things, the research, development, testing, manufacture, storage, recordkeeping, approval, labeling, promotion and marketing, distribution, post-approval monitoring and reporting, sampling, and import and export of pharmaceutical products. Failure to comply with applicable U.S. requirements may subject a company to a variety of administrative or judicial sanctions, such as imposition of clinical holds, refusal by the FDA to approve pending NDAs, warning letters, product recalls, product seizures, total or partial suspension of production or distribution, injunctions, fines, refusals of government contracts, restitution, disgorgement, civil penalties and criminal prosecution.
Pharmaceutical product development in the United States typically involves preclinical or other nonclinical laboratory and animal tests and the submission to the FDA of an Investigational New Drug (IND) application, which must become effective before clinical testing may commence. For commercial approval, the sponsor must submit adequate tests by all methods reasonably applicable to show that the drug is safe for use under the conditions prescribed, recommended or suggested in the proposed labeling. The sponsor must also submit substantial evidence, generally consisting of adequate, well-controlled clinical trials to establish that the drug will have the effect it purports or is represented to have under the conditions of use prescribed, recommended or suggested in the proposed labeling. In certain cases, the FDA may determine that a drug is effective based on one clinical study plus confirmatory evidence. Satisfaction of the FDA pre-market approval requirements typically takes many years and the actual time required may vary substantially based upon the type, complexity and novelty of the product or disease. For botanical drug products in particular, which may be heterogeneous in nature and may carry additional uncertainty about their active constituents in comparison to synthetic small-molecule drug products, one of the critical issues during drug development is ensuring that the therapeutic effect for marketed drug product batches is consistent. FDA has determined that therapeutic consistency can generally be supported by a "totality of the evidence" approach, which the agency has outlined in a 2016 guidance for industry entitled Botanical Drug Development.
Nonclinical tests include laboratory evaluation of product chemistry, formulation and toxicity, as well as animal studies to assess the characteristics and potential safety and efficacy of the product. The conduct of the nonclinical tests must
35
comply with federal requirements, including the FDA's good laboratory practice regulations and the U.S. Department of Agriculture's, or USDA's, regulations implementing the Animal Welfare Act. The results of nonclinical testing are submitted to the FDA as part of an IND along with other information, including information about product chemistry, manufacturing and controls, and a proposed clinical trial protocol. Long-term nonclinical tests, such as animal studies of reproductive toxicity and carcinogenicity, may continue after the IND is submitted.
A 30-day waiting period after the submission of each IND is required prior to the commencement of clinical testing in humans. If the FDA has not imposed a clinical hold on the IND or otherwise commented or questioned the IND within this 30-day period, the clinical trial proposed in the IND may begin.
Clinical trials involve the administration of the investigational new drug to healthy volunteers or patients under the supervision of a qualified investigator. Clinical trials must be conducted: (i) in compliance with federal regulations, (ii) in compliance with good clinical practice (“GCP”), an international standard meant to protect the rights and health of patients and to define the roles of clinical trial sponsors, administrators and monitors (some of which have been codified into U.S. federal regulations), and (iii) under protocols detailing the objectives of the trial, the parameters to be used in monitoring safety and the effectiveness criteria to be evaluated. Each protocol involving testing on U.S. patients and subsequent protocol amendments must be submitted to the FDA as part of the IND.
The FDA may order the temporary, or permanent, discontinuation of a clinical trial at any time or impose other sanctions if it believes that the clinical trial either is not being conducted in accordance with the FDA requirements or presents an unacceptable risk to the clinical trial patients. The trial protocol and informed consent information for patients in clinical trials must also be submitted to an institutional review board, or IRB, at each site where a trial will be conducted for approval. An IRB may also require the clinical trial at the site to be halted, either temporarily or permanently, for failure to comply with the IRB's requirements or may impose other conditions. Clinical trials to support NDAs for marketing approval are typically conducted in three sequential phases, but the phases may overlap. In general, in Phase I, the initial introduction of the drug into healthy human volunteers or, in some cases, patients, the drug is tested to assess metabolism, pharmacokinetics, pharmacological actions, side effects associated with increasing doses and, if possible, early evidence of effectiveness. Phase 2 usually involves trials in a limited patient population to determine the effectiveness of the drug for a particular indication, dosage tolerance and optimum dosage, and to identify common adverse effects and safety risks. If a compound demonstrates evidence of effectiveness and an acceptable safety profile in Phase 2 evaluations, Phase 3 trials are undertaken to obtain the additional information about clinical efficacy and safety in a larger number of patients, typically at geographically dispersed clinical trial sites, to permit the FDA to evaluate the overall benefit-risk relationship of the drug and to provide adequate information for the labeling of the drug. In most cases, the FDA requires two adequate and well-controlled Phase 3 clinical trials to demonstrate the efficacy of the drug. The FDA may, however, determine that a drug is effective based on one clinical trial plus confirmatory evidence. Only a small percentage of investigational drugs complete all three phases and obtain marketing approval. In some cases, the FDA may require post-market studies, known as Phase 4 studies, to be conducted as a condition of approval to gather additional information on the drug's effect in various populations and any side effects associated with long-term use. Depending on the risks posed by the drugs, other post-market requirements may be imposed.
After completion of the required clinical testing, an NDA is prepared and submitted to the FDA. FDA approval of the NDA is required before marketing of the product may begin in the United States. The NDA must include the results of all preclinical, clinical, and other testing and a compilation of data relating to the product's pharmacology, chemistry, manufacture, and controls. The cost of preparing and submitting an NDA is substantial. Under federal law, the submission of most NDAs is additionally subject to a substantial application user fee.
The FDA has 60 days from its receipt of an NDA to determine whether the application will be accepted for filing based on the agency's threshold determination that it is sufficiently complete to permit substantive review. Once the submission is accepted for filing, the FDA begins an in-depth review. Under the statute and implementing regulations, the FDA has 180 days (the initial review cycle) from the date of filing to issue either an approval letter or a complete response letter, unless the review period is adjusted by mutual agreement between the FDA and the applicant or as a result of the applicant submitting a major amendment. In practice, the performance goals established pursuant to the Prescription Drug User Fee Act have effectively extended the initial review cycle beyond 180 days. The FDA's current performance goals call for the FDA to complete review of 90% of standard (non-priority) NDAs within 10 months of receipt and within six months for priority NDAs, but two additional months are added to standard and priority NDAs for
36
a new molecular entity, or NME, such that the 10-month and 6-month action goals for NME applications begin to run from the 60-day filing date rather than from receipt of the original NDA submission.
The FDA may also refer applications for novel drug products, or drug products that present difficult questions of safety or efficacy, to an advisory committee, which is typically a panel that includes clinicians and other experts, for review, evaluation and a recommendation as to whether the application should be approved. The FDA is not bound by the recommendation of an advisory committee, but it generally follows such recommendations. Before approving an NDA, the FDA will typically inspect one or more clinical sites to assure compliance with GCP. Additionally, the FDA will inspect the facility or the facilities at which the drug is manufactured. The FDA will not approve the product unless compliance with current good manufacturing practice (GMP) regulations is satisfactory and the NDA contains data that provide substantial evidence that the drug is safe and effective in the indication studied.
After the FDA evaluates the NDA and the manufacturing facilities, it issues either an approval letter or a complete response letter. A complete response letter (CRL) generally outlines the deficiencies in the submission and may require substantial additional testing, or information, in order for the FDA to reconsider the application. If, or when, those deficiencies have been addressed to the FDA's satisfaction in a resubmission of the NDA, the FDA will issue an approval letter. The FDA has committed to reviewing 90% of NDA resubmissions within two to six months depending on the type of information included in response to the deficiencies identified in the CRL.
An approval letter authorizes commercial marketing of the drug with specific prescribing information for specific indications. As a condition of NDA approval, the FDA may require a risk evaluation and mitigation strategy, or REMS, to help ensure that the benefits of the drug outweigh the potential risks. REMS can include medication guides, communication plans for health care professionals, and/or elements to assure safe use, or ETASU. ETASU can include, but are not limited to, special training or certification for prescribing or dispensing, dispensing only under certain circumstances, special monitoring, and the use of patient registries. The requirement for a REMS can materially affect the potential market and profitability of the drug. Moreover, product approval may require substantial post-approval testing and surveillance to monitor the drug's safety or efficacy. Once granted, product approvals may be withdrawn if compliance with regulatory standards is not maintained or problems are identified following initial marketing.
Fast Track Designation and Accelerated Approval
The FDA is authorized to facilitate the development, and expedite the review, of drugs that are intended for the treatment of a serious or life-threatening disease or condition for which there is no effective treatment and which demonstrate the potential to address unmet medical needs for the condition. These programs include fast track designation, breakthrough therapy designation, priority review designation and other accelerated approvals.
Under the Fast Track Program, the sponsor of a new drug candidate that is intended to treat a serious condition may request that the FDA designate the drug candidate for a specific indication as a Fast Track drug concurrent with, or after, the filing of the IND for the drug candidate. The FDA must determine if the drug candidate qualifies for Fast Track designation within 60 days of receipt of the sponsor's request. In addition to other benefits such as the ability to engage in more frequent interactions with the FDA, the FDA may initiate review of sections of a Fast Track drug's NDA before the application is complete. This rolling review is available if the applicant provides, and the FDA approves, a schedule for the submission of the remaining information and the applicant pays applicable user fees. However, the FDA's time period goal for reviewing an application does not begin until the last section of the NDA is submitted. Additionally, the Fast Track designation may be withdrawn by the FDA if the FDA believes that the designation is no longer supported by data emerging in the clinical trial process.
In 2012, Congress enacted the Food and Drug Administration Safety and Innovation Act, or FDASIA. This law established a new regulatory program for products designated as “breakthrough therapies.” A product may be designated as a breakthrough therapy if it is intended, either alone or in combination with one or more other drugs, to treat a serious or life-threatening disease or condition and preliminary clinical evidence indicates that the product may demonstrate substantial improvement over existing therapies on one or more clinically significant endpoints, such as substantial treatment effects observed early in clinical development. The FDA may take certain actions with respect to designated breakthrough therapies, including: holding meetings with the sponsor throughout the development process; providing timely advice to the product sponsor regarding development and approval; involving more senior staff in the review process; assigning a cross-disciplinary project lead for the review team; and taking other steps to design the clinical trials in an efficient manner.
37
The FDA may also designate a product for priority review if it is a drug that treats a serious condition and, if approved, would provide a significant improvement in safety or effectiveness. The FDA determines at the time that the marketing application is submitted, on a case- by-case basis, whether the proposed drug represents a significant improvement when compared with other available therapies. Significant improvement may be illustrated by evidence of increased effectiveness in the treatment of a condition, elimination or substantial reduction of a treatment-limiting drug reaction, documented enhancement of patient compliance that may lead to improvement in serious outcomes, or evidence of safety and effectiveness in a new subpopulation. A priority review designation is intended to direct overall attention and resources to the evaluation of such applications, and to shorten the FDA's goal for taking action on a marketing application from ten months to six months.
Under the FDA’s accelerated approval regulations, the FDA may approve a drug for a serious or life-threatening illness that provides meaningful therapeutic benefit to patients over existing treatments based upon a surrogate endpoint that is reasonably likely to predict clinical benefit, or on a clinical endpoint that can be measured earlier than irreversible morbidity or mortality, that is reasonably likely to predict an effect on irreversible morbidity or mortality or other clinical benefit, taking into account the severity, rarity or prevalence of the condition and the availability or lack of alternative treatments. The accelerated approval regulations are codified within Title 21 of the Code of Federal Regulations, as Subpart H under Part 314, the part of the FDA regulations covering applications for FDA approval to market a new drug, and as such the accelerated approval pathway is sometimes referred to as approval under “Subpart H.”
In clinical trials, a surrogate endpoint is a measurement of laboratory or clinical signs of a disease or condition that substitutes for a direct measurement of how a patient feels, functions or survives. Surrogate endpoints can often be measured more easily or more rapidly than clinical endpoints. A drug candidate approved under Subpart H is subject to rigorous post-marketing compliance requirements, including the completion of Phase 4 or post-approval clinical trials to confirm the effect on the clinical endpoint. Failure to conduct required post-approval studies, or confirm a clinical benefit during post-marketing studies, will allow the FDA to withdraw the drug from the market on an expedited basis. Unless otherwise informed by the FDA, for an accelerated approval product an applicant must submit to the FDA for consideration during the preapproval review period copies of all promotional materials, including promotional labeling as well as advertisements, intended for dissemination or publication within 120 days following marketing approval. After 120 days following marketing approval, unless otherwise informed by the FDA, the applicant must submit promotional materials at least 30 days prior to the intended time of initial dissemination of the labeling or initial publication of the advertisement. The accelerated approval pathway is most often used in settings in which the course of a disease is long and an extended period of time is required to measure the intended clinical benefit of a drug, even if the effect on the surrogate or intermediate clinical endpoint occurs rapidly. For example, accelerated approval has been used extensively in the development and approval of drugs for treatment of a variety of cancers in which the goal of therapy is generally to improve survival or decrease morbidity and the duration of the typical disease course requires lengthy and sometimes large clinical trials to demonstrate a clinical or survival benefit.
Orphan Drugs
Under the Orphan Drug Act, the FDA may grant orphan drug designation to drugs intended to treat a rare disease or condition—generally a disease or condition that affects fewer than 200,000 individuals. The U.S. Orphan Drug Designation must be requested before submitting an NDA. After the FDA grants orphan drug designation, the generic identity and trade name, if any, of the drug and its designated use are disclosed publicly by the FDA. Orphan drug designation does not convey any advantage in, or shorten the duration of, the regulatory review and approval process. The first NDA applicant to receive FDA approval for a particular active ingredient to treat a particular disease with FDA orphan drug designation is entitled to a seven-year exclusive marketing period in the United States for that product, for that indication. During the seven-year exclusivity period, the FDA may not approve any other applications to market the same drug for the same disease, except in limited circumstances, such as a showing of clinical superiority to the product with orphan drug exclusivity. Orphan drug exclusivity does not prevent the FDA from approving a different drug for the same disease or condition, or the same drug for a different disease or condition. Among the other benefits of orphan drug designation are tax credits for certain research and a waiver of the NDA application user fee.
Nutraceutical Regulation
The FDA regulates foods, food additives, drugs and cosmetics. Unlike pharmaceutical drugs and conventional foods, nutraceuticals are regulated as “dietary supplements” under the Dietary Supplement, Health and Education Act of 1994
38
(DSHEA) as a separate regulatory category of food. Before the DSHEA, dietary supplements were subject to the same regulatory requirements as were other foods. DSHEA amended the FDCA to create a new regulatory framework for the safety and labeling of dietary supplements. Under DSHEA, a company is responsible for determining that the dietary supplements it manufactures or distributes are safe and that any representations or claims made about them are substantiated by adequate evidence to show that they are not false or misleading. Dietary supplements do not need approval from FDA before they are marketed. Except in the case of a “new dietary ingredient,” where pre-market review for safety data and other information is required by law, a firm does not have to provide FDA with the evidence it relies on to substantiate safety or effectiveness before or after marketing a product. In addition, there is a requirement for manufacturers to register pursuant to the Bioterrorism Act with FDA before producing or selling supplements. In June 2007, FDA published regulations for Current Good Manufacturing Practices (“cGMP”) for those who manufacture, package, label or hold dietary supplement products. These regulations focus on practices that ensure the identity, purity, quality, strength and composition of dietary supplements.
Congress defined the term “dietary supplement” in DSHEA as “a product (other than tobacco) intended to supplement the diet that bears or contains one or more of the following dietary ingredients: vitamins, minerals, amino acids, herbs or other botanicals; a concentrate, metabolite, constituent, extract or combination of the ingredients listed above.” A dietary supplement is a product taken by mouth that contains a “dietary ingredient” intended to supplement the diet. The “dietary ingredients” in these products may include vitamins, minerals, herbs or other botanicals, amino acids, and substances such as enzymes, organ tissues, glandulars, and metabolites and can also be extracts or concentrates. Dietary supplements are produced in the form of tablets, capsules, softgels, gelcaps, liquids, or powders. Dietary supplements can also be in other forms, such as a nutrition bar, but if they are in another form, information on their label must not represent the product as a conventional food or a sole item of a meal or diet. Regardless of form, DSHEA places dietary supplements in a special category under the general umbrella of “foods,” not drugs, and requires the product to be labeled as a “dietary supplement.”
According to the FDA, a drug is an article intended to diagnose, cure, mitigate, treat or prevent disease. While nutraceuticals are not intended to cure or treat disease, both dietary supplements and drugs are intended to affect the structure or function of the body. Dietary supplements that contain structure/function claims on their labels must bear the disclaimer: "This statement has not been evaluated by the FDA. This product is not intended to diagnose, treat, cure, or prevent any disease." The manufacturer is responsible for ensuring the accuracy and truthfulness of these claims; they are not approved by FDA. Moreover, dietary supplements are supposed to enhance the diet, not be used as a conventional food or as the sole item of a meal or diet, and not supposed to be taken alone as a substitute for any food or medicine.
The DSHEA requires that a manufacturer or distributor notify FDA if it intends to market a dietary supplement in the U.S. that contains a “new dietary ingredient.” The manufacturer and distributor must demonstrate to FDA why the ingredient is reasonably expected to be safe for use in a dietary supplement, unless it has been recognized as a food substance and is present in the food supply. A new dietary ingredient is an ingredient marketed after October 15, 1994. There is no authoritative list of dietary ingredients that were marketed before October 15, 1994. Therefore, manufacturers and distributors are responsible for determining if a dietary ingredient is “new,” and if it is not, for documenting that the dietary supplements its sells, containing the dietary ingredient, were marketed before October 15, 1994. The DSHEA states that the manufacturer is responsible for the safety evaluation of the product. If the dietary supplement contains a new ingredient, the manufacturer must inform FDA that the new ingredient “can reasonably be expected to be safe” within 75 days of going to market. This notice must provide information that supports the manufacturer’s conclusion that the ingredient is safe. It is up to the FDA to prove that a dietary supplement is unsafe after it is marketed.
A dietary supplement is adulterated if, among other things, it or an ingredient in it presents a “significant or unreasonable risk of illness or injury” when used as directed or contains a new ingredient for which there is insufficient information to provide assurance that the ingredient does not present any significant or unreasonable risk of illness or injury. The DSHEA also has labeling requirements for dietary supplements, including requiring information on the label such as (1) name of each ingredient; (2) quantity of each ingredient; (3) total weight of all ingredients, if a blend; (4) identity of the plant part used; (5) the term “Dietary Supplement;” (6) nutritional labelling information (calories, fat, sodium, etc.).
Pediatric Information
Under the Pediatric Research Equity Act (PREA), NDAs or supplements to NDAs must contain data to assess the safety and effectiveness of the drug for the claimed indications in all relevant pediatric subpopulations and to support dosing and administration for each pediatric subpopulation for which the drug is safe and effective. The FDA may grant full or
39
partial waivers for submission of data, as well as deferrals for several reasons, including a finding that the drug is ready for approval for use in adults before pediatric studies are complete or that additional safety or effectiveness data needs to be collected before the pediatric studies begin. Unless otherwise required by regulation, PREA does not apply to any drug for an indication for which orphan designation has been granted.
The Best Pharmaceuticals for Children Act (BPCA) provides NDA holders a six-month extension of any exclusivity—patent or non-patent—for a drug if certain conditions are met. Conditions for exclusivity include the FDA's determination that information relating to the use of a new drug in the pediatric population may produce health benefits in that population, the FDA making a written request for pediatric studies, and the applicant agreeing to perform, and reporting on, the requested studies within the statutory timeframe. Applications under the BPCA are treated as priority applications, with all of the benefits that designation confers.
Special Protocol Assessment
A company may reach an agreement with the FDA under the Special Protocol Assessment, or SPA, process as to the required design and size of clinical trials intended to form the primary basis of an efficacy claim for a new drug product. According to its performance goals, the FDA seeks to evaluate the protocol within 45 days of the request to assess whether the proposed trial is adequate, and that evaluation may result in discussions and a request for additional information. An SPA request must be made before the proposed trial begins, and all open issues must be resolved before the trial begins. If a written agreement is reached, it will be documented and made part of the administrative record. Under the FDCA and FDA guidance implementing the statutory requirement, an SPA is generally binding upon the FDA except in limited circumstances, such as if the FDA identifies a substantial scientific issue essential to determining safety or efficacy after the study begins, public health concerns emerge that were unrecognized at the time of the protocol assessment, the sponsor and the FDA agree to the change in writing, or if the study sponsor fails to follow the protocol that was agreed upon with the FDA.
Disclosure of Clinical Trial Information
Sponsors of clinical trials of certain FDA-regulated products, including prescription drugs, are required to register and disclose certain clinical trial information on a public website maintained by the U.S. National Institutes of Health (NIH). Information related to the product, patient population, phase of investigation, study sites and investigator, and other aspects of the clinical trial is made public as part of the registration. Sponsors are also obligated to disclose the results of these trials after completion. Disclosure of the results of these trials can be delayed for up to two years if the sponsor certifies that it is seeking approval of an unapproved product or that it will file an application for approval of a new indication for an approved product within one year. Competitors may use this publicly available information to gain knowledge regarding the design and progress of the development programs. Failure to timely register a covered clinical study or to submit study results as provided for in the law can give rise to civil monetary penalties and also prevent the non-compliant party from receiving future grant funds from the federal government. Since the NIH's Final Rule on ClinicalTrials.gov registration and reporting requirements became effective in 2017, both NIH and FDA have signaled the government's willingness to begin enforcing those requirements against clinical trial sponsors who fail to meet those legal obligations, with FDA releasing in late 2018 a proposal for certain procedural steps it intends to take when determining whether and how to assess civil monetary penalties against a non-compliant party.
Post-Approval Requirements
Drugs manufactured, marketed or distributed pursuant to FDA approval decisions are subject to pervasive and continuing regulation by the FDA, including, among other things, requirements relating to recordkeeping, periodic reporting, product sampling and distribution, advertising and promotion, and reporting of adverse experiences with the product. After approval, most changes to the approved product, such as adding new indications or other labeling claims, are subject to FDA review and approval before they can be implemented. There also are continuing, annual user fee requirements for any marketed products and related manufacturing facilities, as well as new application fees for supplemental applications.
In addition, drug manufacturers and other entities involved in the manufacture of approved drugs are required to register their facilities with the FDA and state agencies, and are subject to periodic unannounced inspections by the FDA for compliance with GMP requirements. Prescription drug distribution facilities are also subject to state licensure, including
40
inspections, by the relevant local regulatory authority. Changes to the manufacturing process, specifications or container closure system for an approved drug are strictly regulated and often require prior FDA approval before being implemented. FDA regulations also require investigation and correction of any deviations from GMP and impose reporting and documentation requirements upon the sponsor and others involved in the drug manufacturing process. Accordingly, manufacturers must continue to expend time, money and effort in the area of production and quality control to maintain GMP compliance and ensure ongoing compliance with other statutory requirements the FDCA, such as the requirements for making manufacturing changes to an approved NDA.
Thus, even after new drug approval is granted, Regulatory authorities may withdraw that approval or request product recalls if compliance with regulatory requirements and standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product, including adverse events of unanticipated severity or frequency, or with manufacturing processes, or failure to comply with regulatory requirements, may result in revisions to the approved labeling to add new safety information; imposition of post-market studies or clinical trials to assess new safety risks; or imposition of distribution or other restrictions under a REMS program. Other potential consequences of regulatory non-compliance include, among other things:
| ● | | restrictions on the marketing or manufacturing of the product, complete withdrawal of the product from the market or product recalls; |
| ● | | fines, warning letters or holds on post-approval clinical trials; |
| ● | | refusal of the FDA to approve pending NDAs or supplements to approved NDAs, or suspension or revocation of product license approvals; |
| ● | | product seizure or detention, or refusal to permit the import or export of products; or |
| ● | | injunctions or the imposition of civil or criminal penalties. |
As described further below, the FDA strictly regulates marketing, labeling, advertising and promotion of prescription drug products that are placed on the market. Drugs may be promoted only for the approved indications and in accordance with the provisions of the approved label. The FDA and other agencies actively enforce the laws and regulations prohibiting the promotion of off-label uses, and a company that is found to have improperly promoted off-label uses may be subject to significant penalties.
The Hatch-Waxman Amendments
Orange Book Listing
In 1984, with passage of the Hatch-Waxman Amendments to the FDCA, Congress authorized the FDA to approve generic drugs that are the same as drugs previously approved by the FDA under the NDA provisions of the statute. As part of the marketing application process when seeking approval for a new drug through an NDA, applicants are required to list with the FDA every patent of which claims cover the applicant's product or an approved method of using the product. Upon approval of a drug, approval information about the drug along with each of the applicant's listed patents is then published in the FDA's Approved Drug Products with Therapeutic Equivalence Evaluations, commonly known as the "Orange Book." Pursuant to the Hatch-Waxman Amendments, drugs listed in the Orange Book can, in turn, be cited by potential generic competitors in support of approval of an abbreviated new drug application, or ANDA. An ANDA provides for marketing of a drug product that has the same active ingredients in the same strengths and dosage form as the reference license drug (“RLD”) and has been shown through bioequivalence testing to be bioequivalent to the RLD. The FDA is responsible for determining that the generic drug is "bioequivalent" to the innovator drug, although under the statute, a generic drug is bioequivalent to a RLD if "the rate and extent of absorption of the drug do not show a significant difference from the rate and extent of absorption of the listed drug..."
Other than the requirement for bioequivalence testing, ANDA applicants are not required to conduct, or submit results of, preclinical or clinical tests to prove the safety or effectiveness of their drug product. Drugs approved in this way are most often considered to be therapeutically equivalent to the RLD, are commonly referred to as "generic equivalents" to the RLD, and can often be substituted by pharmacists under prescriptions written for the original RLD in accordance with state law. Specifically, upon approval of an ANDA, the FDA indicates whether the generic product is "therapeutically equivalent" to the RLD in the Orange Book. By operation of certain state laws and numerous health
41
insurance programs, the FDA's designation of therapeutic equivalence in the Orange Book often results in substitution of the generic drug without the knowledge or consent of either the prescribing physician or the patient.
The Hatch-Waxman Amendments also amended the FDCA to enact Section 505(b)(2) of the FDCA, which permits the filing of an NDA where at least some of the information required for approval comes from studies not conducted by or for the applicant and for which the applicant has not obtained a right of reference. A Section 505(b)(2) applicant may eliminate the need to conduct certain preclinical or clinical studies, if it can establish that reliance on studies conducted for a previously-approved product is scientifically appropriate. The FDA may also require companies to perform additional trials or measurements to support the change from the approved product. The FDA may then approve the new product for all or some of the label indications for which the referenced product has been approved, as well as for any new indication sought by the Section 505(b)(2) applicant. With respect to listed patents, patent certification requirements, and the blocking of follow-on marketing applications for the drug product previously approved under an NDA and listed in the Orange Book—known as the reference listed drug, or RLD—505(b)(2) NDA applications and ANDAs are required under the statute and FDA's implementing regulations to follow similar procedures and are subject to similar conditions. However, only in some cases is a 505(b)(2) NDA-approved drug product determined by FDA to be therapeutically equivalent to the original innovator RLD.
As part of its own marketing application process, the ANDA/505(b)(2) applicant is required to certify to the FDA concerning any patents listed for the relevant RLD in the FDA's Orange Book. Specifically, the applicant must certify either that: (i) the required patent information has not been filed; (ii) the listed patent has expired; (iii) the listed patent has not expired, but will expire on a particular date and approval is sought after patent expiration; or (iv) the listed patent is invalid or will not be infringed by the generic product. The ANDA applicant may also elect to submit a section viii statement, certifying that its proposed ANDA or 505(b)(2) labeling does not contain (or carves out) any language regarding the patented method-of-use, rather than certify to a listed method-of-use patent.
If the ANDA/505(b)(2) applicant does not challenge the innovator's listed patents, or indicates that it is not seeking approval of a patented method of use, the ANDA/505(b)(2) application will not be approved by the FDA until all the listed patents claiming the referenced product have expired.
A certification that the new product will not infringe the already approved product's listed patents, or that such patents are invalid, is called a Paragraph IV certification. If the ANDA/505(b)(2) applicant has provided a Paragraph IV certification to the FDA, the applicant must also send notice of that Paragraph IV certification to the NDA sponsor and patent holders once FDA accepts the ANDA/505(b)(2) application for filing. The NDA and patent holders may then initiate a patent infringement lawsuit in response to the notice of the Paragraph IV certification, as provided for in the statute. The filing of a patent infringement lawsuit within 45 days of the receipt of a Paragraph IV certification automatically prevents the FDA from approving the ANDA/505(b)(2) NDA until the earlier of 30 months, expiration of the patent, settlement of the lawsuit, or a decision in the infringement case that is favorable to the ANDA/505(b)(2) applicant.
Non-Patent Exclusivity
Under the Hatch-Waxman Amendments, the FDA also may not approve an ANDA or 505(b)(2) NDA until any applicable period of non-patent exclusivity for the RLD has expired. The FDCA provides a period of five years of non-patent exclusivity for a new drug containing a new chemical entity, or NCE, which is a drug that contains no active moiety that has been approved by the FDA in any other NDA. During this five years of marketing exclusivity, the FDA cannot receive any ANDA or 505(b)(2) application seeking approval of a drug that references a version of the NCE drug.
The FDCA also provides for a period of three years of exclusivity if the NDA includes reports of one or more new clinical investigations, other than bioavailability or bioequivalence studies, that were conducted by or for the applicant and are essential to the approval of the application. This three-year exclusivity period often protects changes to a previously approved drug product, such as a new dosage form, route of administration, combination or the addition of a new indication. During this three-year period of exclusivity, the FDA cannot approve an ANDA or 505(b)(2) application that includes the change.
42
An ANDA or 505(b)(2) application may be submitted one year before NCE exclusivity expires if a Paragraph IV certification is filed. If there is no listed patent in the Orange Book, there may not be a Paragraph IV certification requirement, and in such situations, no ANDA or 505(b)(2) application may be filed before the expiration of the exclusivity period.
For a botanical drug, the FDA may determine that the active moiety is one or more of the principal components, or the complex mixture as a whole. This determination would affect the possibility of any five-year exclusivity as well as the ability of any potential generic competitor to demonstrate that it is the same drug as the original botanical drug. Because the agency has not promulgated specific regulations for botanical drug products and is approaching the development of such products, especially those that are composed of more complex mixtures, on a case-by-case basis, the 2016 Botanical Drug Development guidance for industry represents the best source for the FDA's current thinking on these drug products.
Patent Term Extension
After NDA approval, owners of relevant drug patents may apply for up to a five-year patent extension. The allowable patent term extension is calculated as half of the drug's testing phase—the time between IND submission and NDA submission—and all of the review phase—the time between NDA submission and approval up to a maximum of five years. The time can be shortened if the FDA determines that the applicant did not pursue approval with due diligence. The total patent term after the extension may not exceed 14 years from market approval.
For patents that might expire during the application phase, the patent owner may request an interim patent extension. An interim patent extension increases the patent term by one year and may be renewed up to four times. For each interim patent extension granted, the post-approval patent extension is reduced by one year. The director of the U.S. Patent and Trademark Office must determine that approval of the drug covered by the patent for which a patent extension is being sought is likely. Interim patent extensions are not available for a drug for which an NDA has not been submitted.
Prescription Drug Marketing Act
As part of the sales and marketing process, pharmaceutical companies frequently provide samples of approved drugs to physicians. The Prescription Drug Marketing Act (PDMA) imposes requirements and limitations upon the provision of drug samples to physicians, as well as prohibits states from licensing distributors of prescription drugs unless the state licensing program meets certain federal guidelines that include minimum standards for storage, handling and record keeping. In addition, the PDMA sets forth civil and criminal penalties for violations.
New Legislation and Regulations
From time to time, legislation is drafted, introduced and passed in Congress that could significantly change the statutory provisions governing the testing, approval, manufacturing and marketing of products regulated by the FDA and relevant regulatory authorities outside the United States. In addition to new legislation, regulations and policies are often revised or interpreted by regulatory authorities in ways that may significantly affect our business and its product candidates. It is impossible to predict whether further legislative changes will be enacted or whether regulations, guidance, policies or interpretations will be changed or what the effect of such changes, if any, may be.
Other U.S. Healthcare Laws and Compliance Requirements
If we obtain regulatory approval of our product candidates and launch them commercially in the United States, we may be subject to various federal and state laws targeting fraud and abuse in the healthcare industry. These laws may impact, among other things, our proposed sales, marketing and education programs. In addition, we may be subject to patient privacy regulation by both the federal government and the states in which we conduct our business. Some of the laws that may affect our future ability to operate include:
| ● | | the federal Anti-Kickback Statute, which prohibits, among other things, persons from knowingly and willfully soliciting, receiving, offering or paying remuneration, directly or indirectly, to induce, or in return for, the purchase or recommendation of an item or service reimbursable under a federal healthcare program, such as the Medicare and Medicaid programs; |
43
| ● | | federal civil and criminal false claims laws and civil monetary penalty laws, which prohibit, among other things, individuals or entities from knowingly presenting, or causing to be presented, claims for payment from Medicare, Medicaid, or other third-party payers that are false or fraudulent; |
| ● | | the federal Health Insurance Portability and Accountability Act of 1996, or HIPAA, which created new federal criminal statutes that prohibit executing a scheme to defraud any healthcare benefit program and making false statements relating to healthcare matters; |
| ● | | HIPAA, as amended by the Health Information Technology and Clinical Health Act, or HITECH, and its implementing regulations, which imposes certain requirements relating to the privacy, security and transmission of individually identifiable health information; |
| ● | | the federal transparency requirements under the Physician Payments Sunshine Act require manufacturers of FDA-approved drugs, devices, biologics and medical supplies covered by Medicare or Medicaid to report, on an annual basis, to the Department of Health and Human Services information related to payments and other transfers of value to physicians, teaching hospitals, and certain advanced non-physician health care practitioners and physician ownership and investment interests; and |
| ● | | state law equivalents of each of the above federal laws, such as anti-kickback and false claims laws, which may apply to items or services reimbursed by any third-party payer, including commercial insurers, and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and may not have the same effect, thus complicating compliance efforts. Moreover, some state laws require pharmaceutical companies to comply with the pharmaceutical industry's voluntary compliance guidelines, or the relevant compliance guidance promulgated by the federal government, in addition to requiring drug manufacturers to report information related to payments to physicians and other health care providers or marketing expenditures to the extent that those laws impose requirements that are more stringent than the Physician Payments Sunshine Act. |
Europe/Rest of World Government Regulation
In addition to regulations in the United States, we are and will be subject, either directly or through our distribution partners, to a variety of regulations in other jurisdictions governing, among other things, clinical trials and any commercial sales and distribution of its products, if approved.
Whether or not we obtain FDA approval for a product, we must obtain the requisite approvals from regulatory authorities in non-U.S. countries prior to the commencement of clinical trials or marketing of the product in those countries.
The approval process varies from country to country and can involve additional product testing and additional administrative review periods. The time required to obtain approval in other countries might differ from and be longer than that required to obtain FDA approval. Regulatory approval in one country does not ensure regulatory approval in another, but a failure or delay in obtaining regulatory approval in one country may negatively impact the regulatory process in others.
In the European Union, medicinal products are subject to extensive pre- and post-marketing regulation by regulatory authorities at both the European Union and national levels. Additional rules also apply at the national level to the manufacture, import, export, storage, distribution and sale of controlled substances. In many E.U. member states the regulatory authority responsible for medicinal products is also responsible for controlled substances. Responsibility is, however, split in some member states, such as the United Kingdom. Generally, any company manufacturing or distributing a medicinal product containing a controlled substance in the European Union will need to hold a controlled substances license from the competent national authority and will be subject to specific record-keeping and security obligations. Separate import or export certificates are required for each shipment into or out of the member state.
Clinical Trials and Marketing Approval
Certain countries outside of the United States have a process that requires the submission of a clinical trial application much like an IND prior to the commencement of human clinical trials. In Europe, for example, a clinical trial application, or CTA, must be submitted to the competent national health authority and to independent ethics committees in each country in which a company intends to conduct clinical trials. Once the CTA is approved in accordance with a
44
country's requirements and a company has received favorable ethics committee approval, clinical trial development may proceed in that country.
The requirements and process governing the conduct of clinical trials, product licensing, pricing and reimbursement vary from country to country, even though there is already some degree of legal harmonization in the European Union member states resulting from the national implementation of underlying E.U. legislation. In all cases, the clinical trials must be conducted in accordance with the International Conference on Harmonization, or ICH, guidelines on GCP and other applicable regulatory requirements.
To obtain regulatory approval to place a drug on the market in the European Union, we must submit a marketing authorization application. This application is similar to the NDA in the United States, with the exception of, among other things, country-specific document requirements. All application procedures require an application in the common technical document, or CTD, format, which includes the submission of detailed information about the manufacturing and quality of the product, and non-clinical and clinical trial information. Drugs can be authorized in the European Union by using (i) the centralized authorization procedure, (ii) the mutual recognition procedure, (iii) the decentralized procedure or (iv) national authorization procedures.
The European Commission created the centralized procedure for the approval of human drugs to facilitate marketing authorizations that are valid throughout the European Union and, by extension (after national implementing decisions) in Iceland, Liechtenstein and Norway, which, together with the E.U. member states, comprise the European Economic Area, or EEA. Applicants file marketing authorization applications with the EMA, where they are reviewed by a relevant scientific committee, in most cases the Committee for Medicinal Products for Human Use, or CHMP. The EMA forwards CHMP opinions to the European Commission, which uses them as the basis for deciding whether to grant a marketing authorization. This procedure results in a single marketing authorization granted by the European Commission that is valid across the European Union, as well as in Iceland, Liechtenstein and Norway. The centralized procedure is compulsory for human drugs that are: (i) derived from biotechnology processes, such as genetic engineering, (ii) contain a new active substance indicated for the treatment of certain diseases, such as HIV/AIDS, cancer, diabetes, neurodegenerative diseases, autoimmune and other immune dysfunctions and viral diseases, (iii) officially designated "orphan drugs" (drugs used for rare human diseases) and (iv) advanced-therapy medicines, such as gene-therapy, somatic cell-therapy or tissue-engineered medicines. The centralized procedure may, at the voluntary request of the applicant, also be used for human drugs which do not fall within the above-mentioned categories if the CHMP agrees that (a) the human drug contains a new active substance not yet approved on November 20, 2005; (b) it constitutes a significant therapeutic, scientific or technical innovation or (c) authorization under the centralized procedure is in the interests of patients at the E.U. level.
Under the centralized procedure in the European Union, the maximum timeframe for the evaluation of a marketing authorization application by the EMA is 210 days (excluding clock stops, when additional written or oral information is to be provided by the applicant in response to questions asked by the CHMP), with adoption of the actual marketing authorization by the European Commission thereafter. Accelerated evaluation might be granted by the CHMP in exceptional cases, when a medicinal product is expected to be of a major public health interest from the point of view of therapeutic innovation, defined by three cumulative criteria: the seriousness of the disease to be treated, the absence of an appropriate alternative therapeutic approach, and anticipation of exceptional high therapeutic benefit. In this circumstance, the EMA ensures that the evaluation for the opinion of the CHMP is completed within 150 days and the opinion issued thereafter.
For those medicinal products for which the centralized procedure is not available, the applicant must submit marketing authorization applications to the national medicines regulators through one of three procedures: (i) the mutual recognition procedure (which must be used if the product has already been authorized in at least one other E.U. member state, and in which the E.U. member states are required to grant an authorization recognizing the existing authorization in the other E.U. member state, unless they identify a serious risk to public health), (ii) the decentralized procedure (in which applications are submitted simultaneously in two or more E.U. member states) or (iii) national authorization procedures (which results in a marketing authorization in a single E.U. member state).
45
Mutual Recognition Procedure
The mutual recognition procedure, or MRP, for the approval of human drugs is an alternative approach to facilitate individual national marketing authorizations within the European Union. Basically, the MRP may be applied for all human drugs for which the centralized procedure is not obligatory. The MRP is applicable to the majority of conventional medicinal products and must be used if the product has already been authorized in one or more member states.
The characteristic of the MRP is that the procedure builds on an already—existing marketing authorization in a member state of the European Union that is used as a reference in order to obtain marketing authorizations in other E.U. member states. In the MRP, a marketing authorization for a drug already exists in one or more member states of the European Union and subsequently marketing authorization applications are made in other E.U. member states by referring to the initial marketing authorization. The member state in which the marketing authorization was first granted will then act as the reference member state. The member states where the marketing authorization is subsequently applied for act as concerned member states. The concerned member states are required to grant an authorization recognizing the existing authorization in the reference member state, unless they identify a serious risk to public health.
The MRP is based on the principle of the mutual recognition by E.U. member states of their respective national marketing authorizations. Based on a marketing authorization in the reference member state, the applicant may apply for marketing authorizations in other member states. In such case, the reference member state shall update its existing assessment report about the drug in 90 days. After the assessment is completed, copies of the report are sent to all member states, together with the approved summary of product characteristics, labeling and package leaflet. The concerned member states then have 90 days to recognize the decision of the reference member state and the summary of product characteristics, labeling and package leaflet. National marketing authorizations shall be granted within 30 days after acknowledgement of the agreement.
If any E.U. member state refuses to recognize the marketing authorization by the reference member state, on the grounds of potential serious risk to public health, the issue will be referred to a coordination group. Within a timeframe of 60 days, member states shall, within the coordination group, make all efforts to reach a consensus. If this fails, the procedure is submitted to an EMA scientific committee for arbitration. The opinion of this EMA Committee is then forwarded to the European Commission for the start of the decision making process. As in the centralized procedure, this process entails consulting various European Commission Directorates General and the Standing Committee on Human Medicinal Products.
Data and Market Exclusivity in the European Union
In the European Union, NCEs qualify for eight years of data exclusivity upon marketing authorization and an additional two years of market exclusivity. This data exclusivity, if granted, prevents regulatory authorities in the European Union from referencing the innovator's data to assess a generic (abbreviated) application for eight years, after which generic marketing authorization can be submitted, and the innovator's data may be referenced, but not approved for two years. The overall ten-year period will be extended to a maximum of eleven years if, during the first eight years of those ten years, the marketing authorization (MA) holder obtains an authorization for one or more new therapeutic indications which, during the scientific evaluation prior to their authorization, are held to bring a significant clinical benefit in comparison with existing therapies. Even if a compound is considered to be a NCE and the sponsor is able to gain the prescribed period of data exclusivity, another company nevertheless could also market another version of the drug if such company can complete a full MAA with a complete database of pharmaceutical test, preclinical studies and clinical trials and obtain marketing approval of its product.
Data and Market Exclusivity in Japan
Japan has no established system for data exclusivity or marketing exclusivity. However, the Pharmaceuticals Act of Japan (PAA) provides for a re-examination system after drug approval. This system imposes an obligation on the MA holder to continue to collect clinical data after market approval during a study period. The MA holder must apply for reexamination to the Minster of Health Labor and Welfare within three months of the expiration of the study period. During the study and reexamination period no generic drug may be approved, effectively providing a form of market
46
exclusivity. The study period is determined by the drug category. The study period for an orphan drug is 10 years from MA, the study period for an NCE is eight years from MA, and for an improvement (new indication, formulation, etc.) the study period is four to six years from MA.
Patent Term Extension in Japan
The term of a patent that covers the approved drug may be extended for the shorter of five years, or the period during which the patent could not be worked (exploited) due to obtaining regulatory approval. This period is calculated from the later of the patent registration date (grant date) or the clinical trial start date to the regulatory approval date.
Regulatory Exclusivity in China
China has a six-year regulatory exclusivity period for NCE and orphan drugs, such as NB-01, NB-02 and Gemcabene, which begins at the date of market approval.
Pharmaceutical Coverage, Pricing and Reimbursement
Sales of pharmaceutical products approved for marketing in the United States by the FDA will depend, in part, on the extent to which the costs of the products will be covered by third-party payers, such as government health programs, and commercial insurance and managed health care organizations. These third-party payers are increasingly challenging the prices charged for medical products and services. Additionally, the containment of health care costs has become a priority of federal and state governments, and the prices of drugs have been a focus in this effort. The U.S. government, state legislatures and foreign governments have shown significant interest in implementing cost-containment programs, including price controls, utilization management and requirements for substitution of generic products. Adoption of price controls and cost-containment measures, and adoption of more restrictive policies in jurisdictions with existing controls and measures, could further limit our operating results. If these third-party payers do not consider our products to be cost-effective compared to other available therapies, they may not cover our products after approval as a benefit under their plans or, if they do, the level of payment may not be sufficient to allow us to sell its products on a profitable basis.
The Medicare Prescription Drug, Improvement, and Modernization Act of 2003, or the MMA, imposed requirements for the distribution and pricing of prescription drugs for Medicare beneficiaries and included a major expansion of the prescription drug benefit under Medicare Part D. Under Part D, Medicare beneficiaries may enroll in prescription drug plans offered by private entities that provide coverage of outpatient prescription drugs. Part D is available through both stand-alone prescription drug benefit plans and prescription drug coverage as a supplement to Medicare Advantage plans. Unlike Medicare Parts A and B, Part D coverage is not standardized. Part D prescription drug plan sponsors are not required to pay for all covered Part D drugs, and each drug plan can develop its own drug formulary that identifies which drugs it will cover and at what tier or level. However, Part D prescription drug formularies must include drugs within each therapeutic category and class of covered Part D drugs, though not necessarily all the drugs in each category or class. Any formulary used by a Part D prescription drug plan must be developed and reviewed by a pharmacy and therapeutic committee.
Government payment for some of the costs of prescription drugs may increase demand for products for which we receive marketing approval in the U.S. However, any negotiated prices for our products covered by a Part D prescription drug plan will likely be lower than the prices we might otherwise obtain. Moreover, while the MMA applies only to drug benefits for Medicare beneficiaries, private payers often follow Medicare coverage policy and payment limitations in setting their own payment rates. Any reduction in payment that results from the MMA may result in a similar reduction in payments from non-governmental payers.
The Patient Protection and Affordable Care Act, as amended by the Health Care and Education Affordability Reconciliation Act of 2010 (collectively, the “ACA”), was enacted with the goal of expanding coverage for the uninsured while at the same time containing overall health care costs. With regard to pharmaceutical products, among other things, the ACA expanded and increased industry rebates for drugs covered under Medicaid programs and made changes to the coverage requirements under the Medicare Part D program. We still cannot fully predict the impact of the ACA on pharmaceutical companies as many of the ACA reforms require the promulgation of detailed regulations implementing the statutory provisions which has not yet been completed, and the Centers for Medicare & Medicaid
47
Services has publicly announced that it is analyzing the ACA regulations and policies that have been issued to determine if changes should be made. In addition, although the United States Supreme Court has upheld the constitutionality of most of the ACA, some states have stated their intentions to not implement certain sections of the ACA. These challenges add to the uncertainty of the changes enacted as part of ACA.
In the United States, Medicare covers certain drug purchases by the elderly and eligible disabled people and introduced a reimbursement methodology based on average sales prices for physician-administered drugs. In addition, Medicare may limit the number of drugs that will be covered in any therapeutic class. Ongoing cost reduction initiatives and future laws could decrease the coverage and price that we will receive for any approved products. While Medicare beneficiaries are limited to most elderly and certain disabled individuals, private payors often follow Medicare coverage policy and payment limitations in setting their own payment rates.
Among the provisions of the ACA of importance to our product candidates are the following:
| ● | | an annual, nondeductible fee on any entity that manufactures or imports specified branded prescription drugs and biologic products; |
| ● | | an increase in the statutory minimum rebates a manufacturer must pay under the Medicaid Drug Rebate Program; |
| ● | | expansion of healthcare fraud and abuse laws, including the False Claims Act and the Anti-Kickback Statute, new government investigative powers, and enhanced penalties for noncompliance; |
| ● | | a new Medicare Part D coverage gap discount program, in which manufacturers must agree to offer 50% point-of-sale discounts off negotiated prices; |
| ● | | extension of manufacturers' Medicaid rebate liability; |
| ● | | expansion of eligibility criteria for Medicaid programs; |
| ● | | expansion of the entities eligible for discounts under the Public Health Service Act's pharmaceutical pricing program; |
| ● | | new requirements to report financial arrangements with physicians and teaching hospitals (i.e., the Federal Physician Payment Sunshine Act, which has since been expanded to cover additional specified healthcare providers); |
| ● | | a new requirement to annually report drug samples that manufacturers and distributors provide to physicians; and |
| ● | | a new Patient-Centered Outcomes Research Institute to oversee, identify priorities in, and conduct comparative clinical effectiveness research, along with funding for such research. |
We expect that the ACA, as well as other healthcare reform measures that may be adopted in the future, may result in more rigorous coverage criteria and in additional downward pressure on the price that we will receive for any approved product. Any reduction in payments from Medicare or other government programs may result in a similar reduction in payments from private payors. The implementation of cost containment measures or other healthcare reforms may prevent us from being able to generate revenue, attain profitability, or commercialize our products. The ACA also continues to be the subject of significant political controversy and legal challenges, making its continued implementation uncertain.
In addition, in some foreign countries, the proposed pricing for a drug must be approved before it may be lawfully marketed. The requirements governing drug pricing vary widely from country to country. For example, some E.U. jurisdictions operate positive and negative list systems under which products may only be marketed once a reimbursement price has been agreed. To obtain reimbursement or pricing approval, some of these countries may require the completion of clinical trials that compare the cost-effectiveness of a particular product candidate to currently available therapies. Other member states allow companies to fix their own prices for medicines but monitor and control company profits. Such differences in national pricing regimes may create price differentials between E.U. member states. There can be no assurance that any country that has price controls or reimbursement limitations for pharmaceutical products will allow favorable reimbursement and pricing arrangements for any of our products. Historically, products launched in the European Union do not follow price structures of the United States. In the European Union, the downward pressure on healthcare costs in general, particularly prescription medicines, has become
48
intense. As a result, barriers to entry of new products are becoming increasingly high and patients are unlikely to use a drug product that is not reimbursed by their government.
Human Capital Management
As of December 31, 2020, we had 7 full-time employees. Of these employees, five were engaged in research and development and two were engaged in general and administrative functions. Three of our employees are located in the United States, and four are located in South Korea. We have no collective bargaining agreements with our employees and have not experienced any work stoppages. We consider our relationships with our employees to be good.
Corporate Information
NeuroBo was incorporated under the laws of the State of Delaware in July 2017, and completed the 2019 Merger with Gemphire on December 30, 2019. Our principal executive offices are located at 200 Berkeley Street, 19th Floor, Boston, Massachusetts, 02116. Our website address is www.neurobopharma.com. The information contained on, or that can be accessed through, our website is not a part of this report.
Our business, prospects, financial condition or results of operations could be materially adversely affected by any of the risks and uncertainties set forth below, as well as in any amendments or updates reflected in subsequent filings with the Securities and Exchange Commission (the “SEC”). In assessing these risks, you should also refer to other information contained in this report, including our financial statements and related notes.
Risks Related to our Operations and to Development, Marketing, Commercialization and Regulation of Our Product Candidates
We have incurred losses since inception, we anticipate that we will incur continued losses for the foreseeable future and there is substantial doubt about our ability to continue as a going concern for the full one-year period following the date of this report. We require additional financing to accomplish our long-term business plan and failure to obtain necessary capital when needed on acceptable terms, or at all, could force us to delay, limit, reduce or terminate our operations.
We have experienced net losses and negative cash flows from operating activities since our inception and have an accumulated deficit of $66.5 million as of December 31, 2020. It is possible we will never generate revenue or profit.
As of December 31, 2020, we had cash and cash equivalents of $10.1 million. Operating at the level of scientific activity described in “Management’s Discussion and Analysis of Financial Statements and Results of Operations – Overview - Recent Developments” we expect that our cash and cash equivalents will be adequate to fund operations through into the fourth quarter of 2021. Accordingly, we will need to raise additional capital to fund continued operations at the current level beyond 2021. We have some ability to reduce costs further in 2021 by further curtailing the level of scientific activity planned for 2021, thereby potentially lengthening our operational window into the first quarter of 2022.
Although we are exploring financing opportunities and carefully monitoring the capital markets, we do not yet have any commitments for additional financing and may not be successful in our efforts to raise additional funds. There can be no assurances that additional financing will be available to us on satisfactory terms, or at all. If we are unable to raise sufficient additional capital (which is not assured at this time, particularly as a result of recent depressed capital market conditions), our long-term business plan may not be accomplished, and we may be forced to cease, reduce, or delay operations. For more information about our liquidity and capital resources, see Part II, Item 7 “Management’s Discussion and Analysis of Financial Condition and Results of Operations — Liquidity and Capital Resources.”
The foregoing factors individually and collectively raise substantial doubt about our ability to continue as a going concern for the full one-year period following the date of this report. For more information, see “Management’s Discussion and Analysis of Financial Statements and Results of Operations – Overview - Recent Developments – Going Concern” and “Going Concern” under Note 1 to our audited financial statements which are included elsewhere in this report. The perception that we may not be able to continue as a going concern may cause others to choose not to deal
49
with us due to concerns about our ability to meet our contractual obligations. If we are unable to continue as a going concern, investors could lose all or part of their investment in our Company.
Our pursuit of potential therapeutic and prophylactic treatments for COVID-19 is at an early stage and subject to many risks. We may be unable to receive approval for any of our COVID-19 product candidates a timely manner, if at all, and our COVID-19 product candidate may never be approved.
We may experience difficulties or delays in enrolling patients in clinical trials due to the impact of the global COVID-19 pandemic or other reasons. Many of the risks related to the development of these product candidates are beyond our control, including risks related to clinical development, the regulatory submission process, potential threats to our intellectual property rights and manufacturing delays or difficulties. We may be unable to produce an efficacious and/or approved product for the treatment of patients with early COVID-19 in a timely manner, if at all.
The results of preclinical studies from our COVID-19 product candidates may not be predictive of the results of clinical trials, and the results of any early-stage clinical trials we commence may not be predictive of the results of the later-stage clinical trials. There can be no assurance that any of our clinical trials for our COVID-19 product candidates, or any other of our product candidates, will ultimately be successful or support further clinical development. In addition, the interpretation of the data from our clinical trials of ANA001 or Gemcabene by FDA and other regulatory agencies may differ from our interpretation of such data and the FDA or other regulatory agencies may require that we conduct additional studies or analyses. Any of these factors could delay or prevent us from receiving regulatory approval of ANA001 or Gemcabene and there can be no assurance that our product candidate will be approved in a timely manner, if at all.
If the COVID-19 outbreak is effectively contained or the risk of coronavirus infection is diminished or eliminated before we can successfully develop and manufacture our product candidate, the commercial viability of such product candidate may be diminished or eliminated. We are also committing financial resources and personnel to the development of this product candidate which may cause delays in or otherwise negatively impact our other development programs, despite uncertainties surrounding the longevity and extent of coronavirus as a global health concern. Our business could be negatively impacted by our allocation of significant resources to a global health threat that is unpredictable and could rapidly dissipate or against which our treatment, if successfully developed, may not be effective. In addition, other parties are currently producing therapeutic and vaccine candidates for COVID-19, which may be more efficacious or may be approved prior to our product.
The regulatory pathway for ANA001 and Gemcabene is continually evolving, and may result in unexpected or unforeseen challenges.
The speed at which parties are acting to create and test many therapeutics and vaccines for COVID-19 is unusual, and evolving or changing plans or priorities within the FDA, including those based on new knowledge of COVID-19 and how the disease affects the human body, may significantly affect the regulatory timeline for our product candidates. Results from ongoing clinical trials and discussions with regulatory authorities may raise new questions and require us to redesign proposed clinical trials, including revising proposed endpoints or adding new clinical trial sites or cohorts of subjects. Any such developments could delay the development timeline for our product candidates and materially increase the cost of the development for such candidates.
In light of the COVID-19 pandemic, it is possible that one or more government entities may take actions that directly or indirectly have the effect of abrogating some of our rights or opportunities. If we were to develop a treatment for COVID-19, the economic value of such a therapeutic treatment to us could be limited.
Various government entities, including the U.S. government, are offering incentives, grants and contracts to encourage additional investment by commercial organizations into preventative and therapeutic agents against coronavirus, which may have the effect of increasing the number of competitors and/or providing advantages to known competitors. Accordingly, there can be no assurance that we will be able to successfully establish a competitive market share for our COVID-19 therapeutic treatment, if any.
50
Even if we obtain favorable clinical results, we may not be able to obtain regulatory approval for, or successfully commercialize ANA001 or Gemcabene.
We are not permitted to market ANA001 or Gemcabene in the United States until we receive approval of an NDA from the FDA, or in any foreign countries until we receive the requisite approval from such countries. As a condition to submitting an NDA to the FDA for ANA001, we must complete our ongoing Phase 2 clinical trial, conduct and complete further Phase 3 clinical trials, and any additional nonclinical studies or clinical trials required by the FDA. To date, we have only completed the Phase 1 Single Ascending Dosing (SAD) study. ANA001 may not be successful in clinical trials or receive regulatory approval. Further, ANA001 may not receive regulatory approval even if it is successful in clinical trials. Obtaining approval of an NDA is a complex, lengthy, expensive and uncertain process that typically takes many years following the commencement of clinical trials and depends upon numerous factors, including the substantial discretion of the regulatory authorities. In addition, the policies or regulations, or the type and amount of clinical data necessary to gain approval, may change during the course of a product candidate’s clinical development and may vary among jurisdictions. Our development activities could be harmed or delayed by a partial shutdown of the U.S. government, including the FDA. We have not obtained regulatory approval for any product candidate and it is possible that ANA001 will never obtain regulatory approval. The FDA may delay, limit or deny approval of ANA001 for many reasons, including, among others:
| ● | the results of our clinical trials may not meet the level of statistical or clinical significance required by the FDA for marketing approval; |
| ● | the FDA may disagree with the number, design, size, conduct or implementation of our clinical trials; |
| ● | the FDA may not approve the formulation, labeling or specifications of ANA001; |
| ● | the FDA may require that we conduct additional clinical trials; |
| ● | the contract research organizations (“CROs”) or the clinical investigators that we retain to conduct our clinical trials may take actions outside of our control that materially adversely impact our clinical trials; |
| ● | we, our CROs or clinical investigators may fail to perform in accordance with the FDA’s good clinical practice (“GCP”) requirements; |
| ● | the FDA may disagree with our interpretation of data from our preclinical studies and clinical trials; |
| ● | the FDA may find deficiencies with the manufacturing processes or facilities of third-party manufacturers with which we contract; or |
| ● | the policies or regulations of the FDA may significantly change in a manner that renders our clinical data insufficient for approval or may require that we amend or submit new clinical protocols. |
In addition, similar reasons may cause the EMA or other regulatory authorities to delay, limit or deny approval of ANA001 outside the United States. Any of these factors, many of which are beyond our control, could jeopardize our ability to obtain regulatory approval for and successfully market ANA001.
Alternatively, even if we obtain regulatory approval, that approval may be for indications or patient populations that are not as broad as we intend or desire or may require labeling that includes significant use or distribution restrictions or safety warnings. We may also be required to perform additional, unanticipated clinical trials to obtain approval or be subject to additional post marketing testing requirements to maintain regulatory approval. In addition, regulatory authorities may withdraw their approval of a product or the FDA may require a risk evaluation and mitigation strategy (“REMS”) for a product, which could impose restrictions on its distribution. Any of the foregoing scenarios could materially harm the commercial prospects for our product candidates.
Gemcabene was being evaluated in a Phase 2 randomized, double-blind, placebo-controlled study to assess its efficacy safety and tolerability in patients with severe hypertriglyceridemia. In January 2016, the Gemcabene Phase 2 clinical study was placed on partial clinical hold as the FDA requested 2-year rat and mouse carcinogenicity studies to be completed and submitted. The study currently remains on partial clinical hold for the treatment of dyslipidemia.
51
NeuroBo is currently assessing the path forward for Gemcabene for the indication for COVID-19. As a result, there is a significant uncertainty around our development of Gemcabene as a potential treatment for COVID-19.
We have determined to postpone the initiation of Phase 3 clinical trials of NB-01 under present circumstances and we have terminated all of our agreements with contract research organizations related to NB-01. We may not be able to successfully develop NB-01 pursuant to other alternatives, including as an orphan drug or as a nutraceutical candidate.
NB-01 has successfully completed two Phase 2 proof-of-concept clinical trials for PDN. However, in light of the present business environment including the impact of the COVID-19 disease that emerged in December 2019 as a global pandemic, we have determined that any attempt to conduct Phase 3 clinical trials for NB-01, as previously announced, would be difficult if not impossible in the short or medium term. To conserve financial resources, in the first quarter of 2020 we directed our contract research organization (“CRO”) partners and other vendors working on the Phase 3 clinical trials of NB-01, including Syneos Health, to cease all work and we gave notice of termination of our existing contract arrangements with each of them.
We are currently re-evaluating alternatives to bring the NB-01 asset to the market through a different regulatory pathway. Development of NB-01 as an orphan drug is among the alternatives we are considering, and we may conduct feasibility studies to identify a rare disease relevant to NB-01. Additionally, we are considering marketing the NB-01 product line as nutraceutical (non-pharmaceutical) products. There is no assurance that we will be able to pursue either alternative for NB-01.
Our ability to successfully develop NB-01 as an orphan drug would be subject to the following additional risks, among others:
| ● | the results from different types of animal models could be inconsistent from the previous data we have; |
| ● | a limited number of potential participants could make clinical trials for NB-01 difficult; |
| ● | disparate locations of a limited number of potential participants could make clinical trials difficult; and |
| ● | batch-by-batch consistency is difficult to achieve in clinical trials with small numbers of participants. |
Our ability to successfully develop NB-01 as a nutraceutical product would be subject to the following risks, among others:
| ● | the future growth and profitability of NB-01 would depend in large part upon our ability to successfully hire personnel with requisite marketing expertise, the effectiveness and efficiency of our marketing efforts and our ability to select effective markets and media in which to market and advertise; |
| ● | our inability to properly manage, motivate and retain third party distributors for NB-01, as applicable, could have a material adverse effect on us; |
| ● | the success of NB-01 would likely be linked to the size and growth rate of the vitamin, mineral and dietary supplement market, and an adverse change in the size or growth rate of that market could have a material adverse effect on us; and |
| ● | unfavorable publicity or consumer perception of NB-01 and any similar products distributed by other companies could have a material adverse effect on us. |
We may not be able to successfully obtain regulatory or marketing approval for, or successfully commercialize, any of our product candidates.
Although we currently have no drug product for sale and may never be able to develop marketable drug products, our business depends heavily on the successful clinical development (for our pharmaceutical drug products), regulatory approval and commercialization of our drug candidates.
The clinical trials of our product candidates are, and the manufacturing and marketing of our product candidates will be, subject to extensive and rigorous review and regulation by government authorities in the United States and in other countries where we intend to test and, if approved, market any product candidate. Before obtaining regulatory approvals
52
for the commercial sale of any product candidate as a pharmaceutical product, we must successfully meet a number of critical developmental milestones, including:
The time necessary to achieve these developmental milestones for any individual product candidate is long and uncertain, and we may not successfully complete these milestones for any product candidates that we may develop.
We have not yet completed development of any product candidate. Moreover, both NB-01 and NB-02 are considered a “botanical drug product” by the FDA, which results in the drug candidate having unique features that must be taken into account during the drug development process. Botanical drug products may be heterogeneous in nature and may carry additional uncertainty about their active constituents in comparison to synthetic small-molecule drug products. Accordingly, the FDA may impose additional requirements on us in order to confirm that the final formulation of NB-01 or NB-02 is able to demonstrate the necessary therapeutic consistency to support the marketing of a safe and effective commercial drug product. The complexities of developing botanical drug products may increase the time and costs associated with the development of our product candidates.
In August 2018, the FDA, following submission of a two-year carcinogenicity study, requested additional preclinical studies, including a 13-week PPAR-alpha knockout mouse study with Gemcabene. The FDA stated that there could be no progression to the EOP2 meeting or commencement of the Phase 3 trials, which require more than 6 months of drug exposure, until the partial clinical hold was lifted. This request delayed the timeline for the EOP2 meeting and start of a Phase 3 trial by more than one and a half years. It is possible that the FDA may request additional studies and information prior to lifting the hold which would significantly delay the time and cost to initiating Phase 3 trials and future development of Gemcabene. If the FDA decisions further delay our clinical plans, this could jeopardize our ability to commercialize Gemcabene by April 2024, as required by the Pfizer Agreement. Finally, we cannot assure you that the partial clinical hold will ever be lifted, in which case Gemcabene will never receive NDA approval or be commercialized.
We are continuing to test and develop our product candidates and may explore possible design or formulation changes to address safety, efficacy, manufacturing efficiency and performance issues to the extent any arise. The design of a clinical trial may be able to determine whether its results will support approval of a product, and flaws in the design of a clinical trial may not become apparent until the clinical trial is well advanced or completed. There is no assurance that we will be able to design and complete a clinical trial to support marketing approval. Moreover, nonclinical and clinical data are often susceptible to multiple interpretations and analyses. A number of companies in the pharmaceutical and biotechnology industries have experienced significant setbacks in advanced clinical trials, even after promising results in earlier trials.
We may not be able to complete development of any product candidates that demonstrate safety and efficacy and that will have a commercially reasonable treatment and storage period. If we are unable to complete development of NB-01, NB-02, Gemcabene or any other product candidates that we may develop, we will not be able to commercialize and earn revenue from them.
53
The regulatory review and approval processes of the FDA and comparable foreign regulatory authorities are lengthy, time-consuming and inherently unpredictable, and if we are ultimately unable to obtain regulatory approval for our product candidates, including pursuant to the guidelines applicable to NB-01 and NB-02 as botanical drug products, and the guidelines applicable to Gemcabene, our business will be substantially harmed.
Of the large number of drugs in development in the United States, only a small percentage receive FDA regulatory approval and are commercialized in the United States. We are not permitted to market NB-01, NB-02, Gemcabene or any other product candidate as a pharmaceutical drug in the United States until we receive approval of an NDA from the FDA, or in any foreign countries until we receive the requisite approval from such countries or jurisdictions, such as the marketing authorization application, or MAA, in the European Union from the European Medicines Agency, or EMA.
Successfully completing clinical trials and obtaining approval of an NDA is a complex, lengthy, expensive and uncertain process, and the FDA, or a comparable foreign regulatory authority, may delay, limit or deny approval of an NDA for many reasons, including, among others:
| ● | | disagreement with the design or implementation of our clinical trials; |
| ● | | disagreement with the sufficiency of our clinical trials; |
| ● | | failure to demonstrate the safety and efficacy of the product candidate for the proposed indications; |
| ● | | failure to demonstrate that any clinical and other benefits of the product candidate outweigh their safety risks; |
| ● | | a negative interpretation of the data from our nonclinical studies or clinical trials; |
| ● | | deficiencies in the manufacturing or control processes or failure of third-party manufacturing facilities with which our contracts for clinical and commercial supplies to comply with current Good Manufacturing Practice requirements, or cGMPs; |
| ● | | deficiencies in the harvesting and processing of botanical raw materials under Good Agricultural and Collection Processes, or GACPs, or the inability to demonstrate that the final product is capable of being therapeutically consistent, as applicable to botanical drug products, as applicable; |
| ● | | insufficient data collected from clinical trials or changes in the approval requirements that render our nonclinical and clinical data insufficient to support the filing of an NDA or to obtain regulatory approval; or |
| ● | | changes in clinical practice in or approved products available for the treatment of the target patient population that could have an impact on the indications that we are pursuing for our product candidates. |
Further, the FDA has specific requirements and technical standards for botanical drugs, with which we will be obliged to comply in the clinical development of NB-01 and NB-02 as pharmaceutical drugs, including with respect to the quality and therapeutic consistency standards for the product candidate that will be used in clinical trials. We cannot assure you that it will be able to meet the standards to which it will be held for these purposes.
The FDA or a comparable foreign regulatory authority may also require more information, including additional nonclinical or clinical data to support approval, which may delay or prevent approval and our commercialization plans, or cause us to abandon the development program. Even if we obtain regulatory approval, our product candidates may be approved for fewer or more limited indications than we request, such approval may be contingent on the performance of costly post-marketing clinical trials, or we may not be allowed to include the labeling claims necessary or desirable for the successful commercialization of such product candidate.
Our profits from Gemcabene sales will be limited pursuant to our CVR obligations, and we therefore, may, at any time and in our sole and absolute discretion, discontinue any and all further efforts to develop, divest or otherwise monetize Gemcabene, particularly as a treatment for cardiovascular conditions.
We do not have any further obligation to develop Gemcabene, or to expend any funds or efforts with respect to Gemcabene, to fund, (i) a toxicity study, (ii) a related FDA submission designed to result in the release of the partial clinical hold with respect to Gemcabene, (iii) preparation for an end-of-phase 2 meeting with the FDA, and (iv) consulting costs for up to four former Gemphire employees to support such activities. The expected cost of such activities is based on estimates and assumptions that may prove to be inaccurate.
54
Our profits from Gemcabene sales will be limited pursuant to our CVR obligations. Under the terms of the Current CVR Agreement, CVR holders are entitled to (i) 80% of the Gross Consideration (as defined in the Current CVR Agreement) received from the grant, sale or transfer of rights to Gemcabene as a treatment for cardiovascular conditions and (ii) 10% of the Gross Consideration (as defined in the Current CVR Agreement) received from the grant, sale or transfer of rights to Gemcabene as a treatment for any indication outside of treating cardiometabolic diseases, including COVID-19.
The Phase 2a clinical trial of Gemcabene in Pediatric NAFLD was terminated by the Data and Safety Monitoring Board (DSMB) of the principal investigator following the occurrence of unanticipated problems. This trial termination and the unanticipated problems could have negative impacts on the clinical development of Gemcabene.
On August 10, 2018, the DSMB at Emory University School of Medicine overseeing the non-company, investigator-led open label Phase 2a proof-of-concept trial evaluating Gemcabene in pediatric patients with non-alcoholic fatty liver disease (“NAFLD”) recommended that the trial be terminated due to unanticipated problems. Data on the first three patients who underwent 12 weeks of treatment showed that all three experienced an increase in liver fat content, as measured by MRI-PDFF. Two of the three patients also demonstrated increases in ALT; however, their baseline ALT levels were elevated prior to receiving Gemcabene. The increase in liver fat was deemed an unanticipated problem by the trial investigator because it was an unexpected consistent pattern of worsening of the disease, rather than improvement, creating risk to the patients, which the investigator believed was likely due to the drug. Six subjects had received study medication when the study was halted. Additional data that came to light subsequently showed that during the trial none of the three patients were fully compliant with taking Gemcabene and their life styles could have potentially impacted the findings. Subjects were instructed to self-administer the test-agent daily; however, compliance was significantly compromised as assessed by return of unused tablets and measurement of blood drug levels. All six subjects gained weight and had increased TGs during study treatment. In support of non-compliance, these findings are inconsistent with other Gemcabene trials, and as such, the risk for increased liver fat with Gemcabene treatment is unknown at this time. The six subjects who received Gemcabene were followed in a 12-month safety monitoring period post final-dose, which is now complete. During this follow-on reporting period there were no drug related adverse events reported. There was one serious non-related adverse event of hospitalization of subacute spinal cord infarction/embolism. No deaths or other SAEs were reported.
We cannot assure you that the unanticipated problems observed in the pediatric NAFLD trial will not be seen in future trials nor that serious adverse events (SAEs) will not occur in future trials. We also cannot assure you that the unanticipated problems observed in the pediatric NAFLD trial will not result in the FDA or other regulatory authorities requesting additional analyses of previously completed clinical trials, including the three Phase 2b trials in dyslipidemia completed in 2017 and 2018.
If Gemcabene is associated with adverse effects or undesirable side effects in preclinical testing or clinical trials or has characteristics that are unexpected in preclinical testing or clinical trials, Gemcabene could be less attractive to potential collaborators.
Product candidates may cause undesirable side effects that could delay or prevent their marketing approval, limit the commercial profile of an approved label, or result in significant negative consequences following marketing approval, if any, including marketing withdrawal.
Undesirable side effects caused by any of our product candidates that we may develop or acquire could cause us or the FDA or other regulatory authorities to interrupt, delay or halt our clinical trials and could result in more restrictive labels or the delay or denial of marketing approval by the FDA or other regulatory authorities of such product candidates. Results of our clinical trials could reveal a high and unacceptable severity and prevalence of these or other side effects. In such an event, our trials could be suspended or terminated and the FDA or comparable foreign regulatory authorities could order us to cease further development of or deny approval of our product candidates for any or all targeted indications. In addition, any drug-related side effects could affect patient recruitment or the ability of enrolled patients to complete the trial or result in potential product liability claims. Any of these occurrences may harm our business, financial condition and prospects significantly.
Further, clinical trials by their nature utilize a sample of the potential patient population. With a limited number of patients, rare and severe side effects of our product candidates may only be uncovered with a significantly larger number
55
of patients exposed to the product candidate. If our product candidates receive marketing approval and we or others identify undesirable side effects caused by such product candidates (or any other similar drugs) after such approval, a number of potentially significant negative consequences could result, including:
| ● | | regulatory authorities may withdraw or limit their approval of such product candidates; |
| ● | | regulatory authorities may require the addition of labeling statements, such as a "boxed" warning or a contraindication; |
| ● | | we may be required to recall the product, change the way such product candidates are distributed or administered, conduct additional clinical trials or change the labeling of the product candidates; |
| ● | | regulatory authorities may require a Risk Evaluation and Mitigation Strategy (REMS) plan to mitigate risks, which could include medication guides to be distributed to patients, physician communication plans, or elements to assure safe use, such as restricted distribution methods, patient registries and other risk minimization tools; |
| ● | | we may be subject to regulatory investigations and government enforcement actions; |
| ● | | we may be subject to fines, injunctions or the imposition of civil or criminal penalties; |
| ● | | we may decide to remove such product candidates from the marketplace after they are approved; |
| ● | | the product may be rendered less competitive and sales may decrease; |
| ● | | we could be sued and held liable for injury caused to individuals exposed to or taking its product candidates; and |
| ● | | our reputation may suffer. |
We believe that any of these events could prevent us from achieving or maintaining market acceptance of the affected product candidates and could substantially increase the costs of commercializing our product candidates, if approved, and significantly impact our ability to successfully commercialize our product candidates and generate revenues.
Delays in our clinical trials may lead to a delay in the submission of marketing approval applications and jeopardize our ability to potentially receive approvals and generate revenues from the sale of our products.
We may experience delays in clinical trials. We do not know whether planned clinical trials will begin or enroll subjects on time, need to be redesigned or be completed on schedule, if at all. Clinical trials may be delayed, suspended or terminated for a variety of reasons, such as:
| ● | | delay or failure in reaching agreement with the FDA or a comparable foreign regulatory authority on a trial design that we are able to execute; |
| ● | | delay or failure in obtaining authorization to commence a trial or inability to comply with conditions imposed by a regulatory authority regarding the scope or design of a clinical trial; |
| ● | | inability, delay or failure in identifying and maintaining a sufficient number of trial sites, many of which may already be engaged in competing clinical trial programs; |
| ● | | issues with the manufacture of drug substance for use in clinical trials; |
| ● | | delay or failure in recruiting and enrolling suitable subjects to participate in a trial; |
| ● | | delay or failure in having subjects complete a trial or return for post-treatment follow-up; |
| ● | | clinical sites and investigators deviating from trial protocol, failing to conduct the trial in accordance with regulatory requirements, or dropping out of a trial; |
| ● | | delay or failure in reaching agreement on acceptable terms with prospective clinical research organizations, or CROs, and clinical trial sites, the terms of which can be subject to extensive negotiation and may vary significantly among different CROs and trial sites; |
| ● | | delay or failure in obtaining institutional review board, or IRB, approval to conduct a clinical trial at each site; |
| ● | | delays resulting from negative or equivocal findings of the Data Safety Monitoring Board, or DSMB, if any; |
| ● | | ambiguous or negative results; |
| ● | | decision by the FDA, a comparable foreign regulatory authority, or recommendation by a DSMB to suspend or terminate clinical trials at any time for safety issues or for any other reason; |
56
| ● | | lack of adequate funding to continue the product development program; or |
| ● | | changes in governmental regulations or requirements. |
Any delays in completing our clinical trials will increase our costs, slow down our product candidate development and approval process and jeopardize our ability to commence product sales and generate revenues. Any of these occurrences may significantly harm our business, financial condition and prospects. In addition, many of the factors that cause, or lead to, a delay in the commencement or completion of clinical trials may also ultimately lead to the denial of regulatory approval of our product candidates.
The development of NB-01 is dependent upon securing sufficient quantities of Dioscorea Rhizome and Dioscoreae Nipponicae Rhizoma, which are two plant species native to China.
The therapeutic components of our product candidate, NB-01, consists of Dioscorea Rhizome and Dioscoreae Nipponicae Rhizoma, which are cultivated in China and Korea. We currently secure these components exclusively from Dong-A ST. Our current supply agreement with Dong-A ST expires on September 28, 2023, unless extended by our mutual agreement with Dong-A ST. There can be no assurances that Dioscorea Rhizome and Dioscoreae Nipponicae Rhizoma will continue to grow in sufficient quantities to meet commercial supply requirements or that the countries from which we can secure Dioscorea Rhizome and Dioscoreae Nipponicae Rhizoma will continue to allow the exportation of these components. In the event we are no longer able to obtain these products from Dong-A ST, or in sufficient quantities, we may not be able to produce our proposed products and our business will be adversely affected.
Further, because Dioscorea Rhizome and Dioscoreae Nipponicae Rhizoma are imported from China and Korea, any trade policies or rules that impose conditions or restrictions on the importation of natural products from those regions may restrict or prevent the timely delivery of these products to us, which would adversely affect our business. We may also have difficulty importing these products as a result of the recent COVID-19 pandemic. See the risk factor below entitled “Our business is subject to risks arising from epidemic diseases, such as the recent COVID-19 pandemic.”
We face substantial competition, which may result in others discovering, developing or commercializing products before or more successfully than we do.
The development and commercialization of new products is highly competitive. Our future success depends on our ability to demonstrate and maintain a competitive advantage with respect to the development and commercialization of our product candidates. Our objective is to develop and commercialize new products with superior efficacy, convenience, tolerability and safety. In many cases, the products that we commercialize will compete with existing, market-leading products.
Many of our potential competitors have significantly greater financial, manufacturing, marketing, drug development, technical and human resources than we do. Large pharmaceutical companies, in particular, have extensive experience in clinical testing, obtaining regulatory approvals, recruiting patients and in manufacturing pharmaceutical products. In particular, these companies have greater experience and expertise in securing government contracts and grants to support their research and development efforts, conducting testing and clinical trials, obtaining regulatory approvals to market products, manufacturing such products on a broad scale and marketing approved products. These companies also have significantly greater research and marketing capabilities than we do and may also have products that have been approved or are in late stages of development, and have collaborative arrangements in our target markets with leading companies and research institutions. Established pharmaceutical companies may also invest heavily to accelerate discovery and development of novel compounds or to in-license novel compounds that could make the product that we develop obsolete. As a result of all of these factors, our competitors may succeed in obtaining patent protection and/or FDA approval or discovering, developing and commercializing products before, or more effectively than, we do. In addition, any new product that competes with an approved product must demonstrate compelling advantages in efficacy, convenience, tolerability and safety in order to overcome price competition and to be commercially successful. If we are not able to compete effectively against potential competitors, our business will not grow and our financial condition and operations will suffer.
57
NB-01 and NB-02
There are a number of large pharmaceutical and biotechnology companies that currently market and sell products or are pursuing the development of products for the treatment of painful diabetic neuropathy and for the symptomatic and disease modifying treatment of neurodegenerative diseases, including Alzheimer's disease and tauopathies. Some of these competitive products and therapies are based on scientific approaches that are the same as or similar to our approach and others are based on entirely different approaches. Potential competitors also include academic institutions, government agencies and other public and private research organizations that conduct research, seek patent protection and establish collaborative arrangements for research, development, manufacturing and commercialization.
NB-01 has been in clinical development for the treatment of painful diabetic neuropathy. We are also developing NB-02 for the symptomatic and disease modifying treatment of neurodegenerative diseases, including Alzheimer's disease and tauopathies. For painful diabetic neuropathy, there are no products currently marketed for disease modification, although there are products available to treat painful diabetic neuropathy. For Alzheimer's disease, current symptomatic treatments have limited effectiveness and no disease-modifying therapy is currently available. Some of the currently approved drug therapies are branded and subject to patent protection, and others are available on a generic basis. Many of these approved drugs are well-established therapies and are widely accepted by physicians, patients and third-party payors. Insurers and other third-party payors may also encourage the use of generic products.
Gemcabene —Dyslipidemia
For Gemcabene, the lipid-lowering therapies market is highly competitive, dynamic and dominated by the sale of statin treatments including the cheaper generic versions of statins. Our success will depend, in part, on our ability to obtain a share of the market for our planned indications. Other pharmaceutical companies may develop lipid-lowering therapies for the same indications that compete with Gemcabene, if approved, that do not infringe the claims of our patents, pending patent applications or other proprietary rights, which could adversely affect our business and results of operations. Lipid-lowering therapies currently on the market that would compete with Gemcabene, if approved, are referenced in “Item 1—Business—Competition—Gemcabene—Dyslipidemia” above.
This means that there is significant competition for investigational sites and patients to enroll in clinical studies. Additionally, since some drug candidates may be further along in development, approval of such drug candidates could lead to the FDA and other global health authorities to request and/or require changes to ongoing or future clinical trial designs that could impact timelines and cost.
The biomarkers and pathogenesis of NASH are less understood than the dyslipidemia market and for that reason there are many mechanisms of action under investigation to better understand how to effectively treat the disease. Currently accepted diagnosis of NASH is confirmed through a liver biopsy which is invasive, time consuming and costly. Future growth and evolution of the NASH market may rely on development of less invasive technologies to increase diagnoses rates to broaden the drug treated patient population. Several companies have late stage assets (Phase 3 or outcomes studies) well under way with projected market approval dates in NASH as soon as 2020/2021. For NASH, the market is currently evolving with no approved therapies for the indication across the globe. Current thought leader opinions are pointing to a multiple mechanistic approach to effectively treat NASH.
Several pharmaceutical companies have NASH therapies in development that may be approved for marketing in the United States or outside of the United States. Based on publicly available information, we believe the current therapies in development that would compete with Gemcabene in NASH include but are not limited to:
| ● | | OCALIVA (Obeticholic Acid) (FXR Agonist) being developed by Intercept Pharmaceuticals, Inc.; |
| ● | | Elafibranor (PPAR Agonist) being developed by Genfit SA; |
| ● | | Selonsertib (formerly GS-4997) (ASK1 Inhibitor) being developed by Gilead Sciences, Inc.; |
| ● | | GS-0976 (ACC Inhibitor) being developed by Gilead Sciences, Inc.; |
| ● | | GS-9674 (FXR Agonist) being developed by Gilead; |
| ● | | Cenicriviroc (CVC) (CCR2/CCR5 Inhibitor) being developed by Tobira Therapeutics, Inc. (a wholly-owned subsidiary of Allergan plc); |
58
| ● | | AKR-001 being developed by Akero Therapeutics; |
| ● | | NGM-282 being developed by MGM Biopharmaceuticals; |
| ● | | PXL-770 being developed by Poxel; |
| ● | | EDP-305 being developed by Enanta; |
| ● | | Aramchol (Synthetic Fatty Acid/Bile Acid Conjugate) being developed by Galmed; |
| ● | | MN-001 (5-Lipoxygenase Inhibitor) being developed by MediciNova; |
| ● | | VK2809 (THR-Beta Agonist) being developed by Viking; |
| ● | | BMS-986036 (GFG21) being developed by BMS; |
| ● | | Lanifibranor (PPAR Pan Agonist) being developed by Inventiva; |
| ● | | GR-MD-02 (Galectin-3 Inhibitor) being developed by Galectin Therapeutics; and |
| ● | | MGL-3196 (THR Agonist) being developed by Madrigal. |
ANA001
We expect that, if approved, ANA001 will compete with a number of drugs that are being studied for the treatment of symptoms of COVID-19. In addition to widely distributed vaccines designed to stop the spread of COVID-19, which could adversely affect the addressable population for ANA001, two therapies are currently approved by the FDA for the treatment of symptoms of COVID-19 (remdesivir and Dexamethasone), and three have received EUA from the FDA (baricitinib + remdesivir, Regeneron’s antibody cocktail and bamlanivimab). We are aware of other therapies currently being studied in clinical trials for the treatment of COVID-19, including favipiravir, convalescent plasma, oleandrin, ivermectin and molnupiravir.
In addition to the marketed therapies, we are aware of several companies currently developing and commercializing niclosamide for the treatment of COVID-19 symptoms, including Daewoong, Union Therapeutics, TFF and FirstWave. Approved therapies and additional therapies that may be approved in the near term could significantly and adversely affect the market opportunity for ANA001.
Our commercial success depends upon attaining significant market acceptance of our product candidates, if approved, among hospitals, physicians, patients and healthcare payors.
Even if we obtain regulatory approval for any of our product candidates that we may develop or acquire in the future, the product may not gain market acceptance among hospitals, physicians, health care payors, patients and the medical community. Market acceptance of any of our product candidates for which we receive regulatory approval depends on a number of factors, including:
| ● | | the clinical indications for which the product candidate is approved; |
| ● | | acceptance by major operators of hospitals, physicians and patients of the product candidate as a safe and effective treatment, particularly the ability of our product candidates to establish themselves as a new standard of care in the treatment paradigm for the indications that we are pursuing; |
| ● | | the potential and perceived advantages of our product candidates over alternative treatments as compared to the relative costs of the product candidates and alternative treatments; |
| ● | | the willingness of physicians to prescribe, and patients to take, a product candidate that is based on a botanical source; |
| ● | | the prevalence and severity of any side effects with respect to our product candidates, and any elements that may be imposed by the FDA under a REMS program that could discourage market uptake of the products; |
| ● | | the availability of adequate reimbursement and pricing for any approved products by third party payors and government authorities; |
| ● | | inability of certain types of patients to take our product; |
| ● | | demonstrated ability to treat patients and, if required by any applicable regulatory authority in connection with the approval for target indications, to provide patients with incremental cardiovascular disease benefits, as compared with other available therapies; |
| ● | | the relative convenience and ease of administration of our product candidates, including as compared with other treatments available for approved indications; |
59
| ● | | limitations or warnings contained in the labeling approved by the FDA; |
| ● | | availability of alternative treatments already approved or expected to be commercially launched in the near future; |
| ● | | the effectiveness of our sales and marketing strategies; |
| ● | | guidelines and recommendations of organizations involved in research, treatment and prevention of various diseases that may advocate for alternative therapies; |
| ● | | the willingness of patients to pay out-of-pocket in the absence of third-party coverage; |
| ● | | physicians or patients may be reluctant to switch from existing therapies even if potentially more effective, safe or convenient; |
| ● | | efficacy, safety, and potential advantages compared to alternative treatments; |
| ● | | the ability to offer our product for sale at competitive prices; |
| ● | | the willingness of the target patient population to try new therapies and of physicians to prescribe these therapies; |
| ● | | any restrictions on the use of our product together with other medications; |
| ● | | interactions of our product with other medicines patients are taking; and |
| ● | | the timing of market introduction of our products as well as competitive products. |
There may be delays in getting our product candidates, if approved, on hospital or insurance formularies or limitations on coverages that may be available in the early stages of commercialization for newly approved drugs. If any of our product candidates are approved but fail to achieve market acceptance among hospitals, physicians, patients or health care payors, we will not be able to generate significant revenues, which would have a material adverse effect on our business, prospects, financial condition and results of operations.
Even if we are able to commercialize a future pharmaceutical drug candidate, the profitability of such product candidate will likely depend in significant part on third-party reimbursement practices, which, if unfavorable, would harm our business.
Our ability to commercialize a drug successfully will depend in part on the extent to which coverage and adequate reimbursement will be available from government health administration authorities, private health insurers and other organizations. Government authorities and third-party payors, such as private health insurers and health maintenance organizations, decide which medications they will pay for and establish reimbursement levels. Government authorities and third-party payors have attempted to control costs by limiting coverage and the amount of reimbursement for particular medications. Increasingly, third-party payors are requiring that drug companies provide them with predetermined discounts from list prices and are challenging the prices charged for medical products. We cannot be sure that coverage will be available for any product candidate that we commercialize and, if coverage is available, whether the level of reimbursement will be adequate. Assuming we obtain coverage for our product candidates, if approved, by a third-party payor, the resulting reimbursement payment rates may not be adequate or may require co-payments that patients find unacceptably high. Patients who are prescribed medications for the treatment of their conditions, and their prescribing physicians, generally rely on third-party payors to reimburse all or part of the costs associated with their prescription drugs. Patients are unlikely to use a product candidate, if approved, unless coverage is provided and reimbursement is adequate to cover all or a significant portion of the cost of our products. Therefore, coverage and adequate reimbursement is critical to new product acceptance. If reimbursement is not available or is available only to limited levels, we may not be able to successfully commercialize any product candidate for which we obtain marketing approval.
There may be significant delays in obtaining reimbursement for newly approved drugs, and coverage may be more limited than the purposes for which a product candidate is approved by the FDA or similar regulatory authorities outside the United States. Moreover, eligibility for reimbursement does not imply that any product will be paid for in all cases or at a rate that covers its costs, including research, development, manufacture, sale and distribution. Interim reimbursement levels for a new product, if applicable, may also not be sufficient to cover our costs and may not be made permanent. Reimbursement rates may vary according to the use of the product and the clinical setting in which it is used, may be based on reimbursement levels already set for lower cost medicines and may be incorporated into existing payments for other services. Net prices for products may be reduced by mandatory discounts or rebates required by government healthcare programs or private payors and by any future relaxation of laws that presently restrict imports of
60
medicines from countries where they may be sold at lower prices than in the United States. Third-party payors often rely upon Medicare coverage policy and payment limitations in setting their own reimbursement policies. However, no uniform policy requirement for coverage and reimbursement for drug products exists among third-party payors in the United States. Therefore, coverage and reimbursement for drug products can differ significantly from payor to payor. As a result, the coverage determination process is often a time-consuming and costly process that will require us to provide scientific and clinical support for the use of our products to each payor separately with no assurance that coverage and adequate reimbursement will be applied consistently or obtained in the first instance.
Our inability to promptly obtain coverage and profitable payment rates from both government-funded and private payors for any approved products that we develop could have an adverse effect on our operating results, our ability to raise capital needed to commercialize products and our overall financial condition.
Product liability lawsuits against us could cause us to incur substantial liabilities and could limit commercialization of any product candidate that we may develop.
We face an inherent risk of product liability exposure related to the testing of our product candidates in human clinical trials and will face an even greater risk if we commercially sell any product that we may develop including any nutraceuticals. Product liability claims might be brought against us by patients, healthcare providers or others selling or otherwise coming into contact with any of our products or future product candidate during product testing, manufacturing, marketing or sale. For example, we may be sued on allegations that a product candidate caused injury or that the product is otherwise unsuitable. Any such product liability claims may include allegations of defects in manufacturing, defects in design, a failure to warn of dangers inherent in the product, including as a result of interactions with alcohol or other drugs, negligence, strict liability, and a breach of warranties. Claims could also be asserted under state consumer protection acts.
Nutraceuticals are classified as food ingredients, dietary supplements, or natural health products, and, in most cases, are not necessarily subject to pre-market regulatory approval in the United States. However, if we pursue nutraceutical products, we may, in the future, be subject to various product liability claims, including, among others, claims alleging inadequate instructions for use or inadequate warnings concerning possible side effects and interactions with other substances.
If we cannot successfully defend against claims that our product caused injuries, we could incur substantial liabilities. Regardless of merit or eventual outcome, liability claims may result in:
● | | decreased demand for any product candidate that we are developing; | |
| ● | | injury to our reputation and significant negative media attention; |
| ● | | withdrawal of clinical trial participants; |
| ● | | increased FDA warnings on product labels; |
| ● | | significant costs to defend the related litigation; |
| ● | | substantial monetary awards to trial participants or patients; |
| ● | | distraction of management's attention from our primary business; |
| ● | | loss of revenue; |
| ● | | the inability to commercialize any product candidate that we may develop; |
| ● | | the removal of a product from the market; and |
| ● | | increased insurance costs. |
We do not currently maintain clinical trial insurance coverage for clinical trials. Even if we obtain such insurance in the future, it may not be adequate to cover all liabilities that we may incur. Insurance coverage is increasingly expensive. We may not be able to obtain or maintain insurance coverage at a reasonable cost or in an amount adequate to satisfy any liability that may arise.
61
If we or our third-party manufacturers fail to comply with environmental, health and safety laws and regulations, we could become subject to fines or penalties or incur costs that could have an adverse effect on the success of our business.
Our research and development activities involve the controlled use of potentially hazardous substances, including chemical and biological materials, by us and our third-party manufacturers. Our manufacturers are subject to federal, state and local laws and regulations in the United States and abroad governing laboratory procedures and the use, manufacture, storage, handling and disposal of medical and hazardous materials. Although we believe that our manufacturers' procedures for using, handling, storing and disposing of these materials comply with legally prescribed standards, we cannot completely eliminate the risk of contamination or injury resulting from medical or hazardous materials. As a result of any such contamination or injury, we may incur liability or local, city, state or federal authorities may curtail the use of these materials and interrupt our business operations. In the event of an accident, we could be held liable for damages or penalized with fines, and the liability could exceed our resources. We do not have any insurance for liabilities arising from medical or hazardous materials. Although we maintain workers' compensation insurance to cover us for costs and expenses we may incur due to injuries to our employees resulting from the use of hazardous materials, this insurance may not provide adequate coverage against potential liabilities. Compliance with applicable environmental, health and safety laws and regulations is expensive, and current or future environmental regulations may impair our research, development and production efforts, which could harm our business, prospects, financial condition or results of operations.
If we are unable to establish sales and marketing capabilities to market and sell our product candidates, if they are approved for such marketing, we may be unable to generate any revenue.
In order to market and sell our product candidates in development, we currently intend to build and develop our own sales, marketing and distribution operations. Although our management team has previous experience with such efforts for pharmaceutical products, there can be no assurance that we will be successful in building these operations. The establishment and development of our own commercial sales and marketing teams to discuss any products we may develop will be expensive and time-consuming and could delay any product launch.
If we decide to pursue NB-01 as a nutraceutical product, its success will depend significantly on sales and marketing activities. None of our management team has experience with nutraceutical marketing. Accordingly, our future ability to achieve sales and profits for NB-01 as a nutraceutical product would depend on our ability to attract, train, retain and motivate qualified personnel with sales and marketing expertise. There is a risk that we will be unable to attract, train, retain or motivate such qualified personnel, both near term or in the future, and the failure to do so may severely damage our prospects.
If we are unable to establish adequate sales, marketing and distribution capabilities, we may not be able to generate product revenue and may not become profitable. We will also be competing with many companies that currently have extensive and well-funded sales and marketing operations. If any of our product candidates are approved, we may be unable to compete successfully against these more established companies.
If, in the future, we are unable to establish sales and marketing capabilities or to selectively enter into agreements with third parties to sell and market our product candidates, we may not be successful in commercializing our product candidates if and when they are approved.
We do not have a sales or marketing infrastructure and have no experience in the sale, marketing or distribution of pharmaceutical products. To achieve commercial success for any approved product for which we retain sales and marketing responsibilities, we must either develop a sales and marketing organization or outsource these functions to other third parties. In the future, we may choose to build a focused sales and marketing infrastructure to sell some of our product candidates if and when they are approved.
There are risks involved both with establishing our own sales and marketing capabilities and with entering into arrangements with third parties to perform these services. For example, recruiting and training a sales force is expensive and time consuming and could delay any product launch. If the commercial launch of a product candidate for which we recruit a sales force and establish marketing capabilities is delayed or does not occur for any reason, we would have
62
prematurely or unnecessarily incurred these commercialization expenses. This may be costly, and our investment would be lost if we cannot retain or reposition our sales and marketing personnel.
Factors that may inhibit our efforts to commercialize our product candidates on our own include:
| ● | | our inability to recruit and retain adequate numbers of effective sales and marketing personnel; |
| ● | | the inability of sales personnel to obtain access to physicians or persuade adequate numbers of physicians to prescribe any future pharmaceutical products; and |
| ● | | unforeseen costs and expenses associated with creating an independent sales and marketing organization. |
If we enter into arrangements with third parties to perform sales, marketing and distribution services, our product revenue or the profitability of these product revenue may be lower than if we were to market and sell any products that we develop ourselves. In addition, we may not be successful in entering into arrangements with third parties to sell and market our product candidates or may be unable to do so on terms that are favorable to us. We may have little control over such third parties, and any of them may fail to devote the necessary resources and attention to sell and market our products effectively. If we do not establish sales and marketing capabilities successfully, either on our own or in collaboration with third parties, we will not be successful in commercializing our product candidates.
Any product candidate for which we obtain marketing approval could be subject to marketing restrictions or withdrawal from the market, and we may be subject to penalties if we fail to comply with regulatory requirements or if we experience unanticipated problems with our products.
Any pharmaceutical product candidate for which we obtain marketing approval will be subject to continual requirements of and review by the FDA and other regulatory authorities. These requirements include submissions of safety and other post-marketing information and reports, registration and listing requirements, cGMP requirements, quality assurance and corresponding maintenance of records and documents and requirements regarding the distribution of samples to physicians and recordkeeping. Even if marketing approval of a product candidate is granted, the approval may be subject to limitations on the indicated uses for which the product may be marketed or to the conditions of approval, or contain requirements for costly post-marketing testing and surveillance to monitor the safety or efficacy of the medicine. The FDA closely regulates the post-approval marketing and promotion of drugs to ensure that they are marketed only for the approved indications and in accordance with the provisions of the approved labeling. The FDA imposes stringent restrictions on manufacturers’ communications regarding off-label use and if we do not market our products for their approved indications, we may be subject to enforcement action for off-label marketing and/or promotion.
In addition, later discovery of previously unknown problems with our products, manufacturers or manufacturing processes, or failure to comply with regulatory requirements, may result in, among other things:
| ● | | restrictions on such products, manufacturers or manufacturing processes; |
| ● | | restrictions on the labeling, marketing, distribution or use of a product; |
| ● | | requirements to conduct post-approval clinical trials; |
| ● | | warning or untitled letters; |
| ● | | withdrawal of the products from the market; |
| ● | | refusal to approve pending applications or supplements to approved applications that we submit; |
| ● | | recall of products; |
| ● | | fines, restitution or disgorgement of profits or revenue; |
| ● | | suspension or withdrawal of marketing approvals for the drug products; |
| ● | | refusal to permit the import or export of our products; |
| ● | | product seizure; and |
| ● | | injunctions or the imposition of civil or criminal penalties. |
Any product marketed as a nutraceutical could also be subject to FDA review or adverse action and we could be forced to remove such product from the market.
63
We or any potential collaborator may never receive regulatory approval to market our product candidates outside of the United States.
The activities associated with the development and commercialization of pharmaceutical drugs are subject to comprehensive regulation by the FDA, other regulatory agencies in the United States and by comparable authorities in other countries. Failure to obtain regulatory approval for our product candidates will prevent us or any potential collaborator from commercializing our product candidates as pharmaceutical drugs. We have not received regulatory approval to market any of our product candidates in any jurisdiction, and we do not expect to obtain FDA or any other regulatory approvals to market any of our product candidates for the foreseeable future, if at all. The process of obtaining regulatory approvals is expensive, often takes many years, if approval is obtained at all, and can vary substantially based upon the type, complexity and novelty of the product candidates involved.
We may seek to avail ourselves of mechanisms to expedite and/or reduce the cost for development or approval of any of our product candidates or product candidates we may pursue in the future, such as fast track designation or orphan drug designation, but such mechanisms may not actually lead to a faster or less expensive development or regulatory review or approval process.
We may seek fast track designation, priority review, orphan drug designation, or accelerated approval for any other product candidate we may pursue in the future. For example, if a drug is intended for the treatment of a serious or life-threatening condition and the drug demonstrates the potential to address unmet medical needs for this condition, the drug sponsor may apply for FDA fast track designation. However, the FDA has broad discretion with regard to these mechanisms, and even if we believe a particular product candidate is eligible for any such mechanism, it cannot assure you that the FDA would decide to grant it. Even if we obtain fast track or priority review designation or pursue an accelerated approval pathway, we may not experience a faster and/or less costly development process, review or approval compared to conventional FDA procedures. The FDA may withdraw a particular designation if it believes that the designation is no longer supported by data from our clinical development program.
A breakthrough therapy designation by the FDA for our product candidates may not lead to a faster development or regulatory review or approval process, and it does not increase the likelihood that our product candidates will receive marketing approval.
We may seek a breakthrough designation from FDA for some of our product candidates. Designation as a breakthrough therapy is within the discretion of the FDA. Accordingly, even if we believe one of our product candidates meets the criteria for designation as a breakthrough therapy, the FDA may disagree and instead determine not to make such designation. The receipt of a breakthrough therapy designation for a product candidate may not result in a faster development process, review or approval compared to other drugs and does not assure ultimate approval of the designated product candidate by the FDA. In addition, even if one or more of our product candidates qualifies as breakthrough therapies, the FDA may later decide that the products no longer meet the conditions for qualification or decide that the time period for FDA review or approval will not be shortened.
Current and future legislation may increase the difficulty and cost to obtain marketing approval of and commercialize our product candidates and affect the prices we may obtain.
In the United States and some foreign jurisdictions, there have been a number of legislative and regulatory changes and proposed changes regarding the healthcare system that could prevent or delay marketing approval of our product candidates, restrict post-approval activities and affect our ability to profitably sell any product candidates for which we obtain marketing approval. See the section titled “Item 1—Business—Government Regulation” above.
Legislative and regulatory proposals have been made to expand post-approval requirements and restrict sales and promotional activities for pharmaceutical products. New legislation or regulations may adversely affect the potential for our products as nutraceuticals. We cannot be sure whether additional legislative changes will be enacted, or whether FDA regulations, guidance or interpretations will be changed, or what the impact of such changes on the marketing approvals, if any, of our product candidates may be. In addition, increased scrutiny by the U.S. Congress of the FDA's approval process may significantly delay or prevent marketing approval, as well as subject us to more stringent product labeling and post-marketing conditions and other requirements.
64
If we fail to maintain orphan drug exclusivity for Gemcabene for HoFH, we will have to rely on data and marketing exclusivity for HoFH that is not based on an orphan drug designation, if any, and on our intellectual property rights.
In the United States, we have obtained orphan drug designation for Gemcabene for the treatment of HoFH. We may submit an application to the FDA for other orphan drug designations for Gemcabene such as for the treatment of TG greater than approximately 750 mg/dL (F) or familial partial lipodystrophy under the Orphan Drug Act, the FDA may designate a product as an orphan drug if it is a drug intended to treat a rare disease or condition, defined, in part, as a patient population of fewer than 200,000 in the United States.
In the United States, the company that first obtains FDA approval for a designated orphan drug for the specified rare disease or condition receives orphan drug marketing exclusivity for that drug for a period of seven years. This orphan drug exclusivity prevents the FDA from approving another application, including a full NDA, to market the same drug for the same orphan indication, except in very limited circumstances. For purposes of small molecule drugs, the FDA defines "same drug" as a drug that contains the same active pharmaceutical ingredient (API) and is intended for the same use as the drug in question. A designated orphan drug may not receive orphan drug exclusivity if it is approved for a use that is broader than the indication for which it received orphan designation. In addition, orphan drug exclusive marketing rights in the United States may be lost if the FDA later determines that the request for designation was materially defective or if the manufacturer is unable to assure sufficient quantity of the drug to meet the needs of patients with the rare disease or condition.
The EMA grants orphan drug designation to promote the development of products that may offer therapeutic benefits for life-threatening or chronically debilitating conditions affecting not more than five in 10,000 people in the European Union. Orphan drug designation from the EMA provides ten years of marketing exclusivity following drug approval, subject to reduction to six years if the designation criteria are no longer met.
Even if we are able to obtain and maintain orphan drug exclusivity for Gemcabene for HoFH, the designation may not effectively protect it from competition for HoFH because different drugs can be approved for the same condition. Moreover, even with an orphan drug designation, the FDA can subsequently approve a different formulation of the same API for the same condition if the FDA concludes that the later formulation of the API is safer, more effective or makes a major contribution to patient care.
Governments outside the United States tend to impose strict price controls, which may adversely affect our revenues, if any.
In some countries, particularly the countries of the European Union, the pricing of prescription pharmaceuticals is subject to governmental control. In these countries, pricing negotiations with governmental authorities can take considerable time after the receipt of marketing approval for a product. To obtain reimbursement or pricing approval in some countries, we may be required to conduct a clinical trial that compares the cost-effectiveness of its product candidate to other available therapies. If reimbursement of our products is unavailable or limited in scope or amount, or if pricing is set at unsatisfactory levels, our business could be harmed, possibly materially.
Our relationships with healthcare providers and third-party payors will be subject to applicable anti-kickback, fraud and abuse and other healthcare laws and regulations, which could expose us to criminal sanctions, civil penalties, contractual damages, reputational harm and diminished profits and future earnings, among other penalties and consequences.
Healthcare providers and third-party payors will play a primary role in the recommendation and prescription of any product candidate for which we obtain marketing approval. Our current and future arrangements with third-party payors and customers may expose us to broadly applicable fraud and abuse and other healthcare laws and regulations that may constrain the business or financial arrangements and relationships through which we market, sell and distribute any product candidate for which we obtain marketing approval. Restrictions and obligations under applicable federal and state healthcare laws and regulations are noted in the section “Item 1—Business—Government Regulation” above.
Efforts to ensure that our business arrangements with third parties will comply with applicable healthcare laws and regulations will involve substantial costs. It is possible that governmental authorities will conclude that our business
65
practices may not comply with current or future statutes, regulations or case law involving applicable fraud and abuse or other healthcare laws and regulations. If our operations are found to be in violation of any of these laws or any other governmental regulations that may apply to it, we may be subject to significant civil, criminal and administrative penalties, damages, fines, imprisonment, exclusion of products from government funded healthcare programs, such as Medicare and Medicaid, and the curtailment or restructuring of our operations. If any of the physicians or other healthcare providers or entities with whom we expect to do business is found to be not in compliance with applicable laws, they may be subject to criminal, civil or administrative sanctions, including exclusions from government funded healthcare programs.
We are subject to U.S. and certain foreign export and import controls, sanctions, embargoes, anti-corruption laws, and anti-money laundering laws and regulations. Compliance with these legal standards could impair its ability to compete in domestic and international markets. We can face criminal liability and other serious consequences for violations which can harm its business.
We are subject to export control and import laws and regulations, including the U.S. Export Administration Regulations, U.S. Customs regulations, various economic and trade sanctions regulations administered by the U.S. Treasury Department's Office of Foreign Assets Controls, the U.S. Foreign Corrupt Practices Act of 1977, as amended, the U.S. domestic bribery statute contained in 18 U.S.C. § 201, the U.S. Travel Act, the USA PATRIOT Act, and other state and national anti-bribery and anti-money laundering laws in the countries in which we conduct activities. Anti-corruption laws are interpreted broadly and prohibit companies and their employees, agents, contractors, and other partners from authorizing, promising, offering, or providing, directly or indirectly, improper payments or anything else of value to recipients in the public or private sector. We may engage third parties for clinical trials outside of the United States to sell our products abroad and/or to obtain necessary permits, licenses, patent registrations, and other regulatory approvals. We have direct or indirect interactions with officials and employees of government agencies or government-affiliated hospitals, universities, and other organizations. We can be held liable for the corrupt or other illegal activities of our employees, agents, contractors, and other partners, even if it does not explicitly authorize or have actual knowledge of such activities. Our violations of the laws and regulations described above may result in substantial civil and criminal fines and penalties, imprisonment, the loss of export or import privileges, debarment, tax reassessments, breach of contract and fraud litigation, reputational harm, and other consequences.
Our ability to use our NOLs to offset future taxable income may be subject to certain limitations.
In general, under Section 382 of Internal Revenue Code of 1986, as amended (the “Code”), a corporation that undergoes an "ownership change" is subject to limitations on its ability to utilize its carryforwards to offset future taxable income. Our existing NOL carryforwards, or NOLs, may be subject to limitations arising from previous ownership changes, including in connection with the 2019 and 2020 Mergers. Future changes in our stock ownership, some of which are outside of our control, could result in further ownership changes under Section 382 of the Code. There is also a risk that due to regulatory changes, such as suspensions on the use of NOLs, or other unforeseen reasons, our existing and any future NOLs could expire or otherwise be unavailable to offset future income tax liabilities.
We believe that we have undergone an ownership change as a result of the 2019 and 2020 Mergers. We have begun, but not completed, a preliminary study to confirm whether an ownership change has occurred as a result of the 2019 and 2020 Mergers, but have not conducted a study to assess whether there have been multiple ownership changes since inception due to the significant complexity and cost associated with such a study.
Tax matters, including the changes in corporate tax rates, disagreements with taxing authorities and imposition of new taxes could impact our results of operations and financial condition.
We are subject to income and other taxes in the United States and our operations, plans and results are affected by tax and other initiatives. On December 22, 2017, comprehensive changes to the Code were signed into law, informally titled the Tax Cuts and Jobs Act (the “Tax Act”). The Tax Act included significant changes that could materially impact the taxation of corporations, like us, including among other things, changes to the corporate income tax rate, limitation of the tax deduction for interest expense to business interest income plus 30% of adjusted taxable income (except for certain small businesses), immediate deductions for certain new investments instead of deductions for depreciation expense over time, and modifying or repealing many business deductions and credits (including changes to the orphan
66
drug tax credit and changes to the deductibility of research and experimental expenditures that will be effective in the future). The Tax Act also included a limitation of the deduction for net operating losses (“NOLs”) generated in tax years beginning after December 31, 2017 to 80% of current year taxable income and the general elimination of carrybacks of NOLs generated in taxable years ending after December 31, 2017. However, the Coronavirus Aid, Relief, and Economic Security Act (“CARES Act”) signed into law on March 27, 2020, provided that NOLs generated in a taxable year beginning in 2018, 2019, or 2020, may now be carried back five years. In addition, the 80% taxable income limitation is temporarily removed, allowing NOLs to fully offset net taxable income. Notwithstanding the reduction in the corporate income tax rate, the overall impact of the Tax Act and any future tax reform is uncertain and our business and financial condition could be adversely affected. The impact of the Tax Act and any future tax reform on holders of our common stock is likewise uncertain and could be adverse.
We are also subject to regular reviews, examinations, and audits by the IRS and other taxing authorities with respect to our taxes. Although we believe our tax estimates are reasonable, if a taxing authority disagrees with the positions we have taken, we could face additional tax liability, including interest and penalties. There can be no assurance that payment of such additional amounts upon final adjudication of any disputes will not have a material impact on our results of operations and financial position.
We also need to comply with new, evolving or revised tax laws and regulations. The enactment of or increases in tariffs, or other changes in the application or interpretation of the Tax Act, or on specific products that we may ultimately sell or with which our products compete, may have an adverse effect on our business or on our results of operations.
Inadequate funding for the FDA and other government agencies could hinder their ability to hire and retain key leadership and other personnel, prevent new products and services from being developed or commercialized in a timely manner or otherwise prevent those agencies from performing normal business functions on which the operation of our business may rely, which could negatively impact our business.
The ability of the FDA to review and approve new products can be affected by a variety of factors, including government budget and funding levels, the ability to hire and retain key personnel and accept the payment of user fees, and statutory, regulatory and policy changes. Average review times at the agency have fluctuated in recent years as a result. In addition, government funding of other government agencies on which the combined organization's operations may rely, including those that fund research and development activities is subject to the political process, which is inherently fluid and unpredictable.
Disruptions at the FDA and other agencies may also slow the time necessary for new drugs to be reviewed and/or approved by necessary government agencies, which would adversely affect our business. For example, over the last several years the U.S. government has shut down several times and certain regulatory agencies, such as the FDA, have had to furlough critical FDA and other government employees and stop critical activities. If a prolonged government shutdown occurs, it could significantly impact the ability of the FDA to timely review and process our regulatory submissions, which could have a material adverse effect on our business. Further, upon completion of the 2019 and 2020 Mergers and in our operations as a public company, future government shutdowns could impact our ability to access the public markets and obtain necessary capital in order to properly capitalize and continue our operations.
Federal legislation and actions by state and local governments may permit reimportation of drugs from foreign countries into the United States, including foreign countries where the drugs are sold at lower prices than in the United States, which could adversely affect our operating results.
We may face competition for our product candidates, if approved, from cheaper alternatives sourced from foreign countries that have placed price controls on pharmaceutical products. The Medicare Modernization Act contains provisions that may change U.S. importation laws and expand pharmacists' and wholesalers' ability to import cheaper versions of an approved drug and competing products from Canada, where there are government price controls. These changes to U.S. importation laws will not take effect unless and until the Secretary of Health and Human Services certifies that the changes will pose no additional risk to the public's health and safety and will result in a significant reduction in the cost of products to consumers. The Secretary of Health and Human Services has so far declined to approve a reimportation plan. Proponents of drug reimportation may attempt to pass legislation that would directly allow reimportation under certain circumstances. Legislation or regulations allowing the reimportation of drugs, if enacted,
67
could decrease the price we receive for any product we may develop and adversely affect our future revenues and prospects for profitability.
Risks Related to Dependence on Third Parties
We have relied and will rely on third-party clinical research organizations (CROs) to conduct our preclinical studies and clinical trials. If these CROs do not successfully carry out their contractual duties or meet expected deadlines, we may not be able to obtain regulatory approval for or commercialize our product candidates and our business could be substantially harmed.
We have relied upon and plan to continue to rely upon CROs and clinical data management organizations to monitor and manage data for our ongoing preclinical and clinical programs. Although we control only certain aspects of their activities, we are responsible for ensuring that each of our studies is conducted in accordance with the applicable protocol and legal, regulatory and scientific standards, and our reliance on the CROs does not relieve us of our regulatory responsibilities. We also rely on third parties to conduct our preclinical studies in accordance with Good Laboratory Practice, or GLP, requirements and the Laboratory Animal Welfare Act of 1966 requirements. We, our CROs and our clinical trial sites are required to comply with regulations and current Good Clinical Practices, or GCP, and comparable foreign requirements to ensure that the health, safety and rights of patients are protected in clinical trials, and that data integrity is assured. Regulatory authorities ensure compliance with GCP requirements through periodic inspections of trial sponsors and trial sites. If we, any of our CROs or our clinical trial sites fail to comply with applicable GCP requirements, the clinical data generated in our clinical trials or a specific site may be deemed unreliable and the FDA or comparable foreign regulatory authorities may require us to perform additional clinical trials before approving our marketing applications.
Our CROs are not our employees, and except for remedies available to us under our agreements with such CROs, we cannot control whether or not they devote sufficient time and resources to our ongoing clinical and preclinical programs. If CROs do not successfully carry out their contractual obligations or meet expected timelines or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols, regulatory requirements or for other reasons, our clinical trials may be extended, delayed or terminated and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates. As a result, our results of operations and the commercial prospects for our product candidates would be harmed, our costs could increase and our ability to generate revenues could be delayed.
We rely on third parties to manufacture our product candidates and preclinical and clinical drug supplies.
We have no experience manufacturing our product candidates on a large clinical or commercial scale and have no manufacturing facility. We are currently dependent on Dong-A ST as our sole third party manufacturer for the manufacture of NB-01. We rely completely on third parties to supply and manufacture our preclinical and clinical drug supplies for Gemcabene and ANA001, and we intend to rely on third parties to produce commercial supplies of these product candidates.
We do not own or operate facilities for the manufacture of NB-01 or Gemcabene. We currently have no plans to build our own clinical or commercial scale manufacturing capabilities. We currently work exclusively with Dong-A ST as the sole manufacturer for the production of NB-01 and rely completely on third parties to supply and manufacture our preclinical and clinical drug supplies for Gemcabene and ANA001. To meet our projected needs for clinical supplies to support our activities through regulatory approval and commercial manufacturing, Dong-A ST or our other third party providers will need to provide sufficient scale of production for these projected needs. If any issues arise in the manufacturing and we are unable to arrange for alternative third-party manufacturing sources, we are unable to find an alternative third party capable of reproducing the existing manufacturing method or we are unable to do so on commercially reasonable terms or in a timely manner, we may not be able to complete development of our product candidates, or market or distribute them.
In addition, under FDA's guidelines for botanical drug products, the harvesting and processing of the botanical raw materials that are the basis of our product candidates must be done in compliance with Good Agricultural and Collection
68
Processes, or GACPs. We are relying on Dong-A ST and other third parties to ensure that their practices comply with applicable GACPs.
Reliance on third-party manufacturers entails risks to which we would not be subject if we manufactured our product candidates and preclinical and clinical drug supplies, including:
| ● | | reliance on the third party for regulatory compliance and quality assurance; |
| ● | | the possibility of breach of the manufacturing agreement by the third party because of factors beyond our control (including a failure to synthesize and manufacture our product candidates or any products that we may eventually commercialize in accordance with our specifications); |
| ● | | the possibility of termination or nonrenewal of the agreement by the third party, based on our own business priorities, at a time that is costly or damaging to us; |
| ● | | delay in, or failure to obtain, regulatory approval of any of our product candidates because of the failure by our third-party manufacturer to comply with cGMP or failure to scale up manufacturing processes; and |
| ● | | current manufacturer and any future manufacturers may not be able to manufacture our product candidates at a cost or in quantities or in a timely manner necessary to make commercially successful products. |
If third-party manufacturers do not successfully carry out their contractual obligations or meet expected timelines or if the quality or accuracy of the clinical data they obtain is compromised due to the failure to adhere to our clinical protocols, regulatory requirements or for other reasons, our clinical trials may be extended, delayed or terminated and we may not be able to obtain regulatory approval for or successfully commercialize our product candidates.
We may engage in future acquisitions or in-licenses of technology that could disrupt our business, cause dilution to the combined organization's stockholders and harm our financial condition and operating results.
While we currently have no specific plans to acquire any other businesses or in-license any additional products or technology, we may, in the future, make acquisitions or licenses of, or investments in, companies, products or technologies that we believe are a strategic or commercial fit with its current product candidates and business or otherwise offer opportunities for us. In connection with these acquisitions or investments, the combined organization may:
● | | issue stock that would dilute its stockholders' percentage of ownership; | |
| ● | | expend cash; |
| ● | | incur debt and assume liabilities; and |
| ● | | incur amortization expenses related to intangible assets or incur large and immediate write-offs. |
We also may be unable to find suitable acquisition or license candidates and we may not be able to complete acquisitions or licenses on favorable terms, if at all. If we do complete an acquisition or license, we cannot assure you that it will ultimately strengthen our competitive position or that it will not be viewed negatively by customers, financial markets or investors. Further, future acquisitions or licenses could also pose numerous additional risks to our operations, including:
| ● | | problems integrating the purchased or licensed business, products or technologies; |
| ● | | increases to our expenses; |
| ● | | the failure to have discovered undisclosed liabilities of the acquired or licensed asset or company; |
| ● | | diversion of management's attention from their day-to-day responsibilities; |
| ● | | harm to our operating results or financial condition; |
| ● | | entrance into markets in which we have limited or no prior experience; and |
| ● | | potential loss of key employees, particularly those of the acquired entity. |
We may not be able to complete one or more acquisitions or effectively integrate the operations, products or personnel gained through any such acquisition without a material adverse effect on our business, financial condition and results of operations.
69
We may form or seek strategic alliances or enter into additional licensing arrangements in the future, and we may not realize the benefits of such alliances or licensing arrangements.
We may form or seek strategic alliances, create joint ventures or collaborations or enter into additional licensing arrangements with third parties that we believe will complement or augment our development and commercialization efforts with respect to our products and any future product candidates that we may develop. Any strategic alliance or collaboration may require us to incur non-recurring and other charges, increase our near and long-term expenditures, issue securities that dilute our existing stockholders or disrupt our management and business. Our likely collaborators include large and mid-size pharmaceutical companies, regional and national pharmaceutical companies and biotechnology companies. If we enter into any such arrangements with any third parties, we will likely have limited control over the amount and timing of resources that our collaborators dedicate to the development or commercialization of our products or any future product candidate. Our ability to generate revenues from these arrangements will depend on our collaborators' abilities to successfully perform the functions assigned to them in these arrangements. We cannot be certain that, following a strategic transaction or license, we will achieve the revenue or specific net income that justifies such transaction.
Collaborations involving or product candidates or any future product candidate pose the following risks to us:
| ● | | collaborators have significant discretion in determining the efforts and resources that they will apply to these collaborations; |
| ● | | collaborators may not perform their obligations as expected; |
| ● | | collaborators may not pursue development and commercialization or may elect not to continue or renew development or commercialization programs based on clinical trial results, changes in the collaborator's strategic focus or available funding or external factors such as an acquisition that diverts resources or creates competing priorities; |
| ● | | collaborators may delay clinical trials, provide insufficient funding for a clinical trial program, stop a clinical trial or abandon a product candidate, repeat or conduct new clinical trials or require a new formulation of a product candidate for clinical testing; |
| ● | | collaborators could independently develop, or develop with third parties, products that compete directly or indirectly with our product candidates if the collaborators believe that competitive products are more likely to be successfully developed or can be commercialized under terms that are more economically attractive; |
| ● | | a collaborator with marketing and distribution rights to one or more product candidates may not commit sufficient resources to the marketing and distribution of any such product candidate; |
| ● | | collaborators may not properly maintain or defend our intellectual property rights or may use our proprietary information in such a way as to invite litigation that could jeopardize or invalidate our proprietary information or expose us to potential litigation; |
| ● | | collaborators may infringe the intellectual property rights of third parties, which may expose us to litigation and potential liability; |
| ● | | disputes may arise between the collaborators and us that result in the delay or termination of the research, development or commercialization of our product candidate or that result in costly litigation or arbitration that diverts management’s attention and resources; |
| ● | | we may lose certain valuable rights under circumstances identified in its collaborations, including if it undergoes a change of control; |
| ● | | collaborations may be terminated and, if terminated, may result in a need for additional capital to pursue further development or commercialization of the applicable product candidates; |
| ● | | collaborators may learn about our discoveries and use this knowledge to compete with us in the future; |
| ● | | the results of collaborators' preclinical or clinical studies could harm or impair other development programs; |
| ● | | there may be conflicts between different collaborators that could negatively affect those collaborations and potentially others; |
| ● | | the number and type of our collaborations could adversely affect our attractiveness to future collaborators or acquirers; |
| ● | | collaboration agreements may not lead to development or commercialization of our product candidate in the most efficient manner or at all. If our present or future collaborator were to be involved in a business |
70
combination, the continued pursuit and emphasis on our product development or commercialization program under such collaboration could be delayed, diminished or terminated; and | |||
| ● | | collaborators may be unable to obtain the necessary marketing approvals. |
If future collaboration partners fail to develop or effectively commercialize our product candidates or any future product candidate for any of these reasons, such product candidate may not be approved for sale and our sales of such product candidate, if approved, may be limited, which would have an adverse effect on our operating results and financial condition.
If we are not able to establish new collaborations on commercially reasonable terms, we may have to alter our development and commercialization plans.
We may selectively seek additional third-party collaborators for the development and commercialization of our product candidates. Our likely collaborators for any collaboration arrangements include large and mid-size pharmaceutical companies, regional and national pharmaceutical companies and biotechnology companies. If we enter into any such arrangements with any third parties, we will likely have limited control over the amount and timing of resources that our collaborators dedicate to the development or commercialization of our product candidates. Our ability to generate revenue from these arrangements will depend on our collaborators' abilities to successfully perform the functions assigned to them in these arrangements.
We may be restricted under existing collaboration agreements from entering into future agreements on certain terms with potential collaborators. Collaborations are complex and time-consuming to negotiate and document. In addition, there have been a significant number of recent business combinations among large pharmaceutical companies that have resulted in a reduced number of potential future collaborators.
We may not be able to negotiate collaborations on a timely basis, on acceptable terms, or at all. If we are unable to do so, we may have to curtail the development of the product candidate for which we are seeking to collaborate, reduce or delay our development program or one or more of our other development programs, delay our potential commercialization or reduce the scope of any sales or marketing activities, or increase our expenditures and undertake development or commercialization activities at our own expense. If we elect to increase our expenditures to fund development or commercialization activities on our own, we may need to obtain additional capital, which may not be available to us on acceptable terms or at all. If we do not have sufficient funds, we may not be able to further develop our product candidate or bring it to market and generate product revenue.
Our employees, principal investigators, CROs and consultants may engage in misconduct or other improper activities, including noncompliance with regulatory standards and requirements, which could have a material adverse effect on our business.
We are exposed to the risk that our employees, principal investigators, CROs and consultants may engage in fraudulent conduct or other illegal activity. Misconduct by these parties could include failures to comply with FDA regulations or similar regulations of comparable foreign regulatory authorities, to provide accurate information to the FDA or comparable foreign regulatory authorities, to comply with manufacturing standards we have established, to comply with federal and state healthcare fraud and abuse laws and regulations and similar laws and regulations established and enforced by comparable foreign regulatory authorities, to report financial information or data accurately or to disclose unauthorized activities to us. In particular, sales, marketing and business arrangements in the healthcare industry are subject to extensive laws and regulations intended to prevent fraud, kickbacks, self-dealing and other abusive practices. These laws and regulations may restrict or prohibit a wide range of pricing, discounting, marketing and promotion, sales commission, customer incentive programs and other business arrangements. Employee or third-party misconduct could also involve the improper use of information obtained in the course of clinical trials, which could result in regulatory sanctions and serious harm to our reputation. It is not always possible to identify and deter employee misconduct, and the precautions we take to detect and prevent this activity, such as employee training, may not be effective in controlling unknown or unmanaged risks or losses or in protecting us from governmental investigations or other actions or lawsuits stemming from a failure to be in compliance with such laws or regulations. If any such actions are instituted against us, and we are not successful in defending such action or asserting our rights, those actions could have a significant impact on our business and results of operations, including the imposition of significant fines or other sanctions.
71
Risks Related to Intellectual Property
If we are unable to obtain and maintain sufficient intellectual property rights, our competitive position could be harmed.
We depend on our ability to protect our proprietary technology. We rely on trade secret, patent, copyright and trademark laws, and confidentiality, licensing and other agreements with employees and third parties, all of which offer only limited protection. Our success depends in large part on our ability to obtain and maintain patent protection in the United States and other countries with respect to our proprietary technology and products. Where we have the right to do so under our license agreements, we seek to protect our proprietary position by filing patent applications in the United States and abroad related to our novel technologies and products that are important to our business.
The patent positions of biotechnology and pharmaceutical companies generally are highly uncertain, involve complex legal and factual questions and have in recent years been the subject of much litigation. As a result, the issuance, scope, validity, enforceability and commercial value of our patents, including those patent rights licensed to us by third parties, are highly uncertain.
The steps we have taken to police and protect our proprietary rights may not be adequate to preclude misappropriation of our proprietary information or infringement of our intellectual property rights, both inside and outside the United States. The rights already granted under any of our currently issued patents and those that may be granted under future issued patents may not provide us with the proprietary protection or competitive advantages that we are seeking. If we are unable to obtain and maintain patent protection for our technology and products, or if the scope of the patent protection obtained is not sufficient, our competitors could develop and commercialize technology and products similar or superior to ours, and our ability to successfully commercialize our technology and products may be adversely affected.
With respect to patent rights, we do not know whether any of our pending patent applications for any of our product candidates will result in the issuance of patents that protect our technology or products, or which will effectively prevent others from commercializing competitive technologies and products. Our pending applications cannot be enforced against third parties practicing the technology claimed in such applications unless and until a patent issues from such applications. Further, the examination process may require us or our licensors to narrow the claims, which may limit the scope of patent protection that may be obtained. Although our license agreement with Dong-A ST includes a number of issued patents that are exclusively licensed to us, the issuance of a patent is not conclusive as to its inventorship, scope, validity or enforceability, issued patents that we own or have licensed from third parties may be challenged in the courts or patent offices in the United States and abroad. Such challenges may result in the loss of patent protection, the narrowing of claims in such patents, or the invalidity or unenforceability of such patents, which could limit our ability to stop others from using or commercializing similar or identical technology and products, or limit the duration of the patent protection for our technology and products. Protecting against the unauthorized use of our patented technology, trademarks and other intellectual property rights is expensive, difficult and may, in some cases, not be possible. In some cases, it may be difficult or impossible to detect third party infringement or misappropriation of our intellectual property rights, even in relation to issued patent claims, and proving any such infringement may be even more difficult.
Laws and rulings by U.S. courts make it difficult to predict how patents will be issued or enforced in the biotechnology industry.
Changes in either the patent laws or interpretation of the patent laws in the United States and other countries may have a significant impact on our ability to protect our technology and enforce our intellectual property rights. There have been numerous changes to the patent laws and to the rules of the United States Patent and Trademark Office, or USPTO, which may have a significant impact on our ability to protect our technology and enforce our intellectual property rights. For example, the Leahy-Smith America Invents Act, which was signed into law in 2011, includes a transition from a "first-to-invent" system to a "first- to-file" system, and changes the way issued patents are challenged. Certain changes, such as the institution of inter partes review proceedings, came into effect on September 16, 2012. Substantive changes to patent law associated with the America Invents Act may affect our ability to obtain patents, and, if obtained, to enforce or defend them in litigation or post-grant proceedings, all of which could harm our business.
72
Furthermore, the patent positions of companies engaged in the development and commercialization of biologics and pharmaceuticals are particularly uncertain. Two cases involving diagnostic method claims and "gene patents" have been decided by the Supreme Court. On March 20, 2012, the Supreme Court issued a decision in Mayo Collaborative Services v. Prometheus Laboratories, Inc., or Prometheus, a case involving patent claims directed to measuring a metabolic product in a patient to optimize a drug dosage amount for the patient. According to the Supreme Court, the addition of well-understood, routine or conventional activity such as "administering" or "determining" steps was not enough to transform an otherwise patent ineligible natural phenomenon into patent eligible subject matter. On July 3, 2012, the USPTO issued guidance indicating that process claims directed to a law of nature, a natural phenomenon or an abstract idea that do not include additional elements or steps that integrate the natural principle into the claimed invention such that the natural principle is practically applied and the claim amounts to significantly more than the natural principle itself should be rejected as directed to non-statutory subject matter. On June 13, 2013, the Supreme Court issued its decision in Association for Molecular Pathology v. Myriad Genetics, Inc., or Myriad, a case involving patent claims held by Myriad Genetics, Inc. relating to the breast cancer susceptibility genes BRCA1 and BRCA2. Myriad held that isolated segments of naturally occurring DNA, such as the DNA constituting the BRCA1 and BRCA2 genes, is not patent eligible subject matter, but that complementary DNA, which is an artificial construct that may be created from RNA transcripts of genes, may be patent eligible.
We cannot assure you that our current patent protection and our efforts to seek patent protection for our technology and products will not be negatively impacted by the decisions described above, rulings in other cases or changes in guidance or procedures issued by the USPTO.
Moreover, although the Supreme Court has held in Myriad that isolated segments of naturally occurring DNA are not patent-eligible subject matter, certain third parties could allege that activities that we may undertake infringe other gene-related patent claims, and we may deem it necessary to defend against these claims by asserting non-infringement and/or invalidity positions, or pay to obtain a license to these claims. In any of the foregoing or in other situations involving third-party intellectual property rights, if we are unsuccessful in defending against claims of patent infringement, we could be forced to pay damages or be subjected to an injunction that would prevent us from utilizing the patented subject matter. Such outcomes could harm our business.
We may not be able to protect or practice our intellectual property rights throughout the world.
In jurisdictions where we have not obtained patent protection, competitors may use our intellectual property to develop their own products and further, may export otherwise infringing products to territories where we have patent protection, but where it is more difficult to enforce a patent as compared to the U.S. competitor products may compete with our product candidates, if approved, or any future product candidate in jurisdictions where we do not have issued or granted patents or where we issued or granted patent claims or other intellectual property rights are not sufficient to prevent competitor activities in these jurisdictions. The legal systems of certain countries, particularly certain developing countries, make it difficult to enforce patents and such countries may not recognize other types of intellectual property protection, particularly that relating to pharmaceuticals. This could make it difficult for us to prevent the infringement of its patents or marketing of competing products in violation of our proprietary rights generally in certain jurisdictions. Proceedings to enforce our patent rights in foreign jurisdictions could result in substantial cost and divert its efforts and attention from other aspects of our business.
The laws of some jurisdictions do not protect intellectual property rights to the same extent as the laws in the United States, and many companies have encountered significant difficulties in protecting and defending such rights in such jurisdictions. If we, or our licensors, encounter difficulties in protecting, or are otherwise precluded from effectively protecting, the intellectual property rights important for our business in such jurisdictions, the value of these rights may be diminished and we may face additional competition from others in those jurisdictions. Many countries have compulsory licensing laws under which a patent owner may be compelled to grant licenses to third parties. In addition, many countries limit the enforceability of patents against government agencies or government contractors. In these countries, the patent owner may have limited remedies, which could materially diminish the value of such patent. If we, or any of our licensors, are forced to grant a license to third parties with respect to any patents relevant to our business, our competitive position in the relevant jurisdiction may be impaired and our business and results of operations may be adversely affected.
73
We may become involved in lawsuits to protect or enforce our intellectual property, which could be expensive, time consuming and unsuccessful.
In addition to the possibility of litigation relating to infringement claims asserted against us, we may become a party to other patent litigation and other proceedings, including inter partes review proceedings, post-grant review proceedings, derivation proceedings declared by the USPTO and similar proceedings in foreign countries, regarding intellectual property rights with respect to our current or future technologies or product candidates or products. The cost to us of any patent litigation or other proceeding, even if resolved in our favor, could be substantial. Some of our competitors may be able to sustain the costs of such litigation or proceedings more effectively than we can because of their substantially greater financial resources. Patent litigation and other proceedings may also absorb significant management time. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could impair our ability to compete in the marketplace.
Competitors may infringe or otherwise violate our intellectual property, including patents that may issue to or be licensed by us. As a result, we may be required to file claims in an effort to stop third-party infringement or unauthorized use. Any such claims could provoke these parties to assert counterclaims against us, including claims alleging that we infringe their patents or other intellectual property rights. This can be prohibitively expensive, particularly for a company of our size, and time-consuming, and even if we are successful, any award of monetary damages or other remedy we may receive may not be commercially valuable. In addition, in an infringement proceeding, a court may decide that our asserted intellectual property is not valid or is unenforceable, or may refuse to stop the other party from using the technology at issue on the grounds that our intellectual property does not cover its technology. An adverse determination in any litigation or defense proceedings could put our intellectual property at risk of being invalidated or interpreted narrowly and could put our patent applications at risk of not issuing.
If the breadth or strength of our patent or other intellectual property rights is compromised or threatened, it could allow third parties to commercialize our technology or products or result in our inability to commercialize our technology and products without infringing third-party intellectual property rights. Further, third parties may be dissuaded from collaborating with us.
Interference or derivation proceedings brought by the USPTO or its foreign counterparts may be necessary to determine the priority of inventions with respect to our patent applications, and we may also become involved in other proceedings, such as re-examination proceedings, before the USPTO or its foreign counterparts. Due to the substantial competition in the pharmaceutical space, the number of such proceedings may increase. This could delay the prosecution of our pending patent applications or impact the validity and enforceability of any future patents that we may obtain. In addition, any such litigation, submission or proceeding may be resolved adversely to us and, even if successful, may result in substantial costs and distraction to our management.
Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. Moreover, intellectual property law relating to the fields in which we operate is still evolving and, consequently, patent and other intellectual property positions in our industry are subject to change and are often uncertain. We may not prevail in any of these suits or other efforts to protect its technology, and the damages or other remedies awarded, if any, may not be commercially valuable. During the course of this type of litigation, there could be public announcements of the results of hearings, motions or other interim proceedings or developments. If securities analysts or investors perceive these results to be negative, the market price for the combined organization's common stock could be significantly harmed.
Third parties may initiate legal proceedings alleging that we are infringing their intellectual property rights, the outcome of which would be uncertain and could have a material adverse effect on the success of our business.
Our commercial success depends upon our ability to develop, manufacture, market and sell our product candidates, and to use our proprietary technologies without infringing the proprietary rights of third parties. We may become party to, or threatened with, future adversarial proceedings or litigation regarding intellectual property rights with respect to our products and technology, including interference and various post grant proceedings before the USPTO or non-U.S.
74
opposition proceedings. Third parties may assert infringement claims against us based on existing patents or patents that may be granted in the future.
As a result of any such infringement claims, or to avoid potential claims, we may choose or be compelled to seek intellectual property licenses from third parties. These licenses may not be available on acceptable terms, or at all. Even if we are able to obtain a license, the license would likely obligate us to pay license fees or royalties or both, and the rights granted to us likely would be nonexclusive, which would mean that our competitors also could obtain licenses to the same intellectual property. Ultimately, we could be prevented from commercializing a product candidate or technology or be forced to cease some aspect of our business operations if, as a result of actual or threatened infringement claims, we are unable to enter into licenses of the relevant intellectual property on acceptable terms. Further, if we attempt to modify a product candidate or technology or to develop alternative methods or products in response to infringement claims or to avoid potential claims, we could incur substantial costs, encounter delays in product introductions or interruptions in sales. Ultimately, such efforts could be unsuccessful.
Intellectual property litigation could cause us to spend substantial resources and distract our personnel from their normal responsibilities.
Litigation or other legal proceedings relating to intellectual property claims, with or without merit, is unpredictable and generally expensive and time consuming and is likely to divert significant resources from our core business, including distracting our technical and management personnel from their normal responsibilities. Furthermore, because of the substantial amount of discovery required in connection with intellectual property litigation, there is a risk that some of our confidential information could be compromised by disclosure during this type of litigation. In addition, there could be public announcements of the results of hearings, motions or other interim proceedings or developments and if securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on the price of our common stock and negatively impact our ability to raise additional funds. Such litigation or proceedings could substantially increase our operating losses and reduce the resources available for development activities or any future sales, marketing or distribution activities.
We may not have sufficient financial or other resources to adequately conduct such litigation or proceedings. Some of our competitors may be able to sustain the costs of such litigation or proceedings more effectively than we can because of their greater financial resources and more mature and developed intellectual property portfolios. Accordingly, despite our efforts, we may not be able to prevent third parties from infringing upon or misappropriating or from successfully challenging our intellectual property rights. Uncertainties resulting from the initiation and continuation of patent litigation or other proceedings could have a material adverse effect on our ability to compete in the marketplace.
We may be subject to damages resulting from claims that our employees or we have wrongfully used or disclosed alleged trade secrets of their former employers.
Our employees and consultants have been previously employed at other biotechnology or pharmaceutical companies, including our competitors or potential competitors. Although we are not aware of any claims currently pending against us, we may be subject to claims that these employees, or we have, inadvertently or otherwise used or disclosed trade secrets or other proprietary information or intellectual property of the former employers of our employees. Litigation may be necessary to defend against these claims. Even if we are successful in defending against these claims, litigation could result in substantial costs and be a distraction to management. If we fail in defending such claims, in addition to paying money claims, we may lose valuable intellectual property rights or personnel. A loss of key personnel or their work product could hamper or prevent our ability to commercialize product candidates, which would adversely affect our commercial development efforts.
Our trade secrets are difficult to protect and if we are unable to protect the confidentiality of our trade secrets, our business and competitive position would be harmed.
In addition to seeking patents for some of our technologies and product candidates, we also rely on trade secrets, including unpatented know-how, technology and other proprietary information, to maintain our competitive position. We seek to protect these trade secrets, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, corporate collaborators, outside scientific collaborators, contract
75
manufacturers, consultants, advisors and other third parties. We also enter into confidentiality, non-competition, non-solicitation, and invention assignment agreements with our employees and consultants that obligate them to assign to us any inventions developed in the course of their work for us. However, we cannot guarantee that we have executed these agreements with each party that may have or have had access to our trade secrets or that the agreements we have executed will provide adequate protection. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to seek patent protection on technology relating to our product candidates or obtain adequate remedies for such breaches. As a result, we may be forced to bring claims against third parties, or defend claims that they bring against us, to determine ownership of what we regard as our intellectual property. Monitoring unauthorized disclosure is difficult and we do not know whether the procedures that we have followed to prevent such disclosure are or will be adequate. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the United States may be less willing or unwilling to protect trade secrets. If any of the technology or information that we protect as trade secrets were to be lawfully obtained or independently developed by a competitor, we would have no right to prevent them from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to, or independently developed by, a competitor, our competitive position would be harmed.
Obtaining and maintaining our patent protection depends on compliance with various procedural, documentary, fee payment and other requirements imposed by governmental patent agencies, and our patent protection could be reduced or eliminated for non-compliance with these requirements.
Periodic maintenance fees on any issued patent are due to be paid to the USPTO, and foreign patent agencies in several stages over the lifetime of the patent. The USPTO and various foreign governmental patent agencies require compliance with a number of procedural, documentary, fee payment and other requirements during the patent application process. While an inadvertent lapse can in many cases be cured by payment of a late fee or by other means in accordance with the applicable rules, there are situations in which noncompliance can result in abandonment or lapse of the patent or patent application, resulting in partial or complete loss of patent rights in the relevant jurisdiction. Non-compliance events that could result in abandonment or lapse of a patent or patent application include, but are not limited to, failure to respond to official actions within prescribed time limits, non-payment of fees and failure to properly legalize and submit formal documents. If we or our licensors fail to maintain the patents and patent applications covering our product candidates, our competitors might be able to enter the market, which would have a material adverse effect on our business.
Intellectual property rights do not necessarily address all potential threats to our competitive advantage.
The degree of future protection afforded by our intellectual property rights is uncertain because intellectual property rights have limitations, and may not adequately protect our business or permit us to maintain our competitive advantage. For example:
| ● | | others may be able to make product candidates that are similar to our candidates but that are not covered by the claims of the patents that we own or have exclusively licensed; |
| ● | | we or our future licensors or collaborators might not have been the first to make the inventions covered by the issued patent or pending patent application that we own or have exclusively licensed; |
| ● | | we or our future licensors or collaborators might not have been the first to file patent applications covering certain of our inventions; |
| ● | | others may independently develop similar or alternative technologies or duplicate any of our technologies without infringing our intellectual property rights; |
| ● | | it is possible that our pending patent applications will not lead to issued patents; |
| ● | | issued patents that we own or have exclusively licensed may be held invalid or unenforceable, as a result of legal challenges by our competitors; |
| ● | | our competitors might conduct research and development activities in countries where we do not have patent rights and then use the information learned from such activities to develop competitive products for sale in our major commercial markets; |
| ● | | we may not develop additional proprietary technologies that are patentable; and |
| ● | | the patents of others may have an adverse effect on our business. |
76
Should any of these events occur, they could significantly harm our business, results of operations and prospects.
Risks Related Operations, Employee Matters and Managing Growth
Our business is subject to risks arising from epidemic diseases, such as the recent COVID-19 pandemic.
The recent outbreak of COVID-19 disease, which has been declared by the World Health Organization to be a pandemic, has spread across the globe and is impacting worldwide economic activity. A pandemic, including COVID-19, or other public health epidemic poses the risk that we or our employees, contractors, suppliers, and other partners may be prevented from conducting business activities for an indefinite period of time, including due to spread of the disease within these groups or due to shutdowns that may be requested or mandated by governmental authorities. While it is not possible at this time to estimate the impact that COVID-19 could have on our business, the continued spread of COVID-19 and the measures taken by the governments of countries affected could disrupt the supply chain and the manufacture or shipment of both drug substance and finished drug product for our product candidates for preclinical testing and clinical trials and adversely impact our business, financial condition or results of operations. We often attend and present updates at various medical and investor conferences throughout the year. The COVID-19 pandemic has caused, and is likely to continue to cause, cancellations or reduced attendance of these conferences and we may need to seek alternate methods to present clinical updates and to engage with the medical and investment communities. The spread of COVID-19 may also slow potential enrollment of clinical trials and reduce the number of eligible patients for our clinical trials. The COVID-19 pandemic and mitigation measures may also have an adverse impact on global economic conditions which could have an adverse effect on our business and financial condition and our potential to conduct financings on terms acceptable to us, if at all. The extent to which the COVID-19 pandemic impacts our results will depend on future developments that are highly uncertain and cannot be predicted, including new information that may emerge concerning the severity of the virus and the actions to contain its impact.
We currently have a limited number of employees and our future success depends on our ability to retain our executive officers and to attract, retain and motivate qualified personnel.
Because of the specialized scientific nature of our business, we rely heavily on our ability to attract and retain qualified scientific, technical and managerial personnel. We are highly dependent upon current members of our management team. Our employment relationships with our senior executives are at-will and do not prevent management from terminating their employment with us at any time by providing the requisite advance notice. We intend to increase our technical and management staff as needs arise and supporting resources become available, but the loss of one or more of our senior executive officers could be detrimental to us if we cannot recruit suitable replacements in a timely manner. The competition for qualified personnel in the pharmaceutical field is intense and as a result, we may be unable to continue to attract and retain qualified personnel necessary for the development of our business or to recruit suitable replacement personnel.
We will need to grow the size of our organization, and we may experience difficulties in managing this growth.
As of December 31, 2020, we had 7 full-time employees. As our development and commercialization plans and strategies develop, or as a result of any future acquisitions, we will need additional managerial, operational, development, sales, marketing, financial and other resources. Our management, personnel and systems currently in place will not be adequate to support our future growth. Future growth would impose significant added responsibilities on our employees, including:
| ● | | managing our clinical trials effectively; |
| ● | | identifying, recruiting, maintaining, motivating and integrating additional employees; |
| ● | | managing our internal development efforts effectively while complying with our contractual obligations to licensors, contractors and other third parties; |
| ● | | improving our managerial, development, operational and finance systems; and |
| ● | | expanding our facilities. |
77
As our operations expand, we will need to manage additional relationships with various strategic partners, suppliers and other third parties. Our future financial performance and our ability to commercialize our product candidates and to compete effectively will depend, in part, on our ability to manage any future growth effectively. To that end, we must be able to manage our development efforts and clinical trials effectively and hire, train and integrate additional management, administrative, research and development, and sales and marketing personnel. We may not be able to accomplish these tasks, and our failure to accomplish any of them could prevent us from successfully growing the Company.
We intend to market our product candidates outside of the United States, and if we do, we will be subject to the risks of doing business outside of the United States.
Because we intend to market our product candidates, if approved, outside of the United States, our business is subject to risks associated with doing business outside of the United States. Accordingly, our business and financial results in the future could be adversely affected due to a variety of factors, including:
| ● | | failure to develop an international sales, marketing and distribution system for our products; |
| ● | | changes in a specific country's or region's political and cultural climate or economic condition; |
| ● | | unexpected changes in foreign laws and regulatory requirements; |
| ● | | difficulty of effective enforcement of contractual provisions in local jurisdictions; |
| ● | | inadequate intellectual property protection in foreign countries; |
| ● | | inadequate data protection against unfair commercial use; |
| ● | | trade-protection measures, import or export licensing requirements such as Export Administration Regulations promulgated by the United States Department of Commerce and fines, penalties or suspension or revocation of export privileges; |
| ● | | the effects of applicable foreign tax structures and potentially adverse tax consequences; and |
| ● | | significant adverse changes in foreign currency exchange rates. |
Our business and operations would suffer in the event of system failures or unplanned events.
Despite the implementation of security measures, our internal computer systems and those of our current and future contractors and consultants are vulnerable to damage from computer viruses, unauthorized access, natural disasters, terrorism, war and telecommunication and electrical failures. While we are not aware of any such material system failure, accident or security breach to date, if such an event were to occur and cause interruptions in our operations, it could result in a material disruption of our development programs and our business operations. For example, the loss of clinical trial data from completed or future clinical trials could result in delays in regulatory approval efforts and significantly increase our costs to recover or reproduce the data. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and the further development and commercialization of our product candidates could be delayed.
Furthermore, any unplanned event, such as flood, fire, explosion, tornadoes, earthquake, extreme weather condition, medical epidemics, power shortage, telecommunication failure or other natural or manmade accidents or incidents that result in us being unable to fully utilize the facilities, may have an adverse effect on our ability to operate the business, particularly on a daily basis, and have significant negative consequences on our financial and operating conditions. Loss of access to these facilities may result in increased costs, delays in the development of our product candidates or interruption of our business operations.
We rely significantly on information technology and any failure, inadequacy, interruption or security lapse of that technology or loss of data, including any cyber security incidents, could compromise sensitive information related to our business, prevent us from accessing critical information or expose us to liability which could harm our ability to operate our business effectively and adversely affect our business and reputation.
In the ordinary course of our business, our contract research organizations and other third parties on which we rely collect and store sensitive data, including legally protected patient health information, personally identifiable information about our employees, intellectual property, and proprietary business information. We manage and maintain our
78
applications and data utilizing on-site systems. These applications and data encompass a wide variety of business-critical information, including research and development information and business and financial information.
The secure processing, storage, maintenance and transmission of this critical information is vital to our operations and business strategy. Despite the implementation of security measures, our internal computer systems and those of third parties with which we contract are vulnerable to damage from cyber-attacks, computer viruses, breaches, unauthorized access, interruptions due to employee error or malfeasance or other disruptions, or damage from natural disasters, terrorism, war and telecommunication and electrical failures. Any such event could compromise our networks and the information stored there could be accessed by unauthorized parties, publicly disclosed, lost or stolen. We have measures in place that are designed to detect and respond to such security incidents and breaches of privacy and security mandates. Any such access, disclosure or other loss of information could result in legal claims or proceedings, liability under laws that protect the privacy of personal information, government enforcement actions and regulatory penalties. Unauthorized access, loss or dissemination could also disrupt our operations, including our ability to conduct research, development and commercialization activities, process and prepare Company financial information, manage various general and administrative aspects of our business and damage our reputation, in addition to possibly requiring substantial expenditures of resources to remedy, any of which could adversely affect our business. The loss of clinical trial data could result in delays in our regulatory approval efforts and significantly increase our costs to recover or reproduce the data. In addition, there can be no assurance that we will promptly detect any such disruption or security breach, if at all. To the extent that any disruption or security breach were to result in a loss of, or damage to, our data or applications, or inappropriate disclosure of confidential or proprietary information, we could incur liability and our research, development and commercialization efforts could be delayed.
The pharmaceutical industry is highly competitive and is subject to rapid and significant technological change, which could render our technologies and products obsolete or uncompetitive.
The pharmaceutical industry is highly competitive and is subject to rapid and significant technological change, which could render certain of our products obsolete or uncompetitive. This is particularly true in the development of therapeutics for indications where new products and combinations of products are rapidly being developed that change the treatment paradigm for patients. There is no assurance that our product candidates will be the most effective, have the best safety profile, be the first to market, or be the most economical to make or use. The introduction of competitive therapies as alternatives to our product candidates could dramatically reduce the value of those development projects or chances of successfully commercializing those product candidates, which could have a material adverse effect on our long-term financial success.
We will compete with companies in the United States and internationally, including major pharmaceutical and chemical companies, specialized CROs, research and development firms, universities and other research institutions. Many of our competitors have greater financial resources and selling and marketing capabilities, greater experience in clinical testing and human clinical trials of pharmaceutical products and greater experience in obtaining FDA and other regulatory approvals than we do. In addition, some of our competitors may have lower development and manufacturing costs.
Risks Related to Common Stock
The price of our common stock may be volatile and fluctuate substantially, which could result in substantial losses of our common stock.
The trading price of our common stock is likely to be highly volatile and could be subject to wide fluctuations in response to various factors, some of which are beyond our control, including limited trading volume. In addition to the factors discussed in this "Risk Factors" section, these factors include:
| ● | | adverse results or delays in preclinical studies, clinical trials, regulatory decisions or the development status of our product candidates or any product candidates we may pursue in the future; |
| ● | | our ability to raise sufficient additional funds necessary for the continued development of our product candidates whether through potential collaborative, partnering or other strategic arrangements or otherwise; |
| ● | | our ability to realize any value from Gemcabene, particularly in light of the partial clinical hold and the terminated NAFLD trial; |
79
| ● | | the terms and timing of any future collaborative, licensing or other strategic arrangements that we may establish; |
| ● | | uncertainties created by our future management turnover; |
| ● | | our inability to comply with the minimum listing requirements of the Nasdaq Stock Market LLC; |
| ● | | the timing of achievement of, or failure to achieve, our, or any potential collaborator’s clinical, regulatory and other milestones, such as the commencement of clinical development, the completion of a clinical trial or the receipt of regulatory approval; |
| ● | | decisions to initiate a clinical trial, not initiate a clinical trial, or terminate an existing clinical trial; |
| ● | | adverse regulatory decisions, including failure to receive regulatory approval for our product candidates or regulatory actions requiring or leading to a delay or stoppage of any clinical trials; |
| ● | | the commercial success of any product approved by the FDA or its foreign counterparts; |
| ● | | changes in applicable laws, rules or regulations; |
| ● | | disputes with Pfizer regarding our licensed rights to Gemcabene; |
| ● | | an inability to commercialize Gemcabene before April 16, 2024 as stipulated in the Pfizer license; |
| ● | | adverse developments concerning our manufacturers, suppliers, collaborators and other third parties; |
| ● | | occurrence of health epidemics or contagious diseases, such as COVID-19, and potential effects on our business, clinical trial sites, supply chain and manufacturing facilities; |
| ● | | our failure to commercialize our product candidates; |
| ● | | the success of competitive drugs; |
| ● | | if our patents covering our product candidates expire or are invalidated or are found to be unenforceable, or if some or all of our patent applications do not result in issued patents or result in patents with narrow, overbroad, or unenforceable claims; |
| ● | | additions or departures of key scientific or management personnel; |
| ● | | unanticipated safety concerns related to the use of any product candidates; |
| ● | | our announcements or our competitor's announcements regarding new products, enhancements, significant contracts, acquisitions or strategic partnerships and investments; |
| ● | | the size and growth of our target markets; |
| ● | | our, or companies perceived to be similar to us, failure to meet external expectations or management guidance; |
| ● | | fluctuations in our quarterly financial results or the quarterly financial results of companies perceived to be similar to us; |
| ● | | publication of research reports about us or our industry, recommendations, earning results or estimates or withdrawal of research coverage by securities analysts; |
| ● | | changes in the market valuations of similar companies; |
| ● | | changes in general economic, political and market conditions in any of the regions in which we conduct our business; |
| ● | | changes in our capital structure or dividend policy, future issuances of securities, sales of common stock by officers, directors and significant stockholders or our incurrence of additional debt; |
| ● | | trading volume of our common stock; |
| ● | | changes in accounting practices and ineffectiveness of our internal controls; |
| ● | | disputes, litigation or developments relating to proprietary rights; |
| ● | | timing of milestones and royalty payments; and |
| ● | | other events or factors, many of which are beyond our control. |
In addition, the stock market in general, Nasdaq, and the stock of biopharmaceutical companies in particular, have experienced extreme price and volume fluctuations that have often been unrelated or disproportionate to the operating performance of these companies. Broad market and industry factors may negatively affect the market price of our common stock, regardless of our actual operating performance. In the past, securities class action litigation has often been instituted against companies following periods of volatility in the market price of a company's securities. This type of litigation, if instituted, could result in substantial costs and a diversion of management's attention and resources, which would harm our business, operating results or financial condition.
80
If we are unable to comply with Nasdaq's continued listing requirements, our common stock could be delisted, which could affect our common stock's market price and liquidity and reduce our ability to raise capital.
Our common stock is currently listed on the Nasdaq Capital Market. Nasdaq imposes, among other requirements, continued listing standards including minimum bid, public float and stockholders' equity requirements. If we fail to satisfy the continued listing requirements of the Nasdaq Capital Market, Nasdaq may take steps to delist our common stock. Such a delisting would likely have a negative effect on the price of our common stock and would impair your ability to sell or purchase our common stock when you wish to do so. A delisting would adversely affect the liquidity, trading volume and likely the price of our common stock, causing the value of an investment in us to decrease and having an adverse effect on our business, financial condition and results of operations.
We may enter into financing transactions that are dilutive to our stockholders, impose material restrictions on our business and/or require us to relinquish valuable rights.
Until such time, if ever, as we can generate substantial product revenue, we expect to finance our cash needs through a combination of equity offerings, debt financings, collaborations, strategic alliances, and marketing, distribution or licensing arrangements with third parties. To the extent that we raise additional capital through the sale of equity or convertible debt securities, the ownership interest of current stockholders may be materially diluted, and the terms of such securities could include liquidation or other preferences that adversely affect the rights of our current stockholders. Debt financing and preferred equity financing, if available, may involve agreements that include restrictive covenants that limit our ability to take specified actions, such as incurring additional debt, making capital expenditures or declaring dividends. If we raise funds through collaborations, strategic alliances or marketing, distribution or licensing arrangements with third parties, we may have to relinquish valuable rights to our technologies, future revenue streams, research programs or product candidates or grant licenses on terms that may not be favorable to us. If we are unable to raise additional funds through equity or debt financings or other arrangements when needed, we may be required to delay, scale back or discontinue the development and commercialization of one or more of our product candidates or delay our pursuit of potential in-licenses or acquisitions.
Our share price may decline due to the large number of shares eligible for future sale.
The market price of our common stock could decline as a result of sales of a large number of shares of common stock in the market after the expiration of certain lock-up restrictions imposed on our shareholders in connection with the 2019 Merger, or the perception that such sales could occur. As a condition to the closing of the 2019 Merger, certain stockholders of each of Gemphire and NeuroBo and their affiliates entered into lock-up agreements that restrict their ability to transfer shares of our capital stock for 180 days from the effective time of the 2019 Merger. Such lock-up restrictions expire on or about June 28, 2020.
Upon the expiration of the lock-up agreements described above, all of such shares will be eligible for resale in a public market, subject, in the case of shares held by our affiliates, to volume, manner of sale and other limitations under Rule 144 of the Securities Act. Approximately 87% of our outstanding shares of common stock are currently held by holders we consider to be affiliates.
As restrictions on resale end, the market price of our shares of common stock could drop significantly if the holders of these restricted shares sell them or are perceived by the market as intending to sell them. These factors could also make it more difficult for us to raise additional funds through future offerings of our shares of common stock or other securities.
Our largest shareholder may use its significant interest to take actions not supported by our other shareholders.
As of March 9, 2021, our largest shareholder, E&Investment, Inc. and its affiliates (collectively, the “E&H Entities”), beneficially owned 33.1% of our voting rights. As a result, the E&H Entities may be able to exert a significant influence on the outcome of corporate actions requiring shareholder approval, including mergers, share capital increases and other extraordinary items.
The interests and time horizons of the E&H Entities may differ from those of other shareholders. As a result of its potential influence on our business, the E&H Entities could prevent us from making certain decisions or taking certain actions that would protect the interests of our other shareholders. For example, this concentration of ownership may
81
delay or prevent a change of control of the Company, even in the event that this change of control may benefit other shareholders generally. Similarly, the E&H Entities could prevent us from taking certain actions that would dilute its percentage interest in our shares, even if such actions would generally be beneficial to us and/or to other shareholders. These and other factors related to the E&H Entities’ holding of a significant interest in our shares may reduce the liquidity of our shares and their attractiveness to investors.
Our business could be negatively impacted as a result of shareholder activism, an unsolicited takeover proposal, or a proxy contest.
In recent years, proxy contests and other forms of shareholder activism have been directed against numerous public companies. On March 11, 2021, the E&H Entities publicly disclosed that (i) the E&H Entities entered into a Voting Agreement (the “Voting Agreement”) with Dong-A ST (Dong-A ST, together with the E&H Entities, the “Significant Stockholders”) on March 9, 2021; and (ii) the Significant Stockholders intend, as contemplated by the Voting Agreement, to nominate a slate of directors to be elected to the Board of Directors of NeuroBo (the “Board”) at NeuroBo’s 2021 Annual Meeting of Stockholders and 2022 Annual Meeting of Stockholders and to propose the declassification of the Board. The Significant Shareholders also publicly disclosed that, to accomplish these goals, the Significant Shareholders intend to seek the proxy of a legally sufficient number of shares of NeuroBo’s common stock to take such corporate actions.
If a proxy contest or an unsolicited takeover proposal was made with respect to us, we could incur significant costs in defending the Company, which would have an adverse effect on our financial results. Shareholder activists may also seek to involve themselves in the governance, strategic direction and operations of the Company. Such proposals may disrupt our business and divert the attention of our management and employees, and any perceived uncertainties as to our future direction resulting from such a situation could result in the loss of potential business opportunities, be exploited by our competitors, cause concern to our current or potential customers, and make it more difficult to attract and retain qualified personnel and business partners, all of which could adversely affect our business. In addition, actions of activist shareholders may cause significant fluctuations in our stock price based on temporary or speculative market perceptions or other factors that do not necessarily reflect the underlying fundamentals and prospects of our business.
Provisions in our corporate charter documents and under Delaware law could make an acquisition of us, which may be beneficial to our stockholders, more difficult and may prevent attempts by our stockholders to replace or remove our current management.
Provisions in our certificate of incorporation and the bylaws may discourage, delay or prevent a merger, acquisition or other change in control of us that stockholders may consider favorable, including transactions in which our stockholders might otherwise receive a premium for their shares. These provisions could also limit the price that investors might be willing to pay in the future for shares of our common stock, thereby depressing the market price of our common stock. In addition, because our board is responsible for appointing the members of our management team, these provisions may frustrate or prevent any attempts by stockholders to replace or remove their current management by making it more difficult for stockholders to replace members of our board. Among other things, these provisions:
● | | establish a classified board of directors such that not all members of the board are elected at one time; | |
| ● | | allow the authorized number of our directors to be changed only by resolution of our board of directors; |
| ● | | limit the manner in which our stockholders can remove directors from the board; |
| ● | | establish advance notice requirements for stockholder proposals that can be acted on at stockholder meetings and nominations to our board of directors; |
| ● | | require that stockholder actions must be effected at a duly called stockholder meeting and prohibit actions by our stockholders by written consent; |
| ● | | prohibit our stockholders from calling special meetings; |
| ● | | authorize our board to issue preferred stock without stockholder approval, which preferred stock may include rights superior to the rights of the holders of common stock, and which could be used to institute a shareholder rights plan, or so-called "poison pill," that would work to dilute the stock ownership of a potential hostile acquirer, effectively preventing acquisitions that have not been approved by our board; and |
| ● | | require the approval of the holders of at least two-thirds of the votes that all our stockholders would be entitled to cast to amend or repeal certain provisions of our charter or bylaws. |
82
Moreover, because we are incorporated in Delaware, we are governed by the provisions of Section 203 of the Delaware General Corporation Law, which prohibits a person who owns in excess of 15% of our outstanding voting stock from merging or combining with it for a period of three years after the date of the transaction in which the person acquired in excess of 15% of our outstanding voting stock, unless the merger or combination is approved in a prescribed manner.
An active trading market for our common stock may not be maintained.
Our common stock is currently traded on the Nasdaq Capital Market, but we can provide no assurance that we will be able to maintain an active trading market for our shares on the Nasdaq Capital Market or any other exchange in the future. If there is no active market for our common stock, it may be difficult for our stockholders to sell shares without depressing the market price for the shares or at all.
If securities analysts do not publish research or reports about our business or if they publish negative evaluations of our stock, the price of our stock could decline.
If one or more analysts cover our business and downgrade their evaluations of our stock or publish inaccurate or unfavorable research about our business, the price of our stock could decline. If one or more of these analysts cease to cover our stock, we could lose visibility in the market for our stock, which in turn could cause our stock price and trading volume to decline.
Our executive officers, directors, and their affiliates exercise significant control over us, which will limit the ability of our stockholders to influence corporate matters and could delay or prevent a change in corporate control.
As of December 31, 2020, our officers, directors, and their respective affiliates had beneficial ownership, in the aggregate, of approximately 51.4% of our outstanding common stock. These stockholders, if they act together, may be able to influence our management and affairs and control the outcome of matters submitted to our stockholders for approval, including the election of directors, amendments of our organizational documents, and any merger, consolidation, sale of all or substantially all of our assets or other major corporate transaction. Some of these stockholders acquired some or all of their shares of common stock for substantially less than the current trading price of our common stock, and these stockholders may have interests, with respect to our common stock, that are different from other stockholders. In addition, this concentration of ownership might adversely affect the market price of our common stock, have the effect of delaying, deferring or preventing a change of control of us, or discourage a potential acquirer from making a tender offer or otherwise attempting to obtain control of us.
We are an "emerging growth company" and a "smaller reporting company" and we cannot be certain if the reduced reporting requirements applicable to such companies could make our common stock less attractive to investors.
We are an "emerging growth company", as defined in the JOBS Act. For as long as we continue to be an emerging growth company, we may take advantage of exemptions from various reporting requirements that are applicable to other public companies that are not "emerging growth companies", including exemption from compliance with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act of 2002 (Sarbanes-Oxley Act), reduced disclosure obligations regarding executive compensation in its periodic reports and proxy statements, and exemptions from the requirements of holding a non-binding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. Under the JOBS Act, emerging growth companies can also delay adopting new or revised accounting standards until such time as those standards apply to private companies. We have irrevocably elected not to avail ourselves of this exemption from new or revised accounting standards and, therefore, will be subject to the same new or revised accounting standards as other public companies that are not emerging growth companies.
We will remain an emerging growth company until the earlier of (1) the last day of the fiscal year (a) following the fifth anniversary of the closing of the IPO, (b) in which we have total annual gross revenue of at least $1.07 billion, or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common stock that is held by non-affiliates exceeds $700 million as of the prior June 30th, and (2) the date on which we have issued more than $1 billion in non-convertible debt during the prior three-year period.
83
Even after we no longer qualify as an emerging growth company, we may still qualify as a "smaller reporting company," which would allow us to take advantage of many of the same exemptions from disclosure requirements, including reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements.
We cannot predict if investors will find our common stock less attractive because we may rely on these exemptions. If some investors find our common stock less attractive as a result, there may be a less active trading market for our common stock and our stock price may be more volatile.
We incur increased costs as a result of operating as a public company and our management is required to devote substantial time to compliance initiatives.
The Sarbanes-Oxley Act, the Dodd-Frank Wall Street Reform and Consumer Protection Act, the listing requirements of the stock exchange upon which our common stock is listed and other applicable securities rules and regulations impose various requirements on public companies, including establishment and maintenance of effective disclosure and financial controls and corporate governance practices. Our management and other personnel devote a substantial amount of time to these compliance initiatives. Moreover, these rules and regulations increase our legal and financial compliance costs and make some activities more time-consuming and costly. However, these rules and regulations are often subject to varying interpretations, in many cases due to their lack of specificity, and, as a result, their application in practice may evolve over time as new guidance is provided by regulatory and governing bodies. This could result in continuing uncertainty regarding compliance matters and higher costs necessitated by ongoing revisions to disclosure and governance practices. Stockholder activism, the current political environment and the current high level of government intervention and regulatory reform may lead to substantial new regulations and disclosure obligations, which may lead to additional compliance costs and impact the manner in which we operate our business in ways we cannot currently anticipate.
We are subject to Section 404 of the Sarbanes-Oxley Act and the related rules of the SEC that generally require our management and independent registered public accounting firm to report on the effectiveness of our internal control over financial reporting. However, for so long as we remain an "emerging growth company" as defined in the JOBS Act or a "smaller reporting company", we intend to take advantage of certain exemptions from various reporting requirements that are applicable to public companies that are not emerging growth companies and/or smaller reporting companies, including, but not limited to, for emerging growth companies, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act. Once we are no longer an "emerging growth company" and if our public float is above $75 million as of the last business day of our most recently completed second fiscal quarter or, if before such date, we opt to no longer take advantage of the applicable exemption, we will be required to include an opinion from our independent registered public accounting firm on the effectiveness of our internal control over financial reporting.
To achieve compliance with Section 404, we are required to engage in a process to document and evaluate our internal control over financial reporting, which is both costly and challenging. In this regard, we must dedicate internal resources, hire additional finance and accounting personnel, potentially engage outside consultants and adopt a detailed work plan to assess and document the adequacy of internal control over financial reporting, continue steps to improve control processes as appropriate, validate through testing that controls are functioning as documented and implement a continuous reporting and improvement process for internal control over financial reporting.
During the course of our review and testing, we may identify deficiencies and be unable to remediate them before we must provide the required reports. We or our independent registered public accounting firm may not be able to conclude on an ongoing basis that we have effective internal control over financial reporting, which could harm our operating results, cause investors to lose confidence in our reported financial information and cause the trading price of our stock to fall.
In addition, as a public company we are required to timely file accurate quarterly and annual reports with the SEC under the Exchange Act. In order to report our results of operations and financial statements on an accurate and timely basis, we will depend on CROs to provide timely and accurate notice of their costs to it. Any failure to report our financial results on an accurate and timely basis could result in sanctions, lawsuits, delisting of our shares from Nasdaq or other adverse consequences that would materially harm our business.
84
We have identified material weaknesses in our internal control over financial reporting that could, if not remediated, result in material misstatements in our financial statements or impair our ability to produce accurate and timely consolidated financial statements.
We concluded that there were material weaknesses relating to our internal control over financial reporting relating to accounting for clinical trial expenses and related to accounting for mergers. For more information about these material weaknesses, see Part II, Item 9A (Controls and Procedures) of this report. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting such that there is a reasonable possibility that a material misstatement of the company's annual or interim consolidated financial statements will not be prevented or detected on a timely basis.
Although we have begun to take measures to remediate these material weaknesses, the measures we have taken, and expect to take, to improve our internal controls may not be sufficient to address the issues identified, to ensure that our internal controls are effective or to ensure that the identified material weaknesses will not result in a material misstatement of our annual or interim consolidated financial statements. If we are unable to correct material weaknesses or deficiencies in internal controls in a timely manner, our ability to record, process, summarize and report financial information accurately and within the time periods specified in the rules and forms of the SEC will be adversely affected. This failure could negatively affect the market price and trading liquidity of our common stock, cause investors to lose confidence in our reported financial information, subject us to civil and criminal investigations and penalties, and materially and adversely impact our business and financial condition.
We do not anticipate declaring or paying, in the foreseeable future, any cash dividends on our capital stock and, consequently, the ability of our stockholders to achieve a return on their investment will depend on appreciation in the price of our common stock.
We have never declared or paid any cash dividend on our capital stock and do not currently intend to do so in the foreseeable future. We currently anticipate that we will retain future earnings for the development, operation and expansion of our business. Therefore, the success of an investment in shares of our common stock will depend upon any future appreciation in their value. There is no guarantee that shares of our common stock will appreciate in value or even maintain the price at which you purchased them.
Our Bylaws designate the Court of Chancery of the State of Delaware as the sole and exclusive forum for certain types of actions and proceedings that may be initiated by our stockholders, which could limit our stockholders' ability to obtain a favorable judicial forum for disputes with us or our directors, officers or employees.
Our Bylaws provide that, unless we consent in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware will generally be the sole and exclusive forum for any derivative action or proceeding brought on its behalf, any action asserting a claim of breach of a fiduciary duty owed by any of our directors, officers or other employees to us or our stockholders, any action asserting a claim arising pursuant to any provision of the Delaware General Corporation Law, as amended, the certificate of incorporation or the bylaws or any other action asserting a claim governed by the internal affairs doctrine. This provision does not apply to claims arising under the Securities Act and the Exchange Act or any claim for which the federal courts have exclusive jurisdiction. Any person or entity purchasing or otherwise acquiring any interest in shares of our capital stock shall be deemed to have notice of and to have consented to the provisions of the bylaws described above. This choice of forum provision may limit a stockholder's ability to bring a claim in a judicial forum that it finds favorable for disputes with us or our directors, officers or other employees, which may discourage such lawsuits against us and our directors, officers and employees. Alternatively, if a court were to find this provision inapplicable to, or unenforceable in respect of, one or more of the specified types of actions or proceedings, we may incur additional costs associated with resolving such matters in other jurisdictions, which could adversely affect our business and financial condition.
Unstable market and economic conditions may have serious adverse consequences on our business, financial condition and stock price.
The global credit and financial markets have experienced extreme volatility and disruptions in the past several years, including severely diminished liquidity and credit availability, declines in consumer confidence, declines in economic
85
growth, increases in unemployment rates and uncertainty about economic stability. We cannot assure you that further deterioration in credit and financial markets and confidence in economic conditions will not occur. Our general business strategy may be adversely affected by any such economic downturn, volatile business environment or continued unpredictable and unstable market conditions. If the current equity and credit markets deteriorate, or do not improve, it may make any necessary debt or equity financing more difficult, more costly, and more dilutive. Failure to secure any necessary financing in a timely manner and on favorable terms could have a material adverse effect on our growth strategy, financial performance and stock price and could require it to delay or abandon clinical development plans. In addition, there is a risk that one or more of our current service providers, manufacturers and other partners may not survive these difficult economic times, which could directly affect our ability to attain our operating goals on schedule and on budget.
ITEM 1B. |
Not applicable
ITEM 2. |
We currently lease space in Boston, Massachusetts and in Seoul, South Korea. Effective November 11, 2020 we entered into an amendment to our lease agreement for a new corporate headquarters in Boston, consisting of approximately 80 square feet of office space, which will expire on July 31, 2021. Our research facilities in South Korea, which include lab and office space, consists of approximately 574 square feet.
ITEM 3. |
From time to time, we may be involved in various claims and legal proceedings relating to claims arising out of our operations. We are not currently a party to any legal proceedings that, in the opinion of our management, are likely to have a material adverse effect on our business. Regardless of outcome, litigation can have an adverse impact on us because of defense and settlement costs, diversion of management resources and other factors.
ITEM 4. |
Not applicable.
ITEM 5. | MARKET FOR REGISTRANT’S COMMON EQUITY, RELATED STOCKHOLDER MATTERS AND ISSUER PURCHASES OF EQUITY SECURITIES |
Common Stock
Our common stock is listed on The Nasdaq Capital Market (“Nasdaq”) under the symbol “NRBO.” Before December 31, 2019, our common stock was listed on Nasdaq under the symbol “GEMP.”
Stockholders
On April 9, 2021, we had 22,171,182 shares of common stock outstanding and 95 holders of record of our common stock. The transfer agent and registrar for our common stock is Computershare, Inc.
Dividend Policy
We have never declared or paid any dividends on our common stock, and we do not currently intend to pay any dividends on our common stock for the foreseeable future. Any future determination to pay dividends on our common stock will be, subject to applicable law, at the discretion of our Board of Directors and will depend upon, among other factors, our results of operations, financial condition, capital requirements, and contractual restrictions in loan or other agreements.
86
ITEM 6. |
Not applicable.
ITEM 7 | MANAGEMENT’S DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
The following discussion of our financial condition and results of operations should be read in conjunction with the financial statements and notes included in Part II, Item 8 “Consolidated Financial Statements and Supplementary Data” of this report.
Overview
NeuroBo Pharmaceuticals Inc. (the “Company”, “we”, “us”, or “our”) is a clinical-stage biotechnology company focused on developing and commercializing novel pharmaceuticals to treat neurodegenerative disorders affecting millions of patients worldwide. For more information on our business and our four product candidates, ANA001, NB-01, NB-02 and Gemcabene, see “Business-Overview” in Part I, Item 1 of this report.
Recent Developments
Merger with ANA
On December 31, 2020, we acquired ANA Therapeutics, Inc., a Delaware corporation (“ANA”), pursuant to that certain Agreement and Plan of Merger, dated December 31, 2020 (the “2020 Merger Agreement”), by and among NeuroBo, Shelby Merger Sub 1, Inc., a Delaware corporation (the “First Merger Sub”), Shelby Merger Sub 2, LLC, a Delaware limited liability company (the “Second Merger Sub”), ANA, and Akash Bakshi, solely in his capacity as representative of the securityholders of ANA (the “Representative”).
Pursuant to the 2020 Merger Agreement, First Merger Sub merged with and into ANA, pursuant to which ANA was the surviving entity and became a wholly owned subsidiary of ours (the “First Merger”). Immediately following the First Merger, ANA merged with and into Second Merger Sub, pursuant to which Second Merger Sub was the surviving entity (the “Second Merger,” together with the First Merger, the “2020 Merger”). Second Merger Sub is a wholly-owned subsidiary of ours and changed its name to ANA Therapeutics, LLC (“ANA LLC”). The 2020 Merger is intended to qualify as a tax-free reorganization for U.S. federal income tax purposes. Under the terms of the 2020 Merger Agreement, at the closing of the 2020 Merger, we issued 3,243,875 shares of our common stock to the stockholders of ANA pursuant to the terms of the 2020 Merger Agreement.
87
Pursuant to the 2020 Merger Agreement, following the closing of the 2020 Merger, we are obligated to pay milestone payments (each, a “Milestone Payment”) to certain persons identified in the 2020 Merger Agreement (each a “Stakeholder” and collectively, the “Stakeholders”) in the form, time and manner as set forth in the 2020 Merger Agreement, upon the achievement of the following milestone events set forth below by us or any of its affiliates (each, a “Milestone Event”):
Milestone Event | | | | | | Milestone Payment |
First receipt of Marketing Approval (as defined in the 2020 Merger Agreement) from the FDA for any Niclosamide Product (as defined in the 2020 Merger Agreement) | | | $ | $ | 45.0 million | |
Sales Milestones: | | | | | | |
Milestone Event – Worldwide Cumulative Net Sales of a Niclosamide Product | | | | | | |
equal to or greater than: | | | | | | Milestone Payment |
$500 million | | | | $ | 9.0 million | |
$1 billion | | $ | 13.5 million | |||
$3 billion | | $ | 36.0 million | |||
$5 billion | | $ | 72.0 million | |||
Additionally, pursuant to the 2020 Merger Agreement, we are obligated to pay certain single-digit royalty payments (each, a “Royalty Payment”) to the Stakeholders in the form, time and manner as set forth in the 2020 Merger Agreement, following the first commercial sale of each Niclosamide Product (as defined in the 2020 Merger Agreement) on a country-by-country and Niclosamide Product-by-Niclosamide Product basis.
If, at the closing of the 2020 Merger, the fair market value of the Milestone Payments and Royalty Payments (the “Contingent Consideration”), as determined by an independent valuation firm nationally recognized in valuation matters selected by ANA and reasonably acceptable to us (the “Contingent Consideration Value”), is greater than 60% of the Total Consideration Value (as defined in the 2020 Merger Agreement), no more than 60% of each Milestone Payment may be paid in cash and the remainder shall be paid in NeuroBo common stock (any such shares, “Milestone Consideration Shares”). The Milestone Payments and Royalty Payments were not probable of being triggered as of this report.
The 2020 Merger Agreement further provides that NeuroBo and the Representative may agree that the payment of certain Milestone Payments be in the form of NeuroBo’s common stock. If the Representative and NeuroBo have agreed to pay a portion of a Milestone Payment in shares of NeuroBo common stock, any such payments shall be made in accordance with Nasdaq Listing Rule 5635, and if necessary shall have been approved by the stockholders of NeuroBo prior to issuance.
We will have the option to pay the Contingent Consideration in shares of our common stock, but the number of shares of our common stock to be issued in connection with each Milestone Payment or Royalty Payment, if any, are not currently determinable. The number of shares to be issued in such event will be calculated by dividing the Milestone Payment or Royalty Payment due, as applicable, by the average of the closing sale prices per share of the Company’s common stock as reported on the Nasdaq for the ten (10) trading day period ending on the day before such payment is to be made.
Pursuant to the 2020 Merger Agreement, NeuroBo, the Representative and an escrow agent entered into an Escrow Agreement pursuant to which 405,472 shares of NeuroBo common stock will be held in escrow for a period of fifteen months, to secure the indemnification obligations as contemplated by the 2020 Merger Agreement.
Additionally, pursuant to the 2020 Merger Agreement, NeuroBo and certain Stakeholders entered into indemnification support agreements wherein the Stakeholders agreed (i) to be bound by the indemnification obligations set forth in the 2020 Merger Agreement and (ii) to pay a pro rata share of certain losses as set forth in the 2020 Merger Agreement.
The Board unanimously approved the 2020 Merger Agreement and the related transactions, and the consummation of the 2020 Merger was not subject to approval of the NeuroBo stockholders.
88
COVID-19
We are subject to risks and uncertainties as a result of the COVID-19 pandemic. The extent of the impact of the COVID-19 pandemic on our business is highly uncertain and difficult to predict, as the responses that we, other businesses and governments are taking continue to evolve. Furthermore, capital markets and economies worldwide have also been negatively impacted by the COVID-19 pandemic, and it is possible that it could cause a lasting national or global economic recession. Policymakers around the globe have responded with fiscal policy actions to support the healthcare industry and economy as a whole. The magnitude and overall effectiveness of these actions remain uncertain.
To date, except for the adjustments to scientific activity described under “Current Scientific Activity” below, we have not experienced any significant external changes in our business that would have a significant negative impact on our consolidated statements of operations or cash flows.
Exclusive of the development of certain of our proposed therapies, the severity of the impact of the COVID-19 pandemic on our business will depend on a number of factors, including, but not limited to, the duration and severity of the pandemic and the extent and severity of the impact on our service providers, suppliers, contract research organizations and our clinical trials, all of which are uncertain and cannot be predicted. As of the date of issuance of our financial statements, the extent to which the COVID-19 pandemic may in the future materially impact our financial condition, liquidity or results of operations is uncertain.
Current Scientific Activity
In light of the present business environment, including the impact of the COVID-19 pandemic, we are currently conducting the scientific activities described below with a view toward conserving financial resources.
ANA001, our lead drug candidate, is a proprietary oral niclosamide formulation and was developed as a treatment for patients with moderate COVID-19. Niclosamide is a potential oral antiviral and anti-inflammatory agent with a long history of use and well-understood safety in humans. ANA001 is currently being studied in a 60-subject Phase 2 clinical trial conducted in the United States. We plan to initiate its Phase 3 development program for ANA001 in the third quarter of 2021.
NB-01. For NB-01, we have determined that any attempt to conduct Phase 3 clinical trials, as previously announced, would be difficult if not impossible in the short or medium term. Accordingly, in the first quarter of 2020, we directed our contract research organization (“CRO”) partners and other vendors working on the Phase 3 clinical trials of NB-01 to cease all work and we terminated our existing contract arrangements with each of them.
We are currently evaluating our options regarding the NB-01 asset:
| ● | Orphan drug. Development of NB-01 as an orphan drug is among the alternatives we are considering. We have identified one potential rare disease indication for NB-01, but we have not yet conducted feasibility studies for it. We believe that development for such indication would depend on our ability to renegotiate milestone payments under our exclusive license agreement with Dong-A ST to reflect the potential revenue from such indication. See the risk factor entitled “We have determined to postpone the initiation of Phase 3 clinical trials of NB-01 under present circumstances and we have terminated all of our agreements with contract research organizations related to NB-01. We may not be able to successfully develop NB-01 pursuant to other alternatives, including as an orphan drug or as a nutraceutical candidate” in Part I, Item 1A “Risk Factors” of this report. |
| ● | Nutraceutical. We have considered marketing NB-01 as a nutraceutical (non-pharmaceutical) product, and we may re-explore this pathway if the identified rare disease indication for NB-01 does not proceed. |
NB-02. During the third quarter of 2020, we continued work on preparing an Investigational New Drug (“IND”) application to the FDA for NB-02. In order to preserve operating capital, we have postponed continued work on the IND and the first human clinical trials for NB-02 until global health and macroeconomic conditions improve, with a view toward commencing clinical trial activity in the second half of 2021, subject to improvement of the constraints imposed by the COVID-19 pandemic. We are also considering engaging with a strategic partner to assist with clinical trials for NB-02.
89
Gemcabene. In May 2020, we received written communication from the FDA that the clinical development program for Gemcabene remains on a partial clinical hold. We are currently exploring additional therapeutic indications for Gemcabene that may strengthen our pipeline of assets to treat viral diseases, including COVID-19, either as a stand-alone treatment or in combination with ANA001.
As of December 31, 2020, we had cash and cash equivalents of $10.1 million. Operating at such level of scientific activity, we expect that our cash, including the net proceeds from our private placement in January 2021 (as described below under “Liquidity and Capital Resources”), will be adequate to fund operations into the fourth quarter of 2021.
We will need to raise additional capital to fund continued operations at the current level through the fourth quarter of 2021 and beyond. Although we are exploring financing opportunities and carefully monitoring the capital markets, we do not yet have any commitments for additional financing and may not be successful in our efforts to raise additional funds. Any amounts raised will be used for further development of our product candidates and for other working capital purposes and, depending on the amount raised, for commencing clinical activity on NB-02 in the second half of 2021.
If we are unable to raise additional capital (which is not assured at this time, particularly as a result of recent depressed capital market conditions), our long-term business plan may not be accomplished, and we may be forced to cease, reduce, or delay operations. We have some ability to reduce costs further in 2021, thereby potentially lengthening our operational window further into the first quarter of 2022.
Going Concern
The accompanying consolidated financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America (“GAAP”), which contemplate our continuation as a going concern. We have not established a source of revenues and, as such, have been dependent on funding operations through the sale of equity securities. Since inception, we have experienced significant losses and incurred negative cash flows from operations. We expect to incur further losses over the next several years as we develop our business. We have spent, and expect to continue to spend, a substantial amount of funds in connection with implementing our business strategy.
We will need substantial additional funding to support our continuing operations and to pursue our business strategy and, in the meantime, we have reduced scientific activity (as indicated above) and we are carefully controlling expenses. Until such time as we can generate significant revenue from product sales, if ever, we expect to continue to finance our operations primarily through proceeds derived from the sale of equity.
These factors individually and collectively raise substantial doubt about our ability to continue as a going concern. Our financial statements do not include any adjustments or classifications that may result from our possible inability to continue as a going concern. The report of our independent registered public accounting firm on our financial statements for the year ended December 31, 2020 includes an explanatory paragraph regarding the existence of substantial doubt about our ability to continue as a going concern.
Key operating data
We have incurred significant operating losses since inception. Our ability to generate product revenue sufficient to achieve profitability will depend on the successful development and eventual commercialization of one or more of our current or future product candidates. Our net losses were $29.7 million and $21.3 million for the years ended December 31, 2020 and 2019, respectively. To date, we have not generated any revenue from product sales, collaborations with other companies, government grants or any other source, and do not expect to generate any revenue in the foreseeable future.
90
As of December 31, 2020, we had an accumulated deficit of $66.5 million. We expect to continue to incur significant expenses and increasing operating losses for at least the next several years. We expect that our expenses and capital requirements will increase substantially in connection with our ongoing activities, particularly if and as we:
| ● | pursue clinical development for any of our current product candidates; |
| ● | initiate preclinical studies and clinical trials with respect to any additional indications for our current product candidates and any future product candidates that we may pursue; |
| ● | acquire or in-license other product candidates and/or technologies; |
| ● | develop, maintain, expand and protect our intellectual property portfolio; |
| ● | hire additional clinical, scientific and commercial personnel; |
| ● | establish a commercial manufacturing source and secure supply chain capacity sufficient to provide commercial quantities of any product candidates for which we may obtain regulatory approval; |
| ● | seek regulatory approvals for any product candidates that successfully complete clinical trials; |
| ● | establish a sales, marketing and distribution infrastructure and/or enter into partnership arrangements to commercialize any products for which we may obtain regulatory approval; or |
| ● | add administrative, operational, financial and management information systems and personnel, including personnel to support our product development and planned future commercialization efforts, and to support our transition to a public reporting company. |
Components of Results of Operations
Revenue
To date, we have not generated any revenue from product sales, collaborations with other companies, government grants or any other source, and do not expect to generate any revenue in the foreseeable future. If our product development efforts for our product candidates are successful and result in regulatory approval, we may generate revenue in the future from product sales. We cannot predict if, when, or to what extent we will generate revenue from the commercialization and sale of our product candidates. We may never succeed in obtaining regulatory approval for any of our product candidates or generating revenue through alternative marketing strategies such as nutraceuticals.
Cost of Revenue
To date, we have not generated any revenue and thus have no cost of revenue. If our development efforts for our product candidates are successful and result in regulatory approval, we may generate revenue in the future from product sales and have corresponding cost of revenue. We cannot predict if, when, or to what extent we will incur costs from revenue from the commercialization and sale of our product candidates. If we are successful at commercialization, the cost of revenues would include all costs directly related to providing the commercial asset, which would consist primarily of labor, material, facilities, warehousing and other overhead expenses. Cost of revenues would also include depreciation expense related to certain equipment used as part of the commercial asset.
Operating Expenses
Research and Development Expenses
Research and development expenses consist primarily of costs incurred in connection with the development of our product candidates. We expense research and development costs to operations as incurred. These expenses include:
| ● | employee-related expenses, including salaries, related benefits and stock-based compensation, for employees engaged in research and development functions; |
| ● | expenses incurred in connection with the clinical development of our product candidates, including under agreements with third parties, such as consultants and Clinical Research Organizations (“CROs”); |
| ● | the cost of manufacturing and storing drug products for use in our preclinical studies and clinical trials, including under agreements with third parties, such as consultants and Clinical Manufacturing Organizations (“CMOs”); |
91
| ● | facilities, depreciation and other expenses, which include direct or allocated expenses for rent and maintenance of facilities and insurance; |
| ● | costs related to compliance with regulatory requirements; and |
| ● | payments made under third-party licensing agreements. |
We recognize external development costs based on an evaluation of the progress toward completion of specific tasks using information provided to us by our service providers. This process involves reviewing open contracts and purchase orders, communicating with our personnel to identify services that have been performed on our behalf, and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or otherwise notified of actual costs. Nonrefundable advance payments for goods or services to be received in the future for use in research and development activities are recorded as prepaid expenses. Such amounts are recognized as an expense when the goods have been delivered or the services have been performed, or when it is no longer expected that the goods will be delivered, or the services rendered.
Our direct research and development expenses consist primarily of external costs, such as fees paid to outside consultants, CROs, CMOs and research laboratories in connection with our clinical development, quality assurance and quality control processes, manufacturing, and clinical development activities. Our direct research and development expenses also include fees incurred under third-party license agreements. We use our employee and infrastructure resources across multiple research and development projects. We do not allocate employee costs and costs associated with our facilities, including depreciation or other indirect costs, to specific product candidates because these costs are deployed across multiple programs and, as such, are not separately classified. We use internal resources primarily to conduct manufacturing and clinical development activities. These employees work across multiple programs and, therefore, we do not track our costs by product candidate.
Clinical development activities are central to our business model. We do not believe that our historical costs are indicative of the future costs associated with these programs, nor do they represent the costs of other future programs we may initiate. Product candidates in later stages of clinical development generally have higher development costs than those in earlier stages of clinical development, primarily due to the increased size and duration of later-stage clinical trials. We have some control over the timing of these expenses, but costs may be difficult to control once clinical trials have commenced.
The successful development and commercialization of our product candidates are highly uncertain. At this time, we cannot reasonably estimate or know the nature, timing and costs of the efforts that will be necessary to complete the preclinical and clinical development of any of our product candidates. Additionally, because of the risks inherent in novel treatment discovery and development, we cannot reasonably estimate or know:
| ● | the timing and progress of preclinical and clinical development activities; |
| ● | the number and scope of clinical programs that we decide to pursue; |
| ● | our ability to maintain our current development programs and to establish new ones; |
| ● | establishing an appropriate safety profile with IND-enabling studies; |
| ● | successful patient enrollment in, and the initiation and completion of, clinical trials; |
| ● | the successful completion of clinical trials with safety, tolerability and efficacy profiles that are satisfactory to the FDA or any comparable foreign regulatory authority; |
| ● | the receipt of regulatory approvals from applicable regulatory authorities; |
| ● | the timing, receipt and terms of any marketing approvals from applicable regulatory authorities; |
| ● | our ability to establish new licensing or collaboration arrangements; |
| ● | establishing agreements with third-party manufacturers for clinical supply for our clinical trials and commercial manufacturing, if any of our product candidates is approved; |
| ● | development and timely delivery of clinical-grade and commercial-grade drug formulations that can be used in our clinical trials and for commercial launch; |
| ● | obtaining, maintaining, defending and enforcing patent claims and other intellectual property rights; |
| ● | launching commercial sales of our product candidates, if approved, whether alone or in collaboration with others; |
| ● | maintaining a continued acceptable safety profile of the product candidates following commercialization; or |
| ● | the effect of competing technological and market developments. |
92
A change in the outcome of any of these variables with respect to the development of our product candidates could significantly change the costs and timing associated with the development of that product candidate.
Acquired In-Process Research and Development
We include costs to acquire or in-license product candidates in acquired in-process research and development expenses (“IPR&D”). When we acquire the right to develop and commercialize a new product candidate, any up-front payments, or any future milestone payments that relate to the acquisition or licensing of such a right are immediately expensed as acquired in-process research and development in the period in which they are incurred. These costs are immediately expensed provided that the payments do not also represent processes or activities that would constitute a “business” as defined under GAAP, or provided that the product candidate has not achieved regulatory approval for marketing and, and absent obtaining such approval, has no alternative future use. Royalties owed on future sales of any licensed product will be expensed in the period the related revenues are recognized.
General and Administrative Expenses
General and administrative expenses consist primarily of salaries and related costs, including stock-based compensation, for personnel in executive, finance and administrative functions. General and administrative expenses also include direct and allocated facility-related costs as well as professional fees for legal, patent, consulting, investor and public relations, accounting, and audit services.
We anticipate that our general and administrative expenses will increase in the future as a result of accounting, audit, legal, regulatory, compliance, and director and officer insurance costs as we pursue the development of our product pipeline, as well as investor and public relations expenses associated with being a public company.
Loss on note extinguishment
Loss on note extinguishment consists of the loss associated with debt instrument modifications accounted for as debt extinguishments.
Interest Income (Expense), net
Interest Income
Interest income consists of bank interest earned on our cash and cash equivalents.
Interest Expense
Interest expense consists of the interest calculated at a rate of 5% per annum on the convertible notes issued by the Company, which was a private entity formerly known as NeuroBo Pharmaceuticals, Inc. (“Private NeuroBo”) in February 2018 and debt discount amortization attributed to the underlying beneficial conversion features of the convertible notes. The convertible notes were converted into shares of common stock in connection with the 2019 Merger.
Other Expense, net
Other expense, net reflects non-operating expenses associated mainly with realized foreign currency exchange gains and losses.
Income Taxes
The 2020 Merger was intended to qualify as a tax-free reorganization under Section 368 of the Code, the former ANA shareholders owned approximately 16.5% of the outstanding common stock of the Company immediately after the 2020 Merger.
93
The 2019 Merger in the prior year was intended to qualify as a tax-free reorganization under Section 368 of the Code. Based on the exchange ratio under the 2019 Merger Agreement, the former stockholders of Private NeuroBo owned approximately 96.2% of the outstanding common stock of the Company immediately after the 2019 Merger. Therefore, the 2019 Merger was treated as a reverse acquisition for U.S. federal income tax purposes. As a result of the reverse acquisition, the Company became part of the Private NeuroBo (now NeuroBo Therapeutics) consolidated group with the Company as its new parent. In addition, the Company had a short taxable year in 2019 ending on the date of the 2019 Merger. Also, as a result of the 2019 Merger, for U.S. federal income tax purposes, we believe that the Company underwent an ownership change which places a limit on the amount of a company’s net operating losses that can be deducted annually. We have begun, but have not completed, a preliminary analysis to confirm whether such an ownership change has occurred with respect to the 2019 Merger, but have not conducted any such study to assess whether any prior ownership changes have historically occurred.
Since our inception, we have not recorded any income tax benefits for the NOLs we have incurred in each year or for our earned research and development tax credits, as we believe, based upon the weight of available evidence, that it is more likely than not that all of our NOL carryforwards and tax credits will not be realized. As of December 31, 2020, we had federal, state and foreign NOLs carryforwards of $57.5 million, $25.1 million, and $1.0 million, respectively, which may be available to offset future income tax liabilities and begin to expire in 2034 for federal carryforwards incurred prior to 2018, in 2026 for state carryforwards and in 2028 for the foreign carryforwards. Federal operating loss carryforwards incurred beginning in 2018 do not expire. As of December 31, 2020, we also had federal and state research and development tax credit carryforwards of $0.7 million and $0.5 million, respectively, which may be available to offset future tax liabilities and each begin to expire in 2038 for federal and 2033 for state. We have recorded a full valuation allowance against our net deferred tax assets at each balance sheet date. Utilization of the NOL and research and development credit carryforwards may be subject to a substantial annual limitation due to ownership changes that may have occurred previously or that could occur in the future, as provided by Section 382 of the Code, as well as similar state provisions. Ownership changes may limit the amount of NOL and tax credit carry forwards that can be utilized to offset future taxable income and tax, respectively.
Results of Operations
Comparison of the Years Ended December 31, 2020 and December 31, 2019
The following table summarizes our results of operations for the years ended December 31, 2020 and December 31, 2019 (in thousands):
| | For the Year Ended |
| |||||||
| | December 31, | | |||||||
|
| 2020 |
| 2019 |
| Change | | |||
| | | | | | | | | | |
Operating expenses: | | | | | | | | | | |
Research and development | | $ | 4,531 | | $ | 5,324 | | $ | (793) | |
Acquired in‑process research and development | | | 17,339 | | | 12,151 | | | 5,188 | |
General and administrative | | | 7,846 | | | 2,701 | | | 5,145 | |
Total operating expenses | |
| 29,716 | |
| 20,176 | |
| 9,540 | |
Loss from operations | |
| (29,716) | |
| (20,176) | |
| (9,540) | |
Loss on note extinguishment | | | — | | | (1,114) | | | 1,114 | |
Interest income (expense), net | |
| 39 | |
| (22) | |
| 61 | |
Other expense, net | |
| (1) | |
| — | |
| (1) | |
Loss before income taxes | | | (29,678) | | | (21,312) | | | (8,366) | |
Provision for income taxes | | | — | | | — | | | — | |
Net loss | | $ | (29,678) | | $ | (21,312) | | $ | (8,366) | |
| | | | | | | | | | |
Research and Development Expenses
Research and development expenses were $4.5 million for the year ended December 31, 2020 as compared to $5.3 million for the year ended December 31, 2019. The $0.8 million decrease in 2020 was primarily related to the
94
overall reduction of clinical trial activity, as CRO fees, supply and personnel related costs decreased by approximately $2.5 million, offset in part by CRO termination costs associated with the March 2020 determination to postpone Phase 3 clinical trials of NB-01 in the amount of approximately $1.3 million, and $0.4 million for the development of Gemcabene, which was acquired in the 2019 Merger.
Acquired In-process Research and Development
Acquired in-process research and development expenses for the year ended December 31, 2020 amounted to $17.3 million and was attributable to research and development projects of Niclosamide which were in-process at the 2020 Merger date. Acquired in-process research and development expenses for the year ended December 31, 2019 amounted to $12.2 million and was attributable to research and development projects of Gemphire which were in-process at the 2019 Merger date. Current accounting standards require that the fair value of IPR&D with no alternative future use be charged to expense on the acquisition date.
General and Administrative Expenses
General and administrative expenses were $7.8 million for the year ended December 31, 2020, compared to $2.7 million for the year ended December 31, 2019. The increase of $5.1 million was primarily due to the costs associated with operating as a public company when compared to the comparable prior year. The cost increases in 2020 included legal costs of $1.6 million, $1.5 million in director and officer and other insurance premiums, $0.8 million in accounting and auditing fess, $0.6 million in board of director and other public company costs and $0.6 million in stock-based compensation.
Loss on note extinguishment
Non-cash loss on note extinguishment for the year ended December 31, 2019 was $1.1 million stemming from the modification of our convertible notes in October 2019 that were accounted for as a debt extinguishment. There were no modifications of debt instruments in the current year.
Interest Income (Expense), net
Interest income (expense), net for the year ended December 31, 2020 was $39,000 compared to $(22,000) for the year ended December 31, 2019. Interest income for the year ended December 31, 2020 was $39,000 related to cash deposits. The Company did not incur interest expenses during the year ended December 31, 2020 as there was no debt outstanding during the period.
Interest income (expense), net during the year ended December 31, 2019 included non-cash interest expense in connection with our convertible notes of $129,000, offset in part by interest income of $107,000 million related to cash deposits. Non-cash interest expense during the year ended December 31, 2019 consisted of interest on principal in the amount of $25,000 and costs attributed to the underlying beneficial conversion features of the convertible notes in the amount of $104,000.
Other Expense, net
Other expense, net was $1,000 during the year ended December 31, 2020, compared to less than $1,000 during the year ended December 31, 2019. The net increase in other expense, net was due to a nominal increase in net realized foreign currency exchange losses.
Liquidity and Capital Resources
On January 18, 2021, we entered into a Securities Purchase Agreement (the “2021 Purchase Agreement”) with certain institutional and accredited investors, pursuant to which we, in a private placement (the “2021 Private Placement”), agreed to issue and sell an aggregate of 2,500,000 shares (the “2021 Shares”) of our common stock, par value $0.001 per share at a purchase price of $4.00 per share, and warrants to purchase an aggregate of 2,500,000 shares of common stock (the “2021 Warrants”), resulting in total gross proceeds to us in the amount of $10.0 million, before
95
deducting placement agent fees and offering expenses. The 2021 Warrants have an initial exercise price of $6.03 per share. The 2021 Private Placement closed on January 21, 2021.
On April 13, 2020, we entered into a Securities Purchase Agreement with an institutional investor, pursuant to which we sold in a registered direct offering (the “Registered Offering”) 750,000 shares of our common stock, at an offering price of $10.00 per share. The Registered Offering resulted in gross proceeds of $7.5 million, before deducting the placement agent’s fees and related offering expenses.
Prior to the 2019 Merger, Private NeuroBo funded operations with proceeds from sales of preferred stock and proceeds from the issuance of convertible debt. Prior to the 2019 Merger, Private NeuroBo received net proceeds of $40.9 million from sales of preferred stock and $0.5 million from the sales of convertible notes which were converted into shares of Private NeuroBo common stock, effective immediately prior to the closing of the 2019 Merger.
ln August 2019, Private NeuroBo issued an aggregate of 3,463,593 shares of Series B preferred stock (as adjusted for the exchange ratio in the 2019 Merger Agreement) at a purchase price of $7.00 per share, for aggregate gross consideration of approximately $24.2 million. On December 30, 2019, each share of Series B preferred stock then outstanding was converted into common stock in accordance with the terms of the 2019 Merger Agreement.
Since inception, we have experienced significant losses and incurred negative cash flows from operations. We expect to incur further losses over the next several years as we develop our business. We have spent, and expect to continue to spend, a substantial amount of funds in connection with implementing our business strategy.
We will need substantial additional funding to support our continuing operations and to pursue our business strategy and, in the meantime, we have reduced scientific activity, as described under “Overview–Current Scientific Activity–NB-01” above, and we are carefully controlling expenses. In the first quarter of 2020, in connection with the reduced scientific activity, we directed our CRO partners and other vendors working on the Phase 3 clinical trials of NB-01 to cease all work and have terminated our existing contract arrangements with each of them.
As of December 31, 2020, we had cash and cash equivalents of $10.1 million. Operating at such level of scientific activity, we expect that our cash, including the net proceeds from the Registered Offering and 2021 Private Placement, will be adequate to fund operations into the fourth quarter of 2021.
We will need to raise additional capital to fund continued operations at the current level through the fourth quarter of 2021 and beyond. Although we are exploring financing opportunities and carefully monitoring the capital markets, we do not yet have any commitments for additional financing and may not be successful in our efforts to raise additional funds. If we are unable to raise additional capital (which is not assured at this time), our long-term business plan may not be accomplished, and we may be forced to cease, reduce, or delay operations. We have some ability to reduce costs further in 2021, thereby potentially lengthening our operational window further into the first quarter of 2022.
Cash Flows
The following table summarizes our sources and uses of cash for each of the periods presented (in thousands):
| | For the Year Ended | |||||
| | December 31, | |||||
|
| 2020 | | 2019 |
| ||
| | (in thousands) | |||||
Net cash used in operating activities | | $ | (10,764) | | $ | (7,039) | |
Net cash provided by (used in) investing activities | |
| 69 | | | (6,057) | |
Net cash provided by financing activities | |
| 6,858 | | | 24,167 | |
Net (decrease) increase in cash and restricted cash | | $ | (3,837) | | $ | 11,071 | |
96
Operating Activities
During the year ended December 31, 2020, operating activities used $10.8 million of cash, primarily resulting from our net loss of $29.7 million offset by non-cash expenses related to IPR&D, stock-based compensation and depreciation in the aggregate of $18.1 million. Net cash provided by changes in our operating assets and liabilities for the year ended December 31, 2020 was $0.8 million which primarily consisted of a net increase in accounts payable and accrued expenses. The net increase in accounts payable and accrued expenses was primarily attributed to the timing of vendor invoicing and payments and increases in clinical trial termination costs.
During the year ended December 31, 2019, operating activities used $7.0 million of cash, primarily resulting from our net loss of $21.3 million offset by non-cash expenses related to interest in connection with our convertible notes, the extinguishment loss stemming from the modification of our convertible notes, IPR&D, stock-based compensation and depreciation in the aggregate of $13.5 million. Net cash provided by changes in our operating assets and liabilities for the year ended December 31, 2019 was $0.7 million which consisted of a net decrease in prepaid expenses and other current assets of approximately $0.8 million, offset in part by a net decrease in our accounts payable and accrued expenses of $0.1 million as adjusted for the net liabilities assumed in connection with the 2019 Merger. The net decrease in prepaid expenses and other current assets was primarily due to utilization of clinical research organization deposits for clinical activities. The net decrease in accounts payable and accrued expenses was primarily attributed to the 2019 Merger and the timing of vendor invoicing and payments.
Investing Activities
During the year ended December 31, 2020, net cash provided by investing activities was $0.1 million. Investing activities during the period consisted mainly of cash received, net of transaction costs paid, in connection with the 2020 Merger in the amount of $0.1 million. Purchases of property and equipment in the amount of $4,000 comprised the balance of investing activities during the period.
During the year ended December 31, 2019, net cash used in investing activities was $6.1 million. Investing activities during the period consisted mainly of transaction costs paid in connection with the 2019 Merger, net of cash acquired, in the amount of $5.8 million. Purchases of property and equipment in the amount of $0.2 million comprised the balance of investing activities during the period.
Financing Activities
During the year ended December 31, 2020, net cash provided by financing activities was $6.9 million, consisting of proceeds from the Registered Offering of $6.8 million, net of issuance costs, and from the exercise of stock options of $0.1 million.
During the year ended December 31, 2019, net cash provided by financing activities was $24.2 million, consisting primarily of net proceeds from the sale of Series B preferred stock.
Funding Requirements
We expect our expenses to increase substantially over time in connection with our ongoing activities, particularly as we advance the preclinical activities and clinical trials of our product candidates. The timing and amount of our preclinical and clinical expenditures will depend largely on:
| ● | the availability of capital; |
| ● | the scope, number, initiation, progress, timing, costs, design, duration, any potential delays, and results of clinical trials and nonclinical studies for our current or future product candidates; |
| ● | the clinical development plans we establish for our product candidates; |
| ● | the number and characteristics of product candidates and programs that we develop or may in-license; |
| ● | the outcome, timing and cost of regulatory reviews, approvals or other actions to meet regulatory requirements established by the FDA and comparable foreign regulatory authorities, including the potential for the FDA or |
| comparable foreign regulatory authorities to require that we perform more studies for our product candidates than those that we currently expect; |
| ● | our ability to obtain marketing approval for our product candidates; |
| ● | the cost of filing, prosecuting, defending and enforcing our patent claims and other intellectual property rights covering our product candidates, including any such patent claims and intellectual property rights that we have licensed pursuant to the terms of our license agreement; |
| ● | our ability to maintain, expand and defend the scope of our intellectual property portfolio, including the cost of defending intellectual property disputes, including patent infringement actions brought by third parties against us or our product candidates; |
| ● | the cost and timing of completion of commercial-scale outsourced manufacturing activities with respect to our product candidates; |
| ● | our ability to establish and maintain licensing, collaboration or similar arrangements on favorable terms and whether and to what extent we retain development or commercialization responsibilities under any new licensing, collaboration or similar arrangement; |
| ● | the cost of establishing sales, marketing and distribution capabilities for any product candidates for which we may receive regulatory approval in regions where we choose to commercialize our products on our own; |
| ● | the success of any other business, product or technology that we acquire or in which we invest; |
| ● | the costs of acquiring, licensing or investing in businesses, product candidates and technologies; and |
| ● | our need and ability to hire additional management and scientific and medical personnel. |
We expect that, with current levels of scientific activity, our existing cash and cash equivalents will be sufficient to fund our operating expenses, capital expenditure requirements into the fourth quarter of 2021. We have based this estimate on assumptions that may prove to be wrong, and we could exhaust our available capital resources sooner than we expect.
Critical Accounting Policies and Significant Judgments and Estimates
Our consolidated financial statements are prepared in accordance with U.S. GAAP. The preparation of our consolidated financial statements and related disclosures requires us to make estimates and judgments that affect the reported amounts of assets, liabilities, costs, and expenses, and related disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of expenses during the reporting period. On an on-going basis, our management evaluates its estimates, including those related to accounting for clinical trials, income taxes including the valuation allowance for deferred tax assets, accrued expenses, contingencies and stock-based compensation. We base our estimates on historical experience, known trends and events, and on various other assumptions that are believed to be reasonable under the circumstances, the results of which form the basis for making judgements about the carrying values of assets and liabilities. Actual results could differ from those estimates. Changes in estimates are reflected in reported results in the period in which they become known.
While our significant accounting policies are described in more detail in Note 2 to our consolidated financial statements included elsewhere in this report, we believe that the following accounting policies are those most critical to the judgments and estimates used in the preparation of our consolidated financial statements.
Research and Development Expenses
Research and development costs are charged to expense as incurred. Research and development expenses may comprise of costs incurred in performing research and development activities, including clinical trial costs, manufacturing costs for both clinical and pre-clinical materials as well as other contracted services, license fees, and other external costs. Nonrefundable advance payments for goods and services that will be used in future research and development activities are expensed when the activity is performed or when the goods have been received, rather than when payment is made, in accordance with ASC 730, Research and Development.
As part of the process of preparing our consolidated financial statements, we are required to estimate our accrued research and development expenses. This process involves reviewing open contracts and purchase orders, communicating with applicable personnel to identify services that have been performed on our behalf and estimating the level of service performed and the associated cost incurred for the service when we have not yet been invoiced or
98
otherwise notified of actual costs. Certain of our service providers invoice us in arrears for services performed, on a pre-determined schedule or when contractual milestones are met; however, some service providers require advance payments. We make estimates of our accrued and prepaid expenses as of each balance sheet date in the consolidated financial statements based on facts and circumstances known to us at that time. We periodically confirm the accuracy of these estimates with the service providers and make adjustments, if necessary. Examples of estimated accrued research and development expenses include fees paid to:
| ● | vendors in connection with preclinical development activities; |
| ● | CROs and investigative sites in connection with preclinical studies and clinical trials; and |
| ● | CMOs in connection with the production of preclinical and clinical trial materials. |
We base the expense recorded related to external research and development on our estimates of the services received and efforts expended pursuant to quotes and contracts with multiple CMOs and CROs that supply, conduct and manage preclinical studies and clinical trials on our behalf. The financial terms of these agreements are subject to negotiation, vary from contract to contract and may result in uneven payment flows. There may be instances in which payments made to our vendors will exceed the level of services provided and result in a prepayment of the expense. Payments under some of these contracts depend on factors such as the successful enrollment of patients and the completion of clinical trial milestones. In accruing service fees, we estimate the time period over which services will be performed and the level of effort to be expended in each period. If the actual timing of the performance of services or the level of effort varies from the estimate, we adjust the accrual or the amount of prepaid expenses accordingly. Although we do not expect our estimates to be materially different from amounts actually incurred, our understanding of the status and timing of services performed relative to the actual status and timing of services performed may vary and may result in reporting amounts that are too high or too low in any particular period.
Acquired In-Process Research and Development Expenses
We include costs to acquire or in-license product candidates in acquired in-process research and development expenses. These costs are immediately expensed provided that the payments do not also represent processes or activities that would constitute a “business” as defined under GAAP or provided that the product candidate has not achieved regulatory approval for marketing and absent obtaining such approval, has no alternative future use. Royalties owed on future sales of any licensed product will be expensed in the period the related revenues are recognized.
Stock-Based Compensation
We account for stock-based compensation in accordance with the provisions of ASC 718, Compensation — Stock Compensation (“ASC 718”). Accordingly, compensation costs related to equity instruments granted are recognized at the grant-date fair value. We record forfeitures when they occur. Stock-based compensation arrangements to non-employees are accounted for in accordance with the applicable provisions of ASC 718 using a grant date fair value approach.
The fair value of each stock option grant is estimated on the date of grant using the Black-Scholes option-pricing model, which uses as inputs the fair value of our common stock and assumptions we make for the volatility of the common stock, the expected term of the stock options, the risk-free interest rate for a period that approximates the expected term of the stock options and the expected dividend yield.
We adopted Accounting Standards Update (“ASU”) No. 2016-02, Leases (Topic 842) (“ASU 2016-02”) in the third quarter of 2019. We assess our contracts at inception to determine whether the contract contains a lease, including evaluation of whether the contract conveys the right to control an explicitly or implicitly identified asset for a period of time. We have recognized right-of-use assets and lease liabilities that represent the net present value of future operating lease payments utilizing a discount rate corresponding to our incremental borrowing rate which we amortize over the remaining terms of the leases. For operating leases of a short-term nature, i.e., those with a term of less than twelve months, we recognize lease payments as an expense on a straight-line basis over the remaining lease term.
99
Emerging Growth Company Status
The Jumpstart Our Business Startups (“JOBS”) Act permits an “emerging growth company” such as us to take advantage of an extended transition period to comply with new or revised accounting standards applicable to public companies. We are choosing to take advantage of this provision and, as a result, we will adopt the extended transition period available under the JOBS Act until the earlier of the date we (i) are no longer an emerging growth company or (ii) affirmatively and irrevocably opt out of the extended transition period provided under the JOBS Act.
Off-Balance Sheet Arrangements
We did not have off balance sheet arrangements during the periods presented in this report, and we do not currently have, any off-balance sheet arrangements, as defined in the rules and regulations of the SEC.
Recently Issued Accounting Pronouncements
A description of recently issued accounting pronouncements that may potentially impact our financial position and results of operations disclosed in Note 2 to our consolidated financial statements included in Part II, Item 8 “Consolidated Financial Statements and Supplementary Data” of this report.
ITEM 7A. |
Not applicable.
100
ITEM 8.CONSOLIDATED FINANCIAL STATEMENTS AND SUPPLEMENTARY DATA
INDEX TO FINANCIAL STATEMENTS
| 102 | |
| 103 | |
Consolidated Statements of Operations and Comprehensive Loss | 104 | |
Consolidated Statements of Redeemable Convertible Preferred Stock and Stockholders' Equity (Deficit) | 105 | |
106 | ||
107 |
101
Report of Independent Registered Public Accounting Firm
Stockholders and Board of Directors
NeuroBo Pharmaceuticals, Inc.
Boston, Massachusetts
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated balance sheets of NeuroBo Pharmaceuticals, Inc. (the “Company”) as of December 31, 2020 and 2019, the related consolidated statements of operations and comprehensive loss, redeemable convertible preferred stock and stockholders' equity (deficit), and cash flows for each of the two years in the period ended December 31, 2020, and the related notes (collectively referred to as the “consolidated financial statements”). In our opinion, the consolidated financial statements present fairly, in all material respects, the financial position of the Company at December 31, 2020 and 2019, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2020, in conformity with accounting principles generally accepted in the United States of America.
Going Concern Uncertainty
The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern. As described in Note 1 to the consolidated financial statements, the Company has recurring losses and negative cash flows from operations that raise substantial doubt about the Company’s ability to continue as a going concern. Management's plans in regard to these matters are also described in Note 1. The consolidated financial statements do not include any adjustments that might result from the outcome of this uncertainty.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s consolidated financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (“PCAOB”) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ BDO USA, LLP
We have served as the Company’s auditor since 2019.
Boston, Massachusetts
April 15, 2021
102
(in thousands, except share amounts and par value)
|
| December 31, |
| December 31, |
| ||
| | 2020 | | 2019 |
| ||
| | | | | |
| |
Assets | | | | | | | |
Current assets: | | | | | | | |
Cash | | $ | 10,089 | | $ | 13,908 | |
Restricted cash | | | — | | | 15 | |
Prepaid expenses | | | 546 | | | 153 | |
Other assets | |
| 48 | |
| 42 | |
Total current assets | |
| 10,683 | |
| 14,118 | |
Right-of-use assets and other | | | 130 | | | 150 | |
Property and equipment, net | |
| 155 | |
| 200 | |
Total assets | | $ | 10,968 | | $ | 14,468 | |
Liabilities and stockholders’ equity | | | | | | | |
Current liabilities: | | | | | | | |
Accounts payable | | $ | 2,575 | | $ | 638 | |
Accrued liabilities | |
| 1,096 | |
| 1,422 | |
Lease liability, short-term | | | 24 | | | 22 | |
Total current liabilities | |
| 3,695 | |
| 2,082 | |
Lease liability, long-term | | | 70 | | | 94 | |
Total liabilities | |
| 3,765 | |
| 2,176 | |
Commitments and contingencies (Notes 4, 5, 6 and 14) | | | | | | | |
Stockholders’ equity | | | | | | | |
Preferred stock, $0.001 par value; 10,000,000 shares authorized as of December 31, 2020 and 2019, respectively; no shares issued or outstanding as of December 31, 2020 and 2019. | | | — | | | — | |
Common stock, $0.001 par value per share, 100,000,000 shares authorized as of December 31, 2020 and 2019; 19,671,182 and 15,592,718 shares issued and outstanding as of December 31, 2020 and 2019, respectively. | |
| 20 | |
| 16 | |
Additional paid–in capital | |
| 73,713 | |
| 49,130 | |
Accumulated other comprehensive income | | | 14 | | | 12 | |
Accumulated deficit | |
| (66,544) | |
| (36,866) | |
Total stockholders’ equity | |
| 7,203 | |
| 12,292 | |
Total liabilities and stockholders’ equity | | $ | 10,968 | | $ | 14,468 | |
See accompanying notes.
103
NeuroBo Pharmaceuticals, Inc.
Consolidated Statements of Operations and Comprehensive Loss
(in thousands, except share and per share amounts)
| | For the Year Ended | | ||||
| | December 31, | | ||||
| | 2020 | | 2019 | | ||
Operating expenses: |
| | |
| | |
|
Research and development | | $ | 4,531 | | $ | 5,324 | |
Acquired in‑process research and development | | | 17,339 | | | 12,151 | |
General and administrative | | | 7,846 | | | 2,701 | |
Total operating expenses | |
| 29,716 | |
| 20,176 | |
Loss from operations | |
| (29,716) | |
| (20,176) | |
Loss on note extinguishment | | | — | | | (1,114) | |
Interest income (expense), net | |
| 39 | |
| (22) | |
Other expense, net | |
| (1) | |
| — | |
Loss before income taxes | | | (29,678) | | | (21,312) | |
Provision for income taxes | | | — | |
| — | |
Net loss | |
| (29,678) | |
| (21,312) | |
Other comprehensive income, net of tax | |
| 2 | |
| 10 | |
Comprehensive loss | | $ | (29,676) | | $ | (21,302) | |
Loss per share: | | | | | | | |
Net loss per share, basic and diluted | | $ | (1.83) | | $ | (4.08) | |
Weighted average common shares outstanding: | | | | | | | |
Basic and diluted | |
| 16,217,339 | |
| 5,224,178 | |
| | | | | | | |
See accompanying notes.
104
NeuroBo Pharmaceuticals, Inc.
Consolidated Statements of Redeemable Convertible Preferred Stock and Stockholders’ Equity (Deficit)
(in thousands, except share amounts)
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | Redeemable Convertible | | | | | | | | | Additional | | | Accumulated | | | | | Total | | |||||
| | Preferred Stock | | | | Common Stock | | Paid–In | | | Comprehensive | | Accumulated | | Equity |
| |||||||||
|
| Shares |
| Amount |
| |
| Shares |
| Amount |
| Capital | | | Income |
| Deficit |
| (Deficit) |
| |||||
Balance at December 31, 2018 | | 4,801,020 | | $ | 16,746 | | | | 5,166,812 | | $ | — | | $ | 2,266 | | $ | 2 | | $ | (15,554) | | $ | (13,286) | |
Issuance of Series B redeemable convertible preferred stock, net of issuance costs of $74 | | 3,463,593 | | | 24,166 | | | | — | | | — | | | — | | | | | | — | | | — | |
Conversion of Series A and B redeemable convertible preferred stock into common stock | | (8,264,613) | | | (40,912) | | | | 8,264,613 | | | 1 | | | 40,911 | | | — | | | — | | | 40,912 | |
Extinguishment of related party convertible notes net of substantial premium of $10,620 | | — | | | — | | | | — | | | — | | | 732 | | | — | | | — | | | 732 | |
Conversion of related party convertible notes into common stock | | — | | | — | | | | 1,565,300 | | | — | | | 651 | | | — | | | — | | | 651 | |
Exercise of stock options | | — | | | — | | | | 1,143 | | | — | | | 1 | | | — | | | — | | | 1 | |
Issuance of common stock and warrants to former Gemphire stockholders and effect of reverse asset acquisition | | — | | | — | | | | 594,850 | | | 15 | | | 4,451 | | | — | | | — | | | 4,466 | |
Stock–based compensation | | — | | | — | | | | — | | | — | | | 118 | | | — | | | — | | | 118 | |
Foreign currency translation adjustment | | — | | | — | | | | — | | | — | | | — | | | 10 | | | — | | | 10 | |
Net loss | | — | | | — | | | | — | | | — | | | — | | | — | | | (21,312) | | | (21,312) | |
Balance at December 31, 2019 | | — | | | — | | | | 15,592,718 | | | 16 | | | 49,130 | | | 12 | | | (36,866) | | | 12,292 | |
Issuance of common stock in connection with equity financing | | — | | | — | | | | 750,000 | | | 1 | | | 7,499 | | | — | | | — | | | 7,500 | |
Transaction costs in connection with equity financing | | — | | | — | | | | — | | | — | | | (984) | | | — | | | — | | | (984) | |
Issuance of broker warrants in connection with equity financing | | — | | | — | | | | — | | | — | | | 289 | | | — | | | — | | | 289 | |
Issuance of common stock to former ANA stockholders and effect of asset acquisition | | — | | | — | | | | 3,243,875 | | | 3 | | | 17,027 | | | — | | | — | | | 17,030 | |
Stock–based compensation | | — | | | — | | | | — | | | — | | | 699 | | | — | | | — | | | 699 | |
Exercise of stock options | | — | | | — | | | | 84,589 | | | — | | | 53 | | | — | | | — | | | 53 | |
Foreign currency translation adjustment | | — | | | — | | | | — | | | — | | | — | | | 2 | | | — | | | 2 | |
Net loss | | — | | | — | | | | — | | | — | | | — | | | — | | | (29,678) | | | (29,678) | |
Balance at December 31, 2020 | | — | | $ | — | | | | 19,671,182 | | $ | 20 | | $ | 73,713 | | $ | 14 | | $ | (66,544) | | $ | 7,203 | |
| | | | | | | | | | | | | | | | | | | | | | | | | |
| | | | | | | | | | | | | | | | | | | | | | | | | |
See accompanying notes.
105
NeuroBo Pharmaceuticals, Inc.
Consolidated Statements of Cash Flows
(in thousands)
| | For the Year Ended |
| ||||
| | December 31, |
| ||||
| | 2020 | | 2019 |
| ||
Operating activities | | | | | | | |
Net loss | | $ | (29,678) | | $ | (21,312) | |
Adjustments to reconcile net loss to net cash used in operating activities: | | | | | | | |
In process research and development, non-cash portion | |
| 17,339 | |
| 12,151 | |
Stock-based compensation | |
| 699 | |
| 118 | |
Non-cash interest related to convertible notes - related party | |
| — | |
| 129 | |
Loss on note extinguishment - related party | | | — | | | 1,114 | |
Non-cash lease expense | | | 21 | | | — | |
Depreciation | |
| 46 | |
| 17 | |
Change in assets and liabilities, net of the effects of the asset acquisitions: | | | | | | | |
Prepaid expenses and other assets | |
| 33 | |
| 824 | |
Accounts payable | |
| 1,123 | |
| (1,046) | |
Accrued and other liabilities | |
| (347) | |
| 966 | |
Net cash used in operating activities | |
| (10,764) | |
| (7,039) | |
Investing activities | | | | | | | |
Cash acquired in connection with asset acquisitions | | | 180 | | | 1,525 | |
Transaction costs in connection with asset acquisitions | | | (107) | | | (7,368) | |
Purchases of property and equipment | | | (4) | | | (214) | |
Net cash provided by (used in) investing activities | |
| 69 | |
| (6,057) | |
Financing activities | | | | | | | |
Proceeds from issuance of redeemable preferred stock | | | — | | | 24,240 | |
Proceeds from equity offering | | | 7,500 | | | — | |
Issuance costs | | | (695) | | | (74) | |
Exercise of stock options | | | 53 | | | 1 | |
Net cash provided by financing activities | |
| 6,858 | |
| 24,167 | |
Net (decrease) increase in cash and restricted cash | |
| (3,837) | |
| 11,071 | |
Net foreign exchange difference | | | 3 | | | 7 | |
Cash and restricted cash at beginning of period | |
| 13,923 | |
| 2,845 | |
Cash and restricted cash at end of period | | $ | 10,089 | | $ | 13,923 | |
| | | | | | | |
Reconciliation of cash and restricted cash: | | | | | | | |
Cash | | $ | 10,089 | | $ | 13,908 | |
Restricted cash | | | — | | | 15 | |
Total cash and restricted cash | | $ | 10,089 | | $ | 13,923 | |
| | | | | | | |
Supplemental disclosure of cash flow information: | | | | | | | |
Cash paid for income taxes | | $ | — | | $ | — | |
Cash paid for interest | | $ | — | | $ | 1 | |
| | | | | | | |
Supplemental non-cash investing and financing transactions: | | | | | | | |
Conversion of Series A and Series B preferred stock to common | | $ | — | | $ | 40,912 | |
Conversion of convertible notes to common stock | | $ | — | | $ | 548 | |
Common stock and warrants issued in connection with asset acquisitions | | $ | 17,030 | | $ | 4,466 | |
Net assets (liabilities) assumed in connection with asset acquisitions | | $ | 201 | | $ | (1,537) | |
Unpaid transaction costs in accounts payable and accrued expenses related to asset acquisitions | | $ | 583 | | $ | 306 | |
Beneficial conversion feature related to convertible notes | | $ | — | | $ | 104 | |
Operating lease right of use asset obtained in exchange for operating lease | | $ | — | | $ | 126 | |
Placement warrants issued in connection with equity financing | | $ | 289 | | $ | — | |
See accompanying notes.
106
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements
1. The Company and Basis of Presentation
NeuroBo Pharmaceuticals, Inc. (together with its subsidiaries, the "Company" or "NeuroBo"), formerly known as Gemphire Therapeutics Inc. (“Gemphire”), is a clinical-stage biotechnology company with four therapeutic programs designed to impact a range of indications in neurodegenerative and cardiometabolic disease:
| ● | ANA001, which is focused on the development for coronavirus indications, currently in Phase 2/3 clinical trials as a treatment for COVID-19. |
| ● | NB-01, which is primarily focused on the development of a treatment for painful diabetic neuropathy, but which the Company believes could also treat a range of neuropathic conditions, including chemotherapy-induced peripheral neuropathy and post-traumatic peripheral neuropathy; |
| ● | NB-02, which has the potential to treat the symptoms of cognitive impairment and modify the progression of neurodegenerative diseases associated with the malfunction of a protein called tau, and with amyloid beta plaque deposition; and |
| ● | Gemcabene, which is currently being assessed as an acute indication for COVID-19. Gemcabene was previously focused on developing and commercializing therapies for the treatment of dyslipidemia, a serious medical condition that increases the risk of life-threatening cardiovascular disease, focused on orphan indications such as homozygous familial hypercholesterolemia, as well as nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. |
The Company was originally incorporated as Gemphire Therapeutics Inc. as a C corporation in the state of Delaware. In connection with the closing of the 2019 Merger (as defined below), the Company changed its name to NeuroBo Pharmaceuticals, Inc. The operations have consisted principally of performing research and development activities, clinical development and raising capital. The Company's activities are subject to significant risks and uncertainties, including failing to secure additional funding before sustainable revenues and profit from operations are achieved.
Mergers
2020 Merger with ANA
On December 31, 2020, the Company acquired 100% of ANA Therapeutics, Inc., a Delaware corporation (“ANA”), pursuant to an Agreement and Plan of Merger, dated December 31, 2020 (the “2020 Merger Agreement” or “2020 Merger”). Pursuant to the 2020 Merger Agreement, NeuroBo issued to the stockholders of ANA 3,243,875 shares of its common stock. The 2020 Merger, which closed on December 31, 2020, was accounted for as an asset acquisition pursuant to Topic 805, Business Combinations, as substantially all of the fair value of the assets acquired were concentrated in a group of similar non-financial assets.
2019 Merger with Gemphire
On July 24, 2019, Gemphire Therapeutics Inc. (“Gemphire”), and NeuroBo Pharmaceuticals, Inc. (“Private NeuroBo”) entered into a definitive agreement, which was amended on October 29, 2019 (the “2019 Merger Agreement”). The merger closed on December 30, 2019 (the “2019 Effective Date”), whereby Private NeuroBo merged with a wholly-owned subsidiary of the Company in an all-stock transaction (the “2019 Merger”).
Upon completion of the 2019 Merger, the Company changed its name to NeuroBo Pharmaceuticals, Inc., Private NeuroBo changed its name to NeuroBo Therapeutics, Inc., and the Company changed its ticker symbol on the Nasdaq Capital Market from “GEMP” to "NRBO". Except as otherwise indicated, references herein to “NeuroBo”, “the Company”, the “combined company”, “we”, “us”, and “our”, refer to NeuroBo Pharmaceuticals, Inc. on a post-2019 Merger basis.
107
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Pursuant to the terms of the 2019 Merger Agreement, each outstanding share of Private NeuroBo common stock outstanding immediately prior to the closing of the 2019 Merger was converted into 1.1431 shares of the Company’s common stock (the “Exchange Ratio”). Immediately prior to the closing of the 2019 Merger, all shares of Private NeuroBo redeemable preferred stock then outstanding were exchanged into shares of common stock of Private NeuroBo. In addition, all outstanding options exercisable for common stock of Private NeuroBo converted into options exercisable for shares of the Company’s common stock upon the 2019 Merger. Such options and their related terms were adjusted by the Exchange Ratio. Immediately following the 2019 Merger, the stockholders of Private NeuroBo owned approximately 96.2% of the outstanding common stock of the Company.
The transaction was accounted for as a reverse asset acquisition in accordance with accounting principles generally accepted in the United States of America (“GAAP”). Under this method of accounting, Private NeuroBo was deemed to be the accounting acquirer for financial reporting purposes. This determination was primarily based on the facts that, immediately following the 2019 Merger: (i) Private NeuroBo’s stockholders owned substantially all of the voting rights in the combined company, (ii) Private NeuroBo designated all, but one, of the members of the initial board of directors of the combined company, and (iii) Private NeuroBo’s senior management holds all key positions in the senior management of the combined company. As a result, as of the closing date of the 2019 Merger, the net assets of Gemphire were recorded at their acquisition-date relative fair values in the consolidated financial statements of the Company and the reported operating results prior to the 2019 Merger are those of Private NeuroBo.
Basis of presentation and consolidation principles
The accompanying financial statements were prepared in conformity with GAAP.
On August 11, 2019, Private NeuroBo’s board of directors and stockholders approved an amendment to the restated certificate of incorporation to affect a ten thousand-for-one (10,000-for-1) stock split of Private NeuroBo's common stock and convertible preferred stock. The par value and the authorized shares of the common and convertible preferred stock and the exercise prices of options to purchase common stock were adjusted accordingly as a result of the stock split. All issued and outstanding common stock, options for common stock, convertible preferred stock and convertible notes, as well as the exercise price of each option for common stock and the conversion price for convertible preferred stock and convertible notes, have been retroactively adjusted to reflect this stock split for all periods presented.
The consolidated financial statements of the Company include a South Korean subsidiary, NeuroBo Co., Ltd., which is fully owned by Private NeuroBo. All significant intercompany accounts and transactions have been eliminated in the preparation of the financial statements.
All of the share and per share amounts presented were adjusted, on a retroactive basis, to reflect the ten thousand-for-one (10,000-for-1) stock split and the effect of the exchange of the shares of Private NeuroBo into the shares of the Company at the Exchange Ratio, except for par value and share authorizations of Private NeuroBo for periods presented prior to the 2019 Merger.
Going Concern
From its inception through December 31, 2020, the Company has devoted substantially all of its efforts to drug discovery and development and conducting clinical trials. The Company has a limited operating history and the sales and income potential of the Company's business and market are unproven. Successful transition to attaining profitable operations is dependent upon achieving a level of revenues adequate to support the Company's cost structure. As of December 31, 2020, the Company had $10.1 million in cash. The Company has experienced net losses and negative cash flows from operating activities since its inception and had an accumulated deficit of $66.5 million as of December 31, 2020.
To date, the Company has raised capital principally through the private placements of common stock and redeemable convertible preferred stock as well as via the issuance of convertible notes. Prior to the 2019 Merger, Private NeuroBo had raised a total of $41.0 million from the issuance of redeemable convertible preferred stock and $0.5 million from the issuance of convertible notes. On April 13, 2020, the Company entered into a Securities Purchase Agreement, pursuant to which the Company agreed to issue and sell, in a registered direct offering (the “Registered Offering”), 750,000 shares
108
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
of common stock at an offering price of $10 per share. The Registered Offering resulted in gross proceeds of $7.5 million. In January 2021, the Company entered into a private placement and issued common stock and warrants that resulted in gross proceeds of $10 million. See Note 16 – Subsequent Events. The Company will need to continue to raise a substantial amount of funds until it is able to generate revenues to fund its development activities.
The determination as to whether the Company can continue as a going concern contemplates the realization of assets and the satisfaction of liabilities in the normal course of business. The Company expects to continue to incur net losses and negative cash flows from operations into the foreseeable future. Successful transition to attaining profitable operations is dependent upon achieving a level of revenues adequate to support the Company's cost structure. The Company has incurred net losses since inception and has relied on its ability to fund its operations through debt and equity financings. These conditions raise substantial doubt about the Company's ability to continue as a going concern. The accompanying consolidated financial statements have been prepared assuming that the Company will continue as a going concern and do not include any adjustments that might result from the outcome of this uncertainty. This basis of accounting contemplates the recovery of the Company's assets and the satisfaction of liabilities in the normal course of business.
The Company believes that its existing cash will be sufficient to fund its operations into the fourth quarter of 2021. The Company plans to continue to fund its operations and capital funding needs through a combination of equity offerings, debt financings, or other sources, potentially including collaborations, licenses and other similar arrangements. There can be no assurance that the Company will be able to obtain any sources of financing on acceptable terms, or at all. To the extent that the Company can raise additional funds by issuing equity securities, the Company's stockholders may experience significant dilution. Any debt financing, if available, may involve restrictive covenants that impact the Company's ability to conduct its business.
COVID-19
The Company is subject to risks and uncertainties as a result of the COVID-19 pandemic. The extent of the impact of the COVID-19 pandemic on the Company’s business is highly uncertain and difficult to predict, as the responses that the Company, other businesses and governments are taking continue to evolve. Furthermore, capital markets and economies worldwide have also been negatively impacted by the COVID-19 pandemic, and it is possible that it could cause a lasting national or global economic recession. Policymakers around the globe have responded with fiscal policy actions to support the healthcare industry and economy as a whole. The magnitude and overall effectiveness of these actions remain uncertain.
To date, except for the adjustments to scientific activity described under “Current Scientific Activity” below, the Company has not experienced any significant external changes in our business that would have a significant negative impact on our consolidated statements of operations or cash flows.
Exclusive of the development of certain of the Company’s proposed therapies, the severity of the impact of the COVID-19 pandemic on the Company’s business will depend on a number of factors, including, but not limited to, the duration and severity of the pandemic and the extent and severity of the impact on the Company’s service providers, suppliers, contract research organizations and the Company’s clinical trials, all of which are uncertain and cannot be predicted. As of the date of issuance of Company’s financial statements, the extent to which the COVID-19 pandemic may in the future materially impact the Company’s financial condition, liquidity or results of operations is uncertain.
2. Summary of Significant Accounting Policies
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities, expenses, and related disclosure of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of expenses during the reporting period. The most significant estimates in the Company's consolidated financial statements relate to accrued expenses and the fair value of stock-based compensation and warrant issuances. The Company bases its estimates on historical experience and on various other assumptions that are believed to be reasonable, the results of which form the basis for making judgements about the carrying values of assets and liabilities. Actual results could differ from those estimates. Changes in estimates are reflected in reported results in the period in which they become known.
109
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Concentration of Credit Risk
Financial instruments that potentially subject the Company to a concentration of credit risk consist of cash. The Company’s cash is principally held by one financial institution in the United States. Amounts on deposit may at times exceed federally insured limits. Management believes that the financial institution is financially sound, and accordingly, minimal credit risk exists with respect to the financial institution. As of December 31, 2020, the Company had deposits in excess of federally insured amounts by $9.5 million.
Fair Value of Financial Instruments
The Company’s financial instruments principally include cash, prepaid, other current assets, right of use assets, accounts payable, accrued liabilities, lease liabilities, convertible debt and preferred stock. The carrying amounts of prepaid expenses, accounts payable, and accrued liabilities are reasonable estimates of their fair value because of the short maturity of these items. See Note 12 - Fair Value Measurements with regard to the basis of measurement related to the convertible debt and preferred stock when outstanding.
General and Administrative Expenses
General and administrative expenses consist primarily of personnel-related costs, including salaries and stock-based compensation costs, for personnel in functions not directly associated with research and development activities. Other significant costs include legal fees related to intellectual property and corporate matters and professional fees for accounting and other services.
Research and Development Costs
Research and development costs are charged to expense as incurred. Research and development expenses are comprised of costs incurred in performing research and development activities, including clinical trial costs, manufacturing costs for both clinical and pre-clinical materials as well as other contracted services, license fees, and other external costs. Nonrefundable advance payments for goods and services that will be used in future research and development activities are expensed when the activity is performed or when the goods have been received, rather than when payment is made, in accordance with Accounting Standards Codification (“ASC”) 730, Research and Development.
Acquired In-Process Research and Development
The Company includes costs to acquire or in-license product candidates in acquired in-process research and development expenses (“IPR&D”). When the Company acquires the right to develop and commercialize a new product candidate, any up-front payments, or any future milestone payments that relate to the acquisition or licensing of such a right are immediately expensed as acquired in-process research and development in the period in which they are incurred. These costs are immediately expensed provided that the payments do not also represent processes or activities that would constitute a “business” as defined under GAAP, or provided that the product candidate has not achieved regulatory approval for marketing and, and absent obtaining such approval, has no alternative future use. Royalties owed on future sales of any licensed product will be expensed in the period the related revenues are recognized.
Income Taxes
The Company utilizes the liability method of accounting for income taxes as required by ASC 740, Income Taxes. Under this method, deferred tax assets and liabilities are determined based on differences between financial reporting and the tax basis of assets and liabilities and are measured using enacted tax rates and laws that will be in effect when the differences are expected to reverse. Currently, there is no provision for income taxes, as the Company has incurred operating losses to date, and a full valuation allowance has been provided on the net deferred tax assets.
Stock-Based Compensation
The Company accounts for stock-based compensation in accordance with the provisions of ASC 718, Compensation — Stock Compensation (“ASC 718”). Accordingly, compensation costs related to equity instruments granted are recognized
110
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
at the grant-date fair value. The Company records forfeitures when they occur. Stock-based compensation arrangements to non-employees are accounted for in accordance with the applicable provisions of ASC 718 using a grant date fair value approach.
Convertible Notes
The Company evaluates all conversion and redemption features contained in a debt instrument to determine if there are any embedded features that require bifurcation as a derivative or separation as a beneficial conversion feature. The host debt instrument is discounted for the value of any embedded feature that is accounted for as either a derivative or a beneficial conversion feature. The discount is amortized and recorded to interest expense over the term of the host debt instrument using the effective interest method. The Company’s convertible debt contained an embedded beneficial conversion feature that was separated and recorded as additional paid-in capital.
Fair Value of common stock
In the absence of a public trading market prior to the 2019 Merger, and as a development stage company with no significant revenues, the Company believed that it was appropriate to consider a range of factors to determine the fair value of the common stock at each grant date. In determining the fair value of its common stock, the Company used methodologies, approaches, and assumptions consistent with the American Institute of Certified Public Accountants' (“AICPA”) Audit and Accounting Practice Aid Series: Valuation of Privately Held Company Equity Securities Issued as Compensation (the "AICPA Practice Guide"). The valuations of Private NeuroBo common stock were prepared using a hybrid method, which used market approaches to estimate the enterprise value of Private NeuroBo. The hybrid method is a probability-weighted expected return method ("PWERM"), where the equity value in one or more of the scenarios is calculated using an option pricing method ("OPM"). The PWERM is a scenario-based methodology that estimates the fair value of common stock based upon an analysis of future values for Private NeuroBo, assuming various outcomes. The common stock value was based on the probability-weighted present value of expected future investment returns considering each of the possible outcomes available as well as the rights of each class of stock. The future value of the common stock under each outcome was discounted back to the valuation date at an appropriate risk-adjusted discount rate and probability weighted to arrive at an indication of value for the common stock. A discount for lack of marketability of the common stock was then applied to arrive at an indication of value for the common stock. The OPM treats common stock and preferred stock as call options on the total equity value of a company, with exercise prices based on the value thresholds at which the allocation among the various holders of a company's securities changes. Under this method, the common stock has value only if the funds available for distribution to stockholders exceeded the value of the preferred stock liquidation preferences at the time of the liquidity event, such as a strategic sale or a merger. In addition, the Company considered various objective and subjective factors, along with input from an independent third-party valuation firm. The factors included (1) the achievement of technical and operational milestones by the Company; (2) the status of strategic relationships with collaborators; (3) the significant risks associated with the Company's stage of development; (4) capital market conditions for life science companies and, in particular, similarly situated, privately held, early-stage life science companies; (5) the Company's available cash, financial condition, and results of operations; (6) the most recent sales of the Company's preferred stock to the extent they were with outside parties; and (7) the preferential rights of the outstanding preferred stock.
Leases
On July 1, 2019, the Company adopted Accounting Standards Update (“ASU”) No. 2016-02, Leases (Topic 842) (“ASU 2016-02”). The Company assesses its contracts at inception to determine whether the contract contains a lease, including evaluation of whether the contract conveys the right to control an explicitly or implicitly identified asset for a period of time. The Company has recognized right-of-use assets and lease liabilities that represent the net present value of future operating lease payments utilizing a discount rate corresponding to the Company’s incremental borrowing rate and amortized over the remaining terms of the leases. For operating leases of a short-term nature, i.e., those with a term of less than twelve months, the Company recognizes lease payments as an expense on a straight-line basis over the remaining lease term.
111
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Property and Equipment
Property and equipment is recorded at cost and reduced by accumulated depreciation. Depreciation expense is recognized over the estimated useful lives of the assets using the straight-line method. The estimated useful life for property and equipment ranges from three to five years. Tangible assets acquired for research and development activities and that have an alternative use are capitalized over the useful life of the acquired asset. Estimated useful lives are periodically reviewed, and when appropriate, changes are made prospectively. When certain events or changes in operating conditions occur, asset lives may be adjusted and an impairment assessment may be performed on the recoverability of the carrying amounts. Maintenance and repairs are charged directly to expense as incurred.
Foreign Currency Translation
The foreign subsidiary uses the local currency as the functional currency. The Company translates the assets and liabilities of its foreign operation into U.S. dollars based on the rates of exchange in effect as of the balance sheet date. Expenses are translated into U.S. dollars using average exchange rates for each period. The resulting adjustments from the translation process are included in accumulated other comprehensive (loss) income in the accompanying consolidated balance sheets.
Certain transactions of the Company are settled in foreign currency and are thus translated to U.S. dollars at the rate of exchange in effect at the end of each month. Gains and losses resulting from the translation are included in other income or expense in the accompanying consolidated statements of operations and comprehensive loss.
Patent Costs
Costs related to filing and pursuing patent applications are expensed as incurred, as recoverability of such expenditures is uncertain. These costs are included in general and administrative expenses.
Comprehensive Loss
Comprehensive loss is comprised of net loss and other comprehensive income or loss. Comprehensive loss includes net loss as well as other changes in stockholders' equity (deficit) that result from transactions and economic events other than those with stockholders. Comprehensive loss currently consists of net loss and changes in foreign currency translation adjustments.
Segment Information
Operating segments are components of an enterprise for which separate financial information is available and is evaluated regularly by the Company’s chief operating decision maker in deciding how to allocate resources and assessing performance. The Company’s chief operating decision maker is its Chief Executive Officer. The Company’s Chief Executive Officer views the Company’s operations and manages its business in one operating segment, which is principally the business of development and commercialization of therapeutics.
Recent Accounting Pronouncements Adopted
From time to time, new accounting pronouncements are issued by the Financial Accounting Standards Board ("FASB") or other standard setting bodies that are adopted by the Company as of the specified effective date. Unless otherwise discussed, the Company believes that the impact of recently issued standards that are not yet effective will not have a material impact on its consolidated financial position or results of operations upon adoption.
In August 2018, the FASB issued ASU 2018-13, Fair Value Measurement (Topic 820): Disclosure Framework - Changes to the Disclosure Requirements for Fair Value Measurement (ASU 2018-13). The new guidance modifies the disclosure requirements in Topic 820 as follows:
| ● | Removals: the amount of and reasons for transfers between Level 1 and Level 2 of the fair value hierarchy; the policy for timing of transfers between levels; and the valuation processes for Level 3 fair value measurements. |
112
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
| ● | Modifications: for investments in certain entities that calculate net asset value, an entity is required to disclose the timing of liquidation of an investee’s assets and the date when restrictions from redemption might lapse only if the investee has communicated the timing to the entity or announced the timing publicly; and the amendments clarify that the measurement uncertainty disclosure is to communicate information about the uncertainty in measurement as of the reporting date. |
| ● | Additions: the changes in unrealized gains and losses for the period included in other comprehensive income for recurring Level 3 fair value measurements held at the end of the reporting period; and the range and weighted average of significant unobservable inputs used to develop Level 3 fair value measurements. |
This guidance is effective for all entities for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2019. The amendments on changes in unrealized gains and losses, the range and weighted average of significant unobservable inputs used to develop Level 3 fair value measurements, and the narrative description of measurement uncertainty should all be applied prospectively for only the most recent interim or annual period presented in the initial year of adoption. All other amendments should be applied retrospectively to all periods presented upon their effective date. Early adoption is permitted. The Company adopted the new guidance on January 1, 2020. The guidance did not have a material impact on the consolidated financial statements.
Recent Accounting Pronouncements Not Yet Adopted
In June 2016, the FASB issued ASU 2016-13, “Financial Instruments – Credit Losses”. The ASU sets forth a “current expected credit loss” (CECL) model which requires the Company to measure all expected credit losses for financial instruments held at the reporting date based on historical experience, current conditions, and reasonable supportable forecasts. This replaces the existing incurred loss model and is applicable to the measurement of credit losses on financial assets measured at amortized cost and applies to some off-balance sheet credit exposures. This ASU is effective for fiscal years beginning after December 15, 2019, including interim periods within those fiscal years, with early adoption permitted. Recently, the FASB issued the final ASU to delay adoption for smaller reporting companies to calendar year 2023. The Company is currently assessing the impact of the adoption of this ASU on its consolidated financial statements.
In December 2019, the FASB issued ASU No. 2019-12, Income Taxes (Topic 740) which amends the existing guidance relating to the accounting for income taxes. This ASU is intended to simplify the accounting for income taxes by removing certain exceptions to the general principles of accounting for income taxes and to improve the consistent application of GAAP for other areas of accounting for income taxes by clarifying and amending existing guidance. The ASU is effective for fiscal years beginning after December 15, 2020. The Company does not expect that the adoption of this new guidance will have a material impact on the Company’s consolidated financial statements and plans to adopt this guidance on a prospective basis for the provisions applicable to the Company.
In August 2020, FASB issued ASU 2020-06, Debt—Debt with Conversion and Other Options (Subtopic 470-20) and Derivatives and Hedging—Contracts in Entity’s Own Equity (Subtopic 815-40): Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity, which, among other things, provides guidance on how to account for contracts on an entity’s own equity. This ASU eliminates the beneficial conversion and cash conversion accounting models for convertible instruments. It also amends the accounting for certain contracts in an entity’s own equity that are currently accounted for as derivatives because of specific settlement provisions. In addition, this ASU modifies how particular convertible instruments and certain contracts that may be settled in cash or shares impact the diluted EPS computation. The amendments in this ASU are effective for smaller reporting companies as defined by the SEC for fiscal years beginning after December 15, 2023, including interim periods within those fiscal years. Early adoption is permitted, but no earlier than fiscal years beginning after December 15, 2020. The Company is currently evaluating the impact of ASU 2020-06 on its consolidated financial statements.
113
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
3. Balance Sheet Detail (in thousands)
Property and Equipment
Property and equipment consist of the following:
| | | | | | | | |
| | As of December 31, | | |||||
| | 2020 | | | 2019 | | ||
Research and development equipment | | $ | 158 | | | $ | 158 | |
Office equipment | | | 60 | | | | 59 | |
Total property and equipment | | | 218 | | | | 217 | |
Less accumulated depreciation | | | (63) | | | | (17) | |
Property and equipment, net | | $ | 155 | | | $ | 200 | |
| | | | | | | | |
Depreciation expense was $46 and $17 for the years ended December 31, 2020 and 2019, respectively.
Accrued liabilities
Accrued liabilities consist of the following:
|
| December 31, |
| ||||
| | 2020 | | 2019 |
| ||
External research and development expenses | | $ | 218 | | $ | 915 | |
Payroll related | |
| 277 | |
| 160 | |
Professional services | | | 561 | | | 158 | |
Other | |
| 40 | |
| 189 | |
Total | | $ | 1,096 | | $ | 1,422 | |
| | | | | | | |
4. Mergers
ANA Merger
The 2020 Merger, which closed on December 31, 2020, was accounted for as an asset acquisition pursuant to Topic 805, Business Combinations, as substantially all of the fair value of the assets acquired were concentrated in one asset, and the acquired assets did not have outputs. Because the assets had not yet received regulatory approval, the fair value attributable to the asset was recorded as IPR&D expenses in the Company’s consolidated statements of comprehensive loss for the year ended December 31, 2020.
The total purchase price paid in the 2020 Merger has been allocated to the net assets acquired and liabilities assumed based on their fair values as of the completion of the 2020 Merger. The following summarizes the purchase price paid in the 2020 Merger (in thousands, except share and per share amounts):
Number of shares of the combined organization owned by ANA's pre-merger stockholders | | | 3,243,875 |
Multiplied by the fair value per share of NeuroBo's common stock (1) | | $ | 5.25 |
Fair value of common stock issued to affect the 2020 Merger | | | 17,030 |
Transaction costs | | | 690 |
Purchase price | | $ | 17,720 |
114
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
| (1) | Based on the last reported sale price of the NeuroBo’s common stock on the Nasdaq Capital Market on December 31, 2020, the closing date of the 2020 Merger. |
The allocation of the purchase price is as follows:
Cash acquired | | $ | 180 |
Net assets assumed | | | 201 |
IPR&D (2) | | | 17,339 |
Purchase price | | $ | 17,720 |
| (2) | Represents the pre-2020 Merger research and development projects of ANA which were in-process, but not yet completed, and which the Company plans to advance post-2020 Merger. This consists primarily of technology associated with the Niclosamide drug compound. Current accounting standards require that the fair value of IPR&D projects acquired in an asset acquisition with no alternative future use be allocated a portion of the consideration transferred and charged to expense on the acquisition date. |
Pursuant to the 2020 Merger Agreement, following the closing of the 2020 Merger, the Company is obligated to pay milestone payments (each, a “Milestone Payment”) to certain persons identified in the 2020 Merger Agreement (each a “Stakeholder” and collectively, the “Stakeholders”) in the form, time and manner as set forth in the 2020 Merger Agreement, upon the achievement of the following milestone events set forth below by the Company or any of its affiliates (each, a “Milestone Event”):
Milestone Event | | | | | | Milestone Payment |
First receipt of Marketing Approval (as defined in the 2020 Merger Agreement) from the FDA for any Niclosamide Product (as defined in the 2020 Merger Agreement) | | | $ | | 45.0 million | |
Sales Milestones: | | | | | | |
Milestone Event – Worldwide Cumulative Net Sales of a Niclosamide Product | | | | | | |
equal to or greater than: | | | | | | Milestone Payment |
$500 million | | | | $ | 9.0 million | |
$1 billion | | $ | 13.5 million | |||
$3 billion | | $ | 36.0 million | |||
$5 billion | | $ | 72.0 million | |||
In connection with the acquisition of ANA, the Company assumed a license agreement (the “YourChoice Agreement”) between ANA and YourChoice Therapeutics, Inc. (“YourChoice”). YourChoice granted to ANA, during the term of the YourChoice Agreement, an exclusive, worldwide, fee-bearing license derived from the licensed intellectual property throughout the world. As further discussed in Note 5, pursuant to the YourChoice Agreement, the Company is obligated to pay Milestone Payments to YourChoice.
Additionally, pursuant to the 2020 Merger Agreement, the Company is obligated to pay a royalty of two and a half percent (2.5%) of annual worldwide net sales of each Niclosamide Product (as defined in the Merger Agreement) (each such payment, a “Royalty Payment”) to the Stakeholders in the form, time and manner as set forth in the 2020 Merger Agreement, following the first commercial sale of each Niclosamide Product (as defined in the 2020 Merger Agreement) on a country-by-country and Niclosamide Product-by-Niclosamide Product basis.
As of the December 31, 2020, no Royalty Payments had been accrued as there were no potential milestones yet considered probable.
Pursuant to Topic 805, Business Combinations, in an asset acquisition, contingent consideration is only recognized when it becomes probable or reasonably possible to occur as prescribed under ASC 450, Contingencies. As of the 2020 Merger close date, the contingent consideration outlined above was not deemed probable or reasonably possible to occur, and as such, was excluded from the 2020 Merger purchase price.
115
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Gemphire Merger
The 2019 Merger, which closed on December 30, 2019, was accounted for as a reverse asset acquisition pursuant to Topic 805, Business Combinations, as substantially all of the fair value of the assets acquired were concentrated in a group of similar non-financial assets, and the acquired assets did not have outputs or employees. Because the assets had not yet received regulatory approval, the fair value attributable to these assets was recorded as acquired in-process research and development (“IPR&D”) expenses in the Company’s consolidated statements of comprehensive loss for the year ended December 31, 2019.
Contingent Value Rights Agreement
On December 30, 2019, in connection with the 2019 Merger, the Company, Grand Rapids Holders’ Representative, LLC, as representative of the Company’s stockholders prior to the 2019 Merger (the “Holders’ Representative”), and Computershare Inc. and Computershare Trust Company, N.A. as the rights agent (collectively, the “Rights Agent”), entered into a Contingent Value Rights Agreement (the “Original CVR Agreement”). Under the Original CVR Agreement, the Company’s stockholders of record as of immediately prior to the effective date of the 2019 Merger received one contingent value right (“CVR”) entitling such holders to receive, in the aggregate, 80% of the Gross Consideration less other Permitted Deductions (each as defined in the Original CVR Agreement) received during the 15-year period after the closing of the 2019 Merger (the “CVR Term”) from the grant, sale or transfer of rights to Gemcabene (other than a grant, sale or transfer of rights involving a sale or disposition of the post-2019 Merger combined company) that is entered into during the 10-year period after the closing of the 2019 Merger or pursuant to the Beijing SL Agreement (as defined in Note 6 – License Agreement below), but not including the $2.5 million upfront gross payment pursuant to the Beijing SL Agreement. Under the Original CVR Agreement, the Company agreed to commit up to $1 million to support the further development of Gemcabene, to be funded following execution of the Beijing SL Agreement and the receipt by the Company of the $2.5 million upfront gross payment payable under the Beijing SL Agreement, which the Company received in October 2019. The CVRs are not transferable, except in certain limited circumstances, will not be certificated or evidenced by any instrument, will not accrue interest and will not be registered with the U.S. Securities and Exchange Commission or listed for trading on any exchange. On March 23, 2021, the Company, the Holders’ Representative and the Rights Agent entered into a First Amendment to Contingent Value Rights Agreement to amend the Original CVR Agreement (as amended, the “Current CVR Agreement”). Pursuant to the Current CVR Agreement, the CVR holders are entitled to receive, in the aggregate, (i) 80% of the Gross Consideration less other Permitted Deductions (each as defined in the Current CVR Agreement) received during the CVR Term from the grant, sale or transfer of rights to Gemcabene as a treatment for cardiovascular conditions (other than a grant, sale or transfer of rights involving a sale or disposition of the post-2019 Merger combined company) that is entered into during the 10-year period after the closing of the 2019 Merger or pursuant to the Beijing SL Agreement, but not including the $2.5 million upfront gross payment pursuant to the Beijing SL Agreement; and (ii) 10% of the Gross Consideration less other Permitted Deductions (each as defined in the Current CVR Agreement) received during the CVR Term from the grant, sale or transfer of rights to Gemcabene as a treatment for any indication outside of treating cardiometabolic diseases, including COVID-19 (other than a grant, sale or transfer of rights involving a sale or disposition of the post-2019 Merger combined company), that is entered into during the 10-year period after the closing of the 2019 Merger or pursuant to the Beijing SL Agreement, but not including the $2.5 million upfront gross payment pursuant to the Beijing SL Agreement The Current CVR Agreement will continue in effect until the later of the end of the CVR Term and the payment of all amounts payable thereunder. As of the December 30, 2019, the 2019 Merger closing date, and December 31, 2020, no milestones had been accrued as there were no potential milestones yet considered probable.
116
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
The total purchase price paid in the 2019 Merger has been allocated to the net assets acquired and liabilities assumed based on their fair values as of the completion of the 2019 Merger. The following summarizes the purchase price paid in the 2019 Merger (in thousands, except share and per share amounts):
Number of shares of the combined organization owned by the Company’s pre-2019 Merger stockholders | | | 594,850 |
Multiplied by the fair value per share of GEMP’s common stock (3) | | $ | 7.50 |
Fair value of common stock issued to affect the 2019 Merger | | | 4,461 |
Fair value of warrants issued to affect the 2019 Merger | | | 4 |
Transaction costs | | | 7,674 |
Purchase price | | $ | 12,139 |
| | | |
| (3) | Based on the last reported sale price of the Gemphire’s common stock on the Nasdaq Capital Market on December 30, 2019, the closing date of the 2019 Merger, and gives effect to the Reverse Stock Split. |
The allocation of the purchase price is as follows:
Cash acquired | | $ | 1,525 |
Net liabilities assumed | | | (1,537) |
IPR&D (4) | | | 12,151 |
Purchase price | | $ | 12,139 |
| | | |
| (4) | Represents the pre-2019 Merger research and development projects of Gemphire which were in-process, but not yet completed, and which the Company plans to advance post-2019 Merger. This consists primarily of technology associated with the Gemcabene drug compound. Current accounting standards require that the fair value of IPR&D projects acquired in an asset acquisition with no alternative future use be allocated a portion of the consideration transferred and charged to expense on the acquisition date. The acquired assets did not have outputs or employees. |
5. Commitments and Contingencies (in thousands)
Operating Leases
Boston Leases
In April 2018, the Company entered a non-cancelable operating lease for its headquarters in Boston, MA (the “Boston Lease”). The lease was subsequently amended, and the term was extended to August 2019 with an option to extend the term on a month-to-month basis. The Company exercised the option and extended the lease term on a month-to-month basis through January 15, 2020. The lease is subject to base lease payments and additional charges for common costs related to usage of shared space. Due to its short-term nature, the Company recognizes lease payments as an expense on a straight-line basis over the remaining lease term. For the years ended December 31, 2020 and 2019, the Boston Lease expense was $308 and $134, respectively.
In September 2019, as amended, the Company entered a non-cancelable operating lease for its new corporate headquarters located in Boston, Massachusetts (“New Boston Lease”). The agreement, effective February 1, 2020, has a one-year term, and rental costs of $21 per month prior to the application of certain rent concessions granted by the landlord in the amount of $32.
117
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Future minimum lease payments at December 31, 2020 were as follows under the New Boston Lease (in thousands):
| | December 31, | | |
2021 | | $ | 21 | |
Total minimum payments | | $ | 21 | |
| | | | |
On November 11, 2020, the Company entered a non-cancelable operating lease, for its corporate headquarters. The agreement, effective February 1, 2021, has a six month term, and rental costs of approximately $3 per month prior to the application of certain rent concessions granted by the landlord in the amount of approximately $1 over the term of the lease. No assets and liabilities were recognized for the corporate headquarter leases at December 31, 2020.
Lease in Korea:
In May 2019, the Company entered a non-cancelable operating lease for its new facility in Korea (the “Korea Lease”). The initial lease term is five years with an option to renew for an additional five-year term. The lease commenced on July 2, 2019 and expires on July 1, 2024. The operating lease is subject to a deposit, base rent payments and additional charges for utilities and other common costs. In the third quarter of 2019, the Company recognized a right-of-use asset of $126 as well as a lease liability of $20 in other current liabilities and $106 in other non-current liabilities in conjunction with the commencement of the Korea Lease. The Company’s lease liability represents the net present value of future lease payments utilizing a discount rate of 10%, which corresponds to the Company’s incremental borrowing rate. As of December 31, 2020, the weighted average remaining lease term was 3.5 years. For the years ended December 31, 2020 and 2019, the Company recorded non-cash expense of $21 and $10, respectively, related to the Korea Lease. During the year ended December 31, 2020 and 2019, the Company made cash payments of $32 and $16, respectively, for amounts included in the measurement of lease liabilities.
The following table reconciles the undiscounted lease liabilities to the total lease liabilities recognized on the consolidated balance sheet as of December 31, 2020 (in thousands):
| | As of | | |
| | December 31, | | |
2021 | | $ | 32 | |
2022 | | | 32 | |
2023 | | | 32 | |
2024 | | | 16 | |
Total lease payments | | | 112 | |
Less effect of discounting | | | (18) | |
Total | | | 94 | |
Short-term portion | | | (24) | |
Long-term portion | | $ | 70 | |
| | | | |
Xiehecheng Cultivation Service Agreement
On September 1, 2018, the Company entered into a cultivation service agreement with Xiehecheng Chinese Herm Limited Corporation for the cultivation of two plants used to manufacture the Company's clinical asset, NB-01.
118
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
As of December 31, 2020, future minimum payments under the agreement, which is cancellable annually at the end of each research year, are as follows (in thousands):
| | December 31, | | |
| | | | |
2021 | | $ | 220 | |
2022 | | | 220 | |
| | $ | 440 | |
| | | | |
Pfizer License Agreement
Upon the close of the 2019 Merger, the exclusive license agreement with Pfizer Inc. (“Pfizer”) for the clinical product candidate Gemcabene (the “Pfizer Agreement”) was assumed by the Company. Under the Pfizer Agreement, in exchange for this worldwide exclusive right and license to certain patent rights to make, use, sell, offer for sale and import the clinical product Gemcabene, the Company has agreed to certain milestone and royalty payments on future sales.
The Company agreed to make milestone payments totaling up to $37 million upon the achievement of certain milestones, including the first new drug application (or its foreign equivalent) in any country, regulatory approval in each of the United States, Europe and Japan, the first anniversary of the first regulatory approval in any country, and upon achieving certain aggregate sales levels of Gemcabene. Future milestone payments under the Pfizer Agreement, if any, are not expected to begin for at least several years and extend over a number of subsequent years.
The Company also agreed to pay Pfizer tiered royalties on a country-by-country basis based upon the annual amount of net sales, as specified in the Pfizer Agreement, until the later of: (a) five (5) years after the first commercial sale in such country; (b) the expiration of all regulatory or data exclusivity for Gemcabene in such country; and (c) the expiration or abandonment of the last valid claim of the licensed patents, including any patent term extensions or supplemental protection certificates in such country (collectively, the Royalty Term). Under the Pfizer Agreement, the Company is obligated to use commercially reasonable efforts to develop and commercialize Gemcabene.
None of the future milestone or royalty payments were triggered through December 31, 2020.
The Pfizer Agreement will expire upon expiration of the Royalty Term. On expiration (but not earlier termination), the Company will have a perpetual, exclusive, fully paid-up, royalty-free license under the licensed patent rights and related data to make, use, develop, commercialize, import and otherwise exploit the clinical product candidate Gemcabene. Either party may terminate the Pfizer Agreement for the other party’s material breach following a cure period or immediately upon certain insolvency events relating to the other party. Pfizer may immediately terminate the Pfizer Agreement in the event that (i) the Company or any of its affiliates or sublicenses contests or challenges, or supports or assists any third party to contest or challenge, Pfizer’s ownership of or rights in, or the validity, enforceability or scope of any of the patents licensed under the Pfizer Agreement or (ii) the Company or any of its affiliates or sublicensees fails to achieve the first commercial sale in at least one country by April 16, 2024.
Furthermore, upon termination of the Pfizer Agreement by Pfizer for any of the foregoing reasons, the Company grants Pfizer a non-exclusive, fully paid-up, royalty free, worldwide, transferrable, perpetual and irrevocable license to use any intellectual property rights arising from the development or commercialization of Gemcabene by the Company and any trademarks identifying Gemcabene and agrees to transfer regulatory filings and approvals to Pfizer or permit Pfizer to cross-reference and rely on such regulatory filings and approvals for Gemcabene. The Company may terminate the Pfizer Agreement for convenience upon 90 days’ written notice and payment of an early termination fee of $3.0 million.
As of December 31 2020 and December 31, 2019, there was sufficient uncertainty with regard to both the outcome of the clinical trials and the ability to obtain sufficient funding to support any of the cash milestone payments under the license agreement, and as such, no liabilities were recorded related to the Pfizer Agreement.
119
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
YourChoice License Agreement
As described in Note 4, in connection with the acquisition of ANA, the Company assumed a license agreement (the “YourChoice Agreement”) between ANA and YourChoice Therapeutics, Inc. (“YourChoice”). The fees due under the YourChoice Agreement include royalty payments of 0.5% of annual worldwide net sales of each Niclosamide Product (as defined in the 2020 Merger Agreement) and milestone payments in the aggregate of $19.5 million. The first milestone payment due is $5 million upon first receipt of Marketing Approval (as defined in the 2020 Merger Agreement) for the FDA for any Nicolsamide Product (as defined by the 2020 Merger Agreement), followed by sales milestones of $1 million, $1.5 million, $4 million, and $8 million if worldwide cumulative net sales of a Nicolsamide Product are equal or greater than $500 million, $1, billion, $3, billion, and $5 billion, respectively. The term of the YourChoice Agreement will expire on the expiration or invalidation of the last of the licensed patents under the YourChoice Agreement.
As of December 31 2020, there was sufficient uncertainty with regard to both the outcome of the clinical trials and the ability to obtain sufficient funding to support any of the cash milestone payments under the YourChoice Agreement, and as such, no liabilities were recorded related to the YourChoice Agreement.
Contingencies
From time to time, the Company may be subject to various claims and suits arising in the ordinary course of business. The Company does not expect that the resolution of these matters will have a material adverse effect on its financial position or results of operations.
6. License Agreement
Beijing SL License and Collaboration Agreement
Upon the close of the 2019 Merger, the License and Collaboration Agreement (the “Beijing SL Agreement”) with Beijing SL Pharmaceutical Co., Ltd. (“Beijing SL”) was assumed by the Company, pursuant to which the Company granted Beijing SL an exclusive royalty-bearing license to research, develop, manufacture and commercialize pharmaceutical products comprising, as an active ingredient, Gemcabene in mainland China, Hong Kong, Macau and Taiwan (each, a “region”, and collectively, the “Territory”). The terms of the agreement include payments based upon achievement of milestones and royalties on net product sales. Under the Beijing SL Agreement, the Company has variable consideration in the form of milestone payments. As of December 31, 2020, no revenue under the Beijing SL Agreement has been recognized.
Under the terms of the Beijing SL Agreement, Beijing SL will be responsible, at its expense, for developing and commercializing products containing Gemcabene (each, a “Licensed Product”) in the Territory, with certain assistance from the Company. To the extent mutually agreed to in writing, the Company and Beijing SL will collaborate on the Phase 3 clinical trial for homozygous familial hypercholesterolemia or other clinical trials with the Company as the sponsor designed to enroll patients both inside and outside the Territory (a “Global Study”), but Beijing SL will be responsible, at its expense, for the conduct of any Global Study to the extent solely in the Territory, subject to the Company’s final decision making authority, and the Company will be responsible, at its expense, for the conduct of any Global Study to the extent solely outside of the Territory. Under a territory development plan, the parties shall develop Licensed Products with respect to the Territory. Beijing SL will be responsible for development activities, including non-clinical and clinical studies directed at obtaining regulatory approval of the Licensed Product in the Territory. Beijing SL has agreed to use commercially reasonable efforts to commercialize the Licensed Products for each indication that receives regulatory approval in the Territory and shall prepare and present a commercialization plan that shall be subject to approval by the joint steering committee.
Pursuant to the Beijing SL Agreement, Beijing SL was to make a non-refundable upfront gross payment of $2.5 million to the Company within 45 days of the effective date of the Beijing SL Agreement; the upfront payment was received in October 2019 and such funds were fully expended prior to the close of 2019 Merger. Additionally, with respect to each Licensed Product, the Company is eligible to receive (i) payments for specified developmental and regulatory milestones (including submission of a new drug application to China’s National Medical Product Administration, dosing of the first
120
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
patient in a phase 3 clinical trial in mainland China and regulatory approval for the first and each additional indication of a Licensed Product in the Territory) totaling up to $6 million in the aggregate and (ii) payments for specified global net sales milestones of up to $20 million in the aggregate multiplied by the ratio of the net sales of a Licensed Product sold by Beijing SL in the Territory divided by the global net sales of a Licensed Product, which net sales milestone payments are payable once, upon the first achievement of such milestone.
Beijing SL is also obligated to pay the Company tiered royalties ranging from the mid-teens to twenty percent on the net sales of all Licensed Products in the Territory until the latest of (a) the date on which any applicable regulatory exclusivity with respect to such Licensed Product expires in such region, (b) the expiration or abandonment of the last valid patent claim or joint patent claim covering such Licensed Product in each region and (c) the fifth anniversary of the first commercial sale of such Licensed Product in such region (the “Royalty Term”). Future milestone payments under the Beijing SL Agreement, if any, are not expected to begin for at least one year and will extend over a number of subsequent years. The Company cannot determine the date on which Beijing SL’s potential royalty payment obligations to the Company would expire because Beijing SL has not yet developed any Licensed Products under the Beijing SL Agreement and therefore the Company cannot at this time identify the date of the first commercial sale or the periods of any regulatory exclusivity or patent claims with respect to any Licensed Product.
On a Licensed Product-by-Licensed Product and region-by-region basis upon the expiration of the Royalty Term, the license granted to Beijing SL shall be deemed perpetual, fully paid-up and royalty free with respect to such Licensed Product in such region. Either party may terminate the Agreement (x) with written notice in the event of the other party’s material breach following a cure period or (y) if the other party becomes subject to certain insolvency proceedings. In addition, the Company may terminate the agreement in its entirety if Beijing SL or its affiliates or sublicensees commence a proceeding challenging the validity, enforceability or scope of any of the Company’s patents.
To the extent rights granted to Beijing SL under the Beijing SL Agreement are controlled by the Company pursuant to the Pfizer Agreement, such rights are subject to the terms and conditions of such agreement with Pfizer, and Beijing SL has agreed to comply with such terms and conditions.
The Beijing SL Agreement contemplates that Beijing SL and the Company shall, no later than twelve months prior to the anticipated date of the first commercial sale of a Licensed Product, if any, negotiate in good faith and execute a commercial supply agreement, pursuant to which Beijing SL shall purchase from the Company, and the Company shall use commercially reasonable efforts to supply, Gemcabene or Licensed Product for clinical or commercial purposes, as applicable, until manufacturing and regulatory transfers are complete.
Each of the Company and Beijing SL has agreed to indemnify the other party against certain losses and expenses relating to the development or commercialization of a Licensed Product by the indemnifying party, the negligence or willful misconduct of the indemnifying party or its directors, officers, employees or agents or a breach of the indemnifying party’s representations, warranties or covenants.
7. Debt (in thousands, except share and per share data)
In February 2018, the Company received a total of $500 from the issuance by Private NeuroBo of convertible promissory notes (the "Convertible Notes") with an original maturity date of December 31, 2022. Upon the effective date of the 2019 Merger, the Convertible Notes were converted into 1,565,300 shares of common stock.
Prior to conversion, the lenders had the option to convert all of the then-unpaid note balance including principal and accrued but unpaid interest into common stock, at a conversion price of $0.40 per share after the earlier of (A) the closing of a firmly underwritten public offering pursuant to an effective registration statement under the Securities Act of 1933, as amended, covering the offer and sale of common stock for the account of the Company in the United States of America or similar registration in the Republic of Korea, or (B) January 1, 2020. On October 23, 2019, the Convertible Notes were amended (the “Amended Convertible Notes”) to require mandatory conversion upon the completion of a reverse merger transaction based on the then-unpaid note balance including principal and accrued but unpaid interest into common stock, at a conversion price of $0.40 per share. The amendment was accounted for as an extinguishment of the Convertible Notes. As a result of the extinguishment, the Amended Convertible Notes were recorded at their fair value in the amount of $11.2 million, the underlying beneficial conversion feature was reacquired at its intrinsic value of
121
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
$9.8 million and the remaining debt discount of $0.3 million was reversed. Considering that the Amended Convertible Notes were issued at a substantial premium, the difference between the fair value of the Amended Convertible Notes and principal amount of the Convertible Notes was recorded as a credit to additional paid in capital in the amount of $10.6 million. Consequently, the Company recorded a loss on the extinguishment in the amount of $1.1 million which was reflected in the consolidated statements of operations and comprehensive loss.
The Convertible Notes and Amended Convertible Notes (herein collectively referred to as the “Notes”) accrued interest at a rate of 5.00% per annum. The Company recorded interest on principal of $25 for the year ended December 31, 2019.
The fair value of the common stock, as determined using an option pricing model consistent with the AICPA Practice Guide, was in excess of the conversion price of the Convertible Notes. Accordingly, the Company initially recorded a $401 beneficial conversion feature upon issuance based on the intrinsic value of the conversion feature, which resulted in a debt discount with a corresponding amount to additional paid in capital. Subsequent to the issuance of the Convertible Notes, an additional beneficial conversion feature related to paid-in-kind interest was recorded in the amount of $104.
Debt discount amortization related to the beneficial conversion feature was being amortized over the life of the Convertible Notes using the effective interest method as additional interest expense. Upon the conversion of the Convertible Notes into common stock on December 30, 2019, the remaining debt discount was written off to interest expense. The Company recorded interest expense of $104 for the year ended December 31, 2019 related to the debt discount.
8. Stockholders’ Equity
Common Stock
The voting, dividend, and liquidation rights of the holders of the common stock are subject to and qualified by the rights, powers, and preferences of the holders of the preferred stock when outstanding. The holders of the common stock are entitled to one vote for each share of common stock held at all meetings of stockholders.
Dividend Rights
Common stockholders are entitled to receive dividends at the sole discretion of the board of directors of the Company. There have been no dividends declared on common stock as of December 31, 2020.
Voting Rights
The holders of common stock are entitled to one vote for each share of common stock along with all other classes and series of stock of the Company on all actions to be taken by the stockholders of the Company, including actions that would amend the certificate of incorporation of the Company to increase the number of authorized shares of the common stock.
Liquidation Rights
In the event of any liquidation, dissolution, or winding-up of the Company, the holders of common stock shall be entitled to share in the remaining assets of the Company available for distribution post preferential distributions made to holders of the Company’s preferred stock.
April 2020 Equity Financing
On April 16, 2020, the Company closed on a Securities Purchase Agreement (the “Purchase Agreement”) with an institutional investor, pursuant to which the Company sold, in a registered direct offering (the “Registered Offering”), 750,000 shares (the “Shares”) of the Company’s common stock at an offering price of $10 per share for gross proceeds of $7.5 million.
122
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
In connection with the Registered Offering, the placement agent received a cash commission equal to 7% of the gross proceeds from the sale of the Common Stock and warrants (the “Placement Agent’s Warrants”) to purchase up to 37,500 shares of Common Stock. The Placement Agent’s Warrants have an exercise price of $12.50 per share and a termination date of April 16, 2025. The fair value of the Placement Agent’s Warrants was $289 based on the Black-Scholes pricing model. Input assumptions used were as follows: a risk-free interest rate of 0.4%; expected volatility of 78.0%; expected life of 5 years; and expected dividend yield of 0%. The underlying traded stock price was used in the analysis. The Placement Agent’s Warrants were classified in stockholders’ equity as the number of shares were fixed and determinable and given that the Placement Agent’s Warrants did not require cash settlement or have other provisions precluding equity treatment.
During the year ended December 31, 2020, issuance costs in connection with the Registered Offering were $1.0 million which included cash commissions equal to $0.5 million, legal and other fees of $0.2 million and the value of the Placement Agent’s Warrants of $0.3 million.
Warrants
The following warrants were outstanding as of December 31, 2020 and 2019:
: | | | | | | | | | | | | |
| | | | | | | | | | | | |
| Exercise Price | | Number Outstanding | | | Expiration Date | | Number Exercisable | | |||
| $ | 186.75 | | | 1,440 | | | July 2028 | | | 1,440 | |
| $ | 260.00 | | | 39,115 | | | March 2022 | | | 39,115 | |
Total outstanding December 31, 2019 | | 40,555 | | | | | | 40,555 | | |||
| | | | | | | | | | | | |
| $ | 12.50 | | | 37,500 | | | April 2025 | | | 37,500 | |
Total outstanding December 31, 2020 | | 78,055 | | | | | | 78,055 | | |||
| | | | | | | | | | | | |
| | | | | | | | | | | | |
9. Redeemable Preferred Stock (in thousands, except share and per share data)
Upon close of the 2019 Merger on December 30, 2019, 8,264,613 shares of Private NeuroBo Series A and Series B redeemable preferred stock (as adjusted for the Exchange Ratio) were converted to Private NeuroBo common stock on a 1:1 basis. Previously in April 2018, Private NeuroBo sold and issued in a private placement 4,801,020 shares of Series A redeemable convertible preferred stock (as adjusted for the Exchange Ratio) at $3.50 per share, raising $16,800 in gross proceeds. Subsequently in May and June 2019, Private NeuroBo sold and issued 3,463,593 Series B redeemable convertible preferred stock (as adjusted for the Exchange Ratio) at $7.00 per share, raising $24,240 in gross proceeds.
10. Stock-Based Compensation (in thousands)
Stock-based compensation expense was included in general and administrative and research and development costs as follows in the accompanying statements of comprehensive loss (in thousands):
| | Year Ended | | ||||
| | December 31, | | ||||
|
| | 2020 | |
| 2019 | |
Research and development | | $ | 18 | | $ | 75 | |
General and administrative | | | 681 | | | 43 | |
Total stock-based compensation | | $ | 699 | | $ | 118 | |
123
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Stock Options
2018 Stock Plan
In December 2018, Private NeuroBo adopted the NeuroBo Pharmaceuticals, Inc. 2018 Stock Plan (the "2018 Plan") and in December 2019 in connection with the 2019 Merger, the Company adopted the 2019 Equity Incentive Plan (the “2019 Plan”). 2018 Plan options to purchase Private NeuroBo common stock outstanding as of immediately prior to the 2019 Merger were assumed by the Company upon the 2019 Merger and became options to purchase the Company’s common stock, as adjusted by the Exchange Ratio. The 2018 Plan and 2019 Plan provide for the grant of stock options, restricted stock and other equity awards of the Company's common stock to employees, officers, consultants, and directors. Options expire within a period of not more than ten years from the date of grant. During the years ended December 31, 2020 and 2019, 420,000 and 960,204 stock options were granted, respectively, to employees and non-employee consultants with both service and performance conditions. The options granted with service conditions vest over a period between one year and three years.
As of December 31, 2020, 3,623,708 and 1,497,891 shares were authorized under the 2019 Plan and 2018 Plan, respectively, for issuance under these plans.
The following table summarizes the Company’s stock option plan activity for the years ended December 31, 2020 as follows:
| | | | | | | Weighted‑ | | | | |
| | | | Weighted | | Average | | Aggregate | | ||
| | | | Average | | Remaining | | Intrinsic | | ||
| | Number of | | Exercise | | Contractual | | Value(1) | | ||
| | Options | | Price | | Term (years) | | (in thousands) | | ||
Outstanding at December 31, 2018 | | - | | | — | | — | | | — | |
Granted | | 960,204 | | $ | 0.63 | | — | | | — | |
Exercised | | (1,143) | | $ | 0.63 | | — | | | — | |
Forfeited/Cancelled | | (325,784) | | $ | 0.63 | | — | | | — | |
Outstanding at December 31, 2019 | | 633,277 | | $ | 0.63 | | 9.1 | | $ | 5,142 | |
Granted | | 420,000 | | $ | 8.05 | | — | | | — | |
Exercised | | (84,589) | | $ | 0.63 | | — | | | — | |
Forfeited/Cancelled | | (48,333) | | $ | 8.39 | | — | | | — | |
Outstanding at December 31, 2020 | | 920,355 | | $ | 3.61 | | 8.5 | | $ | 2,535 | |
Vested and expected to vest at December 31, 2020 | | 554,563 | | $ | 5.58 | | 8.8 | | $ | 845 | |
Options exercisable at December 31, 2020 | | 291,229 | | $ | 3.48 | | 8.4 | | $ | 845 | |
| | | | | | | | | | | |
| (1) | The aggregate intrinsic value is calculated as the difference between the exercise price of the underlying options and the fair value of our common stock as of $5.25 per share at December 31, 2020. Options with exercise prices above the fair value of the common stock on December 31, 2020 were excluded from the intrinsic value calculation. |
The weighted average fair value per share of options granted during the year ended December 31, 2020 and 2019 was $5.35 and $0.50, respectively.
The Company measures the fair value of stock options with service-based and performance-based vesting criteria to employees, consultants and directors on the date of grant using the Black-Scholes option pricing model. The Company does not have history to support a calculation of volatility and expected term. As such, the Company has used a weighted-average volatility considering the volatilities of several guideline companies.
For purposes of identifying similar entities, the Company considered characteristics such as industry, length of trading history, and stage of life cycle. The assumed dividend yield was based on the Company’s expectation of not paying
124
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
dividends in the foreseeable future. The average expected life of the options was determined based on the mid-point between the vesting date and the end of the contractual term according to the “simplified method” as described in SEC Staff Accounting Bulletin 110, or the contractual term in cases where the “simplified method” was precluded. The risk-free interest rate is determined by reference to implied yields available from U.S. Treasury securities with a remaining term equal to the expected life assumed at the date of grant. The Company records forfeitures when they occur.
The weighted-average assumptions used in the Black-Scholes option-pricing model are as follows:
| | | Year Ended | ||||||
| | | December 31, | ||||||
|
| | 2020 | | 2019 | ||||
| | | | | | | | | |
Expected stock price volatility | | | | 77.4 | % | | | 75.0 | % |
Expected life of options (years) | | | | 5.8 | | | | 10.0 | |
Expected dividend yield | | | | — | % | | | 0 | % |
Risk free interest rate | | | | 1.51 | % | | | 2.75 | % |
Evergreen provision
Under the 2019 Plan, the shares reserved automatically increase on January 1st of each year, for a period of not more than ten years commencing on January 1, 2020 and ending on (and including) January 1, 2029, to an amount equal to the lesser of 4% of the common shares outstanding as of January 1, or a lesser amount as determined by the Board. The aggregate maximum number of shares of common stock that may be issued pursuant to the 2019 Plan under the evergreen provision is 6,680,000 shares of common stock. On January 1, 2020, 623,708 shares were added to the 2019 Plan as a result of the evergreen provision. See Note 16 - Subsequent Events.
During the years ended December 31, 2020 and 2019, 145,485 and 231,478 stock options vested, respectively. The weighted average fair value per share of options vesting during the years ended December 31, 2020 and 2019 was $4.23 and $0.50, respectively During the years ended December 31, 2020 and 2019, 48,333 and 325,784 stock options were forfeited, respectively. As of December 31, 2020, 4,115,512 shares in the aggregate were available for future issuance under the 2019 Plan and 2018 Plan.
Unrecognized stock-based compensation cost for the stock options issued under the both the Company’s 2019 Plan and 2018 Plan was $1.3 million as of December 31, 2020. The unrecognized stock-based expense is expected to be recognized over a weighted average period of 2.1 years.
11. Net Loss Per Common Share
Basic net loss per share is computed by dividing the net loss by the weighted-average number of common shares outstanding for the period, without consideration for potentially dilutive securities if their effect is antidilutive. Diluted net loss per share is computed by dividing the net loss by the weighted average number of shares of common stock and dilutive common stock equivalents outstanding for the period determined using the treasury stock and if-converted methods. Dilutive common stock equivalents are comprised of convertible preferred stock, convertible notes payable, options outstanding under the Company's stock option plan and warrants during the periods that these instruments are outstanding. For all periods presented, there is no difference in the number of shares used to calculate basic and diluted shares outstanding as inclusion of the potentially dilutive securities would be antidilutive.
125
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
The following potential common shares were not considered in the computation of diluted net loss per share as their effect would have been anti-dilutive:
| | Year ended | | |
| ||
|
| 2020 |
| 2019 | | |
|
Stock options | | 920,355 | | 633,277 | | | |
Warrants | | 78,055 | | 40,568 | | | |
12. Fair Value Measurements
The Company follows accounting guidance that emphasizes that fair value is a market-based measurement, not an entity specific measurement. Fair value is defined as “the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date.” Fair value measurements are defined on a three level hierarchy:
Level 1 inputs: Unadjusted quoted prices for identical assets or liabilities in active markets;
Level 2 inputs: Quoted prices for similar assets and liabilities in active markets, quoted prices in markets that are not active, or inputs which are observable, weather directly or indirectly, for substantially the full term of the asset or liability;
Level 3 inputs: Unobservable inputs that reflect the Company’s own assumptions about the assumptions market participants would use in pricing the asset or liability in which there is little, if any, market activity for the asset or liability at the measurement date.
There were no financial instruments measured on a recurring basis as of December 31, 2020 and 2019 and on a non-recurring basis for any of the periods presented.
13. Income Taxes
The effective tax rate for the years ended December 31, 2020 and 2019 was zero percent. A reconciliation of income tax computed at the statutory federal income tax rate to the provision (benefit) for income taxes included in the accompanying consolidated statements of operations and comprehensive loss is as follows:
| | For the Year Ended December 31, | |||||
| | 2020 | | 2019 | | ||
Income tax (benefit) provision at federal statutory rate | | 21.0 | % | | 21.0 | % | |
State income tax, net of federal benefit | | 1.9 | | | 6.3 | | |
Acquired in-process research and development expense | | (12.3) | | | (15.6) | | |
Valuation allowance | | (44.3) | | | (10.4) | | |
Convertible notes | | — | | | (1.6) | | |
Research credits | | 0.3 | | | 0.8 | | |
Provision to tax return | | 34.2 | | | — | | |
Other | | (0.8) | | | (0.5) | | |
Effective tax rate | | — | % | | — | % | |
| | | | | | | |
126
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Loss before provision for taxes for the years ended December 31, 2020 and 2019 consisted of the following (in thousands):
| | Year Ended December 31, | | ||||
| | 2020 | | 2019 | | ||
| | | | | | | |
Loss before Income taxes: | | | | | | | |
Domestic | | $ | (29,297) | | $ | (20,935) | |
Foreign | | | (381) | | | (377) | |
| | $ | (29,678) | | $ | (21,312) | |
| | | | | | | |
The components of income tax provision (benefit) consisted of the following for the years ended December 31, 2020 and 2019 (in thousands):
| | Year Ended December 31, | | ||||
| | 2020 | | 2019 | | ||
| | | | | | | |
Tax Provision (Benefit): | | | | | | | |
Current | | | | | | | |
Domestic | | $ | — | | $ | — | |
Foreign | | | — | | | — | |
Total current tax provision (benefit) | | | — | | | — | |
| | | | | | | |
Deferred | | | | | | | |
Domestic | | | (13,059) | | | (7,085) | |
Foreign | | | (95) | | | (94) | |
Total deferred tax provision (benefit) | | | (13,154) | | | (7,179) | |
| | | | | | | |
Change in valuation allowance - Domestic | | | 13,059 | | | 7,085 | |
Change in valuation allowance - Foreign | | | 95 | | | 94 | |
| | | | | | | |
Total tax provision (benefit) | | $ | — | | $ | — | |
| | | | | | | |
127
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Significant components of the Company’s deferred tax assets and liabilities are summarized in the tables below as of (in thousands):
| | Year Ended December 31, | | ||||
| | 2020 | | 2019 | | ||
| | | | | | | |
Deferred tax assets: | | | | | | | |
Federal and state operating loss carryforwards | | $ | 14,052 | | $ | 8,805 | |
Foreign operating loss carryforwards | | | 243 | | | 148 | |
Acquired intangibles | | | 11,050 | | | 2,167 | |
Stock-based compensation | | | 161 | | | 32 | |
Lease liability | | | 24 | | | 32 | |
Other | | | 52 | | | 29 | |
Research and development credit carryforwards | | | 1,166 | | | 341 | |
| | | 26,748 | | | 11,554 | |
Valuation allowance - Domestic | | | (26,478) | | | (11,372) | |
Valuation allowance - Foreign | | | (243) | | | (148) | |
Total deferred tax assets, net of valuation allowance | | | 27 | | | 34 | |
| | | | | | | |
Deferred tax liabilities: | | | | | | | |
ROU asset | | | (24) | | | (32) | |
Other | | | (3) | | | (2) | |
Net deferred tax assets | | $ | — | | $ | — | |
| | | | | | | |
As of December 31, 2020 and 2019, the Company had deferred tax assets of approximately $26.7 million and $11.6 million, respectively. Realization of the deferred tax assets is primarily dependent upon future taxable income, if any, the amount and timing of which are uncertain. The Company has had significant pre-tax losses since its inception. The Company has not yet generated revenues and faces significant challenges to becoming profitable. Accordingly, the deferred tax assets have been fully offset by a valuation allowance of $26.7 million and $11.6 million as of December 31, 2020 and 2019, respectively. U.S. deferred tax assets will continue to require a valuation allowance until the Company can demonstrate their realizability through sustained profitability or another source of income.
As of December 31, 2020 and 2019, the Company’s federal net operating loss carryforwards were approximately $57.5 million and $32.1 million, respectively. The Company had federal research credit carryforwards as of December 31, 2020 and 2019 of approximately $0.7 million and $0.1 million, respectively. The federal net operating loss incurred prior to January 1, 2018 will begin to expire in 2034 and tax credit carryforwards will begin to expire in 2038 if not utilized. Federal net operating losses incurred after December 31, 2017 will not expire. As of December 31, 2020 and 2019, the Company had state net operating loss carryforwards of approximately $25.1 million and $32.5 million, respectively. The Company had state research credit carryforwards of $0.5 million and $0.2 million as of December 31, 2020 and 2019, respectively. The state net operating loss carryforwards will begin to expire in 2026, if not utilized, and the state research credit carryforwards will begin to expire in 2033 if not utilized. Lastly, the Company had foreign net operating loss carryforwards of approximately $1.0 million and $0.6 million as of December 31, 2020 and 2019, respectively. The foreign net operating loss carryforwards will begin to expire in 2028.
Utilization of the net operating loss carryforwards and credits may be subject to a substantial annual limitation due to the ownership change limitations provided by Section 382 of the Internal Revenue Code of 1986, as amended, and similar state provisions. Generally, in addition to certain entity reorganizations, the limitation applies when one or more “5-percent shareholders” increase their ownership, in the aggregate, by more than 50 percentage points over a 36-month testing period, or beginning the day after the most recent ownership change, if shorter. The annual limitation may result in the expiration of net operating losses and credits before utilization. As a result of the 2020 Merger, the Company recorded deferred tax assets of $2.0 million which are fully offset by a valuation allowance. As a result of the 2019 Merger, the Company recorded deferred tax assets of $15.0 million which are fully offset by a valuation allowance. The $2.0 million net deferred tax assets recorded in relation to the 2020 Merger and the $15.0 million net deferred tax assets
128
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
recorded in relation to the 2019 Merger do not include federal and state net operating loss carryforwards and federal research and development credit carryforwards that are estimated to expire under Internal Revenue Code Sections 382 and 383 as a result of the 2020 Merger and 2019 Merger. The Company is currently evaluating the impact of Section 382 on its tax attributes.
The Company recognizes interest and/or penalties related to uncertain tax positions in income tax expense. There were no uncertain tax positions as of December 31, 2020 and 2019, and as such, no interest or penalties were recorded to income tax expense.
The Company’s corporate returns are subject to examination beginning with the 2017 tax year for federal and state jurisdictions, and beginning with the 2018 tax year for one foreign jurisdiction.
On March 27, 2020, the United States enacted the Coronavirus Aid, Relief, and Economic Security Act (“CARES Act”). The CARES Act is an emergency economic stimulus package that includes spending and tax breaks to strengthen the United States economy and fund a nationwide effort to curtail the effect of COVID-19. As of December 31, 2020, the Company has analyzed the provisions of the CARES Act and determined it did not have a significant impact to the Company.
On December 27, 2020, the President of the United States signed the Consolidated Appropriations Act, 2021 (“Consolidated Appropriations Act”) into law. The Consolidated Appropriations Act is intended to enhance and expand certain provisions of the CARES Act, allows for the deductions of expenses related to the Paycheck Protection Program funds received by companies, and provides an update to meals and entertainment expensing for 2021. The Consolidated Appropriations Act did not have a material impact to the Company’s income tax provision for 2020.
14. Related Party Transactions (in thousands, except per share data)
Agreements with Dong-A ST
On September 28, 2018, Private NeuroBo entered into a five year manufacturing and supply agreement with Dong-A ST for manufacturing and supply of NB-01 drug substance and placebos for the purpose of research and development to be used in Phase 3 clinical trials (the “Manufacturing Agreement”). The Company recognized $314 product manufacturing related costs under the Manufacturing Agreement for the year ended December 31, 2019. There were no manufacturing related costs under the Manufacturing Agreement for the year ended December 31, 2020. The product manufacturing related costs, when incurred, are reflected as research and development expenses. None of the costs incurred under the Manufacturing Agreement remained unpaid as of December 31, 2020 or 2019.
The Manufacturing Agreement will automatically terminate in the event that the license agreement with Dong-A ST is terminated for any reason. In addition, each of Dong-A ST and NeuroBo may terminate the Manufacturing Agreement (1) upon the material breach by the other party, if the breach is not cured within a specified number of days after receiving notice from the terminating party, or if the breach cannot reasonably be cured within such period and the breaching party has not started to remedy the breach within such period and diligently endeavored to cure the breach within a reasonable time thereafter, or (2) in the event that (i) the other party is the subject of a petition for bankruptcy, reorganization, or arrangement and the same is not dismissed within thirty days thereof, (ii) a receiver or trustee is appointed for all or a substantial portion of the assets of the other party, or (iii) the other party makes an assignment for the benefit of its creditors.
On June 7, 2020, the Company entered into a manufacturing and supply agreement (the “Manufacturing and Supply Agreement”) with Dong-A ST for the manufacturing and supply of NB-02 drug product and placebo for the purpose of research and development of NB-02, including but not limited to, the use in the first NB-02 human clinical trial to be conducted by the Company. Under the terms of the Manufacturing and Supply Agreement, upon receipt of a purchase order from the Company no later than 270 days prior to the requested delivery date, Dong-A ST has agreed to produce for the Company tablets of the NB-02 drug substance and placebos at a specified supply price. The Company is obligated to manufacture, or have manufactured, and supply to Dong-A ST the active pharmaceutical ingredients which are necessary to manufacture the NB-02 drug product. The Manufacturing and Supply Agreement has a five year term, subject to earlier termination under certain circumstances.
129
NeuroBo Pharmaceuticals, Inc.
Notes to Consolidated Financial Statements - continued
Payment of Stockholder Legal Expenses
On December 30, 2019, the board of directors of the Company approved the payment or reimbursement of legal fees and costs incurred jointly and severally by certain stockholders of NeuroBo, including E&Healthcare Fund II, E&Healthcare Fund No. 6, E&Healthcare Fund No. 7 (collectively, the “E&Healthcare Funds”), JK BioPharma Solutions, Inc. and Eun Soo Kang, in connection with the 2019 Merger and certain other matters directly related to the Company’s business that required resolution in connection with the closing of the 2019 Merger. The total amount of such legal fees and costs was $227,000 through December 31, 2019 and was accounted as 2019 Merger transaction costs – see Note 4 – Mergers. Na Yeon (Irene) Kim, is the Chief Executive Officer of the sole general partner of each of the E&Healthcare Funds, and as such may be deemed to have an indirect interest in such matters. Jeong Gyun Oh is the President and Chief Executive Officer of JK BioPharma Solutions, Inc. and the spouse of Eun Soo Kang, and as such may be deemed to have an indirect interest in such matters. There were no additional payments of stockholder legal expenses during the year ended December 31, 2020.
License Agreement with YourChoice
As described in Note 5, in connection with the Company’s acquisition of ANA, the Company assumed the YourChoice Agreement between ANA and YourChoice, effective as of the closing of the 2020 Merger. Pursuant to the YourChoice Agreement, YourChoice granted to ANA, during the term of the YourChoice Agreement, an exclusive, worldwide, fee-bearing license derived from the licensed intellectual property throughout the world. The fees due under the YourChoice Agreement include certain single-digit royalty payments and milestone payments in the aggregate of $19.5 million. The term of the YourChoice Agreement will expire on the expiration or invalidation of the last of the licensed patents under the YourChoice Agreement. Akash Bakshi, the Company’s Chief Operating Officer, and a member of the Board of Directors, serves as Chief Executive Officer and director of YourChoice.
15. Defined Contribution Plan
The Company adopted a 401(k) defined contribution plan in November 2018, effective as of January 1, 2019, for all employees over age 21. Employees can defer up to 90% of their compensation through payroll withholdings into the plan subject to federal law limits. Discretionary employer matches vest over a six-year period beginning on the second anniversary of an employee’s date of hire. Employee contributions and any employer matching contributions made to satisfy certain non-discrimination tests required by the Internal Revenue Code are 100% vested upon contribution. No matching contributions were made during the years ended December 31, 2020 and 2019.
16. Subsequent Events
2021 Private Placement
On January 21, 2021, the Company closed on a Securities Purchase Agreement (the “2021 Purchase Agreement”) with certain institutional and accredited investors, pursuant to which the Company, in a private placement agreed to issue and sell an aggregate of 2,500,000 shares of the Company’s common stock at a purchase price of $4.00 per share, and warrants to purchase an aggregate of 2,500,000 shares of common stock (the “2021 Warrants”), resulting in total gross proceeds to the Company of $10.0 million, before deducting placement agent fees and offering expenses. The 2021 Warrants have an initial exercise price of $6.03 per share. The 2021 Warrants are exercisable beginning six months following the date of issuance and will expire five and one-half years following such date.
2019 Plan Evergreen Provision
On January 1, 2021, 786,847 shares were added to the 2019 Plan as a result of the evergreen provision. See Note 10 -Stock-Based Compensation.
CVR Amendment
As further discussed in Note 4, on March 23, 2021, the Company, the Holders’ Representative and the Rights Agent entered into a First Amendment to Contingent Value Rights Agreement to amend the Original CVR Agreement.
130
ITEM 9.CHANGES IN AND DISAGREEMENTS WITH ACCOUNTANTS ON ACCOUNTING AND FINANCIAL DISCLOSURE
None
ITEM 9A.CONTROLS AND PROCEDURES
Evaluation of Disclosure Controls and Procedures
As required by Rules 13a-15(b) and 15d-15(b) under the Exchange Act, our management, with the participation of our principal executive officer (“PEO”) and principal financial officer (“PFO”), evaluated the effectiveness of our disclosure controls and procedures (as defined in Rules 13a-15(e) and 15(d)-15(e) under the Exchange Act) as of the end of the period covered by this annual report for the Company. Based upon that evaluation, our PEO and PFO concluded that our disclosure controls and procedures were not effective as of the end of the period covered by this annual report, as a result of material weaknesses in our internal control over financial reporting, which is discussed further below.
Management’s Annual Report on Internal Control Over Financial Reporting
Our management is responsible for establishing and maintaining adequate internal control over financial reporting to provide reasonable assurance regarding the reliability of our financial reporting and the preparation of financial statements for external purposes in accordance with U.S. generally accepted accounting principles. Internal control over financial reporting includes those policies and procedures that: (i) pertain to the maintenance of records that, in reasonable detail, accurately and fairly reflect the transactions and dispositions of our assets; (ii) provide reasonable assurance that transactions are recorded as necessary to permit preparation of financial statements in accordance with U.S. generally accepted accounting principles, and that our receipts and expenditures are being made only in accordance with authorizations of our management and Board; and (iii) provide reasonable assurance regarding prevention or timely detection of unauthorized acquisition, use or disposition of our assets that could have a material effect on the financial statements.
The scope of management’s assessment regarding the Company’s internal control over financial reporting includes the criteria set forth by the Internal Control Integrated Framework (2013) issued by the Committee of Sponsoring Organizations of The Treadway Commission.
In connection with the preparation of the audited financial statements included elsewhere in this report, management has identified material weaknesses related to internal control deficiencies relating to accounting for clinical trial costs and accounting for mergers. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of our annual or interim financial statements will not be prevented or detected on a timely basis. Specifically, there were misstatements in clinical accruals and expenses that were discovered during the audit process and would not have been detected by our internal control over financial reporting, and management did not have effective controls to effectively assess the technical accounting related to the accounting for the asset acquisition as well as to identify erroneous inputs and assumptions used to value in-process research and development acquired pursuant to the 2020 Merger. See “Remediation Efforts to Address Material Weaknesses” below for steps we are taking to correct theses material weaknesses.
Remediation Efforts to Address Material Weaknesses
We are in the process of remediating, but have not yet remediated, the material weakness related to clinical trial costs as described above. Under the oversight of the audit committee, management is developing a detailed plan and timetable for the implementation of appropriate remedial measures to address the material weakness. As of the date of this report, we have taken the following actions and are in the process:
| • | we have added more experienced accounting personnel, directly responsible for the oversight of the accounting for clinical trial expenses; |
131
| • | we have improved processes in the area of clinical site expense monitoring, including increasing communication between our accounting and clinical personal, as well as with our clinical vendors; |
| • | we have retained additional qualified outside consultants, where necessary, to advise on highly complex technical accounting matters; and |
| • | We will implement enhanced controls relative to the review and oversight of the accounting for clinical trial expenses as well as the review of technical accounting assessments and review of valuation processes. |
Management may decide to take additional measures to remediate the material weaknesses as necessary.
Notwithstanding the identified material weaknesses, management, including our PEO and PFO, believes the consolidated financial statements included in this annual report fairly represent in all material respects our financial condition, results of operations and cash flows as of and for the periods presented in accordance with GAAP.
Inherent Limitations on Internal Controls
Our management, including our PEO and PFO, does not expect that our disclosure controls and procedures or our internal controls will prevent all error and all fraud. A control system, no matter how well conceived and operated, can provide only reasonable, not absolute, assurance that the objectives of the control system are met. Because of the inherent limitations in all control systems, no evaluation of controls can provide absolute assurance that all control issues and instances of fraud, if any, within the company have been detected. These inherent limitations include the realities that judgments in decision-making can be faulty, and that breakdowns can occur because of simple error or mistake. Additionally, controls can be circumvented by the individual acts of some persons, by collusion of two or more people, or by management override of the control. The design of any system of controls is also based in part upon certain assumptions about the likelihood of future events, and there can be no assurance that any design will succeed in achieving its stated goals under all potential future conditions. Over time, controls may become inadequate because of changes in conditions, or the degree of compliance with the policies or procedures may deteriorate. Because of the inherent limitations in a cost-effective control system, misstatements due to error or fraud may occur and not be detected.
Changes in Internal Control Over Financial Reporting
Other than the material weaknesses and remediation activities noted above, there have been no changes in the Company’s internal control over financial reporting during the quarter ended December 31, 2020 that have materially affected, or are reasonable likely to materially affect, the Company’s internal control over financial reporting.
None
ITEM 10.DIRECTORS, EXECUTIVE OFFICERS AND CORPORATE GOVERNANCE
Directors and Executive Officers
The Board is divided into three classes. Members of each class serve staggered three-year terms. The terms of office of directors in Class I, Class II and Class III expire at the annual meetings of stockholders to be held in 2023, 2021 and
132
2022, respectively. The following table provides information as to each person who is, as of the filing hereof, a director and/or executive officer of the Company.
Name | Position(s) | Age |
Richard Kang | Class III Director, President, Chief Executive Officer, Interim Chief Financial Officer, Secretary and Treasurer | 49 |
Akash Bakshi | Class II Director, Chief Operating Officer and Senior Vice President | 32 |
Jason L. Groves | Class II Director | 49 |
Na Yeon (Irene) Kim | Class I Director | 44 |
Jeong Gyun Oh | Class II Director | 47 |
Michael Salsbury | Class III Director | 71 |
Douglas J. Swirsky | Class I Director and Chair of the Board | 51 |
Business Experience and Background of Directors and Executive Officers
Dr. Richard Kang has served a member of our Board since December 2019. He has served as our President, Chief Executive Officer, Interim Chief Financial Officer, Secretary and Treasurer, effective January 1, 2020. Previously, he served as an officer of NeoImmuneTech, Inc., a biotechnology company developing T cell-centered novel immunotherapeutics, from May 2014 to December 2019, most recently as Co-President and Chief Executive Officer and a member of the board of directors. Dr. Kang held various officer positions at Private NeuroBo, including as President and Chief Operating Officer from September 2017 through February 2019, and also served on Private NeuroBo’s board of directors from July 2017 to February 2019. He was reappointed to Private NeuroBo’s board of directors in December 2019. Dr. Kang also served as President and Chief Executive Officer of JK BioPharma Solutions, Inc. from January 2013 to February 2019. Dr. Kang received a Ph.D. in Molecular Plant Pathology from The University of Edinburgh, an M.S. in Plant Molecular Genetics from Seoul National University and a B.S. in Horticultural Science from Seoul National University. Our Board believes that Dr. Kang’s business experience, executive officer positions at the Company and prior experience as Private NeuroBo's former President, Chief Operating Officer and director qualifies him to serve as a director.
Mr. Akash Bakshi was appointed as a director to our Board and also as our Chief Operating Officer and Senior Vice President in connection with the Company’s acquisition of ANA on December 31, 2020 and has served as a member of our Board since then. Mr. Bakshi served as Chief Executive Officer and a director of ANA, a Silicon Valley-based start-up developing a proprietary niclosamide formulation as a widely accessible antiviral treatment for patients with COVID-19, and has served in these roles since ANA’s inception in March 2020. Mr. Bakshi also serves as CEO and director of YourChoice, a biotech start-up since its inception in May 2018. Prior to joining YourChoice Therapeutics, Mr. Bakshi was an Assistant Director of Technology Analysis at UC Berkley’s Office of Intellectual Property and Industry Research Alliances, a role he began in May 2015 and served until February 2019. Mr. Bakshi received his B.S. in Biochemistry from UC San Diego in 2010 and his Masters of Science from the University of Queensland in 2011. Our Board believes that Mr. Bakshi’s extensive experience in the operational design and regulatory approval of a treatment for COVID-19 and his prior Chief Executive Officer and director roles with ANA, which the Company acquired on December 31, 2020, qualify him to serve as a director.
Mr. Jason L. Groves, Esq. has served a member of our Board since December 2019. He is the Executive Vice President and General Counsel of Medifast, Inc. (NYSE: MED), a publicly held leading manufacturer and distributor of clinically-proven, healthy-living products and programs. He has served in this position since November 2011, and as Corporate Secretary since June 2015. Preceding and during his current position, Mr. Groves was a Medifast, Inc. director from 2009 to 2015, serving on the Audit Committee from 2009 to 2011. Mr. Groves was Assistant Vice President of Government Affairs for Verizon Maryland from 2003 until 2011, after having joined Verizon in 2001. A United States Army veteran, Mr. Groves was a direct-commissioned Judge Advocate in the United States Army Judge Advocate General’s (JAG) Corps. As a JAG officer, he practiced law and had the distinction of prosecuting criminal cases in the District Court of Maryland as a Special Assistant United States Attorney. Mr. Groves recently completed nine years with the Anne Arundel Medical Center Board of Trustees, chairing their international captive insurance company board for eight years. Mr. Groves received his Bachelor of Science degree, cum laude, in Hospitality Management from Bethune-
133
Cookman University, and obtained his Juris Doctor from North Carolina Central University School of Law. Our Board believes that Mr. Grove’s experience serving as an independent director, audit committee member and general counsel of a large corporation and assisting with the initial international introduction of such corporation’s products qualify him to serve as a director.
Ms. Na Yeon (“Irene”) Kim has served as a member of our Board since December 2019 and served as the Chair of our Board from December 2019 to January 2021. Prior to December 2019, she had served on the Board of Private NeuroBo since April 2018. Ms. Kim also currently serves as the Chief Executive Officer of E&Investment, Inc., a South Korean venture capital firm specializing in investments in life sciences companies, a position she has held since March 2018. From October 2015 until March 2018, Ms. Kim was a Representative Director for The SEED Investment Co., Ltd. (formerly known as OST Investment Co., Ltd.), a South Korean investment and fund manager specializing in investments in life sciences companies, and from January 2015 until December 2017, Ms. Kim served as member of the board of directors of Macrogen, Inc., a South Korean, publicly-traded biotechnology company specializing in precision medicine and biotechnology. Ms. Kim also served as an officer of AJUIB Investment, Inc., a venture capital firm headquartered in South Korea specializing in investments in life-science companies from August 2014 until September 2015. Ms. Kim focuses on investment opportunities in a number of industries, particularly in the field of BioPharma, and has more than 15 years of accumulated experience of investment in private equity/venture capital markets. As an investor representative, Ms. Kim has successfully managed more than $400 million in private equity and venture capital funds. Ms. Kim holds an M.S. and B.S. in biomolecular engineering, as well as an M.B.A. from Yonsei University in Korea. Our Board believes that Ms. Kim’s specialized knowledge in building values in life sciences companies and her extensive investment management experience qualify her to serve as a director.
As disclosed above, on March 11, 2021, the E&H Entities (of which Ms. Kim is the Chief Executive Officer, or the Chief Executive Officer of the sole general partner, as applicable) publicly disclosed that (i) the E&H Entities entered into a Voting Agreement with Dong-A ST on March 9, 2021; and (ii) the E&H Entities and Dong-A ST intend, among other things as contemplated by the Voting Agreement, to nominate a slate of directors to be elected to the Board at the Company’s 2021 Annual Meeting of Stockholders and 2022 Annual Meeting of Stockholders and to propose the declassification of the Board.
Mr. Jeong Gyun Oh has served a member of our Board since December 2019. Prior to that, he had served on the Board of Private NeuroBo since March 2019. From January 2017 until February 2019, Mr. Oh served as Chief Financial Officer of JK BioPharma Solutions, Inc., a privately-held, drug development company focused on early stage developmental assets, and has served as the President and Chief Executive Officer of JK BioPharma Solutions, Inc. since March 2019. From August 2001 until December 2016, Mr. Oh held multiple financial positions ultimately culminating in his service as Vice President of Finance with CDNetworks Co., Ltd., a global content delivery network service company. Mr. Oh holds a B.A. in business administration from Seoul National University. Our Board believes that Mr. Oh’s experience as a corporate finance executive with extensive expertise in financial operations, including financial/managerial accounting, business planning and budgeting, qualify him to serve as a director.
Mr. Michael Salsbury has served a member of our Board since December 2019. He has served as Counsel to Verisma Systems, Inc., a provider of cloud-based automated disclosure management systems, since September 2017. From February 2013 to July 2017, he served as Secretary and General Counsel to Best Doctors, Inc., a provider of expert medical opinions. Mr. Salsbury has more than 25 years’ experience as a senior executive with public and private companies and private law practice. Mr. Salsbury received a J.D. and M.B.A. from University of Virginia and a B.A. from Dartmouth College. Our Board believes that Mr. Salsbury’s legal expertise and his experience serving as general counsel and secretary of a Fortune 100 corporation qualifies him to serve as a director.
Mr. Douglas J. Swirsky has served as a member of our Board since September 2020 and as Chair of our Board since January 2021. He has served as the Chief Financial Officer of AavantiBio, Inc., a gene therapy company focused on transforming the lives of patients with rare genetic diseases, since February 2021 and held various other positions with the company between March 2020 and February 2021. He previously served as President and Chief Executive Officer and a director of a publicly traded pharmaceutical company, Rexahn Pharmaceuticals, Inc. (NASDAQ: REXN), from November 2018 until November 2020, when Rexahn merged with Ocuphire Pharma, Inc. He served as Rexahn’s President, Chief Financial Officer, and Corporate Secretary from January 2018 to November 2018. Prior to joining
134
Rexahn, Mr. Swirsky was President, CEO and a director of GenVec, Inc., a publicly traded biotechnology company, a position he held from 2013 through the sale of the company in 2017. He joined GenVec in 2006 as Chief Financial Officer. Prior to joining GenVec, Mr. Swirsky was a Managing Director and the Head of Life Sciences Investment Banking at Stifel Nicolaus from 2005 to 2006 and held investment banking positions at Legg Mason from 2002 until Stifel Financial’s acquisition of the Legg Mason Capital Markets business in 2005. Mr. Swirsky also previously held investment banking positions at UBS, PaineWebber and Morgan Stanley. Mr. Swirsky currently serves as the Chairman of the Board of Cellectar Biosciences, Inc., a publicly traded clinical stage biopharmaceutical company. Within the last five years, Mr. Swirsky also served on the boards of directors of then-publicly traded life sciences companies Pernix Therapeutics Holdings, Inc. and Fibrocell Science, Inc. Mr. Swirsky is a certified public accountant and a CFA® charterholder. He has been recognized as NACD Directorship Certified™ by the National Association of Corporate Directors. He received his B.S. in Business Administration from Boston University and his M.B.A. from the Kellogg School of Management at Northwestern University. Our Board believes that Mr. Swirsky’s experience serving as President and Chief Executive Officer and a director of a publicly traded pharmaceutical company, Rexahn, qualifies him to serve as a director.
Code of Business Conduct and Ethics
Our Board has adopted a code of business conduct and ethics that applies to all of our employees, officers and directors, including our Chief Executive Officer, Chief Financial Officer and other executive officers. We intend to disclose future amendments to certain provisions of our code of business conduct and ethics, or waivers of these provisions, on our website. The full text of our code of conduct is posted on the investor relations section of our website at http://neurobopharma.com/corporate-governance/highlights.
Audit Committee
Our Board has established an audit committee, which is comprised of Mr. Groves, Mr. Oh and Mr. Swirsky, with Mr. Swirsky serving as chair of the committee. Each member of our audit committee meets the requirements for independence under the current Nasdaq and SEC rules and regulations and is financially literate. In addition, our Board has determined that each of Mr. Swirsky and Mr. Oh qualify as an “audit committee financial expert” as defined in Item 407(d)(5)(ii) of Regulation S-K promulgated under the Securities Act. This designation does not impose on either of them any duties, obligations or liabilities that are greater than are generally imposed on members of our audit committee and our Board.
ITEM 11.EXECUTIVE COMPENSATION
Executive Officer Compensation
The following tables and accompanying narrative disclosure discuss the compensation awarded to, earned by, or paid to:
| • | Dr. Richard Kang, our President, Chief Executive Officer, Interim Chief Financial Officer, Secretary and Treasurer; |
| • | Dr. Mark Versavel, our former Chief Medical Officer; and |
| • | Nicola Shannon, our former Vice President, Clinical Operations. |
We refer to these three executive officers as the “named executive officers.”
135
Summary Compensation Table for 2020
The following table presents summary information regarding the total compensation for services rendered in all capacities that was earned by our named executive officers during the fiscal years ended December 31, 2020 and 2019.
NAME AND PRINCIPAL POSITION |
| YEAR |
| SALARY |
| BONUS |
| NON-EQUITY |
| STOCK |
| OPTION |
| ALL OTHER |
| TOTAL |
Richard Kang (1) | | 2020 | | 302,308 | | — | | | | | | — | | — | | 302,308 |
President, Chief Executive Officer, Interim Chief Financial Officer, Secretary and Treasurer | | 2019 | | — | | — | | | | | | — | | — | | — |
Dr. Mark Versavel (2) | | 2020 | | 357,000 | | — | | | | | | — | | — | | 357,000 |
Former Chief Medical Officer | | 2019 | | — | | — | | | | | | — | | — | | — |
Nicola Shannon (3) | | 2020 | | 281,750 | | — | | | | | | — | | — | | 281,750 |
Former Vice President, Clinical Operations | | 2019 | | 66,250 | | 66,250 | | | | | | | |
| (1) | Dr. Kang was appointed as our President, Chief Executive Officer, Interim Chief Financial Officer, Secretary and Treasurer on December 31, 2019 and received no compensation from the Company during the fiscal year ended December 31, 2019. |
| (2) | Dr. Versavel was appointed as our Chief Medical Officer on December 31, 2019 and received no compensation from the Company during the fiscal year ended December 31, 2019. Pursuant to the terms of the consulting agreement between the Company and vZenium LLC, a company owned and managed entirely by Dr. Versavel, the Company provided notice of non-renewal of the consulting agreement, and, consequently, the term of the consulting agreement expired on January 1, 2021. |
| (3) | Ms. Shannon was appointed as our Vice President, Clinical Operations, on December 31, 2019 and received no compensation from the Company during the fiscal year ended December 31, 2019. On January 11, 2021, Ms. Shannon resigned as the Company’s Vice President of Clinical Operations effective January 22, 2021. |
| (4) | Amounts shown in this column for Dr. Versavel represent fees paid to vZenium LLC pursuant to the consulting agreement between the Company and vZenium LLC. |
Agreements with Our Named Executive Officers
We have entered into written agreements with each of our named executive officers.
Dr. Richard Kang
On February 11, 2020, we entered into an Employment Agreement with Dr. Kang, our President and Chief Executive Officer, which was given retroactive effect to January 1, 2020 (the “CEO Employment Agreement”). The CEO Employment Agreement provides for the at-will employment of Dr. Kang as our President and Chief Executive Officer, at a base salary of $300,000 per year. Dr. Kang will be eligible to receive annual bonus compensation with an annual target bonus opportunity of 50% of his base salary, starting with the 2020 fiscal year. Dr. Kang will also be eligible to receive an annual stock option grant and to participate in our employee benefit plans that are in effect for similarly-situated employees.
136
Pursuant to the terms of the CEO Employment Agreement, if Dr. Kang is terminated for any reason, including by us for cause or by Dr. Kang for any reason other than for good reason, Dr. Kang will be eligible to receive any (i) earned or accrued base salary and paid time off through the last day of his employment, (ii) any unreimbursed business expenses incurred through the last day of his employment and (iii) any vested benefits due to Dr. Kang under any Company benefit plan.
In addition, the CEO Employment Agreement provides that if we terminate Dr. Kang’s employment without cause, or if Dr. Kang terminates his employment for good reason, he will be entitled to the following, subject to obtaining from him a general release of claims: (i) severance payments for four months at his then-current base salary payable in accordance with our current payroll practices, (ii) an amount equal to his then-current target bonus prorated through the last day of his employment and payable within 60 days of his last day of employment, (iii) full acceleration of vesting for all stock options as of the last day of his employment and (iv) coverage under the Company’s group health plans for the twelve-month period immediately following the date of his termination for Dr. Kang and his eligible dependents at the same level and at the same cost had Dr. Kang not been terminated.
If Dr. Kang’s employment is terminated due to his disability or death, he or his estate, as applicable, will also be entitled to receive any earned, but unpaid, annual bonus for the fiscal year ending immediately prior to the fiscal year of his termination for death or disability. In the event of a termination due to disability, Dr. Kang will also be entitled to receive any benefits under an applicable long-term disability plan, program or policy.
Under the CEO Employment Agreement, Dr. Kang will also be subject to confidentiality and protection of intellectual property provisions and noncompetition provisions and non-solicitation provisions during his employment and the 12 months thereafter.
Dr. Mark Versavel
We entered into a consulting agreement with vZenium LLC, a Massachusetts limited liability company owned and managed entirely by Dr. Versavel, effective January 1, 2020 (the “Consulting Agreement”). Pursuant to the Consulting Agreement, Dr. Versavel agreed to provide certain professional services, including services as our Chief Medical Officer, for approximately 139 hours per month in exchange for cash compensation of $29,750 per month payable in arrears on the last business day of the month.
Dr. Versavel was also eligible to receive reimbursement for certain out of pocket costs approved in advance by the Company. The Consulting Agreement was to continue on a month to month basis unless and until the death or disability of Dr. Versavel, the date Dr. Versavel ceases to be the sole owner of vZenium LLC, the date it is terminated for cause or until January 1, 2021, provided we and vZenium LLC had not previously agreed to extend the term. The Consulting Agreement was terminable upon the mutual agreement of the parties or upon 10 days’ written notice by either party.
The Consulting Agreement contains non-solicitation provisions applicable during the term and for the one-year period following termination and provisions requiring that intellectual property relating to or resulting from the services provided by Dr. Versavel are the exclusive property of the Company or its affiliates. Dr. Versavel was not eligible to participate in any Company health, life, disability or any insurance plan or retirement plan offered by the Company to its employees.
Pursuant to the terms of the Consulting Agreement, the Company provided notice of non-renewal of the consulting agreement, and, consequently, the term of the Consulting Agreement expired on January 1, 2021.
Nicola Shannon
We entered into an offer letter with Nicola Shannon, effective January 1, 2020. The offer letter provided for the at-will employment of Ms. Shannon as our Vice President of Clinical Operations, reporting to our Chief Medical Officer, at a base salary of $280,000 per year. Ms. Shannon was eligible to receive annual bonus compensation with an annual target bonus opportunity of 25% of her base salary, starting with the 2020 fiscal year. Ms. Shannon was also eligible to
137
participate in our employee benefit plans that were in effect for similarly-situated employees. Ms. Shannon is also subject to confidentiality and protection of intellectual property provisions. Additionally, we granted to Ms. Shannon a discretionary bonus award for the fiscal year ended December 31, 2019 in the amount of $66,250, which was due and payable on or before February 28, 2020.
On January 11, 2021, Nicola Shannon resigned as the Company’s Vice President of Clinical Operations effective January 22, 2021.
Outstanding Equity Awards at Fiscal Year-End 2020
We granted no options or other equity awards to our executive officers in 2020.
As disclosed above, on December 30, 2019, the Company, formerly known as Gemphire Therapeutics Inc., completed its business combination with the private entity formerly known as NeuroBo Pharmaceuticals, Inc. (referred to herein as Private NeuroBo), in accordance with the terms of the Merger Agreement, pursuant to which, among other matters, Merger Sub merged with and into Private NeuroBo, with Private NeuroBo continuing as a wholly owned subsidiary of the Company and the surviving corporation of the Merger. The outstanding stock option awards noted below were granted under the NeuroBo Pharmaceuticals, Inc. 2018 Stock Plan (the “NeuroBo 2018 Plan”) and were converted from Private NeuroBo options into Company options at the effective time of the Merger.
The following table sets forth information regarding outstanding stock options held by our named executive officers as of December 31, 2020:
NAME |
| GRANT DATE (1) |
| VESTING |
| NUMBER OF |
| NUMBER OF |
| OPTION |
| OPTION |
| ||||||||||||||||
Dr. Mark Versavel, |
|
| January 28, 2019 |
|
| January 28, 2019 | |
| 68,596 (2) |
|
| 228,620 (2) |
|
| 0.63 |
|
| January 28, 2029 |
| ||||||||||
Former Chief Medical Officer |
|
| January 31, 2019 |
|
| January 31, 2019 | |
| 45,724 |
|
| - |
|
| 0.63 |
|
| January 31, 2029 |
| ||||||||||
(1) All of the outstanding stock option awards were granted under the NeuroBo 2018 Plan.
(2) Subject to continued service: (a) 80,017 shares underlying the options shall become vested shares upon certain milestones in relation to the Company’s Phase III U.S. NB-01-301 study; (b) 68,586 shares underlying the option shall become vested shares upon certain regulatory milestones related to the Company’s product candidate, NB-02; and (c) an additional 80,017 option shares shall become vested shares upon the occurrence of certain additional milestones related to the Company’s Phase III U.S. trial of NB-01-301. 17,147 shares underlying the options became vested shares on January 28, 2019; and an additional 8,573 of shares underlying the options became vested shares on each of February 1, 2019, May 1, 2019, August 1, 2019, November 1, 2019, February 1, 2020, and May 1, 2020.
Non-Employee Director Compensation
Our non-employee directors receive a mix of cash and share-based compensation intended to encourage non-employee directors to continue to serve on our Board, further align the interests of the directors and stockholders, and attract new
138
non-employee directors with outstanding qualifications. Directors who are employees or officers of the Company do not receive any additional compensation for Board service.
From January 1, 2020 through June 30, 2020, the following compensation schedule was in place for non-employee directors:
| ● | Option to purchase 60,000 shares, vested monthly over 36 months upon election as a director; |
| ● | Annual cash compensation of $20,000 per year; and |
| ● | $20,000 per year for service on a committee, irrespective of the number of committees. |
Effective July 1, 2020 and continuing through December 31, 2020 and thereafter, the Board adopted the following compensation schedule for non-employee directors:
| ● | Option to purchase 60,000 shares, vested monthly over 36 months upon election as a director; |
| ● | Annual cash compensation of $20,000 per year; |
| ● | $20,000 per year for service on a committee, irrespective of the number of committees; |
| ● | $20,000 additional per year for service for each of the Chair of the Nomination Committee and the Compensation Committee; and |
| ● | $40,000 per year additional for service for each of the Chair of the Audit Committee and the Management Committee. |
The following table provides compensation information for the fiscal year ended December 31, 2020 for each non-employee member of our Board.
| | | | | | | |
Name |
| Fees Earned or | Option Awards ($) (1) | Total |
| ||
| | | | | | | |
Ms. Na Yeon (Irene) Kim | | | $70,000 | $335,197 | | 405,197 | |
Jeong Gyun Oh | | | $40,000 | $335,197 | | 375,197 | |
Jason Groves | | | $40,000 | $335,197 | | 375,197 | |
Michael Salsbury | | | $50,000 | $335,197 | | 385,197 | |
Tae Heum (Ted) Jeong (2) | | | $60,000 | $335,197 | | 395,197 | |
Dr. Steven Gullans (3) | | | $23,333 | $335,197 | | 358,530 | |
Douglas Swirsky (4) | | | $13,333 | $234,513 | | 247,846 | |
(1) Options were granted under the 2019 Equity Incentive Plan (“2019 Plan”) to each of the Company’s non-employee directors in January 2020. Each option vests, subject to continuing service, in 36 equal monthly installments beginning on February 13, 2020, except with respect to Mr. Swirsky, whose options were granted on September 1, 2020, such that all such options will be fully vested on the third anniversary of the date of grant. The amounts reported reflect the aggregate grant date fair value of each option granted to the Company’s non-employee directors during the fiscal year ended December 31, 2020, as computed in accordance with ASC 718.
(2) Tae Heum (Ted) Jeong resigned from the Board effective as of January 9, 2021.
(3) Dr. Gullans resigned from the Board effective as of August 30, 2020.
(4) Mr. Swirsky was appointed to the Board effective as of September 1, 2020.
139
Other Director Compensation
Mr. Bakshi was appointed as a director and as the Company’s Chief Operating Officer and Senior Vice President on December 31, 2020, following the effective time of the Company’s acquisition of ANA, and received no compensation from the Company for services as a director for the fiscal year ended December 31, 2020, as shown in the table below:
Name |
| Fees Earned or | Option Awards ($) | Total | ||
Akash Bakshi | | | -- | -- | | -- |
ITEM 12. | SECURITY OWNERSHIP OF CERTAIN BENEFICIAL OWNERS AND MANAGEMENT AND RELATED STOCKHOLDER MATTERS |
The following table sets forth information regarding beneficial ownership of our common stock, as of March 31, 2021 by:
● | each person, or group of affiliated persons, known by us to beneficially own more than 5% of our common stock; |
● | each of our named executive officers; |
● | each of our directors; and |
● | all of our current executive officers and directors as a group. |
The table lists applicable percentage ownership based on 22,171,182 shares of common stock outstanding as of March 31, 2021. In addition, the rules include shares of our common stock issuable pursuant to the exercise of stock options and warrants that are either immediately exercisable or exercisable within 60 days of March 31, 2021. These shares are deemed to be outstanding and beneficially owned by the person holding those options for the purpose of computing the percentage ownership of that person, but they are not treated as outstanding for the purpose of computing the percentage ownership of any other person.
140
We have determined beneficial ownership in accordance with the rules of the SEC. These rules generally attribute beneficial ownership of securities to persons who possess sole or shared voting power or investment power with respect to those securities. Unless otherwise indicated, the persons or entities identified in this table have sole voting and investment power with respect to all shares shown as beneficially owned by them, subject to applicable community property laws. Except as otherwise noted below, the address for each person or entity listed in the table is c/o NeuroBo Pharmaceuticals, Inc., 200 Berkeley Street, 19th Floor, Boston, Massachusetts, 02116.
|
| SHARES |
|
|
|
NAME AND TITLE OF BENEFICIAL OWNER | | NUMBER | | PERCENT | |
Greater than 5% stockholders | | | | | |
JK BioPharma Solutions, Inc. (1) | | 1,817,842 | | 8.2 | % |
Dong-A ST Co., Ltd. (2) | | 2,880,612 | | 13.0 | % |
E&Investment, Inc. (3) | | 7,321,789 | | 33.0 | % |
Roy Lester Freeman (4) | | 1,456,160 | | 6.6 | % |
Directors and Named Executive Officers | | | | | |
Dr. Richard Kang, President, Chief Executive Officer, Interim Chief Financial Officer, Secretary, Treasurer and Director | | — | | — | |
Na Yeon (Irene) Kim, Director (3)(5) | | 7,348,455 | | 33.1 | % |
Jeong Gyun Oh, Director (1)(5) | | 1,843,808 | | 8.3 | % |
Jason Groves, Director (5) | | 26,666 | | * | |
Michael Salsbury, Director (5) | | 26,666 | | * | |
Akash Bakshi, Director (5) | | 884,072 | | 4.0 | % |
Douglas J. Swirsky, Chair of the Board of Directors (6) | | 8,333 | | * | |
Dr. Mark Versavel, former Chief Medical Officer | | 114,310 | | * | |
Nicola Shannon, former Vice President, Clinical Operations | | — | | * | |
All current executive officers and directors as a group (7 persons, not including Dr. Mark Versavel and Nicola Shannon) | | 10,143,000 | | 45.7 | % |
* Represents beneficial ownership of less than one percent.
| (1) | Based on the Company’s review of a filing made on a Schedule 13D on January 10, 2020 with the SEC. JK BioPharma Solutions, Inc. (“JK”) owns 1,817,842 shares of common stock. Mr. Oh may be deemed a beneficial owner of JK’s 1,817,842 shares due to the fact that Mr. Oh is one of two directors of JK. Mr. Oh’s beneficial ownership excludes (and he disclaims beneficial ownership of) 240,051 shares of common stock held by his spouse, Eun Soo Kang. The business address of Mr. Oh and the address of the principal executive offices of JK is 1 Research Court, Suite 370, Rockville, MD 20850. |
| (2) | Solely based on the Company's review of a filing made on a Schedule 13D on March 11, 2021 with the SEC. Dong-A ST Co., Ltd. is a South Korean corporation. The address of Dong-A ST Co., Ltd. Is 64, Cheonho-daero, Dongdaemun-gu, Seoul, Republic of Korea. |
| (3) | Based on the Company’s review of a filing made on an amendment to a Schedule 13D on March 15, 2021 with the SEC. The amendment to the Schedule 13D was filed by The E&Healthcare Investment Fund II (“Fund II”), The E&Healthcare Investment Fund No. 6 (“Fund 6”), The E&Healthcare Investment Fund No. 7 (“Fund 7”), E&Investment, Inc. (“GP”), and Na Yeon Kim. Fund II beneficially owns 4,335,800 shares of common stock, Fund 6 beneficially owns 1,121,190 shares of common stock, Fund 7 beneficially owns 1,864,799 shares of common stock, GP beneficially owns 7,321,789 shares of common stock, and Ms. Kim beneficially owns 7,321,789 shares of Common Stock. The business address of Ms. Kim and the address of the principal office of the entity entities noted in this footnote is 16th floor, Yeoksam I-Tower, 326, Teheran-ro, Gangnam-gu, Seoul, Republic of Korea 06211. |
| (4) | Solely based on the Company’s review of a filing made on a Schedule 13G on February 13, 2020 with the SEC. The address of Mr. Freeman is 200 Berkeley Street, 19th Floor, Boston, Massachusetts, 02116. |
141
| (5) | Each Director of the Company (other than Dr. Kang, Mr. Bakshi and Mr. Swirsky) was issued a stock option to purchase 60,000 shares of common stock on January 13, 2020. The option expires January 12, 2030, has an exercise price of $8.39, and vests in 36 equal monthly installments beginning on February 29, 2020, subject to continued service with the Company, such that the option will be fully vested on the third anniversary of the date of grant. 23,333 shares underlying the option are vested as of March 31, 2021 and an additional 3,333 shares underlying the option will become vested within 60 days of March 31, 2021, subject to continued service with the Company. |
| (6) | Represents shares underlying outstanding stock options that are vested or will become vested within 60 days of March 31, 2021. |
Securities Authorized for Issuance Under Equity Compensation Plans
The following table presents information as of December 31, 2020 with respect to compensation plans under which shares of our common stock may be issued.
Plan Category |
| Number of |
| Weighted-average |
| Number of |
| |||
Equity compensation plans approved by security holders |
|
| 920,355 |
|
| 3.61 |
|
| 5,121,605 | (1) |
Equity compensation plans not approved by security holders |
|
|
|
|
|
|
|
|
|
|
| | | | | | | | | | |
Total |
|
| 920,355 |
| | 3.61 |
|
| 5,121,605 |
|
(1) The number of shares of common stock remaining available for future issuance represent 3,623,708 shares available for issuance under the 2019 Plan and 1,497,891 shares subject to options awarded under the NeuroBo 2018 Plan.
ITEM 13. | CERTAIN RELATIONSHIPS AND RELATED TRANSACTIONS AND DIRECTOR INDEPENDENCE |
Certain Relationships and Related Party Transactions
The following includes a summary of transactions since January 1, 2019 to which we have been a party, in which the amount involved in the transaction exceeded $120,000, and in which any of our directors, executive officers or, to our knowledge, beneficial owners of more than 5% of our capital stock or any member of the immediate family of any of the foregoing persons had or will have a direct or indirect material interest, other than certain equity and other compensation, termination, change of control and other arrangements, which are described under “Executive Compensation.”
Manufacturing Agreement with Dong-A ST
On September 28, 2018, Private NeuroBo entered into a five year manufacturing and supply agreement with Dong-A ST for manufacturing and supply of NB-01 drug substance and placebos for the purpose of research and development to be used in Phase 3 clinical trials (the “Manufacturing Agreement”). As of March 31, 2021, Dong-A ST was the beneficial owner of more than 5% of our capital stock. Under the terms of the Manufacturing Agreement, Dong-A ST has agreed to produce for NeuroBo a specified number of tablets of the NB-01 drug substance and placebos at a supply price to be determined at the time of each individual order. In addition, prices were set for stability testing of the NB-01 drug substance and placebo. The Company recognized approximately $314,000 of product manufacturing related costs within research and development expenses for the year ended December 31, 2019 and did not recognize such expenses for the year ended December 31, 2020.
142
The Manufacturing Agreement will automatically terminate in the event that the license agreement with Dong-A ST is terminated for any reason. In addition, each of Dong-A ST and Private NeuroBo may terminate the Manufacturing Agreement (1) upon the material breach by the other party, if the breach is not cured within a specified number of days after receiving notice from the terminating party, or if the breach cannot reasonably be cured within such period and the breaching party has not started to remedy the breach within such period and diligently endeavored to cure the breach within a reasonable time thereafter, or (2) in the event that (i) the other party is the subject of a petition for bankruptcy, reorganization, or arrangement and the same is not dismissed within thirty days thereof, (ii) a receiver or trustee is appointed for all or a substantial portion of the assets of the other party, or (iii) the other party makes an assignment for the benefit of its creditors.
Convertible Promissory Note Financing
Prior to 2019 Merger, the following holders of more than 5% of Private NeuroBo’s capital stock and Private NeuroBo directors held convertible promissory notes issued by the Company in February 2018 (the “Convertible Notes”):
Related Party |
| | |||
JK BioPharma Solutions, Inc. (1) | | $ | 200,000 | | |
Roy Freeman, M.D. (2) | | $ | 100,000 | | |
The E&Healthcare Investment Fund II (3) |
| $ | 116,000 |
| |
The E&Healthcare Investment Fund No. 6 (3) |
| $ | 32,000 |
| |
The E&Healthcare Investment Fund No. 7 (3) |
| $ | 52,000 |
| |
| (1) | Jeong Gyun Oh, a NeuroBo director, is President and CEO of JK Biopharma Solutions, Inc. JK Biopharma Solutions, Inc. currently assists the Company with certain activities that are primarily related to linguistic translations. All work done to date has been done without compensation. However, Private NeuroBo issued a $32,000 payment to JK BioPharma Solutions, Inc. in February 2018 as reimbursement for payments made to Private NeuroBo vendors during late 2017 and early 2018. |
| (2) | Roy Freeman, M.D. is a co-founder and was a Private NeuroBo director at the time of the Convertible Note purchase. |
| (3) | E&Investment, Inc. is the sole general partner of, and has voting power over the shares held by, each of the E&Healthcare Funds. Na Yeon (Irene) Kim, a NeuroBo director, is the Chief Executive Officer of E&Investment, Inc., and as such has voting and investment control over the shares held by E&Investment, Inc. and its affiliated funds, including the E&Healthcare Funds. |
The Convertible Notes were converted into shares of Private NeuroBo common stock in connection with the 2019 Merger.
Payment of Stockholder Legal Expenses
On December 30, 2019, the Board approved the payment or reimbursement of legal fees and costs incurred jointly and severally by certain stockholders of NeuroBo, including E&Healthcare Fund II, E&Healthcare Fund No. 6, E&Healthcare Fund No. 7 (collectively, the “E&Healthcare Funds”), JK BioPharma Solutions, Inc. and Eun Soo Kang, in connection with the Merger and certain other matters directly related to the Company’s business that required resolution in connection with the closing of the Merger. The total amount of such legal fees and costs was $227,000 through December 31, 2020 and was accounted as Merger transaction costs. Each of Na Yeon (Irene) Kim and Jeong Gyun Oh is a member of our Board. Ms. Kim, is the Chief Executive Officer of the sole general partner of each of the E&Healthcare Funds, and as such may be deemed to have an indirect interest in such matters. Mr. Oh is the President and Chief Executive Officer of JK BioPharma Solutions, Inc. and the spouse of Eun Soo Kang, and as such may be deemed to have an indirect interest in such matters.
ANA Merger and Lock-Up Agreements
As described above in “Management’s Discussion and Analysis of Financial Condition and Results of Operations—Merger with ANA”, on December 31, 2020, the Company acquired ANA pursuant to the 2020 Merger Agreement by
143
and among NeuroBo, the First Merger Sub, the Second Merger Sub, ANA, and Akash Bakshi, solely in his capacity as the representative of the securityholders of ANA. Immediately prior to the 2020 Merger, Mr. Bakshi served as Chief Executive Officer and a director of ANA. In connection with the 2020 Merger, in respect of Mr. Bakshi’s capacity as a former ANA securityholder, (i) the Company issued 884,072 shares to Mr. Bakshi, which, based on the closing price as of the consummation of the 2020 Merger, were collectively worth $4,641,378.00 on such date; and (ii) Mr. Bakshi is also entitled to a pro rata portion of any milestone payments paid under the 2020 Merger Agreement, as described in more detail in “Merger with ANA” above.
Concurrently and in connection with the execution of the 2020 Merger Agreement, Mr. Bakshi, among other persons identified therein, entered into a lock-up agreement with the Company, pursuant to which Mr. Bakshi is subject to a lockup on the sale or transfer of shares of the Company’s common stock held by Mr. Bakshi at the closing of the 2020 Merger, including those shares issued in the 2020 Merger, for a period ending on the earlier of (i) 180 days after the closing date or (ii) approval of a certain milestone payment proposal (described further in the 2020 Merger Agreement) by the Company’s stockholders (the “Lock-Up Agreement”). Mr. Bakshi has served as a member of our Board and as our Chief Operating Officer and Senior Vice President since December 31, 2020.
Akash Bakshi Employment Agreement
Pursuant to an Employment Agreement between the Company and Mr. Bakshi (the “Employment Agreement”), effective as of the closing of the 2020 Merger, Mr. Bakshi has been appointed as Chief Operating Officer and Senior Vice President of the Company, reporting to Company’s Chief Executive Officer and Board of Directors. The Employment Agreement provides for cash compensation of $250,000 per year, plus an opportunity to earn an annual bonus based on the criteria set forth in the Company’s key performance indicators set by the Board. Mr. Bakshi will also eligible to receive a grant of an option to purchase shares the Company’s stock, in an amount consistent with that granted in similarly situated executive officers. The Employment Agreement also includes standard benefits, as well as customary intellectual property assignment and confidentiality provisions that are customary in the Company’s industry.
If Mr. Bakshi’s employment is terminated by the Company without Cause or by Mr. Bakshi for Good Reason (as each term is defined in the Employment Agreement), Mr. Bakshi will be entitled to (a) six months’ base salary and (b) base salary and benefits accrued through the date of termination.
License Agreement with YourChoice
In connection with the Company’s acquisition of ANA, the Company assumed the YourChoice Agreement between ANA and YourChoice, effective as of the closing of the 2020 Merger. Pursuant to the YourChoice Agreement, YourChoice granted to ANA, during the term of the YourChoice Agreement, an exclusive, worldwide, fee-bearing license derived from the licensed intellectual property throughout the world. The fees due under the YourChoice Agreement include certain single-digit royalty payments and milestone payments in the aggregate of $19.5 million. The term of the YourChoice Agreement will expire on the expiration or invalidation of the last of the licensed patents under the YourChoice Agreement. Mr. Bakshi, a member of the Board, serves as Chief Executive Officer and director of YourChoice.
Director Independence
Our common stock is listed on Nasdaq. Under the rules of Nasdaq, independent directors must comprise a majority of a listed company’s board of directors. In addition, the rules of Nasdaq require that, subject to specified exceptions, each member of a listed company’s audit, compensation and nominating and corporate governance committees be independent. Under the rules of Nasdaq, a director will only qualify as an “independent director” if, in the opinion of that company’s board of directors, that person does not have a relationship that would interfere with the exercise of independent judgment in carrying out the responsibilities of a director. Additionally, compensation committee members must not have a relationship with us that is material to the director’s ability to be independent from management in connection with the duties of a compensation committee member.
144
Audit committee members must also satisfy the independence criteria set forth in Rule 10A-3 under the Exchange Act. In order to be considered independent for purposes of Rule 10A-3, a member of an audit committee of a listed company may not, other than in his or her capacity as a member of the audit committee, the board of directors or any other board of directors committee: (i) accept, directly or indirectly, any consulting, advisory or other compensatory fee from the listed company or any of its subsidiaries; or (ii) be an affiliated person of the listed company or any of its subsidiaries.
Our Board has undertaken a review of the independence of each director and considered whether each director has a material relationship with us that could compromise his or her ability to exercise independent judgment in carrying out his or her responsibilities. As a result of this review, our Board affirmatively determined that Na Yeon (Irene) Kim, Jason Groves, Jeong Gyun Oh, Michael Salsbury and Douglas J. Swirsky are “independent directors” as defined under the applicable rules and regulations of the SEC and the listing requirements and rules of Nasdaq. The Board determined that Richard Kang, our Chief Executive Officer, President, Interim Chief Financial Officer, Secretary and Treasurer, and Akash Bakshi, our Chief Operating Officer and a Senior Vice President of the Company, are not independent. In making this determination, our Board considered the current and prior relationships that each non-employee director has with us and all other facts and circumstances that our Board deemed relevant in determining each non-employee director’s independence, including the participation by our non-employee directors, or their affiliates, in certain financing transactions by the Company and the beneficial ownership of our common stock by each non-employee director. See “Certain Relationships and Related Party Transactions” and “Security Ownership of Certain Beneficial Owners and Management and Related Stockholder Matters.”
ITEM 14. |
Service Fees Paid to the Independent Registered Public Accounting Firms
The Audit Committee has considered the scope and fee arrangements for all services provided by BDO USA, LLP, taking into account whether the provision of non-audit-related services is compatible with maintaining BDO USA, LLP independence. The following table presents fees for professional audit services rendered by BDO USA, LLP for the audit of the annual financial statements for the years ended December 31, 2020 and 2019.
FEE CATEGORY |
| FISCAL YEAR |
| FISCAL YEAR |
| ||
Audit fees |
| $ | 458,691 |
| $ | 1,071,164 |
|
Audit-related fees |
| $ |
|
| $ |
|
|
Tax fees |
| $ |
|
| $ |
|
|
All other fees |
| $ |
|
| $ |
|
|
Total fees |
| $ | 458,691 |
| $ | 1,071,164 |
|
Audit fees consist of fees billed for services relating to the audit of our annual financial statement and review of our quarterly financial statements, services that are normally provided in connection with statutory and regulatory filings or engagements, comfort letters, reports on an issuer's internal controls, and review of documents to be filed with the SEC (e.g. periodic filings, registration statements, and company responses to SEC comment letters).
Audit-related fees are related to other assurance and related services that are traditionally performed by an independent accountant such as employee benefit plan audits, due diligence related to mergers and acquisitions, accounting assistance and audits in connection with proposed or consummated acquisitions, attest services that are not required by statute or regulation, and consultations concerning proposed accounting and reporting standards.
Tax fees relate to permissible services for technical tax advice related to federal and state income tax matters.
145
Policy on Audit Committee Pre-Approval of Audit and Permissible Non-Audit Services of Independent Registered Public Accounting Firm
Our audit committee generally pre-approves all audit and permitted non-audit and tax services provided by the independent registered public accounting firm. Pre-approval is detailed as to the particular service or category of services and is generally subject to a specific budget. The independent registered public accounting firm and management are required to periodically report to the audit committee regarding the extent of services provided by the independent registered public accounting firm in accordance with this pre-approval, and the fees for the services performed to date. Our audit committee may also pre-approve particular services on a case-by-case basis. All of the services relating to the fees described in the table above were approved by our audit committee.
ITEM 15. |
(a) The following documents are filed as part of this report:
1. Financial Statements: The information required by this item is contained in Item 8 of this Form 10-K.
2. Financial Statement Schedules:
No financial statement schedules are provided because the information called for is not required or is shown either in the financial statements or notes thereto.
146
3. Exhibits:
EXHIBIT |
| DESCRIPTION OF DOCUMENT |
|
|
|
2.1 | ++ | |
|
|
|
2.2 |
| |
| | |
2.3 | | |
| | |
3.1 |
| |
|
|
|
3.2 |
| |
|
|
|
3.3 |
| |
|
|
|
3.4 | | |
|
|
|
4.1 |
| |
|
|
|
4.2 |
| |
|
|
|
4.3 |
|
|
4.5 | * | |
| | |
10.1 | # | |
| | |
10.9 | + | |
|
|
|
10.10 | # |
147
|
|
|
10.11 | # | |
|
|
|
10.12 | # | |
|
|
|
10.13 | +++ | |
| | |
10.15 | |
|
10.16 | +++ | |
| | |
10.17 | # | |
|
|
|
10.18 | # | |
|
|
|
10.19 |
| |
|
|
|
10.20 | +++ | |
|
|
|
10.21 | +++ | |
|
|
|
10.22 | # | |
|
|
|
10.23 | # | |
|
|
|
10.24 | # | |
| | |
148
10.25 | # | |
|
|
|
10.26 | # | |
|
|
|
10.27 | ++ | |
| | |
10.28 | | |
| | |
10.29# | # ## | |
|
|
|
10.30# | # | |
| | |
10.31 | # | |
| | |
10.32 | # | |
| | |
10.33 | # | |
| | |
10.34 | # | |
| | |
10.35 | # | |
| | |
10.36 | | |
| | |
10.37 | | |
| | |
10.38 | | |
| | |
149
10.39 | | |
| | |
10.40 | * | Employment Agreement, dated as of December 31, 2020, by and between the Registrant and Akash Bakshi. |
| | |
21.1 | * | |
| | |
23.1 | * | |
| | |
31.1 | ** | |
| | |
32.1 | ** | |
| | |
101.INS* |
| XBRL Instance Document |
101.SCH* |
| XBRL Taxonomy Extension Schema Document |
101.CAL* | | XBRL Taxonomy Extension Calculation Linkbase Document |
101.DEF* | | XBRL Taxonomy Extension Definition Linkbase Document |
101.LAB* | | XBRL Taxonomy Extension Label Linkbase Document |
101.PRE* | | XBRL Taxonomy Extension Presentation Linkbase Document |
__________ | | |
# | | Indicates management contract or compensatory plan |
| | |
* | | Filed herewith |
| | |
** | | Furnished herewith |
| | |
+ | | Registrant has omitted and filed separately with the SEC portions of the exhibit pursuant to a confidential treatment request under Rule 406 promulgated under the Securities Act. |
| | |
++ | | Certain schedules and exhibits have been omitted pursuant to Item 601(b)(2) of Regulation S-K. A copy of any omitted schedule and/or exhibit will be furnished to the SEC upon request. |
| | |
+++ | | Certain schedules and exhibits have been omitted pursuant to Item 601(a)(5) of Regulation S-K. A copy of any omitted schedule and/or exhibit will be furnished to the SEC upon request. Certain portions of the exhibits that are not material and would be competitively harmful if publicly disclosed have been redacted pursuant to Item 601(b)(10)(iv) of Regulation S-K. Copies of the unredacted exhibits will be furnished to the SEC upon request. |
150
Pursuant to the requirements of Section 13 or 15(d) of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
|
Date: April 15, 2021 | NEUROBO PHARMACEUTICALS, INC. | |
|
|
|
| By: | /s/ Richard Kang |
|
| Richard Kang |
|
| President and Chief Executive Officer |
Pursuant to the requirements of the Securities Exchange Act of 1934, this report has been signed below by the following persons on behalf of the registrant and in the capacities and on the dates indicated.
|
|
|
|
|
SIGNATURE |
| TITLE |
| DATE |
|
|
|
|
|
/s/ Richard Kang |
|
| ||
Richard Kang |
| President and Chief Executive Officer (Principal Executive Officer, Principal Financial Officer and Principal Accounting Officer) and a Director |
| April 15, 2021 |
|
|
|
|
|
/s/ Douglas J. Swirsky |
|
| ||
Douglas J. Swirsky |
| Chairman of the Board of Directors |
| April 15, 2021 |
|
|
|
|
|
/s/ Jason L. Groves |
|
| ||
Jason L. Groves |
| Director |
| April 15, 2021 |
|
|
|
|
|
/s/ Na Yeon (Irene) Kim |
| |||
Na Yeon (Irene) Kim |
| Director |
| April 15, 2021 |
|
|
|
|
|
/s/ Jeong Gyun Oh |
| |
| |
Jeong Gyun Oh |
| Director |
| April 15, 2021 |
|
|
|
| |
/s/ Michael Salsbury |
| |
| |
Michael Salsbury |
| Director |
| April 15, 2021 |
| | | | |
/s/ Akash Bakshi |
| |
| |
Akash Bakshi |
| Director, Chief Operating Officer and Senior |
| April 15, 2021 |
| | Vice President | | |
151
Similar companies
See also JOHNSON & JOHNSON - Annual report 2023 (10-K 2023-01-01) Annual report 2023 (10-Q 2023-10-01)See also ELI LILLY & Co - Annual report 2024 (10-K 2024-12-31) Annual report 2023 (10-Q 2023-09-30)
See also PFIZER INC - Annual report 2022 (10-K 2022-12-31) Annual report 2023 (10-Q 2023-10-01)
See also AbbVie Inc. - Annual report 2022 (10-K 2022-12-31) Annual report 2023 (10-Q 2023-09-30)
See also NOVO NORDISK A S
