Philip Morris International Inc. - Quarter Report: 2020 September (Form 10-Q)
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 10-Q
(Mark One)
| ☑ | QUARTERLY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | ||||
For the quarterly period ended September 30, 2020
OR
| ☐ | TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | ||||
For the transition period from to
Commission File Number 001-33708
Philip Morris International Inc. | ||||||||||||||
(Exact name of registrant as specified in its charter)
| Virginia | 13-3435103 | ||||
| (State or other jurisdiction of incorporation or organization) | (I.R.S. Employer Identification No.) | ||||
| 120 Park Avenue | New York | New York | 10017 | ||||||||
| (Address of principal executive offices) | (Zip Code) | ||||||||||
| Registrant’s telephone number, including area code | (917) | 663-2000 | ||||||
Former name, former address and former fiscal year, if changed since last report
Securities registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||||||||
| Common Stock, no par value | PM | New York Stock Exchange | ||||||||||||
| 1.875% Notes due 2021 | PM21B | New York Stock Exchange | ||||||||||||
| 1.875% Notes due 2021 | PM21C | New York Stock Exchange | ||||||||||||
| 4.125% Notes due 2021 | PM21 | New York Stock Exchange | ||||||||||||
| 2.900% Notes due 2021 | PM21A | New York Stock Exchange | ||||||||||||
| 2.625% Notes due 2022 | PM22A | New York Stock Exchange | ||||||||||||
| 2.375% Notes due 2022 | PM22B | New York Stock Exchange | ||||||||||||
| 2.500% Notes due 2022 | PM22 | New York Stock Exchange | ||||||||||||
| 2.500% Notes due 2022 | PM22C | New York Stock Exchange | ||||||||||||
| 2.625% Notes due 2023 | PM23 | New York Stock Exchange | ||||||||||||
| 2.125% Notes due 2023 | PM23B | New York Stock Exchange | ||||||||||||
| 3.600% Notes due 2023 | PM23A | New York Stock Exchange | ||||||||||||
| 2.875% Notes due 2024 | PM24 | New York Stock Exchange | ||||||||||||
| 2.875% Notes due 2024 | PM24C | New York Stock Exchange | ||||||||||||
| 0.625% Notes due 2024 | PM24B | New York Stock Exchange | ||||||||||||
| 3.250% Notes due 2024 | PM24A | New York Stock Exchange | ||||||||||||
| Title of each class | Trading Symbol(s) | Name of each exchange on which registered | ||||||||||||
| 2.750% Notes due 2025 | PM25 | New York Stock Exchange | ||||||||||||
| 3.375% Notes due 2025 | PM25A | New York Stock Exchange | ||||||||||||
| 2.750% Notes due 2026 | PM26A | New York Stock Exchange | ||||||||||||
| 2.875% Notes due 2026 | PM26 | New York Stock Exchange | ||||||||||||
| 0.125% Notes due 2026 | PM26B | New York Stock Exchange | ||||||||||||
| 3.125% Notes due 2027 | PM27 | New York Stock Exchange | ||||||||||||
| 3.125% Notes due 2028 | PM28 | New York Stock Exchange | ||||||||||||
| 2.875% Notes due 2029 | PM29 | New York Stock Exchange | ||||||||||||
| 3.375% Notes due 2029 | PM29A | New York Stock Exchange | ||||||||||||
| 0.800% Notes due 2031 | PM31 | New York Stock Exchange | ||||||||||||
| 3.125% Notes due 2033 | PM33 | New York Stock Exchange | ||||||||||||
| 2.000% Notes due 2036 | PM36 | New York Stock Exchange | ||||||||||||
| 1.875% Notes due 2037 | PM37A | New York Stock Exchange | ||||||||||||
| 6.375% Notes due 2038 | PM38 | New York Stock Exchange | ||||||||||||
| 1.450% Notes due 2039 | PM39 | New York Stock Exchange | ||||||||||||
| 4.375% Notes due 2041 | PM41 | New York Stock Exchange | ||||||||||||
| 4.500% Notes due 2042 | PM42 | New York Stock Exchange | ||||||||||||
| 3.875% Notes due 2042 | PM42A | New York Stock Exchange | ||||||||||||
| 4.125% Notes due 2043 | PM43 | New York Stock Exchange | ||||||||||||
| 4.875% Notes due 2043 | PM43A | New York Stock Exchange | ||||||||||||
| 4.250% Notes due 2044 | PM44 | New York Stock Exchange | ||||||||||||
Indicate by check mark whether the registrant (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days. Yes þ No ¨
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files). Yes þ No ¨
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” “smaller reporting company,” and "emerging growth company" in Rule 12b-2 of the Exchange Act.
Large accelerated filer þ Accelerated filer ☐
Non-accelerated filer ☐ Smaller reporting company ☐
Emerging growth company ☐
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ¨
Indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act). Yes ☐ No þ
At October 22, 2020, there were 1,557,315,937 shares outstanding of the registrant’s common stock, no par value per share.
1
PHILIP MORRIS INTERNATIONAL INC.
TABLE OF CONTENTS
| Page No. | ||||||||
| PART I - | ||||||||
| Item 1. | ||||||||
| Condensed Consolidated Statements of Earnings for the | ||||||||
| Nine Months Ended September 30, 2020 and 2019 | ||||||||
| Three Months Ended September 30, 2020 and 2019 | ||||||||
| Condensed Consolidated Statements of Comprehensive Earnings for the | ||||||||
| Nine Months Ended September 30, 2020 and 2019 | ||||||||
| Three Months Ended September 30, 2020 and 2019 | ||||||||
| Condensed Consolidated Balance Sheets at | ||||||||
| September 30, 2020 and December 31, 2019 | ||||||||
| Condensed Consolidated Statements of Cash Flows for the | ||||||||
| Nine Months Ended September 30, 2020 and 2019 | ||||||||
| Condensed Consolidated Statements of Stockholders’ (Deficit) Equity for the | ||||||||
| Nine Months Ended September 30, 2020 and 2019 | ||||||||
| Three Months Ended September 30, 2020 and 2019 | ||||||||
| Item 2. | ||||||||
| Item 4. | ||||||||
| PART II - | ||||||||
| Item 1. | ||||||||
| Item 1A. | ||||||||
| Item 2. | ||||||||
| Item 6. | ||||||||
In this report, “PMI,” “we,” “us” and “our” refer to Philip Morris International Inc. and its subsidiaries.
Trademarks and service marks in this report are the registered property of, or licensed by, the subsidiaries of Philip Morris International Inc. and are italicized.
2
PART I – FINANCIAL INFORMATION
Item 1. Financial Statements.
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Statements of Earnings
(in millions of dollars, except per share data)
(Unaudited)
| For the Nine Months Ended September 30, | |||||||||||
| 2020 | 2019 | ||||||||||
| Revenues including excise taxes | $ | 56,516 | $ | 58,072 | |||||||
| Excise taxes on products | 35,266 | 35,980 | |||||||||
| Net revenues | 21,250 | 22,092 | |||||||||
| Cost of sales | 6,997 | 7,735 | |||||||||
| Gross profit | 14,253 | 14,357 | |||||||||
| Marketing, administration and research costs (Notes 8, 18 & 19) | 5,435 | 6,282 | |||||||||
| Amortization of intangibles | 55 | 50 | |||||||||
| Operating income | 8,763 | 8,025 | |||||||||
| Interest expense, net | 454 | 434 | |||||||||
| Pension and other employee benefit costs (Note 3) | 68 | 61 | |||||||||
| Earnings before income taxes | 8,241 | 7,530 | |||||||||
| Provision for income taxes | 1,764 | 1,670 | |||||||||
| Equity investments and securities (income)/loss, net | 4 | (86) | |||||||||
| Net earnings | $ | 6,473 | $ | 5,946 | |||||||
| Net earnings attributable to noncontrolling interests | 393 | 377 | |||||||||
| Net earnings attributable to PMI | $ | 6,080 | $ | 5,569 | |||||||
Per share data (Note 6): | |||||||||||
| Basic earnings per share | $ | 3.90 | $ | 3.57 | |||||||
| Diluted earnings per share | $ | 3.90 | $ | 3.57 | |||||||
See notes to condensed consolidated financial statements.
3
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Statements of Earnings
(in millions of dollars, except per share data)
(Unaudited)
| For the Three Months Ended September 30, | |||||||||||
| 2020 | 2019 | ||||||||||
| Revenues including excise taxes | $ | 20,444 | $ | 20,380 | |||||||
| Excise taxes on products | 12,998 | 12,738 | |||||||||
| Net revenues | 7,446 | 7,642 | |||||||||
| Cost of sales | 2,416 | 2,605 | |||||||||
| Gross profit | 5,030 | 5,037 | |||||||||
| Marketing, administration and research costs (Notes 8 & 18) | 1,769 | 2,234 | |||||||||
| Amortization of intangibles | 18 | 15 | |||||||||
| Operating income | 3,243 | 2,788 | |||||||||
| Interest expense, net | 163 | 132 | |||||||||
| Pension and other employee benefit costs (Note 3) | 23 | 20 | |||||||||
| Earnings before income taxes | 3,057 | 2,636 | |||||||||
| Provision for income taxes | 640 | 635 | |||||||||
| Equity investments and securities (income)/loss, net | (20) | (45) | |||||||||
| Net earnings | 2,437 | 2,046 | |||||||||
| Net earnings attributable to noncontrolling interests | 130 | 150 | |||||||||
| Net earnings attributable to PMI | $ | 2,307 | $ | 1,896 | |||||||
Per share data (Note 6): | |||||||||||
| Basic earnings per share | $ | 1.48 | $ | 1.22 | |||||||
| Diluted earnings per share | $ | 1.48 | $ | 1.22 | |||||||
See notes to condensed consolidated financial statements.
4
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Statements of Comprehensive Earnings
(in millions of dollars)
(Unaudited)
| For the Nine Months Ended September 30, | ||||||||||||||
| 2020 | 2019 | |||||||||||||
| Net earnings | $ | 6,473 | $ | 5,946 | ||||||||||
| Other comprehensive earnings (losses), net of income taxes: | ||||||||||||||
Change in currency translation adjustments: | ||||||||||||||
Unrealized gains (losses), net of income taxes of $38 in 2020 and $(230) in 2019 | (1,373) | 363 | ||||||||||||
(Gains)/losses transferred to earnings - deconsolidation of RBH, net of income taxes of $0 in 2020 and 2019 (Note 19) | — | 502 | ||||||||||||
Change in net loss and prior service cost: | ||||||||||||||
Net gains (losses) and prior service costs, net of income taxes of $0 in 2020 and $133 in 2019 | — | 133 | ||||||||||||
Amortization of net losses, prior service costs and net transition costs, net of income taxes of $(49) in 2020 and $(43) in 2019 | 223 | 180 | ||||||||||||
(Gains)/losses transferred to earnings - deconsolidation of RBH, net of income taxes of $0 in 2020 and $(15) in 2019 (Note 19) | — | 27 | ||||||||||||
Change in fair value of derivatives accounted for as hedges: | ||||||||||||||
Gains (losses) recognized, net of income taxes of $8 in 2020 and $5 in 2019 | (37) | (34) | ||||||||||||
(Gains) losses transferred to earnings, net of income taxes of $1 in 2020 and $3 in 2019 | (17) | (12) | ||||||||||||
| Total other comprehensive earnings (losses) | (1,204) | 1,159 | ||||||||||||
| Total comprehensive earnings | 5,269 | 7,105 | ||||||||||||
| Less comprehensive earnings attributable to: | ||||||||||||||
| Noncontrolling interests | 393 | 391 | ||||||||||||
| Comprehensive earnings attributable to PMI | $ | 4,876 | $ | 6,714 | ||||||||||
See notes to condensed consolidated financial statements.
5
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Statements of Comprehensive Earnings
(in millions of dollars)
(Unaudited)
| For the Three Months Ended September 30, | ||||||||||||||
| 2020 | 2019 | |||||||||||||
| Net earnings | $ | 2,437 | $ | 2,046 | ||||||||||
| Other comprehensive earnings (losses), net of income taxes: | ||||||||||||||
Change in currency translation adjustments: | ||||||||||||||
Unrealized gains (losses), net of income taxes of $78 in 2020 and $(163) in 2019 | (588) | 12 | ||||||||||||
Change in net loss and prior service cost: | ||||||||||||||
Net gains (losses) and prior service costs, net of income taxes of $0 in 2020 and $(2) in 2019 | — | (2) | ||||||||||||
Amortization of net losses, prior service costs and net transition costs, net of income taxes of $(15) in 2020 and $(13) in 2019 | 74 | 61 | ||||||||||||
Change in fair value of derivatives accounted for as hedges: | ||||||||||||||
Gains (losses) recognized, net of income taxes of $7 in 2020 and $2 in 2019 | (31) | (14) | ||||||||||||
(Gains) losses transferred to earnings, net of income taxes of $(1) in 2020 and $0 in 2019 | (2) | 13 | ||||||||||||
| Total other comprehensive earnings (losses) | (547) | 70 | ||||||||||||
Total comprehensive earnings | 1,890 | 2,116 | ||||||||||||
| Less comprehensive earnings attributable to: | ||||||||||||||
| Noncontrolling interests | 141 | 135 | ||||||||||||
| Comprehensive earnings attributable to PMI | $ | 1,749 | $ | 1,981 | ||||||||||
See notes to condensed consolidated financial statements.
6
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Balance Sheets
(in millions of dollars)
(Unaudited)
| September 30, 2020 | December 31, 2019 | ||||||||||
| ASSETS | |||||||||||
| Cash and cash equivalents | $ | 4,821 | $ | 6,861 | |||||||
Trade receivables (less allowances of $21 in 2020 and $20 in 2019) | 3,080 | 3,080 | |||||||||
| Other receivables | 632 | 637 | |||||||||
Inventories: | |||||||||||
| Leaf tobacco | 1,996 | 2,052 | |||||||||
| Other raw materials | 1,757 | 1,596 | |||||||||
| Finished product | 4,407 | 5,587 | |||||||||
| 8,160 | 9,235 | ||||||||||
| Other current assets | 760 | 701 | |||||||||
Total current assets | 17,453 | 20,514 | |||||||||
Property, plant and equipment, at cost | 14,293 | 14,446 | |||||||||
| Less: accumulated depreciation | 8,159 | 7,815 | |||||||||
| 6,134 | 6,631 | ||||||||||
| Goodwill (Note 4) | 5,647 | 5,858 | |||||||||
| Other intangible assets, net (Note 4) | 1,926 | 2,113 | |||||||||
| Investments in unconsolidated subsidiaries and equity securities (Notes 11&14) | 4,579 | 4,635 | |||||||||
| Deferred income taxes | 1,225 | 1,153 | |||||||||
| Other assets | 2,165 | 1,971 | |||||||||
| TOTAL ASSETS | $ | 39,129 | $ | 42,875 | |||||||
See notes to condensed consolidated financial statements.
Continued
7
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Balance Sheets (Continued)
(in millions of dollars, except share data)
(Unaudited)
| September 30, 2020 | December 31, 2019 | ||||||||||
| LIABILITIES | |||||||||||
| Short-term borrowings (Note 10) | $ | 152 | $ | 338 | |||||||
| Current portion of long-term debt (Note 10) | 1,992 | 4,051 | |||||||||
| Accounts payable | 2,155 | 2,299 | |||||||||
| Accrued liabilities: | |||||||||||
| Marketing and selling | 752 | 666 | |||||||||
| Taxes, except income taxes | 4,870 | 5,837 | |||||||||
| Employment costs | 981 | 1,042 | |||||||||
| Dividends payable | 1,879 | 1,831 | |||||||||
| Other | 1,897 | 1,973 | |||||||||
| Income taxes | 847 | 796 | |||||||||
| Total current liabilities | 15,525 | 18,833 | |||||||||
Long-term debt (Note 10) | 27,346 | 26,656 | |||||||||
| Deferred income taxes | 661 | 908 | |||||||||
| Employment costs | 3,589 | 3,634 | |||||||||
| Income taxes and other liabilities | 2,253 | 2,443 | |||||||||
| Total liabilities | 49,374 | 52,474 | |||||||||
Contingencies (Note 8) | |||||||||||
STOCKHOLDERS’ (DEFICIT) EQUITY | |||||||||||
Common stock, no par value (2,109,316,331 shares issued in 2020 and 2019) | — | — | |||||||||
| Additional paid-in capital | 2,071 | 2,019 | |||||||||
| Earnings reinvested in the business | 31,537 | 30,987 | |||||||||
| Accumulated other comprehensive losses | (10,567) | (9,363) | |||||||||
| 23,041 | 23,643 | ||||||||||
Less: cost of repurchased stock (552,016,777 and 553,421,668 shares in 2020 and 2019, respectively) | 35,133 | 35,220 | |||||||||
| Total PMI stockholders’ deficit | (12,092) | (11,577) | |||||||||
| Noncontrolling interests | 1,847 | 1,978 | |||||||||
| Total stockholders’ deficit | (10,245) | (9,599) | |||||||||
| TOTAL LIABILITIES AND STOCKHOLDERS’ (DEFICIT) EQUITY | $ | 39,129 | $ | 42,875 | |||||||
See notes to condensed consolidated financial statements.
8
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Statements of Cash Flows
(in millions of dollars)
(Unaudited)
| For the Nine Months Ended September 30, | ||||||||||||||
| 2020 | 2019 | |||||||||||||
| CASH PROVIDED BY (USED IN) OPERATING ACTIVITIES | ||||||||||||||
| Net earnings | $ | 6,473 | $ | 5,946 | ||||||||||
| Adjustments to reconcile net earnings to operating cash flows: | ||||||||||||||
| Depreciation and amortization | 709 | 709 | ||||||||||||
| Deferred income tax (benefit) provision | (187) | (124) | ||||||||||||
| Asset impairment and exit costs, net of cash paid (Note 18) | (44) | 42 | ||||||||||||
| Cash effects of changes in: | ||||||||||||||
| Receivables, net | (141) | (506) | ||||||||||||
| Inventories | 823 | (64) | ||||||||||||
| Accounts payable | (22) | (31) | ||||||||||||
| Accrued liabilities and other current assets | (1,026) | 292 | ||||||||||||
| Income taxes | (181) | (6) | ||||||||||||
| Pension plan contributions | (62) | (89) | ||||||||||||
| Other | 308 | 597 | (1) | |||||||||||
| Net cash provided by operating activities | 6,650 | 6,766 | ||||||||||||
| CASH PROVIDED BY (USED IN) INVESTING ACTIVITIES | ||||||||||||||
| Capital expenditures | (462) | (696) | ||||||||||||
| Investments in unconsolidated subsidiaries and equity securities | (3) | (31) | ||||||||||||
| Deconsolidation of RBH (Note 19) | — | (1,346) | (2) | |||||||||||
| Net investment hedges | (138) | 429 | ||||||||||||
| Other | 35 | 17 | ||||||||||||
| Net cash used in investing activities | (568) | (1,627) | ||||||||||||
See notes to condensed consolidated financial statements.
Continued
9
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Statements of Cash Flows (Continued)
(in millions of dollars)
(Unaudited)
| For the Nine Months Ended September 30, | |||||||||||
| 2020 | 2019 | ||||||||||
| CASH PROVIDED BY (USED IN) FINANCING ACTIVITIES | |||||||||||
| Short-term borrowing activity by original maturity: | |||||||||||
| Net issuances (repayments) - maturities of 90 days or less | $ | (159) | $ | (340) | |||||||
| Issuances - maturities longer than 90 days | 45 | 989 | |||||||||
| Repayments - maturities longer than 90 days | (45) | (989) | |||||||||
| Long-term debt proceeds | 2,230 | 3,819 | |||||||||
| Long-term debt repaid | (3,999) | (2,971) | |||||||||
| Dividends paid | (5,485) | (5,336) | |||||||||
| Sale (purchase) of subsidiary shares to/(from) noncontrolling interests | 3 | 47 | |||||||||
| Other | (621) | (321) | |||||||||
| Net cash used in financing activities | (8,031) | (5,102) | |||||||||
| Effect of exchange rate changes on cash, cash equivalents and restricted cash | (89) | (146) | |||||||||
Cash, cash equivalents and restricted cash(3): | |||||||||||
| Increase (Decrease) | (2,038) | (109) | |||||||||
| Balance at beginning of period | 6,865 | 6,620 | |||||||||
| Balance at end of period | $ | 4,827 | $ | 6,511 | |||||||
(1) Includes the Loss on Deconsolidation of RBH ($239 million) and the Canadian tobacco litigation-related charge ($194 million) that were included in marketing, administration and research costs in the condensed consolidated statements of earnings for the nine months ended September 30, 2019. For further details on these charges, see Note 19. Deconsolidation of RBH.
(2) Includes deconsolidation of RBH cash and cash equivalents of $1,323 million and restricted cash of $23 million.
(3) The amounts for cash and cash equivalents shown above include restricted cash of $6 million and $4 million as of September 30, 2020 and 2019, respectively, and $4 million and $27 million as of December 31, 2019, and 2018, respectively, which were included in other current assets in the condensed consolidated balance sheets.
See notes to condensed consolidated financial statements.
10
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Statements of Stockholders’ (Deficit) Equity
For the Nine Months Ended September 30, 2020 and 2019
(in millions of dollars, except per share amounts)
(Unaudited)
| PMI Stockholders’ (Deficit) Equity | |||||||||||||||||||||||||||||||||||||||||
| Common Stock | Additional Paid-in Capital | Earnings Reinvested in the Business | Accumulated Other Comprehensive Losses | Cost of Repurchased Stock | Noncontrolling Interests | Total | |||||||||||||||||||||||||||||||||||
| Balances, January 1, 2019 | $ | — | $ | 1,939 | $ | 31,014 | $ | (10,111) | $ | (35,301) | $ | 1,720 | $ | (10,739) | |||||||||||||||||||||||||||
| Net earnings | 5,569 | 377 | 5,946 | ||||||||||||||||||||||||||||||||||||||
| Other comprehensive earnings (losses), net of income taxes | 616 | 14 | 630 | ||||||||||||||||||||||||||||||||||||||
| Issuance of stock awards | 44 | 79 | 123 | ||||||||||||||||||||||||||||||||||||||
Dividends declared ($3.45 per share) | (5,386) | (5,386) | |||||||||||||||||||||||||||||||||||||||
| Payments to noncontrolling interests | (305) | (305) | |||||||||||||||||||||||||||||||||||||||
| Deconsolidation of RBH (Note 19) | 529 | 529 | |||||||||||||||||||||||||||||||||||||||
| Other | (2) | 49 | 47 | ||||||||||||||||||||||||||||||||||||||
| Balances, September 30, 2019 | $ | — | $ | 1,981 | $ | 31,197 | $ | (8,966) | $ | (35,222) | $ | 1,855 | $ | (9,155) | |||||||||||||||||||||||||||
| Balances, January 1, 2020 | $ | — | $ | 2,019 | $ | 30,987 | $ | (9,363) | $ | (35,220) | $ | 1,978 | $ | (9,599) | |||||||||||||||||||||||||||
| Net earnings | 6,080 | 393 | 6,473 | ||||||||||||||||||||||||||||||||||||||
| Other comprehensive earnings (losses), net of income taxes | (1,204) | — | (1,204) | ||||||||||||||||||||||||||||||||||||||
| Issuance of stock awards | 35 | 87 | 122 | ||||||||||||||||||||||||||||||||||||||
Dividends declared ($3.54 per share) | (5,530) | (5,530) | |||||||||||||||||||||||||||||||||||||||
| Payments to noncontrolling interests | (510) | (510) | |||||||||||||||||||||||||||||||||||||||
| Other | 17 | — | (14) | 3 | |||||||||||||||||||||||||||||||||||||
| Balances, September 30, 2020 | $ | — | $ | 2,071 | $ | 31,537 | $ | (10,567) | $ | (35,133) | $ | 1,847 | $ | (10,245) | |||||||||||||||||||||||||||
See notes to condensed consolidated financial statements.
11
Philip Morris International Inc. and Subsidiaries
Condensed Consolidated Statements of Stockholders’ (Deficit) Equity
For the Three Months Ended September 30, 2020 and 2019
(in millions of dollars, except per share amounts)
(Unaudited)
| PMI Stockholders’ (Deficit) Equity | |||||||||||||||||||||||||||||||||||||||||
| Common Stock | Additional Paid-in Capital | Earnings Reinvested in the Business | Accumulated Other Comprehensive Losses | Cost of Repurchased Stock | Noncontrolling Interests | Total | |||||||||||||||||||||||||||||||||||
| Balances, July 1, 2019 | $ | — | $ | 1,948 | $ | 31,128 | $ | (9,051) | $ | (35,224) | $ | 1,790 | $ | (9,409) | |||||||||||||||||||||||||||
| Net earnings | 1,896 | 150 | 2,046 | ||||||||||||||||||||||||||||||||||||||
| Other comprehensive earnings (losses), net of income taxes | 85 | (15) | 70 | ||||||||||||||||||||||||||||||||||||||
| Issuance of stock awards | 35 | 2 | 37 | ||||||||||||||||||||||||||||||||||||||
Dividends declared ($1.17 per share) | (1,827) | (1,827) | |||||||||||||||||||||||||||||||||||||||
| Payments to noncontrolling interests | (74) | (74) | |||||||||||||||||||||||||||||||||||||||
| Other | (2) | 4 | 2 | ||||||||||||||||||||||||||||||||||||||
| Balances, September 30, 2019 | $ | — | $ | 1,981 | $ | 31,197 | $ | (8,966) | $ | (35,222) | $ | 1,855 | $ | (9,155) | |||||||||||||||||||||||||||
| Balances, July 1, 2020 | $ | — | $ | 2,044 | $ | 31,103 | $ | (10,009) | $ | (35,135) | $ | 1,877 | $ | (10,120) | |||||||||||||||||||||||||||
| Net earnings | 2,307 | 130 | 2,437 | ||||||||||||||||||||||||||||||||||||||
| Other comprehensive earnings (losses), net of income taxes | (558) | 11 | (547) | ||||||||||||||||||||||||||||||||||||||
| Issuance of stock awards | 26 | 2 | 28 | ||||||||||||||||||||||||||||||||||||||
Dividends declared ($1.20 per share) | (1,873) | (1,873) | |||||||||||||||||||||||||||||||||||||||
| Payments to noncontrolling interests | (171) | (171) | |||||||||||||||||||||||||||||||||||||||
| Other | 1 | 1 | |||||||||||||||||||||||||||||||||||||||
| Balances, September 30, 2020 | $ | — | $ | 2,071 | $ | 31,537 | $ | (10,567) | $ | (35,133) | $ | 1,847 | $ | (10,245) | |||||||||||||||||||||||||||
See notes to condensed consolidated financial statements.
12
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Note 1. Background and Basis of Presentation:
Background
Philip Morris International Inc. is a holding company incorporated in Virginia, U.S.A., whose subsidiaries and affiliates and their licensees are engaged in the manufacture and sale of cigarettes and other nicotine-containing products, including reduced-risk products, in markets outside of the United States of America. In addition, PMI ships a version of its Platform 1 device and its consumables authorized by the U.S. Food and Drug Administration ("FDA") to Altria Group, Inc., for sale in the United States under license. Throughout these financial statements, the term "PMI" refers to Philip Morris International Inc. and its subsidiaries.
Reduced-risk products ("RRPs") is the term PMI uses to refer to products that present, are likely to present, or have the potential to present less risk of harm to smokers who switch to these products versus continuing smoking. PMI has a range of RRPs in various stages of development, scientific assessment and commercialization.
"Platform 1" is the term PMI uses to refer to PMI’s reduced-risk product that uses a precisely controlled heating device incorporating our IQOS HeatControl technology, into which a specially designed and proprietary tobacco unit is inserted and heated to generate an aerosol.
Basis of Presentation
The interim condensed consolidated financial statements of PMI are unaudited. These interim condensed consolidated financial statements have been prepared in conformity with accounting principles generally accepted in the United States of America ("U.S. GAAP") and such principles are applied on a consistent basis. It is the opinion of PMI’s management that all adjustments necessary for a fair statement of the interim results presented have been reflected therein. All such adjustments were of a normal recurring nature. Net revenues and net earnings attributable to PMI for any interim period are not necessarily indicative of results that may be expected for the entire year.
PMI has analyzed the impact of the Coronavirus pandemic ("COVID-19") on its financial statements as of September 30, 2020. PMI has determined that the changes to its significant judgments and estimates did not have a material impact with respect to goodwill, intangible assets, long-lived assets or its hedge accounting activities.
As of March 22, 2019, PMI deconsolidated the financial results of its Canadian subsidiary, Rothmans, Benson & Hedges Inc. ("RBH") from PMI's financial statements. For further details, see Note 19. Deconsolidation of RBH.
These statements should be read in conjunction with the audited consolidated financial statements and related notes, which appear in PMI’s Annual Report on Form 10-K for the year ended December 31, 2019.
13
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Note 2. Stock Plans:
In May 2017, PMI’s shareholders approved the Philip Morris International Inc. 2017 Performance Incentive Plan (the “2017 Plan”). Under the 2017 Plan, PMI may grant to eligible employees restricted shares and restricted share units, performance-based cash incentive awards and performance-based equity awards. Up to 25 million shares of PMI’s common stock may be issued under the 2017 Plan. At September 30, 2020, shares available for grant under the 2017 Plan were 17,277,020.
In May 2017, PMI’s shareholders also approved the Philip Morris International Inc. 2017 Stock Compensation Plan for Non-Employee Directors (the “2017 Non-Employee Directors Plan”). A non-employee director is defined as a member of the PMI Board of Directors who is not a full-time employee of PMI or of any corporation in which PMI owns, directly or indirectly, stock possessing at least 50% of the total combined voting power of all classes of stock entitled to vote in the election of directors in such corporation. Up to 1 million shares of PMI common stock may be awarded under the 2017 Non-Employee Directors Plan. At September 30, 2020, shares available for grant under the plan were 934,372.
Restricted share unit (RSU) awards
During the nine months ended September 30, 2020 and 2019, shares granted to eligible employees and the weighted-average grant date fair value per share related to RSU awards were as follows:
| Number of Shares Granted | Weighted-Average Grant Date Fair Value Per RSU Award Granted | ||||||||||
| 2020 | 1,712,750 | $ 85.81 | |||||||||
| 2019 | 1,717,230 | $ 77.25 | |||||||||
Compensation expense related to RSU awards was as follows:
| Compensation Expense Related to RSU Awards | ||||||||
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | ||||||
| 2020 | $ 98 | $ 30 | ||||||
| 2019 | $ 91 | $ 27 | ||||||
As of September 30, 2020, PMI had $164 million of total unrecognized compensation cost related to non-vested RSU awards. The cost is recognized over the original restriction period of the awards, which is typically three years after the date of the award, or upon death, disability or reaching the age of 58.
During the nine months ended September 30, 2020, 1,128,308 RSU awards vested. The grant date fair value of all the vested awards was approximately $110 million. The total fair value of RSU awards that vested during the nine months ended September 30, 2020 was approximately $97 million.
Performance share unit (PSU) awards
During the nine months ended September 30, 2020 and 2019, PMI granted PSU awards to certain executives. The PSU awards require the achievement of certain performance factors, which are predetermined at the time of grant, typically over a three-year performance cycle. The performance metrics for such PSU's granted during the nine months ended September 30, 2020 consisted of PMI's Total Shareholder Return ("TSR") relative to a predetermined peer group and on an absolute basis (40% weight), PMI’s currency-neutral compound annual adjusted diluted earnings per share growth rate (30% weight), and PMI’s performance against specific measures of PMI’s transformation, defined as net revenues from PMI's RRPs and any other non-combustible products as a percentage of PMI's total net revenues in the last year of the performance cycle (30% weight). The performance metrics for such PSUs granted during the nine months ended September 30, 2019 consisted of PMI’s TSR relative to a predetermined peer group and on an absolute basis (50% weight), PMI’s currency-neutral compound annual adjusted operating income growth rate, excluding acquisitions (30% weight), and PMI’s performance against specific measures of PMI’s transformation (20% weight).
14
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
The aggregate of the weighted performance factors for the three metrics in each such PSU award determines the percentage of PSUs that will vest at the end of the three-year performance cycle. The minimum percentage of such PSUs that can vest is zero, with a target percentage of 100 and a maximum percentage of 200. Each such vested PSU entitles the participant to one share of common stock. An aggregate weighted PSU performance factor of 100 will result in the targeted number of PSUs being vested. At the end of the performance cycle, participants are entitled to an amount equivalent to the accumulated dividends paid on common stock during the performance cycle for the number of shares earned.
During the nine months ended September 30, 2020 and 2019, shares granted to eligible employees and the grant date fair value per share related to PSU awards were as follows:
| Number of Shares Granted | PSU Grant Date Fair Value Subject to Other Performance Factors Per Share | PSU Grant Date Fair Value Subject to TSR Performance Factor Per Share | |||||||||
| 2020 | 671,220 | $ 86.04 | $ 80.36 | ||||||||
| 2019 | 647,700 | $ 77.23 | $ 83.59 | ||||||||
The grant date fair value of the PSU awards subject to the other performance factors was determined by using the average of the high and low market price of PMI’s stock at the date of the grant. The grant date fair value of the PSU market based awards subject to the TSR performance factor was determined by using the Monte Carlo simulation model. The following assumptions were used to determine the grant date fair value of the PSU awards subject to the TSR performance factor:
| 2020 | 2019 | ||||||||||
Risk-free interest rate (a) | 1.4 | % | 2.4 | % | |||||||
Expected volatility (b) | 23.5 | % | 21.4 | % | |||||||
(a) Based on the U.S. Treasury yield curve.
(b) Determined using the observed historical volatility.
Compensation expense related to PSU awards was as follows:
| Compensation Expense Related to PSU Awards | ||||||||
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | ||||||
| 2020 | $ 30 | ($ 2) | ||||||
| 2019 | $ 43 | $ 10 | ||||||
As of September 30, 2020, PMI had $47 million of total unrecognized compensation cost related to non-vested PSU awards. The cost is recognized over the performance cycle of the awards, or upon death, disability or reaching the age of 58.
During the nine months ended September 30, 2020, 343,806 PSU awards vested. The grant date fair value of all the vested awards was approximately $35 million. The total fair value of PSU awards that vested during the nine months ended September 30, 2020 was approximately $30 million.
Note 3. Benefit Plans:
Pension coverage for employees of PMI’s subsidiaries is provided, to the extent deemed appropriate, through separate plans, many of which are governed by local statutory requirements. In addition, PMI provides health care and other benefits to substantially all U.S. retired employees and certain non-U.S. retired employees. In general, health care benefits for non-U.S. retired employees are covered through local government plans.
15
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Pension and other employee benefit costs per the condensed consolidated statements of earnings consisted of the following:
| For the Nine Months Ended September 30, | For the Three Months Ended September 30, | ||||||||||||||||||||||
| (in millions) | 2020 | 2019 | 2020 | 2019 | |||||||||||||||||||
| Net pension costs (income) | $ | (13) | $ | (16) | $ | (5) | $ | (6) | |||||||||||||||
| Net postemployment costs | 75 | 72 | 26 | 25 | |||||||||||||||||||
| Net postretirement costs | 6 | 5 | 2 | 1 | |||||||||||||||||||
| Total pension and other employee benefit costs | $ | 68 | $ | 61 | $ | 23 | $ | 20 | |||||||||||||||
Pension Plans
Components of Net Periodic Benefit Cost
Net periodic pension cost consisted of the following:
Pension (1) | |||||||||||||||||||||||
| For the Nine Months Ended September 30, | For the Three Months Ended September 30, | ||||||||||||||||||||||
| (in millions) | 2020 | 2019 | 2020 | 2019 | |||||||||||||||||||
| Service cost | $ | 198 | $ | 161 | $ | 68 | $ | 54 | |||||||||||||||
| Interest cost | 54 | 89 | 19 | 29 | |||||||||||||||||||
| Expected return on plan assets | (265) | (247) | (92) | (82) | |||||||||||||||||||
| Amortization: | |||||||||||||||||||||||
| Net loss | 197 | 142 | 68 | 47 | |||||||||||||||||||
| Prior service cost | 1 | — | — | — | |||||||||||||||||||
| Net periodic pension cost | $ | 185 | $ | 145 | $ | 63 | $ | 48 | |||||||||||||||
(1) Primarily non-U.S. based defined benefit retirement plans.
Employer Contributions
PMI makes, and plans to make, contributions, to the extent that they are tax deductible and to meet specific funding requirements of its funded pension plans. Employer contributions of $62 million were made to the pension plans during the nine months ended September 30, 2020. Currently, PMI anticipates making additional contributions during the remainder of 2020 of approximately $49 million to its pension plans, based on current tax and benefit laws. However, this estimate is subject to change as a result of changes in tax and other benefit laws, as well as asset performance significantly above or below the assumed long-term rate of return on pension assets, or changes in interest and currency rates.
Note 4. Goodwill and Other Intangible Assets, net:
The movements in goodwill were as follows:
| (in millions) | European Union | Eastern Europe | Middle East & Africa | South & Southeast Asia | East Asia & Australia | Latin America & Canada | Total | ||||||||||||||||
| Balances, December 31, 2019 | $ | 1,338 | $ | 300 | $ | 89 | $ | 2,898 | $ | 551 | $ | 682 | $ | 5,858 | |||||||||
| Changes due to: | |||||||||||||||||||||||
| Currency | 18 | 3 | (15) | (116) | (13) | (88) | (211) | ||||||||||||||||
| Balances, September 30, 2020 | $ | 1,356 | $ | 303 | $ | 74 | $ | 2,782 | $ | 538 | $ | 594 | $ | 5,647 | |||||||||
At September 30, 2020, goodwill primarily reflects PMI’s acquisitions in Colombia, Greece, Indonesia, Mexico, Pakistan and Serbia, as well as the business combination in the Philippines.
16
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Details of other intangible assets were as follows:
| September 30, 2020 | December 31, 2019 | |||||||||||||||||||||||||
| (in millions) | Weighted-Average Remaining Useful Life | Gross Carrying Amount | Accumulated Amortization | Net | Gross Carrying Amount | Accumulated Amortization | Net | |||||||||||||||||||
| Non-amortizable intangible assets | $ | 1,199 | $ | 1,199 | $ | 1,319 | $ | 1,319 | ||||||||||||||||||
| Amortizable intangible assets: | ||||||||||||||||||||||||||
| Trademarks | 13 years | 1,197 | $ | 562 | 635 | 1,217 | $ | 526 | 691 | |||||||||||||||||
| Distribution networks | 8 years | 109 | 73 | 36 | 113 | 72 | 41 | |||||||||||||||||||
| Other* | 8 years | 104 | 48 | 56 | 106 | 44 | 62 | |||||||||||||||||||
| Total other intangible assets | $ | 2,609 | $ | 683 | $ | 1,926 | $ | 2,755 | $ | 642 | $ | 2,113 | ||||||||||||||
* Primarily includes intellectual property rights
Non-amortizable intangible assets substantially consist of trademarks from PMI’s acquisitions in Indonesia and Mexico. The decrease since December 31, 2019 was due to currency movements of ($120 million).
The decrease in the gross carrying amount of amortizable intangible assets from December 31, 2019, was mainly due to currency movements of ($27 million).
The change in the accumulated amortization from December 31, 2019, was mainly due to the 2020 amortization of $55 million, partially offset by currency movements of ($14 million).
Amortization expense for each of the next five years is estimated to be $73 million or less, assuming no additional transactions occur that require the amortization of intangible assets.
During the second quarter of 2020, PMI completed its annual review of goodwill and non-amortizable intangible assets for potential impairment, and no impairment charges were required as a result of this review.
Note 5. Financial Instruments:
Overview
PMI operates in markets outside of the United States of America, with manufacturing and sales facilities in various locations around the world. PMI utilizes certain financial instruments to manage foreign currency and interest rate exposure. Derivative financial instruments are used by PMI principally to reduce exposures to market risks resulting from fluctuations in foreign currency exchange and interest rates by creating offsetting exposures. PMI is not a party to leveraged derivatives and, by policy, does not use derivative financial instruments for speculative purposes. Financial instruments qualifying for hedge accounting must maintain a specified level of effectiveness between the hedging instrument and the item being hedged, both at inception and throughout the hedged period. PMI formally documents the nature and relationships between the hedging instruments and hedged items, as well as its risk-management objectives, strategies for undertaking the various hedge transactions and method of assessing hedge effectiveness. Additionally, for hedges of forecasted transactions, the significant characteristics and expected terms of the forecasted transaction must be specifically identified, and it must be probable that each forecasted transaction will occur. If it were deemed probable that the forecasted transaction would not occur, the gain or loss would be recognized in earnings.
17
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
PMI uses deliverable and non-deliverable forward foreign exchange contracts, foreign currency swaps and foreign currency options, collectively referred to as foreign exchange contracts ("foreign exchange contracts"), and interest rate contracts to mitigate its exposure to changes in exchange and interest rates from third-party and intercompany actual and forecasted transactions. Both foreign exchange contracts and interest rate contracts are collectively referred to as derivative contracts ("derivative contracts"). The primary currencies to which PMI is exposed include the Euro, Indonesian rupiah, Japanese yen, Mexican peso, Philippine peso, Russian ruble and Swiss franc. At September 30, 2020, PMI had contracts with aggregate notional amounts of $24.1 billion of which $5.8 billion related to cash flow hedges, $8.3 billion related to hedges of net investments in foreign operations and $10.0 billion related to other derivatives that primarily offset currency exposures on intercompany financing.
The fair value of PMI’s derivative contracts included in the condensed consolidated balance sheets as of September 30, 2020 and December 31, 2019, were as follows:
| Derivative Assets | Derivative Liabilities | |||||||||||||||||||||||||||||||||||||
| Fair Value | Fair Value | |||||||||||||||||||||||||||||||||||||
| At | At | At | At | |||||||||||||||||||||||||||||||||||
| (in millions) | Balance Sheet Classification | September 30, 2020 | December 31, 2019 | Balance Sheet Classification | September 30, 2020 | December 31, 2019 | ||||||||||||||||||||||||||||||||
Derivative contracts designated as hedging instruments | Other current assets | $ | 164 | $ | 319 | Other accrued liabilities | $ | 89 | $ | 23 | ||||||||||||||||||||||||||||
| Other assets | 35 | 21 | Income taxes and other liabilities | 351 | 301 | |||||||||||||||||||||||||||||||||
Derivative contracts not designated as hedging instruments | Other current assets | 55 | 50 | Other accrued liabilities | 90 | 70 | ||||||||||||||||||||||||||||||||
| Other assets | — | — | Income taxes and other liabilities | 35 | 25 | |||||||||||||||||||||||||||||||||
| Total derivatives | $ | 254 | $ | 390 | $ | 565 | $ | 419 | ||||||||||||||||||||||||||||||
18
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
For the nine months and three months ended September 30, 2020 and 2019, PMI's cash flow and net investment hedging instruments impacted the condensed consolidated statements of earnings and comprehensive earnings as follows:
| (pre-tax, in millions) | For the Nine Months Ended September 30, | ||||||||||||||||||||||||||||
| Amount of Gain/(Loss) Recognized in Other Comprehensive Earnings/(Losses) on Derivatives | Statement of Earnings Classification of Gain/(Loss) Reclassified from Other Comprehensive Earnings/(Losses) into Earnings | Amount of Gain/(Loss) Reclassified from Other Comprehensive Earnings/(Losses) into Earnings | |||||||||||||||||||||||||||
| 2020 | 2019 | 2020 | 2019 | ||||||||||||||||||||||||||
| Derivatives in Cash Flow Hedging Relationship | |||||||||||||||||||||||||||||
| Derivative contracts | $ | (45) | $ | (39) | |||||||||||||||||||||||||
| Net revenues | $ | 4 | $ | 22 | |||||||||||||||||||||||||
| Cost of sales | 7 | — | |||||||||||||||||||||||||||
| Marketing, administration and research costs | 15 | (3) | |||||||||||||||||||||||||||
| Interest expense, net | (8) | (4) | |||||||||||||||||||||||||||
| Derivatives in Net Investment Hedging Relationship | |||||||||||||||||||||||||||||
| Derivative contracts | (52) | 564 | |||||||||||||||||||||||||||
| Total | $ | (97) | $ | 525 | $ | 18 | $ | 15 | |||||||||||||||||||||
| (pre-tax, in millions) | For the Three Months Ended September 30, | ||||||||||||||||||||||||||||
| Amount of Gain/(Loss) Recognized in Other Comprehensive Earnings/(Losses) on Derivatives | Statement of Earnings Classification of Gain/(Loss) Reclassified from Other Comprehensive Earnings/(Losses) into Earnings | Amount of Gain/(Loss) Reclassified from Other Comprehensive Earnings/(Losses) into Earnings | |||||||||||||||||||||||||||
| 2020 | 2019 | 2020 | 2019 | ||||||||||||||||||||||||||
| Derivatives in Cash Flow Hedging Relationship | |||||||||||||||||||||||||||||
| Derivative contracts | $ | (38) | $ | (16) | |||||||||||||||||||||||||
| Net revenues | $ | — | $ | (7) | |||||||||||||||||||||||||
| Cost of sales | — | — | |||||||||||||||||||||||||||
| Marketing, administration and research costs | 4 | (4) | |||||||||||||||||||||||||||
| Interest expense, net | (3) | (2) | |||||||||||||||||||||||||||
| Derivatives in Net Investment Hedging Relationship | |||||||||||||||||||||||||||||
| Derivative contracts | (321) | 419 | |||||||||||||||||||||||||||
| Total | $ | (359) | $ | 403 | $ | 1 | $ | (13) | |||||||||||||||||||||
Cash Flow Hedges
PMI has entered into derivative contracts to hedge the foreign currency exchange and interest rate risks related to certain forecasted transactions. Gains and losses associated with qualifying cash flow hedge contracts are deferred as components of accumulated other comprehensive losses until the underlying hedged transactions are reported in PMI’s condensed consolidated statements of earnings. As of September 30, 2020, PMI has hedged forecasted transactions for periods not exceeding the next twenty-one months with the exception of one derivative contract that expires in May 2024. The impact of these hedges is primarily included in operating cash flows on PMI’s condensed consolidated statements of cash flows.
19
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Hedges of Net Investments in Foreign Operations
PMI designates certain foreign currency denominated debt and derivative contracts as net investment hedges, primarily of its Euro net assets. For the nine months ended September 30, 2020 and 2019, these hedges of net investments resulted in gains (losses), net of income taxes, of $(276) million and $771 million, respectively, principally related to changes in the exchange and interest rates between the Euro and U.S. dollar. For the three months ended September 30, 2020 and 2019, these hedges of net investments resulted in gains (losses), net of income taxes, of $(402) million and $598 million, respectively, principally related to changes in the exchange and interest rates between the Euro and U.S. dollar. These gains (losses) were reported as a component of accumulated other comprehensive losses within currency translation adjustments. Gains (losses) attributable to changes in foreign currency exchange rates substantially offset the losses and gains generated on the underlying assets. For the nine months ended September 30, 2020 and 2019, the gains for amounts excluded from the effectiveness testing recognized in earnings were $150 million and $171 million, respectively. For the three months ended September 30, 2020 and 2019, the gains for amounts excluded from the effectiveness testing recognized in earnings were $46 million and $54 million, respectively. These gains were accounted for in interest expense, net, on the condensed consolidated statement of earnings. The premiums paid for, and settlements of, net investment hedges are included in investing cash flows on PMI’s condensed consolidated statements of cash flows.
Other Derivatives
PMI has entered into derivative contracts to hedge the foreign currency exchange and interest rate risks related to intercompany loans between certain subsidiaries, and third-party loans. While effective as economic hedges, no hedge accounting is applied for these contracts; therefore, the unrealized gains (losses) relating to these contracts are reported in marketing, administration and research costs in PMI’s condensed consolidated statements of earnings. For the nine months ended September 30, 2020 and 2019, the gains (losses) from contracts for which PMI did not apply hedge accounting were $(105) million and $25 million, respectively. For the three months ended September 30, 2020 and 2019, the gains (losses) from contracts for which PMI did not apply hedge accounting were $(79) million and $86 million, respectively. The gains (losses) from these contracts attributable to changes in foreign currency exchange rates substantially offset the losses and gains generated by the underlying intercompany and third-party loans being hedged.
For the nine months and three months ended September 30, 2020 and 2019, these items impacted the condensed consolidated statement of earnings as follows:
| (pre-tax, in millions) | ||||||||||||||||||||||||||||||||
| Derivatives not Designated as Hedging Instruments | Statement of Earnings Classification of Gain/(Loss) | Amount of Gain/(Loss) Recognized in Earnings | ||||||||||||||||||||||||||||||
| For the Nine Months Ended September 30, | For the Three Months Ended September 30, | |||||||||||||||||||||||||||||||
| 2020 | 2019 | 2020 | 2019 | |||||||||||||||||||||||||||||
| Derivative contracts | ||||||||||||||||||||||||||||||||
| Interest expense, net | $ | 59 | $ | 79 | $ | 11 | $ | 31 | ||||||||||||||||||||||||
| Total | $ | 59 | $ | 79 | $ | 11 | $ | 31 | ||||||||||||||||||||||||
20
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Qualifying Hedging Activities Reported in Accumulated Other Comprehensive Losses
Derivative gains or losses reported in accumulated other comprehensive losses are a result of qualifying hedging activity. Transfers of these gains or losses to earnings are offset by the corresponding gains or losses on the underlying hedged item. Hedging activity affected accumulated other comprehensive losses, net of income taxes, as follows:
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | |||||||||||||||
| 2020 | 2019 | 2020 | 2019 | ||||||||||||||
| Gain/(loss) at beginning of period, | $ | 3 | $ | 35 | $ | (18) | $ | (10) | |||||||||
| Derivative (gains)/losses transferred to earnings | (17) | (12) | (2) | 13 | |||||||||||||
| Change in fair value | (37) | (34) | (31) | (14) | |||||||||||||
| Gain/(loss) as of September 30, | $ | (51) | $ | (11) | $ | (51) | $ | (11) | |||||||||
At September 30, 2020, PMI expects $12 million of derivative losses that are included in accumulated other comprehensive losses to be reclassified to the condensed consolidated statement of earnings within the next 12 months. These losses are expected to be substantially offset by the statement of earnings impact of the respective hedged transactions.
Contingent Features
PMI’s derivative instruments do not contain contingent features.
Credit Exposure and Credit Risk
PMI is exposed to credit loss in the event of non-performance by counterparties. While PMI does not anticipate non-performance, its risk is limited to the fair value of the financial instruments less any cash collateral received or pledged. PMI actively monitors its exposure to credit risk through the use of credit approvals and credit limit and by selecting and continuously monitoring a diverse group of major international banks and financial institutions as counterparties.
Fair Value
See Note 11. Fair Value Measurements and Note 13. Balance Sheet Offsetting for additional discussion of derivative financial instruments.
Note 6. Earnings Per Share:
Basic and diluted earnings per share (“EPS”) were calculated using the following:
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | |||||||||||||||
| 2020 | 2019 | 2020 | 2019 | ||||||||||||||
| Net earnings attributable to PMI | $ | 6,080 | $ | 5,569 | $ | 2,307 | $ | 1,896 | |||||||||
Less distributed and undistributed earnings attributable to share-based payment awards | 15 | 13 | 5 | 5 | |||||||||||||
| Net earnings for basic and diluted EPS | $ | 6,065 | $ | 5,556 | $ | 2,302 | $ | 1,891 | |||||||||
| Weighted-average shares for basic EPS | 1,557 | 1,556 | 1,558 | 1,556 | |||||||||||||
| Plus contingently issuable performance stock units (PSUs) | — | — | — | — | |||||||||||||
| Weighted-average shares for diluted EPS | 1,557 | 1,556 | 1,558 | 1,556 | |||||||||||||
Unvested share-based payment awards that contain non-forfeitable rights to dividends or dividend equivalents are participating securities and therefore are included in PMI’s earnings per share calculation pursuant to the two-class method.
For the 2020 and 2019 computations, there were no antidilutive stock awards.
21
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Note 7. Segment Reporting:
PMI’s subsidiaries and affiliates are engaged in the manufacture and sale of cigarettes and other nicotine-containing products, including RRPs, in markets outside of the United States of America. In addition, PMI ships a version of its Platform 1 device and its consumables authorized by the FDA to Altria Group, Inc. for sale in the United States under license. Operating segments for PMI are organized by geographic region and managed by segment managers who are responsible for the operating and financial results of the regions inclusive of all product categories sold in the region. PMI’s operating segments are the European Union; Eastern Europe; Middle East & Africa; South & Southeast Asia; East Asia & Australia; and Latin America & Canada. PMI records net revenues and operating income to its segments based upon the geographic area in which the customer resides. Revenues from shipments of Platform 1 devices, heated tobacco units and accessories to Altria Group, Inc. for sale under license in the United States are included in Net Revenues of the Latin America & Canada segment.
PMI’s chief operating decision maker evaluates segment performance and allocates resources based on regional operating income, which includes results from all product categories sold in each region.
PMI disaggregates its net revenue from contracts with customers by both geographic location and product category for each of PMI's six operating segments, as PMI believes this best depicts how the nature, amount, timing and uncertainty of its revenue and cash flows are affected by economic factors.
Segment data were as follows:
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | |||||||||||||||
| 2020 | 2019 | 2020 | 2019 | ||||||||||||||
| Net revenues: | |||||||||||||||||
| European Union | $ | 7,960 | $ | 7,381 | $ | 2,950 | $ | 2,645 | |||||||||
| Eastern Europe | 2,470 | 2,300 | 899 | 899 | |||||||||||||
| Middle East & Africa | 2,348 | 3,058 | 768 | 1,127 | |||||||||||||
| South & Southeast Asia | 3,211 | 3,607 | 1,071 | 1,246 | |||||||||||||
| East Asia & Australia | 4,045 | 4,094 | 1,358 | 1,252 | |||||||||||||
Latin America & Canada (1) | 1,216 | 1,652 | 400 | 473 | |||||||||||||
| Net revenues | $ | 21,250 | $ | 22,092 | $ | 7,446 | $ | 7,642 | |||||||||
| Operating income (loss): | |||||||||||||||||
| European Union | $ | 3,924 | $ | 3,346 | $ | 1,588 | $ | 1,255 | |||||||||
| Eastern Europe | 610 | 284 | 245 | (101) | |||||||||||||
| Middle East & Africa | 819 | 1,304 | 261 | 519 | |||||||||||||
| South & Southeast Asia | 1,290 | 1,471 | 402 | 539 | |||||||||||||
| East Asia & Australia | 1,792 | 1,520 | 637 | 451 | |||||||||||||
Latin America & Canada (1) | 328 | 100 | 110 | 125 | |||||||||||||
| Operating income | $ | 8,763 | $ | 8,025 | $ | 3,243 | $ | 2,788 | |||||||||
(1) As of March 22, 2019, PMI deconsolidated the financial results of its Canadian subsidiary, Rothmans, Benson & Hedges Inc. ("RBH") from PMI's financial statements. For further details, see Note 19. Deconsolidation of RBH.
Items affecting the comparability of results from operations were as follows:
•Russia excise and VAT audit charge - See Note 8. Contingencies for details of the $374 million pre-tax charge included in the Eastern Europe segment for the nine months and three months ended September 30, 2019.
•Canadian tobacco litigation-related expense - See Note 8. Contingencies and Note 19. Deconsolidation of RBH for details of the $194 million pre-tax charge included in the Latin America & Canada segment for the nine months ended September 30, 2019.
22
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
•Loss on deconsolidation of RBH - See Note 19. Deconsolidation of RBH for details of the $239 million loss included in the Latin America & Canada segment for the nine months ended September 30, 2019.
•Asset impairment and exit costs - See Note 18. Asset Impairment and Exit Costs for a breakdown of these costs by segment for the nine months and three months ended September 30, 2020 and 2019.
PMI's net revenues by product category were as follows:
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | |||||||||||||||
| 2020 | 2019 | 2020 | 2019 | ||||||||||||||
| Net revenues: | |||||||||||||||||
| Combustible products: | |||||||||||||||||
| European Union | $ | 6,099 | $ | 6,139 | $ | 2,244 | $ | 2,178 | |||||||||
| Eastern Europe | 1,681 | 1,774 | 636 | 664 | |||||||||||||
| Middle East & Africa | 2,296 | 2,810 | 768 | 1,064 | |||||||||||||
| South & Southeast Asia | 3,211 | 3,607 | 1,071 | 1,246 | |||||||||||||
| East Asia & Australia | 1,876 | 2,074 | 605 | 680 | |||||||||||||
| Latin America & Canada | 1,196 | 1,634 | 393 | 466 | |||||||||||||
| Total combustible products | $ | 16,360 | $ | 18,039 | $ | 5,716 | $ | 6,298 | |||||||||
| Reduced-risk products: | |||||||||||||||||
| European Union | $ | 1,861 | $ | 1,242 | $ | 706 | $ | 467 | |||||||||
| Eastern Europe | 789 | 526 | 263 | 235 | |||||||||||||
| Middle East & Africa | 52 | 248 | — | 63 | |||||||||||||
| South & Southeast Asia | — | — | — | — | |||||||||||||
| East Asia & Australia | 2,169 | 2,020 | 753 | 572 | |||||||||||||
| Latin America & Canada | 20 | 18 | 7 | 7 | |||||||||||||
| Total reduced-risk products | $ | 4,890 | $ | 4,053 | $ | 1,730 | $ | 1,344 | |||||||||
| Total PMI net revenues | $ | 21,250 | $ | 22,092 | $ | 7,446 | $ | 7,642 | |||||||||
Note: Sum of product categories or Regions might not foot to total PMI due to roundings.
Net revenues related to combustible products refer to the operating revenues generated from the sale of these products, including shipping and handling charges billed to customers, net of sales and promotion incentives, and excise taxes. These net revenue amounts consist of the sale of PMI's cigarettes and other tobacco products combined. Other tobacco products primarily include roll-your-own and make-your-own cigarettes, pipe tobacco, cigars and cigarillos and do not include reduced-risk products.
Net revenues related to reduced-risk products refer to the operating revenues generated from the sale of these products, including shipping and handling charges billed to customers, net of sales and promotion incentives, and excise taxes. These net revenue amounts consist of the sale of PMI's heated tobacco units, IQOS devices and related accessories, and other nicotine-containing products, which primarily include PMI's e-vapor products.
PMI recognizes revenue, when control is transferred to the customer, typically either upon shipment or delivery of goods.
Note 8. Contingencies:
Tobacco-Related Litigation
Legal proceedings covering a wide range of matters are pending or threatened against us, and/or our subsidiaries, and/or our indemnitees in various jurisdictions. Our indemnitees include distributors, licensees, and others that have been named as parties
23
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
in certain cases and that we have agreed to defend, as well as to pay costs and some or all of judgments, if any, that may be entered against them. Pursuant to the terms of the Distribution Agreement between Altria Group, Inc. (“Altria”) and PMI, PMI will indemnify Altria and Philip Morris USA Inc. (“PM USA”), a U.S. tobacco subsidiary of Altria, for tobacco product claims based in substantial part on products manufactured by PMI or contract manufactured for PMI by PM USA, and PM USA will indemnify PMI for tobacco product claims based in substantial part on products manufactured by PM USA, excluding tobacco products contract manufactured for PMI.
It is possible that there could be adverse developments in pending cases against us and our subsidiaries. An unfavorable outcome or settlement of pending tobacco-related litigation could encourage the commencement of additional litigation.
Damages claimed in some of the tobacco-related litigation are significant and, in certain cases in Brazil, Canada and Nigeria, range into the billions of U.S. dollars. The variability in pleadings in multiple jurisdictions, together with the actual experience of management in litigating claims, demonstrate that the monetary relief that may be specified in a lawsuit bears little relevance to the ultimate outcome. Much of the tobacco-related litigation is in its early stages, and litigation is subject to uncertainty. However, as discussed below, we have to date been largely successful in defending tobacco-related litigation.
We and our subsidiaries record provisions in the consolidated financial statements for pending litigation when we determine that an unfavorable outcome is probable and the amount of the loss can be reasonably estimated. At the present time, except as stated otherwise in this Note 8. Contingencies, while it is reasonably possible that an unfavorable outcome in a case may occur, after assessing the information available to it (i) management has not concluded that it is probable that a loss has been incurred in any of the pending tobacco-related cases; (ii) management is unable to estimate the possible loss or range of loss for any of the pending tobacco-related cases; and (iii) accordingly, no estimated loss has been accrued in the consolidated financial statements for unfavorable outcomes in these cases, if any. Legal defense costs are expensed as incurred.
It is possible that our consolidated results of operations, cash flows or financial position could be materially affected in a particular fiscal quarter or fiscal year by an unfavorable outcome or settlement of certain pending litigation. Nevertheless, although litigation is subject to uncertainty, we and each of our subsidiaries named as a defendant believe, and each has been so advised by counsel handling the respective cases, that we have valid defenses to the litigation pending against us, as well as valid bases for appeal of adverse verdicts. All such cases are, and will continue to be, vigorously defended. However, we and our subsidiaries may enter into settlement discussions in particular cases if we believe it is in our best interests to do so.
CCAA Proceedings and Stay of Tobacco-Related Cases Pending in Canada
As a result of the Court of Appeal of Quebec’s decision in both the Létourneau and Blais cases described below, our subsidiary, Rothmans, Benson & Hedges Inc. (“RBH”), and the other defendants, JTI Macdonald Corp., and Imperial Tobacco Canada Limited, sought protection in the Ontario Superior Court of Justice under the Companies’ Creditors Arrangement Act (“CCAA”) on March 22, March 8, and March 12, 2019 respectively. CCAA is a Canadian federal law that permits a Canadian business to restructure its affairs while carrying on its business in the ordinary course. The initial CCAA order made by the Ontario Superior Court on March 22, 2019 authorizes RBH to pay all expenses incurred in carrying on its business in the ordinary course after the CCAA filing, including obligations to employees, vendors, and suppliers. As further described in Note 19. Deconsolidation of RBH, RBH is now deconsolidated from our consolidated financial statements. As part of the CCAA proceedings, there is currently a comprehensive stay up to and including March 31, 2021 of all tobacco-related litigation pending in Canada against RBH and the other defendants, including PMI and our indemnitees (PM USA and Altria), namely, the smoking and health class actions filed in various Canadian provinces and health care cost recovery actions. These proceedings are presented below under the caption “Stayed Litigation — Canada.” Ernst & Young Inc. has been appointed as monitor of RBH in the CCAA proceedings. In accordance with the CCAA process, as the parties work towards a plan of arrangement or compromise in a confidential mediation, it is anticipated that the court will set additional hearings and further extend the stay of proceedings. On April 17, 2019, the Ontario Superior Court ruled that RBH and the other defendants will not be allowed to file an application to the Supreme Court of Canada for leave to appeal the Court of Appeal’s decision in the Létourneau and the Blais cases so long as the comprehensive stay of all tobacco-related litigation in Canada remains in effect and that the time period to file the application would be extended by the stay period. While RBH believes that the findings of liability and damages in both Létourneau and the Blais cases were incorrect, the CCAA proceedings will provide a forum for RBH to seek resolution through a plan of arrangement or compromise of all tobacco-related litigation pending in Canada. It is not possible to predict the resolution of the underlying legal proceedings or the length of the CCAA process.
24
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Stayed Litigation — Canada
Smoking and Health Litigation — Canada
In the first class action pending in Canada, Conseil Québécois Sur Le Tabac Et La Santé and Jean-Yves Blais v. Imperial Tobacco Ltd., Rothmans, Benson & Hedges Inc. and JTI-Macdonald Corp., Quebec Superior Court, Canada, filed in November 1998, RBH and other Canadian manufacturers (Imperial Tobacco Canada Ltd. and JTI-Macdonald Corp.) are defendants. The plaintiffs, an anti-smoking organization and an individual smoker, sought compensatory and punitive damages for each member of the class who allegedly suffers from certain smoking-related diseases. The class was certified in 2005. The trial court issued its judgment on May 27, 2015. The trial court found RBH and two other Canadian manufacturers liable and found that the class members’ compensatory damages totaled approximately CAD 15.5 billion, including pre-judgment interest (approximately $11.8 billion). The trial court awarded compensatory damages on a joint and several liability basis, allocating 20% to our subsidiary (approximately CAD 3.1 billion, including pre-judgment interest (approximately $2.4 billion)). In addition, the trial court awarded CAD 90,000 (approximately $68,440) in punitive damages, allocating CAD 30,000 (approximately $22,810) to RBH. The trial court estimated the disease class at 99,957 members. RBH appealed to the Court of Appeal of Quebec. In October 2015, the Court of Appeal ordered RBH to furnish security totaling CAD 226 million (approximately $171.9 million) to cover both the Létourneau and Blais cases, which RBH has paid in installments through March 2017. The Court of Appeal ordered Imperial Tobacco Canada Ltd. to furnish security totaling CAD 758 million (approximately $576 million) in installments through June 2017. JTI Macdonald Corp. was not required to furnish security in accordance with plaintiffs’ motion. The Court of Appeal ordered that the security is payable upon a final judgment of the Court of Appeal affirming the trial court’s judgment or upon further order of the Court of Appeal. On March 1, 2019, the Court of Appeal issued a decision largely affirming the trial court’s findings of liability and the compensatory and punitive damages award while reducing the total amount of compensatory damages to approximately CAD 13.5 billion including interest (approximately $10.3 billion) due to the trial court’s error in the calculation of interest. The compensatory damages award is on a joint and several basis with an allocation of 20% to RBH (approximately CAD 2.7 billion, including pre-judgment interest (approximately $2.05 billion)). The Court of Appeal upheld the trial court’s findings that defendants violated the Civil Code of Quebec, the Quebec Charter of Human Rights and Freedoms, and the Quebec Consumer Protection Act by failing to warn adequately of the dangers of smoking and by conspiring to prevent consumers from learning of the dangers of smoking. The Court of Appeal further held that the plaintiffs either need not prove, or had adequately proven, that these faults were a cause of the class members’ injuries. In accordance with the judgment, defendants are required to deposit their respective portions of the damages awarded in both the Létourneau case described below and the Blais case, approximately CAD 1.1 billion (approximately $836 million), into trust accounts within 60 days. RBH’s share of the deposit is approximately CAD 257 million (approximately $194 million). PMI recorded a pre-tax charge of $194 million in its consolidated results, representing $142 million net of tax, as tobacco litigation-related expense, in the first quarter of 2019. The charge reflects PMI’s assessment of the portion of the judgment that represents probable and estimable loss prior to the deconsolidation of RBH and corresponds to the trust account deposit required by the judgment.
In the second class action pending in Canada, Cecilia Létourneau v. Imperial Tobacco Ltd., Rothmans, Benson & Hedges Inc. and JTI-Macdonald Corp., Quebec Superior Court, Canada, filed in September 1998, RBH and other Canadian manufacturers (Imperial Tobacco Canada Ltd. and JTI-Macdonald Corp.) are defendants. The plaintiff, an individual smoker, sought compensatory and punitive damages for each member of the class who is deemed addicted to smoking. The class was certified in 2005. The trial court issued its judgment on May 27, 2015. The trial court found RBH and two other Canadian manufacturers liable and awarded a total of CAD 131 million (approximately $99.6 million) in punitive damages, allocating CAD 46 million (approximately $35 million) to RBH. The trial court estimated the size of the addiction class at 918,000 members but declined to award compensatory damages to the addiction class because the evidence did not establish the claims with sufficient accuracy. The trial court found that a claims process to allocate the awarded punitive damages to individual class members would be too expensive and difficult to administer. On March 1, 2019, the Court of Appeal issued a decision largely affirming the trial court’s findings of liability and the total amount of punitive damages awarded allocating CAD 57 million including interest (approximately $43.4 million) to RBH. See the Blais description above and Note 19. Deconsolidation of RBH below for further detail concerning the security order pertaining to both Létourneau and Blais cases and the impact of the decision on PMI’s financial statements.
RBH and PMI believe the findings of liability and damages in both Létourneau and the Blais cases were incorrect and in contravention of applicable law on several grounds including the following: (i) defendants had no obligation to warn class
25
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
members who knew, or should have known, of the risks of smoking; (ii) defendants cannot be liable to class members who would have smoked regardless of what warnings were given; and (iii) defendants cannot be liable to all class members given the individual differences between class members.
In the third class action pending in Canada, Kunta v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Winnipeg, Canada, filed June 12, 2009, we, RBH, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and chronic obstructive pulmonary disease (“COPD”), severe asthma, and mild reversible lung disease resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, as well as restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products.
In the fourth class action pending in Canada, Adams v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Saskatchewan, Canada, filed July 10, 2009, we, RBH, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and COPD resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who have smoked a minimum of 25,000 cigarettes and have allegedly suffered, or suffer, from COPD, emphysema, heart disease, or cancer, as well as restitution of profits.
In the fifth class action pending in Canada, Semple v. Canadian Tobacco Manufacturers' Council, et al., The Supreme Court (trial court), Nova Scotia, Canada, filed June 18, 2009, we, RBH, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges his own addiction to tobacco products and COPD resulting from the use of tobacco products. He is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, as well as restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products.
In the sixth class action pending in Canada, Dorion v. Canadian Tobacco Manufacturers' Council, et al., The Queen's Bench, Alberta, Canada, filed June 15, 2009, we, RBH, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and chronic bronchitis and severe sinus infections resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers, their estates, dependents and family members, restitution of profits, and reimbursement of government health care costs allegedly caused by tobacco products. To date, we, our subsidiaries, and our indemnitees have not been properly served with the complaint.
In the seventh class action pending in Canada, McDermid v. Imperial Tobacco Canada Limited, et al., Supreme Court, British Columbia, Canada, filed June 25, 2010, we, RBH, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges his own addiction to tobacco products and heart disease resulting from the use of tobacco products. He is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who were alive on June 12, 2007, and who suffered from heart disease allegedly caused by smoking, their estates, dependents and family members, plus disgorgement of revenues earned by the defendants from January 1, 1954, to the date the claim was filed.
In the eighth class action pending in Canada, Bourassa v. Imperial Tobacco Canada Limited, et al., Supreme Court, British Columbia, Canada, filed June 25, 2010, we, RBH, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, the heir to a deceased smoker, alleges that the decedent was addicted to tobacco products and suffered from emphysema resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who were alive on June 12, 2007, and who suffered from chronic respiratory diseases allegedly caused by smoking, their estates, dependents and family members, plus disgorgement of revenues earned by the defendants from January 1, 1954, to the date the claim was filed. In December 2014, plaintiff filed an amended statement of claim.
In the ninth class action pending in Canada, Suzanne Jacklin v. Canadian Tobacco Manufacturers' Council, et al., Ontario Superior Court of Justice, filed June 20, 2012, we, RBH, and our indemnitees (PM USA and Altria), and other members of the industry are defendants. The plaintiff, an individual smoker, alleges her own addiction to tobacco products and COPD resulting from the use of tobacco products. She is seeking compensatory and punitive damages on behalf of a proposed class comprised of all smokers who have smoked a minimum of 25,000 cigarettes and have allegedly suffered, or suffer, from COPD, heart disease, or cancer, as well as restitution of profits.
26
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Health Care Cost Recovery Litigation — Canada
In the first health care cost recovery case pending in Canada, Her Majesty the Queen in Right of British Columbia v. Imperial Tobacco Limited, et al., Supreme Court, British Columbia, Vancouver Registry, Canada, filed January 24, 2001, we, RBH, our indemnitee (PM USA), and other members of the industry are defendants. The plaintiff, the government of the province of British Columbia, brought a claim based upon legislation enacted by the province authorizing the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, resulting from a “tobacco related wrong.”
In the second health care cost recovery case filed in Canada, Her Majesty the Queen in Right of New Brunswick v. Rothmans Inc., et al., Court of Queen's Bench of New Brunswick, Trial Court, New Brunswick, Fredericton, Canada, filed March 13, 2008, we, RBH, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of New Brunswick based on legislation enacted in the province. This legislation is similar to the law introduced in British Columbia that authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
In the third health care cost recovery case filed in Canada, Her Majesty the Queen in Right of Ontario v. Rothmans Inc., et al., Ontario Superior Court of Justice, Toronto, Canada, filed September 29, 2009, we, RBH, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Ontario based on legislation enacted in the province. This legislation is similar to the laws introduced in British Columbia and New Brunswick that authorize the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
In the fourth health care cost recovery case filed in Canada, Attorney General of Newfoundland and Labrador v. Rothmans Inc., et al., Supreme Court of Newfoundland and Labrador, St. Johns, Canada, filed February 8, 2011, we, RBH, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Newfoundland and Labrador based on legislation enacted in the province that is similar to the laws introduced in British Columbia, New Brunswick and Ontario. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
In the fifth health care cost recovery case filed in Canada, Attorney General of Quebec v. Imperial Tobacco Limited, et al., Superior Court of Quebec, Canada, filed June 8, 2012, we, RBH, our indemnitee (PM USA), and other members of the industry are defendants. The claim was filed by the government of the province of Quebec based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
In the sixth health care cost recovery case filed in Canada, Her Majesty in Right of Alberta v. Altria Group, Inc., et al., Supreme Court of Queen's Bench Alberta, Canada, filed June 8, 2012, we, RBH, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Alberta based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
In the seventh health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Manitoba v. Rothmans, Benson & Hedges, Inc., et al., The Queen's Bench, Winnipeg Judicial Centre, Canada, filed May 31, 2012, we, RBH, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Manitoba based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
In the eighth health care cost recovery case filed in Canada, The Government of Saskatchewan v. Rothmans, Benson & Hedges Inc., et al., Queen's Bench, Judicial Centre of Saskatchewan, Canada, filed June 8, 2012, we, RBH, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Saskatchewan based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
27
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
In the ninth health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Prince Edward Island v. Rothmans, Benson & Hedges Inc., et al., Supreme Court of Prince Edward Island (General Section), Canada, filed September 10, 2012, we, RBH, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Prince Edward Island based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
In the tenth health care cost recovery case filed in Canada, Her Majesty the Queen in Right of the Province of Nova Scotia v. Rothmans, Benson & Hedges Inc., et al., Supreme Court of Nova Scotia, Canada, filed January 2, 2015, we, RBH, our indemnitees (PM USA and Altria), and other members of the industry are defendants. The claim was filed by the government of the province of Nova Scotia based on legislation enacted in the province that is similar to the laws enacted in several other Canadian provinces. The legislation authorizes the government to file a direct action against cigarette manufacturers to recover the health care costs it has incurred, and will incur, as a result of a “tobacco related wrong.”
__________
The table below lists the number of tobacco-related cases pertaining to combustible products pending against us and/or our subsidiaries or indemnitees as of October 23, 2020, October 22, 2019 and October 23, 2018:¹
| Type of Case | Number of Cases Pending as of October 23, 2020 | Number of Cases Pending as of October 22, 2019 | Number of Cases Pending as of October 23, 2018 | |||||||||||||||||
| Individual Smoking and Health Cases | 43 | 49 | 63 | |||||||||||||||||
| Smoking and Health Class Actions | 9 | 10 | 10 | |||||||||||||||||
| Health Care Cost Recovery Actions | 17 | 17 | 16 | |||||||||||||||||
| Label-Related Class Actions | — | — | 1 | |||||||||||||||||
| Individual Label-Related Cases | 5 | 5 | 1 | |||||||||||||||||
| Public Civil Actions | 2 | 2 | 2 | |||||||||||||||||
Since 1995, when the first tobacco-related litigation was filed against a PMI entity, 509 Smoking and Health, Label-Related, Health Care Cost Recovery, and Public Civil Actions in which we and/or one of our subsidiaries and/or indemnitees were a defendant have been terminated in our favor. Thirteen cases have had decisions in favor of plaintiffs. Ten of these cases have subsequently reached final resolution in our favor and three remain on appeal.
The table below lists the verdict and significant post-trial developments in the three pending cases where a verdict was returned in favor of the plaintiff:
______
¹ Includes cases pending in Canada.
28
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
| Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||||||||||||||||||||
| May 27, 2015 | Canada/Conseil Québécois Sur Le Tabac Et La Santé and Jean-Yves Blais | Class Action | On May 27, 2015, the Superior Court of the District of Montreal, Province of Quebec ruled in favor of the Blais class on liability and found the class members’ compensatory damages totaled approximately CAD 15.5 billion (approximately $11.8 billion), including pre-judgment interest. The trial court awarded compensatory damages on a joint and several liability basis, allocating 20% to our subsidiary (approximately CAD 3.1 billion including pre-judgment interest (approximately $2.4 billion)). The trial court awarded CAD 90,000 (approximately $68,440) in punitive damages, allocating CAD 30,000 (approximately $22,810) to our subsidiary. The trial court ordered defendants to pay CAD 1 billion (approximately $760 million) of the compensatory damage award, CAD 200 million (approximately $152.1 million) of which is our subsidiary’s portion, into a trust within 60 days. | In June 2015, RBH commenced the appellate process with the Court of Appeal of Quebec. On March 1, 2019, the Court of Appeal issued a decision largely affirming the trial court's decision. (See “Stayed Litigation — Canada” for further detail.) | ||||||||||||||||||||||
29
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
| Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||||||||||||||||||||
| May 27, 2015 | Canada/Cecilia Létourneau | Class Action | On May 27, 2015, the Superior Court of the District of Montreal, Province of Quebec ruled in favor of the Létourneau class on liability and awarded a total of CAD 131 million (approximately $99.6 million) in punitive damages, allocating CAD 46 million (approximately $35 million) to RBH. The trial court ordered defendants to pay the full punitive damage award into a trust within 60 days. The court did not order the payment of compensatory damages. | In June 2015, RBH commenced the appellate process with the Court of Appeal of Quebec. On March 1, 2019, the Court of Appeal issued a decision largely affirming the trial court's decision. (See “Stayed Litigation — Canada” for further detail.) | ||||||||||||||||||||||
| Date | Location of Court/Name of Plaintiff | Type of Case | Verdict | Post-Trial Developments | ||||||||||||||||||||||
| August 5, 2016 | Argentina/Hugo Lespada | Individual Action | On August 5, 2016, the Civil Court No. 14 - Mar del Plata, issued a verdict in favor of plaintiff, an individual smoker, and awarded him ARS 110,000 (approximately $1,414), plus interest, in compensatory and moral damages. The trial court found that our subsidiary failed to warn plaintiff of the risk of becoming addicted to cigarettes. | On August 23, 2016, our subsidiary filed its notice of appeal. On October 31, 2017, the Civil and Commercial Court of Appeals of Mar del Plata ruled that plaintiff's claim was barred by the statute of limitations and it reversed the trial court's decision. On November 28, 2017, plaintiff filed an extraordinary appeal of the reversal of the trial court's decision to the Supreme Court of the Province of Buenos Aires. | ||||||||||||||||||||||
Pending claims related to tobacco products generally fall within the following categories:
Smoking and Health Litigation: These cases primarily allege personal injury and are brought by individual plaintiffs or on behalf of a class or purported class of individual plaintiffs. Plaintiffs' allegations of liability in these cases are based on various theories of recovery, including negligence, gross negligence, strict liability, fraud, misrepresentation, design defect, failure to warn, breach of express and implied warranties, violations of deceptive trade practice laws and consumer protection statutes. Plaintiffs in these cases seek various forms of relief, including compensatory and other damages, and injunctive and equitable relief. Defenses raised in these cases include licit activity, failure to state a claim, lack of defect, lack of proximate cause, assumption of the risk, contributory negligence, and statute of limitations.
As of October 23, 2020, there were a number of smoking and health cases pending against us, our subsidiaries or indemnitees, as follows:
30
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
•43 cases brought by individual plaintiffs in Argentina (31), Brazil (3), Canada (2), Chile (3), Italy (1), the Philippines (1), Turkey (1) and Scotland (1), compared with 49 such cases on October 22, 2019, and 63 cases on October 23, 2018; and
•9 cases brought on behalf of classes of individual plaintiffs in Canada, compared with 10 such cases on October 22, 2019 and 10 such cases on October 23, 2018.
The class actions pending in Canada are described above under the caption “Smoking and Health Litigation — Canada.”
In a class action in Brazil, The Smoker Health Defense Association (ADESF) v. Souza Cruz, S.A. and Philip Morris Marketing, S.A., Nineteenth Lower Civil Court of the Central Courts of the Judiciary District of São Paulo, Brazil, filed July 25, 1995, our subsidiary and another member of the industry are defendants. The plaintiff, a consumer organization, sought damages for all addicted smokers and former smokers, and injunctive relief. In 2004, the trial court found defendants liable without hearing evidence and awarded “moral damages” of R$1,000 (approximately $179) per smoker per full year of smoking plus interest at the rate of 1% per month, as of the date of the ruling. The court did not award actual damages, which were to be assessed in the second phase of the case. The size of the class was not estimated. Defendants appealed to the São Paulo Court of Appeals, which annulled the ruling in November 2008, finding that the trial court had inappropriately ruled without hearing evidence and returned the case to the trial court for further proceedings. In May 2011, the trial court dismissed the claim. In February 2015, the appellate court unanimously dismissed plaintiff's appeal. In September 2015, plaintiff appealed to the Superior Court of Justice. In February 2017, the Chief Justice of the Superior Court of Justice denied plaintiff's appeal. Plaintiff filed a further appeal. In August 2020, the Superior Court of Justice confirmed the denial of plaintiff's appeal finally dismissing the plaintiff's claim.
Health Care Cost Recovery Litigation: These cases, brought by governmental and non-governmental plaintiffs, seek reimbursement of health care cost expenditures allegedly caused by tobacco products. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including unjust enrichment, negligence, negligent design, strict liability, breach of express and implied warranties, violation of a voluntary undertaking or special duty, fraud, negligent misrepresentation, conspiracy, public nuisance, defective product, failure to warn, sale of cigarettes to minors, and claims under statutes governing competition and deceptive trade practices. Plaintiffs in these cases seek various forms of relief including compensatory and other damages, and injunctive and equitable relief. Defenses raised in these cases include lack of proximate cause, remoteness of injury, failure to state a claim, adequate remedy at law, “unclean hands” (namely, that plaintiffs cannot obtain equitable relief because they participated in, and benefited from, the sale of cigarettes), and statute of limitations.
As of October 23, 2020, there were 17 health care cost recovery cases pending against us, our subsidiaries or indemnitees in Brazil (1), Canada (10), Korea (1) and Nigeria (5), compared with 17 such cases on October 22, 2019 and 16 such cases on October 23, 2018.
The health care cost recovery actions pending in Canada are described above under the caption “Health Care Cost Recovery Litigation — Canada.”
In the health care cost recovery case in Brazil, The Attorney General of Brazil v. Souza Cruz Ltda., et al., Federal Trial Court, Porto Alegre, Rio Grande do Sul, Brazil, filed May 21, 2019, we, our subsidiaries, and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past six years, payment of anticipated costs of treating future alleged smoking-related diseases, and moral damages. Defendants filed answers to the complaint in May 2020.
In the first health care cost recovery case in Nigeria, The Attorney General of Lagos State v. British American Tobacco (Nigeria) Limited, et al., High Court of Lagos State, Lagos, Nigeria, filed March 13, 2008, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. We are in the process of making challenges to service and the court's jurisdiction. Currently, the case is stayed in the trial court pending the appeals of certain co-defendants relating to service objections.
In the second health care cost recovery case in Nigeria, The Attorney General of Kano State v. British American Tobacco (Nigeria) Limited, et al., High Court of Kano State, Kano, Nigeria, filed May 9, 2007, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive
31
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
relief, plus punitive damages. We are in the process of making challenges to service and the court's jurisdiction. Currently, the case is stayed in the trial court pending the appeals of certain co-defendants relating to service objections.
In the third health care cost recovery case in Nigeria, The Attorney General of Gombe State v. British American Tobacco (Nigeria) Limited, et al., High Court of Gombe State, Gombe, Nigeria, filed October 17, 2008, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. In February 2011, the court ruled that the plaintiff had not complied with the procedural steps necessary to serve us. As a result of this ruling, plaintiff must re-serve its claim. We have not yet been re-served.
In the fourth health care cost recovery case in Nigeria, The Attorney General of Oyo State, et al., v. British American Tobacco (Nigeria) Limited, et al., High Court of Oyo State, Ibadan, Nigeria, filed May 25, 2007, we and other members of the industry are defendants. Plaintiffs seek reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. We challenged service as improper. In June 2010, the court ruled that plaintiffs did not have leave to serve the writ of summons on the defendants and that they must re-serve the writ. We have not yet been re-served.
In the fifth health care cost recovery case in Nigeria, The Attorney General of Ogun State v. British American Tobacco (Nigeria) Limited, et al., High Court of Ogun State, Abeokuta, Nigeria, filed February 26, 2008, we and other members of the industry are defendants. Plaintiff seeks reimbursement for the cost of treating alleged smoking-related diseases for the past 20 years, payment of anticipated costs of treating alleged smoking-related diseases for the next 20 years, various forms of injunctive relief, plus punitive damages. In May 2010, the trial court rejected our service objections. We have appealed.
In the health care cost recovery case in Korea, the National Health Insurance Service v. KT&G, et. al., filed April 14, 2014, our subsidiary and other Korean manufacturers are defendants. Plaintiff alleges that defendants concealed the health hazards of smoking, marketed to youth, added ingredients to make their products more harmful and addictive, and misled consumers into believing that Lights cigarettes are safer than regular cigarettes. The National Health Insurance Service seeks to recover damages allegedly incurred in treating 3,484 patients with small cell lung cancer, squamous cell lung cancer, and squamous cell laryngeal cancer from 2003 to 2012. The case is awaiting a court decision in November 2020.
Label-Related Cases: These cases, now brought only by individual plaintiffs, allege that the use of the descriptor “Lights” or other alleged misrepresentations or omissions of labeling information constitute fraudulent and misleading conduct. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including misrepresentation, deception, and breach of consumer protection laws. Plaintiffs seek various forms of relief including restitution, injunctive relief, and compensatory and other damages. Defenses raised include lack of causation, lack of reliance, assumption of the risk, and statute of limitations.
As of October 23, 2020, there were 5 label-related cases brought by individual plaintiffs in Italy (1) and Chile (4) pending against our subsidiaries, compared with 5 such cases on October 22, 2019, and 1 such case on October 23, 2018.
Public Civil Actions: Claims have been filed either by an individual, or a public or private entity, seeking to protect collective or individual rights, such as the right to health, the right to information or the right to safety. Plaintiffs' allegations of liability in these cases are based on various theories of recovery including product defect, concealment, and misrepresentation. Plaintiffs in these cases seek various forms of relief including injunctive relief such as banning cigarettes, descriptors, smoking in certain places and advertising, as well as implementing communication campaigns and reimbursement of medical expenses incurred by public or private institutions.
As of October 23, 2020, there were 2 public civil actions pending against our subsidiaries in Argentina (1) and Venezuela (1), compared with 2 such cases on October 22, 2019, and 2 such cases on October 23, 2018.
In the public civil action in Argentina, Asociación Argentina de Derecho de Danos v. Massalin Particulares S.A., et al., Civil Court of Buenos Aires, Argentina, filed February 26, 2007, our subsidiary and another member of the industry are defendants. The plaintiff, a consumer association, seeks the establishment of a relief fund for reimbursement of medical costs associated with diseases allegedly caused by smoking. Our subsidiary filed its answer in September 2007. In March 2010, the case file was transferred to the Federal Court on Administrative Matters after the Civil Court granted plaintiff's request to add the national government as a co-plaintiff in the case. The case is currently awaiting a court decision on the merits.
32
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
In the public civil action in Venezuela, Federation of Consumers and Users Associations (“FEVACU”), et al. v. National Assembly of Venezuela and the Venezuelan Ministry of Health, Constitutional Chamber of the Venezuelan Supreme Court, filed April 29, 2008, we were not named as a defendant, but the plaintiffs published a notice pursuant to court order, notifying all interested parties to appear in the case. In January 2009, our subsidiary appeared in the case in response to this notice. The plaintiffs purport to represent the right to health of the citizens of Venezuela and claim that the government failed to protect adequately its citizens' right to health. The claim asks the court to order the government to enact stricter regulations on the manufacture and sale of tobacco products. In addition, the plaintiffs ask the court to order companies involved in the tobacco industry to allocate a percentage of their “sales or benefits” to establish a fund to pay for the health care costs of treating smoking-related diseases. In October 2008, the court ruled that plaintiffs have standing to file the claim and that the claim meets the threshold admissibility requirements. In December 2012, the court admitted our subsidiary and BAT's subsidiary as interested third parties. In February 2013, our subsidiary answered the complaint.
Reduced-Risk Products
In Colombia, an individual filed a purported class action, Ana Ferrero Rebolledo v. Philip Morris Colombia S.A., et al., in April 2019 against our subsidiaries with the Civil Court of Bogota related to the marketing of our Platform 1 product. Plaintiff alleged that our subsidiaries advertise the product in contravention of law and in a manner that misleads consumers by portraying the product in a positive light, and further asserts that the Platform 1 vapor contains many toxic compounds, creates a high level of dependence, and has damaging second-hand effects. Plaintiff sought injunctive relief and damages on her behalf and on a behalf of two classes (class 1 - all Platform 1 consumers in Colombia who seek damages for the purchase price of the product and personal injuries related to the alleged addiction, and class 2 - all residents of the neighborhood where the advertising allegedly took place who seek damages for exposure to the alleged illegal advertising). Our subsidiaries answered the complaint in January 2020, and in February 2020, plaintiff filed an amended complaint. The amended complaint modifies the relief sought on behalf of the named plaintiff and on behalf of a single class (all consumers of Platform 1 products in Colombia who seek damages for the product purchase price and personal injuries related to the use of an allegedly harmful product). The amended complaint has not yet been served on our subsidiaries.
Other Litigation
The Department of Special Investigations of the government of Thailand ("DSI") conducted an investigation into alleged underpayment by our subsidiary, Philip Morris (Thailand) Limited ("PM Thailand"), of customs duties and excise taxes relating to imports from the Philippines covering the period 2003-2007. On January 18, 2016, the Public Prosecutor filed charges against our subsidiary and seven former and current employees in the Bangkok Criminal Court alleging that PM Thailand and the individual defendants jointly and with the intention to defraud the Thai government, under-declared import prices of cigarettes to avoid full payment of taxes and duties in connection with import entries of cigarettes from the Philippines during the period of July 2003 to June 2006. The government is seeking a fine of approximately THB 80.8 billion (approximately $2.6 billion). In May 2017, Thailand enacted a new customs act. The new act, which took effect in November 2017, substantially limits the amount of fines that Thailand could seek in these proceedings. PM Thailand believes that its declared import prices are in compliance with the Customs Valuation Agreement of the World Trade Organization and Thai law and that the allegations of the Public Prosecutor are inconsistent with several decisions already taken by Thai Customs and other Thai governmental agencies. Trial in the case began in November 2017 and concluded in September 2019. In November 2019, the trial court found our subsidiary guilty of under-declaration of the prices and imposed a fine of approximately THB 1.2 billion (approximately $38.3 million). The trial court dismissed all charges against the individual defendants. In December 2019, as required by the Thai law, our subsidiary paid the fine. This payment is included in other assets on the condensed consolidated balance sheets and negatively impacted net cash provided by operating activities in the condensed consolidated statements of cash flows in the period of payment. Our subsidiary filed an appeal of the trial court's decision. In addition, the Public Prosecutor filed an appeal of the trial court's decision challenging the dismissal of charges against the individual defendants and the amount of the fine imposed. If our subsidiary ultimately prevails on appeal, then Thailand will be required to return this payment to our subsidiary.
The DSI also conducted an investigation into alleged underpayment by PM Thailand of customs duties and excise taxes relating to imports from Indonesia covering the period 2000-2003. On January 26, 2017, the Public Prosecutor filed charges against PM Thailand and its former Thai employee in the Bangkok Criminal Court alleging that PM Thailand and its former employee
33
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
jointly and with the intention to defraud the Thai government under-declared import prices of cigarettes to avoid full payment of taxes and duties in connection with import entries during the period from January 2002 to July 2003. The government is seeking a fine of approximately THB 19.8 billion (approximately $632 million). In May 2017, Thailand enacted a new customs act. The new act, which took effect in November 2017, substantially limits the amount of fines that Thailand could seek in these proceedings. PM Thailand believes that its declared import prices are in compliance with the Customs Valuation Agreement of the World Trade Organization and Thai law, and that the allegations of the Public Prosecutor are inconsistent with several decisions already taken by Thai Customs and a Thai court. Trial in the case began in November 2018 and concluded in December 2019. In March 2020, the trial court found our subsidiary guilty of under-declaration of the prices and imposed a fine of approximately THB 130 million (approximately $4.1 million). The trial court dismissed all charges against the individual defendant. In April 2020, as required by Thai law, our subsidiary paid the fine. Our subsidiary will appeal the trial court's decision. If our subsidiary ultimately prevails on appeal, then Thailand will be required to return this payment to our subsidiary.
The South Korean Board of Audit and Inspection (“BAI”) conducted an audit of certain Korean government agencies and the tobacco industry into whether inventory movements ahead of the January 1, 2015 increase of cigarette-related taxes by tobacco companies, including Philip Morris Korea Inc. ("PM Korea"), our South Korean subsidiary, were in compliance with South Korean tax laws. In November 2016, the tax authorities completed their audit and assessed allegedly underpaid taxes and penalties. In order to avoid nonpayment financial costs, PM Korea paid approximately KRW 272 billion (approximately $240 million), of which KRW 100 billion (approximately $88.1 million) was paid in 2016 and KRW 172 billion (approximately $151.5 million) was paid in the first quarter of 2017. These paid amounts are included in other assets in the condensed consolidated balance sheets and negatively impacted net cash provided by operating activities in the condensed consolidated statements of cash flows in the period of payment. PM Korea appealed the assessments. In January 2020, a trial court ruled that PM Korea did not underpay taxes in the amount of approximately KRW 218 billion (approximately $192 million). The tax authorities appealed this decision to the appellate court. In September 2020, the appellate court upheld the trial court's decision. The tax authorities have appealed to the Supreme Court of South Korea. In June 2020, another trial court ruled that PM Korea did not underpay approximately KRW 54 billion (approximately $48 million) of alleged underpayments. The government agencies appealed this decision. If the tax authorities and government agencies ultimately lose, then they would be required to return the paid amounts to PM Korea.
The Moscow Tax Inspectorate for Major Taxpayers (“MTI”) conducted an audit of AO Philip Morris Izhora (“PM Izhora”), our Russian subsidiary, for the 2015-2017 financial years. On July 26, 2019, MTI issued its initial assessment, claiming that intercompany sales of cigarettes between PM Izhora and another Russian subsidiary prior to excise tax increases and submission by PM Izhora of the maximum retail sales price notifications for cigarettes to the tax authorities were improper under Russian tax laws and resulted in underpayment of excise taxes and VAT. In August 2019, PM Izhora submitted its objections disagreeing with MTI’s allegations set forth in the initial assessment and MTI’s methodology for calculating the alleged underpayments. MTI accepted some of PM Izhora’s arguments and in September 2019, issued the final tax assessment claiming an underpayment of RUB 24.3 billion (approximately $374 million), including penalties and interest. In accordance with Russian tax laws, PM Izhora paid the entire amount of MTI’s final assessment. This amount was neither imposed on, nor concurrent with, the specific revenue-producing transaction, nor was it collected from customers of our Russian subsidiaries. In the third quarter of 2019, PMI recorded a pre-tax charge of $374 million, in marketing, administration and research costs in the condensed consolidated statements of earnings, representing $315 million net of an associated income tax benefit of $59 million.
The Saudi Arabia Customs General Authority issued its assessments requiring our distributors (one former and one current) to pay additional customs duties in an amount of approximately 1.5 billion Saudi Riyal, or approximately $396 million, in relation to the fees paid by these distributors under their agreements with our subsidiary for exclusive rights to distribute our products in Saudi Arabia during the period of 2014 through 2018. In order to challenge these assessments, the distributors posted bank guarantees equaling the amount of the above assessments. To enable the distributors' challenge, our subsidiary agreed with the banks to bear 80 percent of the amount the authority may draw on the bank guarantees. In September and October 2020, respectively, the distributors lost their challenges of the assessments; one of the distributors appealed, and the other may appeal by November 6, 2020. Our subsidiary and our distributors believe that customs duties paid in Saudi Arabia were in compliance with the applicable law and the WTO Customs Valuation Agreement.
A putative shareholder class action lawsuit, In re Philip Morris International Inc. Securities Litigation, is pending in the United States District Court for the Southern District of New York, purportedly on behalf of purchasers of Philip Morris International Inc. stock between July 26, 2016 and April 18, 2018. The lawsuit names Philip Morris International Inc. and certain officers
34
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
and employees as defendants and includes allegations that the defendants made false and/or misleading statements and/or failed to disclose information about PMI’s business, operations, financial condition, and prospects, related to product sales of, and alleged irregularities in clinical studies of, PMI’s Platform 1 product. The lawsuit seeks various forms of relief, including damages. In November 2018, the court consolidated three putative shareholder class action lawsuits with similar allegations previously filed in the Southern District of New York (namely, City of Westland Police and Fire Retirement System v. Philip Morris International Inc., et al, Greater Pennsylvania Carpenters’ Pension Fund v. Philip Morris International Inc., et al., and Gilchrist v. Philip Morris International Inc., et al.) into these proceedings. A putative shareholder class action lawsuit, Rubenstahl v. Philip Morris International Inc., et al., that had been previously filed in December 2017 in the United States District Court for the District of New Jersey, was voluntarily dismissed by the plaintiff due to similar allegations in these proceedings. On February 4, 2020, the court granted defendants’ motion in its entirety, dismissing all but one of the plaintiffs’ claims with prejudice. The court noted that one of plaintiffs’ claims (allegations relating to four non-clinical studies of PMI’s Platform 1 product) did not state a viable claim but allowed plaintiffs to replead that claim by March 3, 2020. On February 18, 2020, the plaintiffs filed a motion for reconsideration of the court's February 4th decision; this motion was denied on September 21, 2020. On September 28, 2020, plaintiffs filed an amended complaint seeking to replead allegations relating to four non-clinical studies of PMI's Platform 1 product. We believe that this lawsuit is without merit and will continue to defend it vigorously.
In April 2020, affiliates of British American Tobacco plc (“BAT”) commenced patent infringement proceedings, RAI Strategic Holdings, Inc., et al. v. Altria Client Services LLC, et al., in the federal court in the Eastern District of Virginia, where PMI's subsidiary, Philip Morris Products S.A., as well as Altria Group, Inc.'s subsidiaries, are defendants. Plaintiffs seek damages and injunctive relief against the commercialization of the Platform 1 products in the United States. In April 2020, BAT affiliates filed a complaint against PMI, Philip Morris Products S.A., Altria Group, Inc., and its subsidiaries before the International Trade Commission. Plaintiffs seek an order to prevent the importation of Platform 1 products into the United States. In June 2020, defendants filed their responses in both proceedings. The defendants also counterclaimed that BAT infringed their patents relating to certain e-vapor products, seeking damages for the commercialization of these products by BAT.
In April 2020, BAT’s affiliate commenced patent infringement proceedings, Nicoventures Trading Limited v. PM GmbH, et al., against PMI’s German subsidiary, Philip Morris GmbH, and Philip Morris Products S.A., in the Regional Court in Munich, Germany. Plaintiffs seek damages and injunctive relief against the commercialization of the Platform 1 products in Germany.
In July 2020, in response to a challenge in the United Kingdom by PMI’s subsidiary to patents related to the BAT patents in the German proceedings, BAT affiliates brought a patent infringement action, Nicoventures Trading Limited, et al. v. Philip Morris Products S.A., et al., against Philip Morris Products S.A. and PMI’s U.K. subsidiary, Philip Morris Limited, in the English High Court, seeking damages and injunctive relief against the commercialization of the Platform 1 products in the United Kingdom.
In September 2020, BAT’s affiliates commenced patent infringement and unfair competition proceedings, RAI Strategic Holdings, Inc., et al. v. Philip Morris Products S.A., et al., against Philip Morris Products S.A. and PMI’s Italian subsidiaries, Philip Morris Manufacturing & Technology Bologna S.p.A. and Philip Morris Italia S.R.L., in the Court of Milan, Italy. Plaintiffs seek damages, as well as injunctive relief against the manufacture in Italy of the Platform 1 heated tobacco units allegedly infringing the asserted patents and the commercialization of the Platform 1 products in Italy.
In October 2020, BAT’s affiliate commenced patent infringement proceedings, Nicoventures Trading Limited v. Philip Morris CR a.s., et al., against PMI's Czech subsidiary, Philip Morris CR a.s., and Philip Morris Products S.A., in the Town Court of Prague, Czech Republic, seeking preliminary injunctive relief against the commercialization of the Platform 1 products in the Czech Republic. In October 2020, the Court dismissed plaintiff’s request for preliminary injunction in its entirety. Plaintiff may appeal the decision.
In October 2020, BAT’s affiliate commenced patent infringement proceedings, RAI Strategic Holdings, Inc. v. Philip Morris Polska Distribution sp. z o.o., against PMI’s Polish subsidiary, Philip Morris Polska Distribution Sp. z o.o., in the Regional Court in Warsaw, IP Division. Plaintiff seeks preliminary injunctive relief against the commercialization of the Platform 1 products in Poland.
Other patent challenges by both parties are pending in various jurisdictions.
We believe that the foregoing proceedings by the affiliates of BAT are without merit and will defend them vigorously.
We are also involved in additional litigation arising in the ordinary course of our business. While the outcomes of these proceedings are uncertain, management does not expect that the ultimate outcomes of other litigation, including any reasonably
35
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
possible losses in excess of current accruals, will have a material adverse effect on our consolidated results of operations, cash flows or financial position.
Third-Party Guarantees
On October 17, 2020, Medicago Inc., an equity method investee of Philip Morris Investments B.V. (“PMIBV”), a PMI subsidiary, entered into a contribution agreement with the Canadian government (the “Contribution Agreement”) whereby the Canadian government agreed to contribute up to CAD 173 million (approximately $131 million on the date of signing) to Medicago Inc., to support its on-going COVID-19 vaccine development and clinical trials, and for the construction of its Quebec City manufacturing facility (the “Project”). PMIBV and the majority shareholder of Medicago Inc. are also parties to the Contribution Agreement as guarantors of Medicago Inc.’s obligations thereunder on a joint and several basis (“Co-Guarantors”). The Co-Guarantors agreed to repay amounts contributed by the Canadian government plus interest, if Medicago Inc. fails to do so, and could be responsible for the costs of other Medicago’s obligations (such as the achievement of specific milestones of the Project).
In connection with the Contribution Agreement, PMIBV and the majority shareholder of Medicago Inc. entered into a guarantors’ agreement that apportions Co-Guarantors’ obligations and limits those of PMIBV to its then share of holdings in Medicago Inc., which as of September 30, 2020 was approximately 32%.
The guarantees are in effect through March 31, 2026. PMI is currently assessing the impact of this transaction on its consolidated financial statements and at the inception of these guarantees does not expect the impact to be material.
Note 9. Income Taxes:
Income tax provisions for jurisdictions outside the United States of America, as well as state and local income tax provisions, were determined on a separate company basis, and the related assets and liabilities were recorded in PMI’s condensed consolidated balance sheets.
On March 27, 2020, the Coronavirus Aid, Relief, and Economic Security Act (“CARES Act”) was signed into law in the U.S. to provide certain relief as a result of the COVID-19 pandemic. In addition, governments around the world have enacted or implemented various forms of tax relief measures in response to the economic conditions in the wake of COVID-19. As of September 30, 2020, PMI has determined that neither the CARES Act nor changes to income tax laws or regulations in other jurisdictions had a significant impact on PMI’s effective tax rate, with the exception of the corporate income tax rate reduction in Indonesia.
On July 20, 2020, the U.S. Department of the Treasury and the Internal Revenue Service released final and proposed regulations under the Global Intangible Low-Taxed Income (“GILTI”) and other provisions of the Internal Revenue Code. PMI has analyzed these elective regulations and recorded the impact in its condensed consolidated financial statements, as described below.
PMI’s effective tax rates for the nine months and three months ended September 30, 2020 were 21.4% and 20.9%, respectively. PMI’s effective tax rates for the nine months and three months ended September 30, 2019 were 22.2% and 24.1%, respectively. The effective tax rate for the nine months ended September 30, 2020 was favorably impacted by a reduction of estimated U.S. federal and state income tax liabilities for years 2018 and 2019 mostly due to the GILTI regulations mentioned above ($93 million), a decrease in deferred tax liabilities related to the fair value adjustment of equity securities held by PMI (for further details, see Note 11. Fair Value Measurements) and a decrease in deferred tax liabilities related to the corporate income tax rate reduction in Indonesia, partially offset by a decrease in deductions related to foreign-derived intangible income for the years 2018 and 2019. The effective tax rate for the nine months ended September 30, 2019 was favorably impacted by the reversal of a deferred tax liability on the unremitted earnings of PMI's Canadian subsidiary, RBH ($49 million), a reduction of estimated U.S. federal income tax on dividend repatriation for the years 2015-2018 ($67 million) and by the Tax Cuts and Jobs Act. PMI estimates that its full-year 2020 effective tax rate will be 22% to 23%, excluding the discrete tax events mentioned above. Changes in currency exchange rates, earnings mix by taxing jurisdiction or future regulatory developments may have an
36
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
impact on the effective tax rates, which PMI monitors each quarter. Significant judgment is required in determining income tax provisions and in evaluating tax positions.
PMI is regularly examined by tax authorities around the world and is currently under examination in a number of jurisdictions. The U.S. federal statute of limitations remains open for the years 2015 and onward. Foreign and U.S. state jurisdictions have statutes of limitations generally ranging from to five years.
It is reasonably possible that within the next 12 months certain tax examinations will close, which could result in a change in unrecognized tax benefits along with related interest and penalties. An estimate of any possible change cannot be made at this time.
Note 10. Indebtedness:
Short-term Borrowings:
PMI's short-term borrowings, consisting of bank loans to certain PMI subsidiaries at September 30, 2020 and December 31, 2019, had a carrying value of $152 million and $338 million, respectively. The fair value of PMI’s short-term borrowings, based on current market interest rates, approximates carrying value.
Long-term Debt:
At September 30, 2020 and December 31, 2019, PMI’s long-term debt consisted of the following:
| (in millions) | September 30, 2020 | December 31, 2019 | ||||||||||||
U.S. dollar notes, 1.125% to 6.375% (average interest rate 3.342%), due through 2044 | $ | 19,732 | $ | 19,783 | ||||||||||
| Foreign currency obligations: | ||||||||||||||
Euro notes, 0.125% to 3.125% (average interest rate 1.983%), due through 2039 | 8,811 | 9,822 | ||||||||||||
Swiss franc notes, 1.625% to 2.000% (average interest rate 1.830%), due through 2024 | 597 | 899 | ||||||||||||
Other (average interest rate 3.019%), due through 2025 | 198 | 203 | ||||||||||||
| 29,338 | 30,707 | |||||||||||||
| Less current portion of long-term debt | 1,992 | 4,051 | ||||||||||||
| $ | 27,346 | $ | 26,656 | |||||||||||
Other foreign currency debt above includes mortgage debt in Switzerland and finance lease obligations at September 30, 2020 and December 31, 2019.
PMI's debt issuances in the first nine months of 2020 were as follows:
| (in millions) | ||||||||||||||||||||||||||
| Type | Face Value | Interest Rate | Issuance | Maturity | ||||||||||||||||||||||
| U.S. dollar notes | (a) | $750 | 1.125% | May 2020 | May 2023 | |||||||||||||||||||||
| U.S. dollar notes | (a) | $750 | 1.500% | May 2020 | May 2025 | |||||||||||||||||||||
| U.S. dollar notes | (a) | $750 | 2.100% | May 2020 | May 2030 | |||||||||||||||||||||
(a) Interest on these notes is payable semi-annually in arrears beginning in November 2020.
Credit Facilities:
On January 31, 2020, PMI entered into an agreement to amend and extend the term of its $2.0 billion 364-day revolving credit facility from February 4, 2020, to February 2, 2021.
37
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
On February 10, 2020, PMI entered into a new $2.0 billion multi-year revolving credit facility, expiring on February 10, 2025. The new credit facility replaced the $2.5 billion multi-year revolving credit facility, which was terminated effective February 10, 2020. PMI had no borrowings outstanding under the terminated facility, which was due to expire on February 28, 2021.
At September 30, 2020, PMI's total committed credit facilities were as follows:
(in billions)
Type | Committed Credit Facilities | |||||||
364-day revolving credit, expiring February 2, 2021 | $ | 2.0 | ||||||
Multi-year revolving credit, expiring October 1, 2022 | 3.5 | |||||||
Multi-year revolving credit, expiring February 10, 2025 | 2.0 | |||||||
Total facilities | $ | 7.5 | ||||||
At September 30, 2020, there were no borrowings under these committed credit facilities, and the entire committed amounts were available for borrowing.
Note 11. Fair Value Measurements:
The authoritative guidance defines fair value as the exchange price that would be received for an asset or paid to transfer a liability (an exit price) in the principal or most advantageous market for the asset or liability in an orderly transaction between market participants on the measurement date. The guidance also establishes a fair value hierarchy, which requires an entity to maximize the use of observable inputs and minimize the use of unobservable inputs when measuring fair value. The guidance describes three levels of input that may be used to measure fair value, which are as follows:
Level 1 -Quoted prices in active markets for identical assets or liabilities;
Level 2 -Observable inputs other than Level 1 prices, such as quoted prices for similar assets or liabilities; quoted prices in markets that are not active; or other inputs that are observable or can be corroborated by observable market data for substantially the full term of the assets or liabilities; and
Level 3 -Unobservable inputs that are supported by little or no market activity and that are significant to the fair value of the assets or liabilities.
Equity Securities
The fair value of PMI’s equity securities, which are determined by using quoted prices in active markets, have been classified within Level 1.
Derivative Financial Instruments
PMI assesses the fair value of its foreign exchange contracts and interest rate contracts using standard valuation models that use, as their basis, readily observable market inputs. The fair value of PMI’s foreign exchange forward contracts, foreign currency swaps and interest rate contracts is determined by using the prevailing foreign exchange spot rates and interest rate differentials, and the respective maturity dates of the instruments. The fair value of PMI’s currency options is determined by using a Black-Scholes methodology based on foreign exchange spot rates and interest rate differentials, currency volatilities and maturity dates. PMI’s derivative financial instruments have been classified within Level 2 in the table shown below. See Note 5. Financial Instruments for additional discussion of derivative financial instruments.
38
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Debt
The fair value of PMI’s outstanding debt, which is utilized solely for disclosure purposes, is determined using quotes and market interest rates currently available to PMI for issuances of debt with similar terms and remaining maturities. The aggregate carrying value of PMI’s debt, excluding short-term borrowings and $45 million of finance leases, was $29,293 million at September 30, 2020. The fair value of PMI’s outstanding debt, excluding the aforementioned short-term borrowings and finance leases, was classified within Level 1 and Level 2 in the table shown below.
The aggregate fair values of PMI's investments in equity securities, derivative financial instruments and PMI's debt as of September 30, 2020, were as follows:
| (in millions) | Fair Value at September 30, 2020 | Quoted Prices in Active Markets for Identical Assets/Liabilities (Level 1) | Significant Other Observable Inputs (Level 2) | Significant Unobservable Inputs (Level 3) | ||||||||||||||||||||||
| Assets: | ||||||||||||||||||||||||||
Equity securities (1) | $ | 244 | $ | 244 | $ | — | $ | — | ||||||||||||||||||
| Derivative contracts | 254 | — | 254 | — | ||||||||||||||||||||||
| Total assets | $ | 498 | $ | 244 | $ | 254 | $ | — | ||||||||||||||||||
| Liabilities: | ||||||||||||||||||||||||||
| Debt | $ | 32,934 | $ | 32,763 | $ | 171 | $ | — | ||||||||||||||||||
| Derivative contracts | 565 | — | 565 | — | ||||||||||||||||||||||
| Total liabilities | $ | 33,499 | $ | 32,763 | $ | 736 | $ | — | ||||||||||||||||||
(1) Unrealized pre-tax loss of $78 million ($62 million net of tax) on equity securities was recorded in the condensed consolidated statements of earnings for the nine months ended September 30, 2020. During the three months ended September 30, 2020, PMI did not record any unrealized gains or losses on equity securities in the condensed consolidated statements of earnings.
Note 12. Accumulated Other Comprehensive Losses:
PMI’s accumulated other comprehensive losses, net of taxes, consisted of the following:
| At | At | At | ||||||||||||||||||
| (in millions) | September 30, 2020 | December 31, 2019 | September 30, 2019 | |||||||||||||||||
| Currency translation adjustments | $ | (6,910) | $ | (5,537) | $ | (5,649) | ||||||||||||||
| Pension and other benefits | (3,606) | (3,829) | (3,306) | |||||||||||||||||
| Derivatives accounted for as hedges | (51) | 3 | (11) | |||||||||||||||||
| Total accumulated other comprehensive losses | $ | (10,567) | $ | (9,363) | $ | (8,966) | ||||||||||||||
Reclassifications from Other Comprehensive Earnings
The movements in accumulated other comprehensive losses and the related tax impact, for each of the components above, that are due to current period activity and reclassifications to the income statement, including those related to the deconsolidation of RBH, are shown on the condensed consolidated statements of comprehensive earnings for the nine months and three months ended September 30, 2020 and 2019. For additional information, see Note 3. Benefit Plans for disclosures related to PMI's pension and other benefits, Note 5. Financial Instruments for disclosures related to derivative financial instruments and Note 19. Deconsolidation of RBH for disclosures related to the deconsolidation of RBH.
39
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Note 13. Balance Sheet Offsetting:
Derivative Financial Instruments
PMI uses foreign exchange contracts and interest rate contracts to mitigate its exposure to changes in exchange and interest rates from third-party and intercompany actual and forecasted transactions. Substantially all of PMI's derivative financial instruments are subject to master netting arrangements, whereby the right to offset occurs in the event of default by a participating party. While these contracts contain the enforceable right to offset through close-out netting rights, PMI elects to present them on a gross basis in the condensed consolidated balance sheets. Collateral associated with these arrangements is in the form of cash and is unrestricted. See Note 5. Financial Instruments for disclosures related to PMI's derivative financial instruments.
The effects of these derivative financial instrument assets and liabilities on PMI's condensed consolidated balance sheets were as follows:
| (in millions) | Gross Amounts Recognized | Gross Amount Offset in the Condensed Consolidated Balance Sheet | Net Amounts Presented in the Condensed Consolidated Balance Sheet | Gross Amounts Not Offset in the Condensed Consolidated Balance Sheet | ||||||||||||||||
| Financial Instruments | Cash Collateral Received/Pledged | Net Amount | ||||||||||||||||||
| At September 30, 2020 | ||||||||||||||||||||
| Assets | ||||||||||||||||||||
| Derivative contracts | $ | 254 | $ | — | $ | 254 | $ | (185) | $ | (54) | $ | 15 | ||||||||
| Liabilities | ||||||||||||||||||||
| Derivative contracts | $ | 565 | $ | — | $ | 565 | $ | (185) | $ | (368) | $ | 12 | ||||||||
| At December 31, 2019 | ||||||||||||||||||||
| Assets | ||||||||||||||||||||
| Derivative contracts | $ | 390 | $ | — | $ | 390 | $ | (297) | $ | (91) | $ | 2 | ||||||||
| Liabilities | ||||||||||||||||||||
| Derivative contracts | $ | 419 | $ | — | $ | 419 | $ | (297) | $ | (59) | $ | 63 | ||||||||
Note 14. Related Parties - Investments in Unconsolidated Subsidiaries, Equity Securities and Other:
Investments in unconsolidated subsidiaries:
At September 30, 2020 and December 31, 2019, PMI had total investments in unconsolidated subsidiaries of $918 million and $1,053 million, respectively, which were accounted for under the equity method of accounting. Equity method investments are initially recorded at cost. Under the equity method of accounting, the investment is adjusted for PMI's proportionate share of earnings or losses, dividends, capital contributions, changes in ownership interests and movements in currency translation adjustments. The carrying value of our equity method investments at September 30, 2020 and December 31, 2019 exceeded our share of the unconsolidated subsidiaries' book value by $762 million and $901 million, respectively. The difference between the investment carrying value and the amount of underlying equity in net assets, excluding $733 million and $863 million attributable to goodwill as of September 30, 2020 and December 31, 2019, respectively, is being amortized on a straight-line basis over the underlying assets' estimated useful lives of 10 to 20 years. At September 30, 2020 and December 31, 2019, PMI received year-to-date dividends from unconsolidated subsidiaries of $53 million and $100 million, respectively.
PMI holds a 23% equity interest in Megapolis Distribution BV, the holding company of CJSC TK Megapolis, PMI's distributor in Russia (Eastern Europe segment).
40
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
PMI holds a 49% equity interest in United Arab Emirates-based Emirati Investors-TA (FZC) (“EITA”). PMI holds an approximate 25% economic interest in Société des Tabacs Algéro-Emiratie (“STAEM”), an Algerian joint venture that is 51% owned by EITA and 49% by the Algerian state-owned enterprise Management et Développement des Actifs et des Ressources Holding ("MADAR Holding"), which is part of the Middle East & Africa segment, manufactures and distributes under license some of PMI’s brands.
The initial investments in Megapolis Distribution BV and EITA were recorded at cost and are included in investments in unconsolidated subsidiaries and equity securities on the condensed consolidated balance sheets.
Equity securities:
Following the deconsolidation of RBH on March 22, 2019, PMI recorded the continuing investment in RBH, PMI's wholly owned subsidiary in Canada, at fair value of $3,280 million at the date of deconsolidation, within investments in unconsolidated subsidiaries and equity securities. For further details, see Note 19. Deconsolidation of RBH. Transactions between PMI and RBH are considered to be related party transactions from the date of deconsolidation and are included in the tables below.
Other related parties:
United Arab Emirates-based Trans-Emirates Trading and Investments (FZC) ("TTI") holds a 33% non-controlling interest in Philip Morris Misr LLC ("PMM"), an entity incorporated in Egypt which is consolidated in PMI’s financial statements in the Middle East & Africa segment. PMM sells, under license, PMI brands in Egypt through an exclusive distribution agreement with a local entity that is also controlled by TTI.
Godfrey Phillips India Ltd ("GPI") is one of the non-controlling interest holders in IPM India, which is a 56.3% owned PMI consolidated subsidiary in the South & Southeast Asia segment. GPI also acts as contract manufacturer and distributor for IPM India. Amounts in the tables below include transactions between these related parties.
Financial activity with the above related parties:
PMI’s net revenues and expenses with the above related parties were as follows:
| For the Nine Months Ended September 30, | For the Three Months Ended September 30, | ||||||||||||||||
| (in millions) | 2020 | 2019 | 2020 | 2019 | |||||||||||||
| Net revenues: | |||||||||||||||||
| Megapolis Group | $ | 1,609 | $ | 1,537 | $ | 583 | $ | 622 | |||||||||
| Other | 813 | 770 | 290 | 279 | |||||||||||||
Net revenues (a) | $ | 2,422 | $ | 2,307 | $ | 873 | $ | 901 | |||||||||
| Expenses: | |||||||||||||||||
| Other | $ | 38 | $ | 41 | $ | 11 | $ | 14 | |||||||||
| Expenses | $ | 38 | $ | 41 | $ | 11 | $ | 14 | |||||||||
(a) Net revenues exclude excise taxes and VAT billed to customers.
41
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
PMI’s balance sheet activity with the above related parties was as follows:
| (in millions) | At September 30, 2020 | At December 31, 2019 | |||||||||
| Receivables: | |||||||||||
| Megapolis Group | $ | 534 | $ | 375 | |||||||
| Other | 239 | 148 | |||||||||
| Receivables | $ | 773 | $ | 523 | |||||||
| Payables: | |||||||||||
| Other | $ | 19 | $ | 20 | |||||||
| Payables | $ | 19 | $ | 20 | |||||||
The activities with the above related parties are in the ordinary course of business, and are primarily for distribution, service fees, contract manufacturing and license agreements. PMI eliminated its respective share of all significant intercompany transactions with the equity method investees.
Note 15. Sale of Accounts Receivable:
To mitigate risk and enhance cash and liquidity management PMI sells trade receivables to unaffiliated financial institutions. These arrangements allow PMI to sell, on an ongoing basis, certain trade receivables without recourse. The trade receivables sold are generally short-term in nature and are removed from the condensed consolidated balance sheets. PMI sells trade receivables under two types of arrangements, servicing and non-servicing. For servicing arrangements, PMI continues to service the sold trade receivables on an administrative basis and does not act on behalf of the unaffiliated financial institutions. When applicable, a servicing liability is recorded for the estimated fair value of the servicing. The amounts associated with the servicing liability were not material as of September 30, 2020 and September 30, 2019. Under the non-servicing arrangements, PMI does not provide any administrative support or servicing after the trade receivables have been sold to the unaffiliated financial institutions.
Cumulative trade receivables sold, including excise taxes, for the nine months ended September 30, 2020 and 2019, were $8.4 billion and $7.8 billion, respectively. PMI’s operating cash flows were positively impacted by the amount of the trade receivables sold and derecognized from the condensed consolidated balance sheets, which remained outstanding with the unaffiliated financial institutions. The trade receivables sold that remained outstanding under these arrangements as of September 30, 2020 and September 30, 2019, were $0.6 billion, and $0.6 billion, respectively. The net proceeds received are included in cash provided by operating activities in the condensed consolidated statements of cash flows. The difference between the carrying amount of the trade receivables sold and the sum of the cash received is recorded as a loss on sale of trade receivables within marketing, administration and research costs in the condensed consolidated statements of earnings. For the nine months and three months ended September 30, 2020 and 2019, the loss on sale of trade receivables was immaterial.
42
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Note 16. Product Warranty:
PMI's IQOS devices are subject to standard product warranties generally for a period of 12 months from the date of purchase or such other periods as required by law. PMI generally provides in cost of sales for the estimated cost of warranty in the period the related revenue is recognized. PMI assesses the adequacy of its accrued product warranties and adjusts the amounts as necessary based on actual experience and changes in future estimates. Factors that affect product warranties may vary across markets but typically include device version mix, product failure rates, logistics and service delivery costs, and warranty policies. PMI accounts for its product warranties within other accrued liabilities. At September 30, 2020 and December 31, 2019, these amounts were as follows:
| (in millions) | At September 30, 2020 | At December 31, 2019 | |||||||||
| Balance at beginning of period | $ | 140 | $ | 67 | |||||||
| Changes due to: | |||||||||||
| Warranties issued | 204 | 303 | |||||||||
| Settlements | (189) | (230) | |||||||||
| Currency | 4 | — | |||||||||
| Balance at end of period | $ | 159 | $ | 140 | |||||||
Note 17. Leases:
The components of PMI’s lease cost were as follows for the nine months and three months ended September 30, 2020 and 2019:
| For the Nine Months Ended September 30, | For the Three Months Ended September 30, | ||||||||||||||||
| (in millions) | 2020 | 2019 | 2020 | 2019 | |||||||||||||
| Operating lease cost | $ | 177 | $ | 180 | $ | 60 | $ | 61 | |||||||||
| Short-term lease cost | 37 | 45 | 12 | 15 | |||||||||||||
| Variable lease cost | 23 | 20 | 8 | 10 | |||||||||||||
| Total lease cost | $ | 237 | $ | 245 | $ | 80 | $ | 86 | |||||||||
For the nine months ended September 30, 2020 and 2019, lease cost of $55 million and $59 million were recorded in cost of sales, respectively, and $182 million and $186 million were recorded in marketing, administration and research costs, respectively. For the three months ended September 30, 2020 and 2019, lease costs of $19 million and $21 million were recorded in cost of sales, respectively, and $61 million and $65 million were recorded in marketing, administration and research costs, respectively.
Note 18. Asset Impairment and Exit Costs:
Organizational Design Optimization
As part of PMI’s transformation to a smoke-free future, PMI seeks to optimize its organizational design, which includes the elimination, relocation and outsourcing of certain operations center and centralized activities. In January 2020, PMI commenced the first phase of a multi-phase restructuring project in Switzerland. PMI initiated the employee consultation procedure, as required under Swiss law, for the impacted employees in the first phase. This consultation was completed in April 2020; however, in light of the COVID-19 pandemic, PMI management temporarily suspended its restructuring plans. Additionally, in February 2020, PMI launched a voluntary separation program in Switzerland for certain eligible employees.
43
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
In June 2020, PMI resumed its restructuring activities in Switzerland and additionally, announced the outsourcing of certain activities in New York, U.S.A. These activities are expected to impact approximately 240 positions that will be either eliminated, relocated or outsourced. For the nine months ended September 30, 2020, PMI recorded pre-tax asset impairment and exit costs of $71 million. During the three months ended September 30, 2020, PMI did not record any pre-tax asset impairment and exit costs.
In August 2020, PMI commenced the second phase of a multi-phase restructuring project in Switzerland. This second phase is expected to impact approximately 200 existing positions that will be either eliminated or relocated. PMI initiated the employee consultation procedure, as required under Swiss law, for the impacted employees in the second phase. The third phase is expected to commence in the beginning of 2021. Until the consultation process for the respective phase is concluded, such phase is not considered probable (under U.S. GAAP), and the total potential costs cannot be determined. As a result, no related costs were recorded for the nine months ended September 30, 2020 related to the second and third phases of the restructuring project.
Additionally, the amounts related to the pension accounting impacts of the restructuring, which could be significant, have not been reflected in the third quarter of 2020 as the thresholds for accounting for any related pension curtailment or settlement were not exceeded by September 30, 2020.
Global Manufacturing Infrastructure Optimization
In light of declining PMI cigarette volumes resulting from lower total industry volumes and the shift to smoke-free alternatives, PMI continues to optimize its global manufacturing infrastructure. During 2019, PMI recorded asset impairment and exit costs related to plant closures in Argentina, Colombia, Germany and Pakistan as part of its global manufacturing infrastructure optimization.
For the nine months and three months ended September 30, 2019, PMI recorded pre-tax asset impairment and exit costs of $65 million and $22 million, respectively. For the nine months ended September 30, 2019, these costs were related to cigarette plant closures in Pakistan ($20 million) and Colombia ($45 million); and for the three months ended September 30, 2019, these costs were related to the plant closure in Colombia.
Asset Impairment and Exit Costs by Segment
PMI recorded the following pre-tax asset impairment and exit costs by segment:
44
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | |||||||||||||||
| 2020 | 2019 | 2020 | 2019 | ||||||||||||||
Separation programs: (1) | |||||||||||||||||
| European Union | $ | 24 | $ | — | $ | — | $ | — | |||||||||
| Eastern Europe | 6 | — | — | — | |||||||||||||
| Middle East & Africa | 8 | — | — | — | |||||||||||||
| South & Southeast Asia | 10 | 3 | — | — | |||||||||||||
| East Asia & Australia | 12 | — | — | — | |||||||||||||
| Latin America & Canada | 4 | 37 | — | 22 | |||||||||||||
| Total separation programs | 64 | 40 | — | 22 | |||||||||||||
Asset impairment charges: (1) | |||||||||||||||||
| European Union | 3 | — | — | — | |||||||||||||
| Eastern Europe | 1 | — | — | — | |||||||||||||
| Middle East & Africa | 1 | — | — | — | |||||||||||||
| South & Southeast Asia | 1 | 17 | — | — | |||||||||||||
| East Asia & Australia | 1 | — | — | — | |||||||||||||
| Latin America & Canada | — | 8 | — | — | |||||||||||||
| Total asset impairment charges | 7 | 25 | — | — | |||||||||||||
| Asset impairment and exit costs | $ | 71 | $ | 65 | $ | — | $ | 22 | |||||||||
(1) Organizational design optimization pre-tax charges were allocated across all operating segments.
The total pre-tax asset impairment and exit costs above were included in marketing, administration and research costs on the condensed consolidated statements of earnings.
Movement in Exit Cost Liabilities
The movement in exit cost liabilities for the nine months ended September 30, 2020 was as follows:
| (in millions) | |||||
| Liability balance, January 1, 2020 | $ | 191 | |||
| Charges, net | 64 | ||||
| Cash spent | (115) | ||||
| Currency/other | 4 | ||||
| Liability balance, September 30,2020 | $ | 144 | |||
Future cash payments for exit costs incurred to date are anticipated to be substantially paid by the end of 2021, with approximately $31 million expected to be paid in the remainder of 2020.
45
Philip Morris International Inc. and Subsidiaries
Notes to Condensed Consolidated Financial Statements
(Unaudited)
Note 19. Deconsolidation of RBH:
As discussed in Note 8. Contingencies, following the March 1, 2019 judgment of the Court of Appeal of Québec in two class action lawsuits against PMI's Canadian subsidiary, Rothmans, Benson & Hedges Inc. ("RBH"), PMI recorded in its consolidated results a pre-tax charge of $194 million, representing $142 million net of tax, in the first quarter of 2019. This pre-tax Canadian tobacco litigation-related expense was included in marketing, administration and research costs on PMI's condensed consolidated statement of earnings for the nine months ended September 30, 2019. The charge reflects PMI’s assessment of the portion of the judgment that represents probable and estimable loss prior to the deconsolidation of RBH and corresponds to the trust account deposit required by the judgment. RBH’s share of the deposit is approximately CAD 257 million.
On March 22, 2019, RBH obtained an initial order from the Ontario Superior Court of Justice granting it protection under the Companies’ Creditors Arrangement Act ("CCAA"), which is a Canadian federal law that permits a Canadian business to restructure its affairs while carrying on its business in the ordinary course with minimal disruption to its customers, suppliers and employees.
The administration of the CCAA process, principally relating to the powers provided to the court and the court appointed monitor, removes certain elements of control of the business from both PMI and RBH. As a result, PMI has determined that it no longer has a controlling financial interest over RBH as defined in ASC 810 (Consolidation), and PMI deconsolidated RBH as of the date of the CCAA filing. PMI has also determined that it does not exert "significant influence" over RBH as that term is defined in ASC 323 (Investments-Equity Method and Joint Ventures). Therefore, as of March 22, 2019, PMI accounted for its continuing investment in RBH in accordance with ASC 321 (Investments-Equity Securities) as an equity security, without readily determinable fair value.
Following the deconsolidation, the carrying value of assets and liabilities of RBH was removed from the consolidated balance sheet of PMI, and the continuing investment in RBH was recorded at fair value at the date of deconsolidation. The total amount deconsolidated from PMI’s balance sheet was $3,519 million, including $1,323 million of cash, $1,463 million of goodwill, $529 million of accumulated other comprehensive earnings, primarily related to historical currency translation and $204 million of other assets and liabilities, net. While PMI is accounting for its investment in RBH as an equity security, PMI would recognize dividends as income upon receipt. However, while it remains under creditor protection, RBH does not anticipate paying dividends.
The fair value of PMI’s continuing investment in RBH of $3,280 million was determined at the date of deconsolidation, recorded within Investments in unconsolidated subsidiaries and equity securities and is assessed for impairment on an ongoing basis. The estimated fair value of the underlying business was determined based on an income approach using a discounted cash flow analysis, as well as a market approach for certain contingent liabilities. The information used in the estimate includes observable inputs, primarily a discount rate of 8%, a terminal growth rate of 2.5% and information about total tobacco market size in Canada and RBH’s share of the market, as well as unobservable inputs such as operating budgets and strategic plans, various inflation scenarios, estimated shipment volumes, and expected product pricing and projected margins.
The difference between the carrying value of the assets and liabilities of RBH that were deconsolidated, and the fair value of the continuing investment, as determined at the date of deconsolidation, was $239 million, before tax, and this loss on deconsolidation is reflected within marketing, administration and research costs on PMI’s condensed consolidated statement of earnings for the nine months ended September 30, 2019. PMI also recorded a tax benefit of $49 million within the provision for income taxes for the nine months ended September 30, 2019, related to the reversal of a deferred tax liability on unremitted earnings of RBH.
RBH is party to transactions with PMI and its consolidated subsidiaries entered into in the normal course of business; these transactions include royalty payments and recharge of various corporate expenses for services benefiting RBH. Up to the date of the CCAA filing, these transactions were eliminated on consolidation and had no impact on PMI’s consolidated statement of earnings. After deconsolidating RBH, these transactions are treated as third-party transactions in PMI’s financial statements. The amount of these related party transactions is included within Note 14. Related Parties - Investments in Unconsolidated Subsidiaries, Equity Securities and Other.
Developments in the CCAA process, including resolution through a plan of arrangement or compromise of all pending tobacco-related litigation currently stayed in Canada, as discussed in Note 8. Contingencies, could result in a material change in the fair value of PMI’s continuing investment in RBH.
46
Item 2.
MANAGEMENT’S DISCUSSION AND ANALYSIS OF
FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Description of Our Company
We are leading a transformation in the tobacco industry to create a smoke-free future and ultimately replace cigarettes with smoke-free products to the benefit of adults who would otherwise continue to smoke, society, the company and its shareholders. We are a leading international tobacco company engaged in the manufacture and sale of cigarettes, as well as smoke-free products and associated electronic devices and accessories, and other nicotine-containing products in markets outside the United States. In addition, we ship a version of our Platform 1 device and its consumables to Altria Group, Inc. for sale under license in the United States, where the U.S. Food and Drug Administration ("FDA") has authorized their marketing as a modified risk tobacco product ("MRTP"), finding that an exposure modification order for these products is appropriate for the promotion of public health. We are building a future on a new category of smoke-free products that, while not risk-free, are a much better choice than continuing to smoke. Through multidisciplinary capabilities in product development, state-of-the-art facilities and scientific substantiation, we aim to ensure that our smoke-free products meet adult consumer preferences and rigorous regulatory requirements. Our IQOS smoke-free product brand portfolio includes heat-not-burn tobacco and nicotine-containing vapor products.
We manage our business in six operating segments:
•European Union ("EU");
•Eastern Europe ("EE");
•Middle East & Africa ("ME&A"), which includes our international duty free business;
•South & Southeast Asia ("S&SA");
•East Asia & Australia ("EA&A"); and
•Latin America & Canada ("LA&C"), which includes transactions under license with Altria Group, Inc. for the distribution of our Platform 1 product in the United States.
Our cigarettes are sold in more than 180 markets, and in many of these markets they hold the number one or number two market share position. We have a wide range of premium, mid-price and low-price brands. Our portfolio comprises both international and local brands. In addition to the manufacture and sale of cigarettes, we are engaged in the development and commercialization of reduced-risk products ("RRPs"). RRPs is the term we use to refer to products that present, are likely to present, or have the potential to present less risk of harm to smokers who switch to these products versus continuing smoking.
We use the term net revenues to refer to our operating revenues from the sale of our products, including shipping and handling charges billed to customers, net of sales and promotion incentives, and excise taxes. Our net revenues and operating income are affected by various factors, including the volume of products we sell, the price of our products, changes in currency exchange rates and the mix of products we sell. Mix is a term used to refer to the proportionate value of premium-price brands to mid-price or low-price brands in any given market (product mix). Mix can also refer to the proportion of shipment volume in more profitable markets versus shipment volume in less profitable markets (geographic mix).
Our cost of sales consists principally of: tobacco leaf, non-tobacco raw materials, labor and manufacturing costs; shipping and handling costs; and the cost of the IQOS devices produced by third-party electronics manufacturing service providers. Estimated costs associated with IQOS warranty programs are generally provided for in cost of sales in the period the related revenues are recognized.
Our marketing, administration and research costs include the costs of marketing and selling our products, other costs generally not related to the manufacture of our products (including general corporate expenses), and costs incurred to develop new products. The most significant components of our marketing, administration and research costs are marketing and sales expenses and general and administrative expenses.
Philip Morris International Inc. is a legal entity separate and distinct from its direct and indirect subsidiaries. Accordingly, our right, and thus the right of our creditors and stockholders, to participate in any distribution of the assets or earnings of any subsidiary is subject to the prior rights of creditors of such subsidiary, except to the extent that claims of our company itself as a creditor may be recognized. As a holding company, our principal sources of funds, including funds to make payment on our
47
debt securities, are from the receipt of dividends and repayment of debt from our subsidiaries. Our principal wholly owned and majority-owned subsidiaries currently are not limited by long-term debt or other agreements in their ability to pay cash dividends or to make other distributions with respect to their common stock that are otherwise compliant with law.
Executive Summary
The following executive summary provides the business update and significant highlights from the "Discussion and Analysis" that follows.
Consolidated Operating Results for the Nine Months Ended September 30, 2020
•Net Revenues - Net revenues of $21.3 billion for the nine months ended September 30, 2020 decreased by $842 million, or 3.8%, from the comparable 2019 amount, and were impacted by the effects of the COVID-19 pandemic, particularly in the second quarter of 2020. The change in our net revenues from the comparable 2019 amount was driven by the following (variances not to scale with year-to-date results):
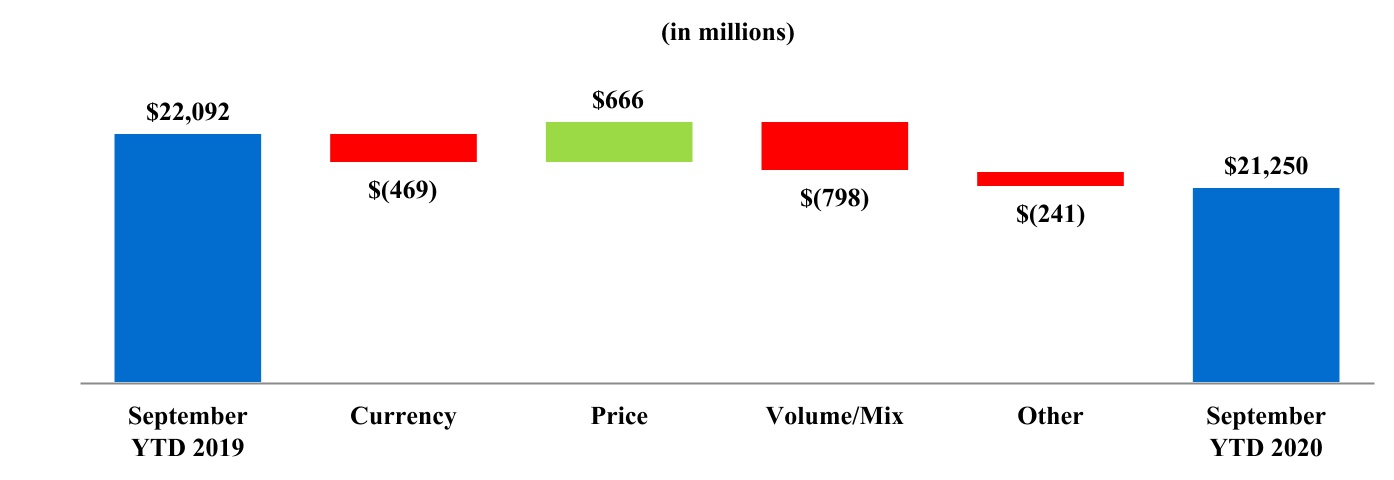
For the nine months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 1.7%, reflecting: unfavorable volume/mix, primarily due to lower cigarette volume (mainly in Argentina, Indonesia, Italy, Japan, Mexico, the Philippines, PMI Duty Free, Poland, Russia, Spain, Turkey and Ukraine, partly offset by Germany), partially offset by higher heated tobacco unit volume (notably in the EU, Japan, Russia and Ukraine, partly offset by PMI Duty Free); and the unfavorable impact of $241 million, shown in "Cost/Other," mainly resulting from the deconsolidation of RBH; partly offset by a favorable pricing variance (notably driven by Australia, the GCC, Germany, Japan, Mexico, the Philippines, PMI Duty Free and Russia, partially offset by Indonesia and Turkey). For further details on the deconsolidation of RBH, see Note 8. Contingencies and Note 19. Deconsolidation of RBH.
48
Net revenues by product category for the nine months ended September 30, 2020 and 2019 are shown below:

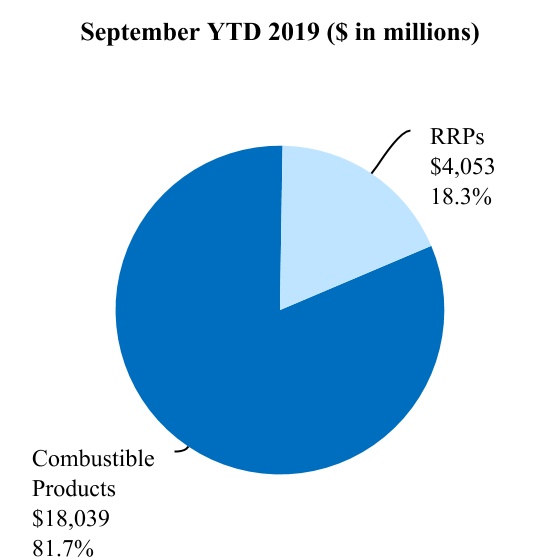
•Diluted Earnings Per Share - The changes in our reported diluted earnings per share (“diluted EPS”) for the nine months ended September 30, 2020, from the comparable 2019 amounts, were as follows:
| Diluted EPS | % Growth (Decline) | |||||||||||||
| For the nine months ended September 30, 2019 | $ | 3.57 | ||||||||||||
| 2019 Asset impairment and exit costs | 0.03 | |||||||||||||
| 2019 Canadian tobacco litigation-related expense | 0.09 | |||||||||||||
| 2019 Loss on deconsolidation of RBH | 0.12 | |||||||||||||
| 2019 Russia excise and VAT audit charge | 0.20 | |||||||||||||
| 2019 Tax items | (0.04) | |||||||||||||
| Subtotal of 2019 items | 0.40 | |||||||||||||
| 2020 Asset impairment and exit costs | (0.04) | |||||||||||||
| 2020 Fair value adjustment for equity security investments | (0.04) | |||||||||||||
| 2020 Tax items | 0.06 | |||||||||||||
| Subtotal of 2020 items | (0.02) | |||||||||||||
| Currency | (0.28) | |||||||||||||
| Interest | (0.01) | |||||||||||||
| Change in tax rate | — | |||||||||||||
| Operations | 0.24 | |||||||||||||
| For the nine months ended September 30, 2020 | $ | 3.90 | 9.2 | % | ||||||||||
Asset impairment and exit costs – We recorded pre-tax asset impairment and exit costs of $71 million (or $0.04 per share impact on diluted EPS) during the nine months ended September 30, 2020, related to the organizational design optimization plan primarily in Switzerland. We recorded pre-tax asset impairment and exit costs of $65 million (or $0.03 per share impact on diluted EPS) during the nine months ended September 30, 2019, related to cigarette plant closures in Colombia and Pakistan as part of the optimization of our global manufacturing footprint. The total pre-tax charges were included in marketing, administration and research costs on the condensed consolidated statements of earnings. For further details, see Note 18. Asset Impairment and Exit Costs.
Canadian tobacco litigation-related expense – In the first quarter of 2019, we recorded a pre-tax charge of $194 million, representing $142 million net of tax, relating to the judgment against our Canadian subsidiary, Rothmans, Benson & Hedges Inc. ("RBH"), in two Québec smoking and health class actions. The charge of $0.09 per share reflects our assessment of the portion of the judgment that represents probable and estimable loss prior to the deconsolidation of RBH and corresponds to the
49
trust account deposit required by the judgment. The total pre-tax charge was included in marketing, administration and research costs on the condensed consolidated statements of earnings and was included in the operating income of the Latin America & Canada segment. For further details, see Note 8. Contingencies and Note 19. Deconsolidation of RBH.
Loss on deconsolidation of RBH – Following the judgment in the two Québec smoking and health class actions, RBH obtained an initial order from the Ontario Superior Court of Justice granting it protection under the Companies’ Creditors Arrangement Act (“CCAA”), which is a Canadian federal law that permits a Canadian business to restructure its affairs while carrying on its business in the ordinary course with minimal disruption to its customers, suppliers and employees. The administration of the CCAA process, principally relating to the powers provided to the court and the court appointed monitor, removes certain elements of control of the business from both PMI and RBH. As a result, we have determined that we no longer have a controlling financial interest over RBH and that we do not exert "significant influence" over RBH under U.S. GAAP. Therefore, we deconsolidated RBH as of the date of the CCAA filing on March 22, 2019, and will account for our continuing investment in RBH as an equity security, without readily determinable fair value.
A loss on the deconsolidation of RBH of $239 million was included in marketing, administration and research costs on the condensed consolidated statements of earnings for the nine months ended September 30, 2019 and was included in the operating income of the Latin America & Canada segment. The $0.12 per share impact also included a tax benefit of $49 million within the provision for income taxes, as discussed above, related to the reversal of a deferred tax liability on the unremitted earnings of RBH. For further details, see Note 8. Contingencies and Note 19. Deconsolidation of RBH.
Russia excise and VAT audit charge – As a result of the final tax assessment for the 2015-2017 financial years received by our Russian affiliate, in the third quarter of 2019, PMI recorded a pre-tax charge of $374 million in marketing, administration and research costs in the condensed consolidated statements of earnings, representing $315 million net of income tax and a diluted EPS charge of $0.20. The pre-tax charge of $374 million was included in the operating income of the Eastern Europe segment. For further details, see Note 8. Contingencies.
Fair Value adjustment for equity security investments – During the nine months ended September 30, 2020, we recorded an unfavorable fair value adjustment for our equity security investments of $62 million after tax (or $0.04 per share decrease in diluted EPS). The fair value adjustment for our equity security investments was included in equity investments and securities (income)/loss, net ($78 million loss) and provision for income taxes ($16 million benefit) on the condensed consolidated statements of earnings. For further details, see Note 11. Fair Value Measurements.
Income taxes – The 2019 Tax items that increased our 2019 diluted EPS by $0.04 per share in the table above were primarily due to a reduction in estimated U.S. federal income tax on dividend repatriation for the years 2015 - 2018 ($67 million). The 2020 Tax items that increased our 2020 diluted EPS by $0.06 per share in the table above were due to final U.S. tax regulations under the Global Intangible Low-Taxed Income ("GILTI") provisions of the Internal Revenue Code for years 2018 and 2019 ($93 million). For further details, see Note 9. Income Taxes.
Currency – The unfavorable impact of $0.28 per share during the reporting period results from the fluctuations of the U.S. dollar, especially against the Brazilian real, Euro, Mexican pesos, Russian ruble, Swiss franc and Turkish lira. This unfavorable currency movement has impacted our profitability across our primary revenue markets and local currency cost bases.
Operations – The increase in diluted EPS of $0.24 from our operations in the table above was due primarily to the following segments:
•European Union: Favorable volume/mix, favorable pricing and lower manufacturing costs, partially offset by higher marketing, administration and research costs;
•East Asia & Australia: Lower marketing, administration and research costs, lower manufacturing costs and favorable pricing, partially offset by unfavorable volume/mix; and
•Eastern Europe: Favorable volume/mix and favorable pricing, partially offset by higher marketing, administration and research costs and higher manufacturing costs;
partially offset by
•Middle East & Africa: Unfavorable volume/mix and lower fees for certain distribution rights billed to customers in certain markets, partially offset by favorable pricing and lower marketing, administration and research costs;
•South & Southeast Asia: Unfavorable volume/mix, partially offset by favorable pricing and lower marketing, administration and research costs; and
50
•Latin America & Canada: Unfavorable volume/mix, as well as the unfavorable impact resulting from the deconsolidation of RBH, partially offset by favorable pricing and lower marketing, administration and research costs.
Consolidated Operating Results for the Three Months Ended September 30, 2020
•Net Revenues - Net revenues of $7.4 billion for the three months ended September 30, 2020 decreased by $0.2 billion, or 2.6%, from the comparable 2019 amount, and were impacted by the effects of the COVID-19 pandemic. The change in our net revenues from the comparable 2019 amount was driven by the following (variances not to scale with quarterly results):

During the quarter, net revenues, excluding unfavorable currency, decreased by 1.5%, mainly reflecting: unfavorable volume/mix, primarily due to lower cigarette volume (mainly in Indonesia, Japan and PMI Duty Free), partially offset by higher heated tobacco unit volume (notably in the EU, Japan, Russia and Ukraine, partly offset by PMI Duty Free); partially offset by a favorable pricing variance (notably driven by Germany, Japan, Russia and Saudi Arabia, partly offset by Indonesia and Turkey).
Net revenues by product category for the three months ended September 30, 2020 and 2019, are shown below:


51
•Diluted Earnings Per Share - The changes in our reported diluted EPS for the three months ended September 30, 2020, from the comparable 2019 amounts, were as follows:
| Diluted EPS | % Growth (Decline) | |||||||
| For the three months ended September 30, 2019 | $ | 1.22 | ||||||
| 2019 Asset impairment and exit costs | 0.01 | |||||||
| 2019 Russia excise and VAT audit charge | 0.20 | |||||||
| 2019 Tax items | — | |||||||
| Subtotal of 2019 items | 0.21 | |||||||
| 2020 Asset impairment and exit costs | — | |||||||
| 2020 Fair value adjustment for equity security investments | — | |||||||
| 2020 Tax items | 0.06 | |||||||
| Subtotal of 2020 items | 0.06 | |||||||
| Currency | (0.09) | |||||||
| Interest | (0.01) | |||||||
| Change in tax rate | (0.02) | |||||||
| Operations | 0.11 | |||||||
| For the three months ended September 30, 2020 | $ | 1.48 | 21.3 | % | ||||
Asset impairment and exit costs – We recorded pre-tax asset impairment and exit costs of $22 million (or $0.01 per share impact on diluted EPS) during the three months ended September 30, 2019, related to the cigarette plant closure in Colombia as a part of the optimization of our global manufacturing footprint. The total pre-tax charge was included in marketing, administration and research costs on the condensed consolidated statements of earnings. For further details, see Note 18. Asset Impairment and Exit Costs.
Russia excise and VAT audit charge – As a result of the final tax assessment for the 2015-2017 financial years received by our Russian affiliate, in the third quarter of 2019, PMI recorded a pre-tax charge of $374 million in marketing, administration and research costs in the condensed consolidated statements of earnings, representing $315 million net of income tax and a diluted EPS charge of $0.20. The pre-tax charge of $374 million was included in the operating income of the Eastern Europe segment. For further details, see Note 8. Contingencies.
Income Taxes – The 2020 Tax items that increased our 2020 diluted EPS by $0.06 per share in the table above were due to final U.S. tax regulations under the GILTI provisions of the Internal Revenue Code for years 2018 and 2019 ($93 million). The change in the tax rate that decreased our diluted EPS by $0.02 per share in the table above was primarily due to changes in earnings mix by taxing jurisdiction and an increase of estimated U.S. federal income tax liabilities for years 2018 and 2019 due to a decrease in deductions related to foreign-derived intangible income. For further details, see Note 9. Income Taxes.
Currency – The unfavorable impact of $0.09 per share in the third quarter results from the fluctuations of the U.S. dollar, especially against the Russian ruble and Swiss franc. This unfavorable currency movement has impacted our profitability across our primary revenue markets and local currency cost bases.
Operations – The increase in diluted EPS of $0.11 from our operations in the table above was due primarily to the following segments:
•European Union: Favorable volume/mix, favorable pricing, lower manufacturing costs and lower marketing, administration and research costs;
•East Asia & Australia: Favorable pricing, lower manufacturing costs and lower marketing, administration and research costs, partially offset by unfavorable volume/mix;
•Eastern Europe: Favorable pricing, lower manufacturing costs and favorable volume/mix, partially offset by higher marketing, administration and research costs; and
•Latin America & Canada: Lower marketing, administration and research costs and favorable pricing, partially offset by
52
unfavorable volume/mix;
partially offset by
•Middle East & Africa: Unfavorable volume/mix, partially offset by lower marketing, administration and research costs, lower manufacturing costs and favorable pricing; and
•South & Southeast Asia: Unfavorable pricing and unfavorable volume/mix, partially offset by lower marketing, administration and research costs.
For further details, see the “Consolidated Operating Results” and “Operating Results by Business Segment” sections of the following “Discussion and Analysis.”
COVID-19 Impact on Our Business
COVID-19: Business Continuity Update
Since the onset of the COVID-19 pandemic, PMI has undertaken a number of business continuity measures to mitigate potential disruption to its operations and route-to-market in order to preserve the availability of products to its customers and adult consumers.
Currently:
•PMI has sufficient access to the inputs for its products and is not facing any significant business continuity issues with respect to key suppliers;
•The large majority of PMI's manufacturing facilities globally are currently operational, including all heated tobacco unit factories. Certain cigarette production facilities—accounting for less than 5% of PMI's total cigarette production capacity worldwide—are temporarily impacted by government-mandated shutdowns or production limitations;
•There are adequate inventories, based on existing sales trends, of PMI finished goods across all key markets for cigarettes and across all IQOS markets for heated tobacco units and tobacco heating devices;
•PMI does not anticipate out-of-stock situations in any major operating income markets and generally expects consumers to have adequate access to its products; and
•PMI has sufficient liquidity resources through cash on hand, the ongoing cash generation of its business, and its access to the commercial paper and debt markets.
Nonetheless, significant uncertainty remains as the spread of the disease is increasing in a number of markets, resulting in additional restrictions and increasing risk of disruptions.
53
Discussion and Analysis
Consolidated Operating Results
See pages 92-97 for a discussion of our "Cautionary Factors That May Affect Future Results." Our net revenues and operating income by segment are shown in the table below:
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | ||||||||||||
| 2020 | 2019 | 2020 | 2019 | |||||||||||
| Net revenues: | ||||||||||||||
| European Union | $ | 7,960 | $ | 7,381 | $ | 2,950 | $ | 2,645 | ||||||
| Eastern Europe | 2,470 | 2,300 | 899 | 899 | ||||||||||
| Middle East & Africa | 2,348 | 3,058 | 768 | 1,127 | ||||||||||
| South & Southeast Asia | 3,211 | 3,607 | 1,071 | 1,246 | ||||||||||
| East Asia & Australia | 4,045 | 4,094 | 1,358 | 1,252 | ||||||||||
Latin America & Canada (1) | 1,216 | 1,652 | 400 | 473 | ||||||||||
| Net revenues | $ | 21,250 | $ | 22,092 | $ | 7,446 | $ | 7,642 | ||||||
| Operating income (loss): | ||||||||||||||
| European Union | $ | 3,924 | $ | 3,346 | $ | 1,588 | $ | 1,255 | ||||||
| Eastern Europe | 610 | 284 | 245 | (101) | ||||||||||
| Middle East & Africa | 819 | 1,304 | 261 | 519 | ||||||||||
| South & Southeast Asia | 1,290 | 1,471 | 402 | 539 | ||||||||||
| East Asia & Australia | 1,792 | 1,520 | 637 | 451 | ||||||||||
Latin America & Canada (1) | 328 | 100 | 110 | 125 | ||||||||||
| Operating income | $ | 8,763 | $ | 8,025 | $ | 3,243 | $ | 2,788 | ||||||
(1) As of March 22, 2019, PMI deconsolidated the financial results of its Canadian subsidiary, Rothmans, Benson & Hedges Inc. ("RBH") from PMI's financial statements. For further details, see Note 19. Deconsolidation of RBH.
Items affecting the comparability of results from operations were as follows:
•Russia excise and VAT audit charge - See Note 8. Contingencies for details of the $374 million pre-tax charge included in the Eastern Europe segment for the nine months and three months ended September 30, 2019.
•Canadian tobacco litigation-related expense - See Note 8. Contingencies and Note 19. Deconsolidation of RBH for details of the $194 million pre-tax charge included in the Latin America & Canada segment for the nine months ended September 30, 2019.
•Loss on deconsolidation of RBH - See Note 19. Deconsolidation of RBH for details of the $239 million loss included in the Latin America & Canada segment for the nine months ended September 30, 2019.
•Asset impairment and exit costs - See Note 18. Asset Impairment and Exit Costs for a breakdown of these costs by segment for the nine months and three months ended September 30, 2020 and 2019.
54
Our net revenues by product category are shown in the table below:
| PMI Net Revenues by Product Category | ||||||||||||||||||||
| (in millions) | For the Nine Months Ended September 30, | For the Three Months Ended September 30, | ||||||||||||||||||
| 2020 | 2019 | Change | 2020 | 2019 | Change | |||||||||||||||
| Combustible Products | ||||||||||||||||||||
| European Union | $ | 6,099 | $ | 6,139 | (0.7) | % | $ | 2,244 | $ | 2,178 | 3.0 | % | ||||||||
| Eastern Europe | 1,681 | 1,774 | (5.3) | % | 636 | 664 | (4.2) | % | ||||||||||||
| Middle East & Africa | 2,296 | 2,810 | (18.3) | % | 768 | 1,064 | (27.8) | % | ||||||||||||
| South & Southeast Asia | 3,211 | 3,607 | (11.0) | % | 1,071 | 1,246 | (14.1) | % | ||||||||||||
| East Asia & Australia | 1,876 | 2,074 | (9.5) | % | 605 | 680 | (11.0) | % | ||||||||||||
| Latin America & Canada | 1,196 | 1,634 | (26.8) | % | 393 | 466 | (15.6) | % | ||||||||||||
| Total Combustible Products | $ | 16,360 | $ | 18,039 | (9.3) | % | $ | 5,716 | $ | 6,298 | (9.2) | % | ||||||||
| Reduced-Risk Products | ||||||||||||||||||||
| European Union | $ | 1,861 | $ | 1,242 | 49.9 | % | $ | 706 | $ | 467 | 51.3 | % | ||||||||
| Eastern Europe | 789 | 526 | 50.1 | % | 263 | 235 | 11.9 | % | ||||||||||||
| Middle East & Africa | 52 | 248 | (79.2) | % | — | 63 | -(100)% | |||||||||||||
| South & Southeast Asia | — | — | — | % | — | — | — | % | ||||||||||||
| East Asia & Australia | 2,169 | 2,020 | 7.3 | % | 753 | 572 | 31.6 | % | ||||||||||||
| Latin America & Canada | 20 | 18 | 12.0 | % | 7 | 7 | (6.1) | % | ||||||||||||
| Total Reduced-Risk Products | $ | 4,890 | $ | 4,053 | 20.6 | % | $ | 1,730 | $ | 1,344 | 28.6 | % | ||||||||
| Total PMI Net Revenues | $ | 21,250 | $ | 22,092 | (3.8) | % | $ | 7,446 | $ | 7,642 | (2.6) | % | ||||||||
Note: Sum of product categories or Regions might not foot to total PMI due to roundings.
Net revenues related to combustible products refer to the operating revenues generated from the sale of these products, including shipping and handling charges billed to customers, net of sales and promotion incentives, and excise taxes. These net revenue amounts consist of the sale of our cigarettes and other tobacco products combined. Other tobacco products primarily include roll-your-own and make-your-own cigarettes, pipe tobacco, cigars and cigarillos and do not include reduced-risk products.
Net revenues related to reduced-risk products refer to the operating revenues generated from the sale of these products, including shipping and handling charges billed to customers, net of sales and promotion incentives, and excise taxes. These net revenue amounts consist of the sale of our heated tobacco units, IQOS devices and related accessories, and other nicotine-containing products, which primarily include our e-vapor products.
We recognize revenue when control is transferred to the customer, typically either upon shipment or delivery of goods.
Revenues from shipments of Platform 1 devices, heated tobacco units and accessories to Altria Group, Inc., commencing in the third quarter of 2019, for sale under license in the United States are included in Net Revenues of the Latin America & Canada segment.
References to "Cost/Other" in the Consolidated Financial Summary table of total PMI and the six operating segments throughout this "Discussion and Analysis" reflects the currency-neutral variances of: cost of sales (excluding the volume/mix cost component); marketing, administration and research costs (including asset impairment and exit costs, the Canadian tobacco litigation-related expense, the charge related to the deconsolidation of RBH in Canada, and the Russia excise and VAT audit charge); and amortization of intangibles. “Cost/Other” also includes the currency-neutral net revenue variance, unrelated to volume/mix and price components, attributable to fees for certain distribution rights billed to customers in certain markets in the ME&A Region, as well as the impact of the deconsolidation in RBH.
55
Our shipment volume by segment for cigarettes and heated tobacco units is shown in the table below:
| PMI Shipment Volume (Million Units) | ||||||||||||||||||||
| For the Nine Months Ended September 30, | For the Three Months Ended September 30, | |||||||||||||||||||
| 2020 | 2019 | Change | 2020 | 2019 | Change | |||||||||||||||
| Cigarettes | ||||||||||||||||||||
| European Union | 126,142 | 133,093 | (5.2) | % | 45,179 | 47,238 | (4.4) | % | ||||||||||||
| Eastern Europe | 70,737 | 74,779 | (5.4) | % | 25,661 | 27,379 | (6.3) | % | ||||||||||||
| Middle East & Africa | 88,087 | 101,957 | (13.6) | % | 30,903 | 36,994 | (16.5) | % | ||||||||||||
| South & Southeast Asia | 108,179 | 130,230 | (16.9) | % | 37,238 | 42,362 | (12.1) | % | ||||||||||||
| East Asia & Australia | 35,154 | 38,650 | (9.0) | % | 10,784 | 12,692 | (15.0) | % | ||||||||||||
| Latin America & Canada | 45,542 | 52,906 | (13.9) | % | 15,699 | 16,854 | (6.9) | % | ||||||||||||
| Total Cigarettes | 473,841 | 531,615 | (10.9) | % | 165,464 | 183,519 | (9.8) | % | ||||||||||||
| Heated Tobacco Units | ||||||||||||||||||||
| European Union | 14,069 | 8,810 | 59.7 | % | 5,181 | 3,474 | 49.1 | % | ||||||||||||
| Eastern Europe | 14,374 | 8,213 | 75.0 | % | 4,882 | 3,858 | 26.5 | % | ||||||||||||
| Middle East & Africa | 834 | 2,061 | (59.5) | % | 179 | 588 | (69.6) | % | ||||||||||||
| South & Southeast Asia | 10 | — | — | % | 10 | — | — | % | ||||||||||||
| East Asia & Australia | 24,799 | 23,253 | 6.6 | % | 8,601 | 7,976 | 7.8 | % | ||||||||||||
Latin America & Canada (1) | 316 | 202 | 56.4 | % | 114 | 89 | 28.1 | % | ||||||||||||
| Total Heated Tobacco Units | 54,402 | 42,539 | 27.9 | % | 18,967 | 15,985 | 18.7 | % | ||||||||||||
| Cigarettes and Heated Tobacco Units | ||||||||||||||||||||
| European Union | 140,211 | 141,903 | (1.2) | % | 50,360 | 50,712 | (0.7) | % | ||||||||||||
| Eastern Europe | 85,111 | 82,992 | 2.6 | % | 30,543 | 31,237 | (2.2) | % | ||||||||||||
| Middle East & Africa | 88,921 | 104,018 | (14.5) | % | 31,082 | 37,582 | (17.3) | % | ||||||||||||
| South & Southeast Asia | 108,189 | 130,230 | (16.9) | % | 37,248 | 42,362 | (12.1) | % | ||||||||||||
| East Asia & Australia | 59,953 | 61,903 | (3.2) | % | 19,385 | 20,668 | (6.2) | % | ||||||||||||
| Latin America & Canada | 45,858 | 53,108 | (13.7) | % | 15,813 | 16,943 | (6.7) | % | ||||||||||||
| Total Cigarettes and Heated Tobacco Units | 528,243 | 574,154 | (8.0) | % | 184,431 | 199,504 | (7.6) | % | ||||||||||||
(1) Includes shipments to Altria Group, Inc., commencing in the third quarter of 2019, for sale in the United States under license.
Following the deconsolidation of our Canadian subsidiary, we will continue to report the volume of brands sold by RBH for which other PMI subsidiaries are the trademark owners. These include HEETS, Next, Philip Morris and Rooftop.
Heated tobacco units ("HTU") is the term we use to refer to heated tobacco consumables, which for us include our HEETS, HEETS Creations, HEETS Dimensions, HEETS Marlboro and HEETS FROM MARLBORO (defined collectively as HEETS), Marlboro HeatSticks and Parliament HeatSticks, as well as the KT&G-licensed brand, Fiit (outside of Korea).
Market share for HTUs is defined as the total sales volume for HTUs as a percentage of the total estimated sales volume for cigarettes and HTUs.
Shipment volume of heated tobacco units to the United States is included in the heated tobacco unit shipment volume of the Latin America & Canada segment.
References to total international market, defined as worldwide cigarette and heated tobacco unit volume excluding the United States, total industry, total market and market shares throughout this "Discussion and Analysis" are our estimates for tax-paid products based on the latest available data from a number of internal and external sources and may, in defined instances,
56
exclude the People's Republic of China and/or our duty free business. In addition, to reflect the deconsolidation of RBH, effective March 22, 2019, PMI's total market share has been restated for previous periods.
2020 third-quarter and nine months year-to-date estimates for total industry volume and market share in certain geographies reflect limitations on the availability and accuracy of industry data during pandemic-related restrictions.
In-market sales ("IMS") is defined as sales to the retail channel, depending on the market and distribution model.
North Africa is defined as Algeria, Egypt, Libya, Morocco and Tunisia.
The Gulf Cooperation Council ("GCC") is defined as Bahrain, Kuwait, Oman, Qatar, Saudi Arabia and the United Arab Emirates (UAE).
Unless otherwise stated, references to total industry, total market, our shipment volume and our market share performance reflect cigarettes and heated tobacco units.
From time to time, PMI’s shipment volumes are subject to the impact of distributor inventory movements, and estimated total industry/market volumes are subject to the impact of inventory movements in various trade channels that include estimated trade inventory movements of PMI’s competitors arising from market-specific factors that significantly distort reported volume disclosures. Such factors may include changes to the manufacturing supply chain, shipment methods, consumer demand, timing of excise tax increases or other influences that may affect the timing of sales to customers. In such instances, in addition to reviewing PMI shipment volumes and certain estimated total industry/market volumes on a reported basis, management reviews these measures on an adjusted basis that excludes the impact of distributor and/or estimated trade inventory movements. Management also believes that disclosing PMI shipment volumes and estimated total industry/market volumes in such circumstances on a basis that excludes the impact of distributor and/or estimated trade inventory movements, such as on an IMS basis, improves the comparability of performance and trends for these measures over different reporting periods.
57
Key market data regarding total market size, our shipments and market share are shown in the tables below:
| For the Nine Months Ended September 30, | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PMI Shipments (billion units) | PMI Market Share (%)(1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Market | Total Market (billion units) | Total | Cigarette | Heated Tobacco Unit | Total | Heated Tobacco Unit | ||||||||||||||||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | 2020 | 2019 | 2020 | 2019 | 2020 | 2019 | 2020 | 2019 | 2020 | 2019 | |||||||||||||||||||||||||||||||||||||||||||||
| Total | 1,892.9 | 2,018.7 | 528.2 | 574.2 | 473.8 | 531.6 | 54.4 | 42.5 | 27.9 | 28.4 | 2.9 | 2.1 | ||||||||||||||||||||||||||||||||||||||||||||
| European Union | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| France | 28.0 | 29.1 | 12.7 | 13.1 | 12.5 | 13.1 | 0.1 | 0.1 | 44.9 | 44.9 | 0.5 | 0.2 | ||||||||||||||||||||||||||||||||||||||||||||
| Germany | 56.4 | 55.1 | 22.0 | 20.8 | 20.8 | 20.2 | 1.2 | 0.6 | 38.9 | 37.7 | 2.1 | 1.1 | ||||||||||||||||||||||||||||||||||||||||||||
| Italy | 50.8 | 51.1 | 26.7 | 26.6 | 22.8 | 24.1 | 4.0 | 2.5 | 52.0 | 51.6 | 7.6 | 4.3 | ||||||||||||||||||||||||||||||||||||||||||||
| Poland | 35.0 | 35.4 | 13.6 | 14.5 | 12.1 | 13.8 | 1.6 | 0.7 | 38.9 | 41.1 | 4.5 | 2.1 | ||||||||||||||||||||||||||||||||||||||||||||
| Spain | 31.7 | 34.4 | 10.1 | 11.3 | 9.8 | 11.1 | 0.3 | 0.2 | 31.5 | 31.5 | 1.0 | 0.7 | ||||||||||||||||||||||||||||||||||||||||||||
| Eastern Europe | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Russia | 163.6 | 168.1 | 51.6 | 48.8 | 42.3 | 43.4 | 9.3 | 5.3 | 32.3 | 29.7 | 6.0 | 3.3 | ||||||||||||||||||||||||||||||||||||||||||||
| Middle East & Africa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Saudi Arabia | 15.8 | 16.1 | 6.2 | 6.7 | 6.2 | 6.7 | — | — | 38.5 | 40.5 | 0.2 | — | ||||||||||||||||||||||||||||||||||||||||||||
| Turkey | 86.6 | 93.4 | 35.3 | 40.6 | 35.3 | 40.6 | — | — | 40.7 | 43.4 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| South & Southeast Asia | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Indonesia | 201.7 | 222.7 | 58.3 | 72.1 | 58.3 | 72.1 | — | — | 28.9 | 32.4 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| Philippines | 47.0 | 52.7 | 32.1 | 37.2 | 32.1 | 37.2 | — | — | 68.4 | 70.7 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| East Asia & Australia | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Australia | 8.3 | 9.2 | 2.5 | 2.6 | 2.5 | 2.6 | — | — | 29.6 | 27.6 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| Japan | 111.3 | 120.1 | 38.8 | 40.5 | 17.7 | 20.9 | 21.1 | 19.5 | 36.7 | 34.3 | 20.0 | 16.9 | ||||||||||||||||||||||||||||||||||||||||||||
| Korea | 54.8 | 51.7 | 11.3 | 11.8 | 7.8 | 8.2 | 3.5 | 3.6 | 20.7 | 22.9 | 6.4 | 6.9 | ||||||||||||||||||||||||||||||||||||||||||||
| Latin America & Canada | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Argentina | 24.1 | 24.7 | 14.9 | 17.6 | 14.9 | 17.6 | — | — | 61.9 | 71.2 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| Mexico | 21.9 | 25.1 | 13.6 | 16.6 | 13.6 | 16.6 | — | — | 62.0 | 65.9 | 0.2 | — | ||||||||||||||||||||||||||||||||||||||||||||
| (1) Market share estimates are calculated using IMS data | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: % change for Total Market and PMI shipments is computed based on millions of units; PMI Market Share estimates for previous periods are restated to reflect RBH deconsolidation and exclude RBH-owned brands. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
58
| For the Three Months Ended September 30, | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PMI Shipments (billion units) | PMI Market Share (%)(1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Market | Total Market (billion units) | Total | Cigarette | Heated Tobacco Unit | Total | Heated Tobacco Unit | ||||||||||||||||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | 2020 | 2019 | 2020 | 2019 | 2020 | 2019 | 2020 | 2019 | 2020 | 2019 | |||||||||||||||||||||||||||||||||||||||||||||
| Total | 669.8 | 693.5 | 184.4 | 199.5 | 165.5 | 183.5 | 19.0 | 16.0 | 28.2 | 28.9 | 3.1 | 2.3 | ||||||||||||||||||||||||||||||||||||||||||||
| European Union | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| France | 9.8 | 10.2 | 4.2 | 4.5 | 4.1 | 4.5 | — | — | 45.3 | 45.0 | 0.5 | 0.2 | ||||||||||||||||||||||||||||||||||||||||||||
| Germany | 20.4 | 20.7 | 7.4 | 7.4 | 7.0 | 7.2 | 0.4 | 0.2 | 36.4 | 35.7 | 1.9 | 1.1 | ||||||||||||||||||||||||||||||||||||||||||||
| Italy | 18.8 | 18.3 | 9.7 | 9.5 | 8.2 | 8.5 | 1.5 | 1.1 | 52.1 | 52.0 | 7.8 | 4.6 | ||||||||||||||||||||||||||||||||||||||||||||
| Poland | 13.5 | 12.5 | 5.1 | 5.3 | 4.5 | 5.0 | 0.6 | 0.3 | 37.8 | 42.5 | 4.7 | 2.4 | ||||||||||||||||||||||||||||||||||||||||||||
| Spain | 11.7 | 12.6 | 3.7 | 3.8 | 3.6 | 3.8 | 0.1 | 0.1 | 32.2 | 31.6 | 0.9 | 0.7 | ||||||||||||||||||||||||||||||||||||||||||||
| Eastern Europe | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Russia | 60.0 | 61.8 | 18.6 | 18.9 | 15.5 | 16.2 | 3.1 | 2.7 | 31.7 | 30.7 | 5.8 | 4.0 | ||||||||||||||||||||||||||||||||||||||||||||
| Middle East & Africa | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Saudi Arabia | 5.5 | 5.5 | 2.4 | 2.0 | 2.4 | 2.0 | — | — | 36.9 | 40.8 | 0.4 | — | ||||||||||||||||||||||||||||||||||||||||||||
| Turkey | 32.3 | 31.8 | 13.5 | 14.2 | 13.5 | 14.2 | — | — | 41.7 | 44.5 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| South & Southeast Asia | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Indonesia | 70.2 | 77.5 | 19.8 | 25.0 | 19.8 | 25.0 | — | — | 28.2 | 32.3 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| Philippines | 17.5 | 17.3 | 11.7 | 12.4 | 11.7 | 12.4 | — | — | 66.8 | 71.4 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| East Asia & Australia | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Australia | 3.2 | 3.2 | 1.0 | 0.9 | 1.0 | 0.9 | — | — | 29.8 | 27.7 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| Japan | 40.7 | 41.9 | 11.9 | 13.3 | 4.6 | 6.5 | 7.3 | 6.8 | 36.9 | 34.5 | 20.5 | 17.1 | ||||||||||||||||||||||||||||||||||||||||||||
| Korea | 20.2 | 18.4 | 3.9 | 4.1 | 2.7 | 2.9 | 1.2 | 1.1 | 19.5 | 22.2 | 6.0 | 6.2 | ||||||||||||||||||||||||||||||||||||||||||||
| Latin America & Canada | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Argentina | 8.4 | 8.4 | 4.9 | 5.9 | 4.9 | 5.9 | — | — | 58.1 | 69.6 | — | — | ||||||||||||||||||||||||||||||||||||||||||||
| Mexico | 7.7 | 7.7 | 4.8 | 4.9 | 4.8 | 4.9 | — | — | 62.7 | 63.4 | 0.2 | — | ||||||||||||||||||||||||||||||||||||||||||||
| (1) Market share estimates are calculated using IMS data | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Note: % change for Total Market and PMI shipments is computed based on millions of units; PMI Market Share estimates for previous periods are restated to reflect RBH deconsolidation and exclude RBH-owned brands. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Consolidated Operating Results for the Nine Months Ended September 30, 2020
The following discussion compares our consolidated operating results for the nine months ended September 30, 2020, with the nine months ended September 30, 2019.
Our total shipment volume decreased by 8.0%, due to:
•the EU, reflecting lower cigarette shipment volume, notably in Italy, Poland and Spain, partly offset by higher heated tobacco unit shipment volume across the Region, particularly in Italy and Poland;
•Middle East & Africa, reflecting lower cigarette shipment volume, primarily in PMI Duty Free and Turkey, as well as lower heated tobacco unit shipment volume due to PMI Duty Free;
•South & Southeast Asia, reflecting lower cigarette shipment volume, primarily in Indonesia, Pakistan and the Philippines;
•East Asia & Australia, reflecting lower cigarette shipment volume, predominantly in Japan, partly offset by higher heated tobacco unit shipment volume driven by Japan; and
59
•Latin America & Canada, reflecting lower cigarette shipment volume, primarily in Argentina, Canada (due to the impact of the deconsolidation of RBH), and Mexico, partially offset by Brazil. Excluding the volume impact from the RBH deconsolidation, our total shipment volume in the Region decreased by 12.0%;
partly offset by
•Eastern Europe, reflecting higher heated tobacco unit shipment volume across the Region, notably in Russia and Ukraine, partly offset by lower cigarette shipment volume, mainly in Russia and Ukraine.
Excluding the volume impact from the RBH deconsolidation of approximately 1.0 billion units (reflecting first quarter 2019 volume of RBH-owned brands and including Duty-Free sales of these brands in Canada), PMI's total shipment volume decreased by 7.8%.
Impact of Inventory Movements
The net impact of estimated distributor inventory movements was immaterial. Excluding the volume impact from the deconsolidation of RBH, our total in-market sales declined by 7.7%.
Our cigarette shipment volume by brand and heated tobacco units shipment volume are shown in the table below:
| PMI Shipment Volume by Brand (Million Units) | |||||||||||
| Nine Months Year-to-Date | |||||||||||
| 2020 | 2019 | Change | |||||||||
| Cigarettes | |||||||||||
| Marlboro | 175,638 | 196,883 | (10.8) | % | |||||||
| L&M | 69,215 | 69,765 | (0.8) | % | |||||||
| Chesterfield | 39,274 | 43,502 | (9.7) | % | |||||||
| Philip Morris | 34,823 | 36,949 | (5.8) | % | |||||||
| Parliament | 25,575 | 29,085 | (12.1) | % | |||||||
| Sampoerna A | 23,801 | 26,012 | (8.5) | % | |||||||
| Bond Street | 18,481 | 21,099 | (12.4) | % | |||||||
| Dji Sam Soe | 18,344 | 23,089 | (20.6) | % | |||||||
| Lark | 12,059 | 15,575 | (22.6) | % | |||||||
| Fortune | 7,008 | 9,702 | (27.8) | % | |||||||
| Others | 49,623 | 59,954 | (17.2) | % | |||||||
| Total Cigarettes | 473,841 | 531,615 | (10.9) | % | |||||||
Heated Tobacco Units (1) | 54,402 | 42,539 | 27.9 | % | |||||||
| Total Cigarettes and Heated Tobacco Units | 528,243 | 574,154 | (8.0) | % | |||||||
(1) Includes shipments to Altria Group, Inc., commencing in the third quarter of 2019, for sale in the United States under license.
Note: Sampoerna A includes Sampoerna; Philip Morris includes Philip Morris/Dubliss; and Lark includes Lark Harmony.
Our cigarette shipment volume of the following brands decreased:
•Marlboro, mainly due to Indonesia, Japan, Mexico, the Philippines, PMI Duty Free and Turkey, partly offset by Russia;
•L&M, notably due to PMI Duty Free and Poland, partly offset by Mexico and Turkey;
•Chesterfield, mainly due to Poland, Russia and Turkey, partly offset by Brazil and Saudi Arabia;
•Philip Morris, primarily due to Argentina and Italy, partly offset by Japan, the Philippines and Russia;
•Parliament, mainly due to PMI Duty Free, Russia and Turkey;
•Sampoerna A in Indonesia, mainly due to premium A Mild;
•Bond Street, largely due to Russia and Ukraine;
60
•Dji Sam Soe in Indonesia, mainly due to Dji Sam Soe Magnum Mild;
•Lark, primarily due to Japan and Turkey;
•Fortune in the Philippines; and
•"Others," notably due to: the impact of the deconsolidation of RBH in Canada; mid-price Hope in the Philippines, Muratti in Turkey and Sampoerna U in Indonesia; and low-price Baronet (morphed to L&M) in Mexico, Jackpot in the Philippines and Morven in Pakistan; partly offset by mid-price Sampoerna Hijau in Indonesia.
The increase in our heated tobacco unit shipment volume was mainly driven by the EU (notably Italy and Poland), Eastern Europe (notably Russia and Ukraine) and Japan, partly offset by PMI Duty Free.
International Share of Market (excluding China and the United States)
Our total international market share (excluding China and the United States), defined as our cigarette and heated tobacco unit sales volume as a percentage of total industry cigarette and heated tobacco unit sales volume, decreased by 0.5 points to 27.9%, reflecting:
•Total international market share for cigarettes of 25.0%, down by 1.2 points; and
•Total international market share for heated tobacco units of 2.9%, up by 0.8 points.
Our total international cigarette sales volume as a percentage of total industry cigarette sales volume was down by 1.1 points to 25.9%, mainly reflecting: out-switching to heated tobacco units, as well as lower cigarette market share and/or an unfavorable geographic mix impact, notably in Indonesia, Mexico, Pakistan, the Philippines, PMI Duty Free and Turkey, partly offset by Germany and Russia.
| Financial Summary | ||||||||||||||||||||||||||||||||||||||
| Financial Summary - Nine Months Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other(1) | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 21,250 | $ | 22,092 | (3.8) | % | (1.7) | % | $ | (842) | $ | (469) | $ | 666 | $ | (798) | $ | (241) | ||||||||||||||||||||
| Cost of Sales | (6,997) | (7,735) | 9.5 | % | 7.3 | % | 738 | 173 | — | 318 | 247 | |||||||||||||||||||||||||||
Marketing, Administration and Research Costs (2) | (5,435) | (6,282) | 13.5 | % | 15.6 | % | 847 | (131) | — | — | 978 | |||||||||||||||||||||||||||
| Amortization of Intangibles | (55) | (50) | (10.0) | % | (10.0) | % | (5) | — | — | — | (5) | |||||||||||||||||||||||||||
| Operating Income | $ | 8,763 | $ | 8,025 | 9.2 | % | 14.5 | % | $ | 738 | $ | (427) | $ | 666 | $ | (480) | $ | 979 | ||||||||||||||||||||
(1) Cost/Other variance includes the impact of the RBH deconsolidation.
(2) Favorable Cost/Other variance includes the 2019 Russia excise and VAT audit charge of $374 million, the 2019 Canadian tobacco litigation-related expense of $194 million, the 2019 loss on deconsolidation of RBH of $239 million, the 2019 asset impairment and exit costs of $65 million and the 2020 asset impairment and exit costs of ($71 million), as well as the impact of the RBH deconsolidation.
Note: Net Revenues include revenues from shipments of the Platform 1 device, heated tobacco units and accessories to Altria Group, Inc., commencing in the third quarter of 2019, for sale under license in the United States.
For the nine months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 1.7%, reflecting: unfavorable volume/mix, primarily due to lower cigarette volume (mainly in Argentina, Indonesia, Italy, Japan, Mexico, the Philippines, PMI Duty Free, Poland, Russia, Spain, Turkey and Ukraine, partly offset by Germany), partially offset by higher heated tobacco unit volume (notably in the EU, Japan, Russia and Ukraine, partly offset by PMI Duty Free); and the unfavorable impact of $241 million, shown in "Cost/Other," mainly resulting from the deconsolidation of RBH; partly offset by a favorable pricing variance (notably driven by Australia, the GCC, Germany, Japan, Mexico, the Philippines, PMI Duty Free and Russia, partially offset by Indonesia and Turkey).
The unfavorable currency in net revenues was due primarily to the Brazilian real, Euro, Indonesian rupiah, Mexican pesos, Russian ruble and Turkish lira, partially offset by the Philippine peso.
61
Net revenues include $4,890 million in 2020 and $4,053 million in 2019 related to the sale of RRPs. IQOS devices accounted for approximately 8% of RRP net revenues for the nine months ended September 30, 2020, mainly due to a naturally lower ratio of new users to existing users, longer replacement cycles and geographic mix, particularly in the third quarter.
Operating income, excluding unfavorable currency, increased by 14.5%, notably reflecting a favorable comparison, shown in "Cost/Other," of charges recorded September year-to-date 2020 of $71 million, related to asset impairment and exit costs associated with organizational design optimization, to charges recorded in the same period in 2019 of $872 million, related to: asset impairment and exit costs ($65 million) associated with plant closures in Colombia and Pakistan, the loss on the deconsolidation of RBH ($239 million), the Canadian tobacco litigation-related expense ($194 million), and the Russia excise and VAT audit charge ($374 million).
Excluding these 2019 and 2020 charges, and unfavorable currency of $427 million, operating income increased by 4.1%, primarily reflecting: a favorable pricing variance; lower manufacturing costs (driven by productivity gains related to reduced-risk and combustible products) and lower marketing, administration and research costs (partly driven by cost efficiencies); partially offset by unfavorable volume/mix, mainly due to lower cigarette volume (primarily in Argentina, Indonesia, Italy, Japan, Mexico, the Philippines, PMI Duty Free, Poland and Spain, partly offset by Germany), partly offset by higher heated tobacco unit volume (notably in the EU, Japan, Russia and Ukraine, partly offset by PMI Duty Free); and the unfavorable impact of the deconsolidation of RBH, included in "Cost/Other."
Interest expense, net, of $454 million increased by $20 million (4.6%).
Our effective tax rate decreased by 0.8 percentage points to 21.4%. The effective tax rate for the nine months ended September 30, 2020 was favorably impacted by a reduction of estimated U.S. federal and state income tax liabilities for years 2018 and 2019 mostly due to final regulations under the GILTI provisions of the Internal Revenue Code ($93 million), a decrease in deferred tax liabilities related to the fair value adjustment of equity securities held by PMI and a decrease in deferred tax liabilities related to the corporate income tax rate reduction in Indonesia, partially offset by a decrease in deductions related to foreign-derived intangible income for the years 2018 and 2019. The effective tax rate for the nine months ended September 30, 2019, was favorably impacted by the reversal of a deferred tax liability on the unremitted earnings of our Canadian subsidiary, RBH ($49 million), a reduction of estimated U.S. federal income tax on dividend repatriation for the years 2015-2018 ($67 million) and by the Tax Cuts and Jobs Act. We estimate that our full-year 2020 effective tax rate will be 22% to 23%, excluding the discrete tax events mentioned above. Changes in currency exchange rates, earnings mix by taxing jurisdiction or future regulatory developments may have an impact on the effective tax rates, which we monitor each quarter. Significant judgment is required in determining income tax provisions and in evaluating tax positions. For further details, see Note 9. Income Taxes.
We are regularly examined by tax authorities around the world, and we are currently under examination in a number of jurisdictions. It is reasonably possible that within the next 12 months certain tax examinations will close, which could result in a change in unrecognized tax benefits along with related interest and penalties. An estimate of any possible change cannot be made at this time.
Net earnings attributable to PMI of $6.1 billion increased by $511 million or 9.2%. This increase was due primarily to higher operating income as discussed above. Diluted and basic EPS of $3.90 increased by 9.2%. Excluding an unfavorable currency impact of $0.28, diluted EPS increased by 17.1%.
Consolidated Operating Results for the Three Months Ended September 30, 2020
The following discussion compares our consolidated operating results for the three months ended September 30, 2020, with the three months ended September 30, 2019.
Our total shipment volume decreased by 7.6%, due to:
•the EU, reflecting lower cigarette shipment volume, notably in France, Italy and Poland, partly offset by Austria, partially offset by higher heated tobacco unit shipment volume across the Region, notably in Italy and Poland;
•Eastern Europe, reflecting lower cigarette shipment volume, particularly in Russia and Ukraine, partly offset by higher heated tobacco unit shipment volume across the Region, primarily in Russia and Ukraine;
62
•Middle East & Africa, reflecting lower cigarette shipment volume, mainly in North Africa, PMI Duty Free and Turkey, as well as lower heated tobacco unit shipment volume due to PMI Duty Free;
•South & Southeast Asia, reflecting lower cigarette shipment volume, primarily in Indonesia and the Philippines, partly offset by Pakistan and Thailand;
•East Asia & Australia, reflecting lower cigarette shipment volume, mainly in Japan, partly offset by higher heated tobacco unit shipment volume, primarily in Japan; and
•Latin America & Canada, reflecting lower cigarette shipment volume, primarily in Argentina, partly offset by Brazil.
Impact of Inventory Movements
Excluding the net unfavorable impact of estimated distributor inventory movements of approximately 3.4 billion units, our total in-market sales declined by 5.8%, due to a 8.8% decline in cigarettes, partly offset by a 28.5% increase in heated tobacco units.
The net unfavorable impact of estimated distributor inventory movements of approximately 3.4 billion units reflected a net unfavorable impact of 1.8 billion cigarettes, mainly due to Japan and PMI Duty Free, partly offset by Saudi Arabia, and a net unfavorable impact of 1.6 billion heated tobacco units, primarily due to Japan and Russia.
Our cigarette shipment volume by brand and heated tobacco units shipment volume are shown in the table below:
| PMI Shipment Volume by Brand (Million Units) | |||||||||||
| Third-Quarter | |||||||||||
| 2020 | 2019 | Change | |||||||||
| Cigarettes | |||||||||||
| Marlboro | 61,581 | 68,859 | (10.6) | % | |||||||
| L&M | 24,189 | 24,428 | (1.0) | % | |||||||
| Chesterfield | 13,768 | 15,001 | (8.2) | % | |||||||
| Philip Morris | 12,254 | 13,275 | (7.7) | % | |||||||
| Parliament | 9,540 | 10,407 | (8.3) | % | |||||||
| Sampoerna A | 7,999 | 8,756 | (8.6) | % | |||||||
| Bond Street | 6,441 | 7,687 | (16.2) | % | |||||||
| Dji Sam Soe | 6,372 | 8,599 | (25.9) | % | |||||||
| Lark | 3,846 | 4,955 | (22.4) | % | |||||||
| Fortune | 2,263 | 3,215 | (29.6) | % | |||||||
| Others | 17,211 | 18,337 | (6.1) | % | |||||||
| Total Cigarettes | 165,464 | 183,519 | (9.8) | % | |||||||
Heated Tobacco Units (1) | 18,967 | 15,985 | 18.7 | % | |||||||
| Total Cigarettes and Heated Tobacco Units | 184,431 | 199,504 | (7.6) | % | |||||||
(1) Includes shipments to Altria Group, Inc., commencing in the third quarter of 2019, for sale in the United States under license.
Note: Sampoerna A includes Sampoerna; Philip Morris includes Philip Morris/Dubliss; and Lark includes Lark Harmony.
Our cigarette shipment volume of the following brands decreased:
•Marlboro, mainly due to Indonesia, Japan and PMI Duty Free, partly offset by the Philippines and Russia;
•L&M, notably due to Egypt, Germany, PMI Duty Free and Poland, partly offset by Mexico, Thailand and Turkey;
•Chesterfield, mainly due to Poland, Russia and Turkey, partly offset by Brazil and Saudi Arabia;
•Philip Morris, primarily due to Argentina and Italy, partly offset by the Philippines and Russia;
•Parliament, mainly due to Japan, Korea, Kuwait, PMI Duty Free and Russia;
•Sampoerna A in Indonesia, mainly due to premium A Mild;
63
•Bond Street, notably due to Russia;
•Dji Sam Soe in Indonesia, mainly due to Dji Sam Soe Magnum Mild;
•Lark, primarily due to Japan;
•Fortune in the Philippines; and
•"Others," notably due to: mid-price Sampoerna U in Indonesia, partly offset by mid-price Sampoerna Hijau in Indonesia and low-price Morven in Pakistan.
The increase in our heated tobacco unit shipment volume was mainly driven by the EU (notably Italy and Poland), Eastern Europe (notably Russia and Ukraine) and Japan, partly offset by PMI Duty Free.
International Share of Market (excluding China and the United States)
Our total international market share (excluding China and the United States), decreased by 0.7 point to 28.2%, reflecting:
•Total international market share for cigarettes of 25.1%, down by 1.5 points; and
•Total international market share for heated tobacco units of 3.1%, up by 0.8 points.
Our total international cigarette sales volume as a percentage of total industry cigarette sales volume was down by 1.3 points to 26.1%, mainly reflecting: out-switching to heated tobacco units, as well as lower cigarette market share and/or an unfavorable geographic mix impact, notably in Argentina, Indonesia, PMI Duty Free and Ukraine, partly offset by Brazil and Pakistan.
| Financial Summary | ||||||||||||||||||||||||||||||||||||||
| Financial Summary - Quarters Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 7,446 | $ | 7,642 | (2.6) | % | (1.5) | % | $ | (196) | $ | (78) | $ | 171 | $ | (275) | $ | (14) | ||||||||||||||||||||
| Cost of Sales | (2,416) | (2,605) | 7.3 | % | 6.8 | % | 189 | 13 | — | 50 | 126 | |||||||||||||||||||||||||||
Marketing, Administration and Research Costs(1) | (1,769) | (2,234) | 20.8 | % | 23.5 | % | 465 | (61) | — | — | 526 | |||||||||||||||||||||||||||
| Amortization of Intangibles | (18) | (15) | (20.0) | % | (13.3) | % | (3) | (1) | — | — | (2) | |||||||||||||||||||||||||||
| Operating Income | $ | 3,243 | $ | 2,788 | 16.3 | % | 20.9 | % | $ | 455 | $ | (127) | $ | 171 | $ | (225) | $ | 636 | ||||||||||||||||||||
(1) Favorable Cost/Other variance includes the 2019 Russia excise and VAT audit charge of $374 million and the 2019 asset impairment and exit costs of $22 million.
Note: Net Revenues include revenues from shipments of Platform 1 devices, heated tobacco units and accessories to Altria Group, Inc., commencing in the third quarter of 2019, for sale under license in the United States.
For the three months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 1.5%, mainly reflecting: unfavorable volume/mix, primarily due to lower cigarette volume (mainly in Indonesia, Japan and PMI Duty Free), partially offset by higher heated tobacco unit volume (notably in the EU, Japan, Russia and Ukraine, partly offset by PMI Duty Free); partially offset by a favorable pricing variance (notably driven by Germany, Japan, Russia and Saudi Arabia, partly offset by Indonesia and Turkey).
The unfavorable currency in net revenues was due primarily to the Mexican pesos, Russian ruble and Turkish lira, partially offset by the Euro and Japanese yen.
Net revenues include $1,730 million in 2020 and $1,344 million in 2019 related to the sale of RRPs.
Operating income, excluding unfavorable currency, increased by 20.9%, notably reflecting a favorable comparison, shown in "Cost/Other," due to charges recorded in the in the third quarter of 2019 of $396 million, related to: asset impairment and exit
64
costs ($22 million) associated with a plant closure in Colombia and the Russia excise and VAT audit ($374 million).
Excluding these charges and unfavorable currency of $127 million, operating income increased by 5.8%, primarily reflecting: a favorable pricing variance; lower manufacturing costs (driven by productivity gains related to reduced-risk and combustible products); and lower marketing, administration and research costs (partly driven by cost efficiencies); partially offset by unfavorable volume/mix, due to the same factors as for net revenues noted above.
Interest expense, net, of $163 million increased by $31 million (23.5%).
Our effective tax rate decreased by 3.2 percentage points to 20.9%. The effective tax rate for the three months ended September 30, 2020 was favorably impacted by a reduction of estimated U.S. federal and state income tax liabilities for years 2018 and 2019 mostly due to final regulations under the GILTI provisions of the Internal Revenue Code ($93 million), partially offset by a decrease in deductions related to foreign-derived intangible income for the years 2018 and 2019. For further details, see Note 9. Income Taxes.
Net earnings attributable to PMI of $2.3 billion increased by $411 million or 21.7%. This increase was due primarily to higher operating income as discussed above. Diluted and basic EPS of $1.48 increased by 21.3%. Excluding an unfavorable currency impact of $0.09, diluted EPS increased by 28.7%.
Operating Results by Business Segment
Business Environment
Taxes, Legislation, Regulation and Other Matters Regarding the Manufacture, Marketing, Sale and Use of Tobacco Products
The tobacco industry and our company face a number of challenges that may adversely affect our business, volume, results of operations, cash flows and financial position. These challenges, which are discussed below and in “Cautionary Factors That May Affect Future Results,” include:
•regulatory restrictions on our products, including restrictions on the packaging, marketing, and sale of tobacco or other nicotine-containing products that could reduce our competitiveness, eliminate our ability to communicate with adult consumers, or even ban certain of our products;
•fiscal challenges, such as excessive excise tax increases and discriminatory tax structures;
•illicit trade in cigarettes and other tobacco products, including counterfeit, contraband and so-called “illicit whites”
•intense competition, including from non-tax paid volume by certain local manufacturers;
•pending and threatened litigation as discussed in Note 8. Contingencies; and
•governmental investigations.
Regulatory Restrictions: The tobacco industry operates in a highly regulated environment. The well-known risks of smoking have led regulators to impose significant restrictions and high excise taxes on cigarettes.
Much of the regulation that shapes the business environment in which we operate is driven by the World Health Organization's (“WHO”) Framework Convention on Tobacco Control (“FCTC”), which entered into force in 2005. The FCTC has as its main objective to establish a global agenda for tobacco regulation, with the purpose of reducing tobacco use. To date, 181 countries and the European Union are Parties to the FCTC. The treaty requires Parties to have in place various tobacco control measures and recommends others. The FCTC governing body, the Conference of the Parties (“CoP”), has also adopted non-binding guidelines and policy recommendations related to certain articles of the FCTC that go beyond the text of the treaty. In October 2018, the CoP recognized the need for more scientific assessment and improved reporting to define policy on heated tobacco products. Similar to its previous policy recommendations on e-cigarettes, the CoP invited countries to regulate, restrict or prohibit heated tobacco products, as appropriate under their national laws.
In July 2019, the WHO issued the Report on the Global Tobacco Epidemic 2019. While citing insufficient independent studies regarding the benefits and the unknown long-term health impacts of electronic nicotine delivery systems and heated tobacco products, the WHO has taken the position that such products are not risk-free and should be regulated in the same manner as
65
cigarettes and in line with the FCTC provisions. It is not possible to predict whether or to what extent measures recommended by the WHO, including the FCTC guidelines, will be implemented.
We believe that when better alternatives to cigarettes exist, the discussion should not be whether these alternatives should be made available to the more than one billion men and women who smoke today, but how fast, and within what regulatory framework to maximize their adoption while minimizing unintended use. Therefore, we advocate for regulatory frameworks that recognize a significant difference on a risk continuum between combustible tobacco on the one hand and non-combustible tobacco and other nicotine-containing products on the other. Regulation should include measures that will accelerate switching to non-combustible products, for example, by allowing adult consumers who would not otherwise quit to receive truthful and non-misleading information about such products to enable them to make informed decisions and by applying uniform product standards to enable manufacturers to demonstrate the safety of these products, as well as the absence of combustion. Regulation should also include specific rules for ingredients, labeling and consumer communication, and should ensure that the public is informed about the health risks of all combustible and non-combustible tobacco and nicotine-containing products. Importantly, regulation must include measures designed to prevent initiation by youth and non-smokers. We support mandated health warnings, minimum age laws, restrictions on advertising, and public place smoking restrictions. We also support regulatory measures that help reduce illicit trade.
Certain measures are discussed in more detail below and in the Reduced-Risk Products (RRPs) section.
Fiscal Challenges: Excessive and disruptive excise, sales and other tax increases and discriminatory tax structures are expected to continue to have an adverse impact on our profitability, due to lower consumption and consumer down-trading to non-premium, discount, other low-price or low-taxed combustible tobacco products such as fine cut tobacco and illicit cigarettes. In addition, in certain jurisdictions, some of our combustible products are subject to tax structures that discriminate against premium-price products and manufactured cigarettes. We believe that such tax policies undermine public health by encouraging consumers to turn to illicit trade, and ultimately undercut government revenue objectives, disrupt the competitive environment, and encourage criminal activity. Other jurisdictions have imposed, or are seeking to impose, levies or other taxes specifically on tobacco companies, such as taxes on revenues and/or profits.
EU Tobacco Products Directive: In April 2014, the EU adopted a significantly revised EU Tobacco Products Directive (TPD), which entered into force in May 2016. All Member States have adopted laws transposing the TPD. The TPD sets forth a comprehensive set of regulatory requirements for tobacco products, including:
•health warnings covering 65% of the front and back panels of cigarette packs, with an option for Member States to further standardize tobacco packaging, including the introduction of plain packaging;
•a ban on characterizing flavors in some tobacco products, with a transition period for menthol that expired in May 2020;
•security features and tracking and tracing measures that became effective on May 20, 2019; and
•a framework for the regulation of novel tobacco products and e-cigarettes, including requirements for health warnings and information leaflets, a prohibition on product packaging text related to reduced risk, and the introduction of notification requirements or authorization procedures in advance of commercialization.
The EU Commission’s Directorate General for Health and Food Safety is preparing a report on the implementation of the TPD, including the evaluation of whether the TPD has achieved its objectives and is still relevant considering scientific, international and technical developments, including in novel tobacco products and e-cigarettes. The report is expected to include recommendations on potential revisions of the TPD to account for such developments. The report is due by May 2021.
EU Tobacco Excise Directive: In addition, the EU Commission is preparing a legislative proposal for the revision of the 2011 EU Tobacco Excise Directive that would include definitions and tax treatment of novel tobacco products and e-cigarettes. The proposal is expected to be finalized by the end of 2021.
Plain Packaging and Other Packaging Restrictions: Plain packaging legislation bans the use of branding, logos and colors on packaging other than the brand name and variant that may be printed only in specified locations and in a uniform font. To date, plain packaging laws have been adopted in certain markets in all of our operating segments, including the key markets of Australia, France, Saudi Arabia and Turkey. Some countries, such as Canada, New Zealand and Israel, adopted plain packaging regulations that apply to all tobacco products, including RRPs. Other countries are also considering plain packaging legislation.
66
Some countries have adopted, or are considering adopting, packaging restrictions that could have an impact similar to plain packaging. Examples of such restrictions include standardizing the shape and size of packages, prohibiting certain colors or the use of certain descriptive phrases on packaging, and requiring very large graphic health warnings that leave little space for branding.
Restrictions and Bans on the Use of Ingredients: The WHO and others in the public health community have recommended restrictions or total bans on the use of some or all ingredients in tobacco products, including menthol. Broad restrictions and ingredient bans would require us to reformulate our American blend tobacco products and could reduce our ability to differentiate these products in the market in the long term. In many countries, menthol bans would eliminate the entire category of mentholated tobacco products. The European Union banned cigarettes and roll-your-own tobacco products with characterizing flavors. Other tobacco products, including heated tobacco products, are exempted from this flavor ban. The EU Commission is required to withdraw this exemption for a particular product category if it determines that there is a substantial change of circumstances, such as a significant increase of EU-wide sales volumes in such product category. It is not possible to predict how or when this determination would be made. Other countries may follow the EU’s approach. For instance, Turkey banned menthol as of May 2020. Broader ingredient bans have been adopted by Canada and Brazil. In Brazil, an ingredient ban is currently on appeal by a tobacco industry union, of which our Brazilian subsidiary is a member. It is not possible to predict the outcome of these legal proceedings.
Bans on Display of Tobacco Products at Retail: In a number of our markets, including, but not limited to, Australia and Russia, governments have banned the display of tobacco products at the point of sale. Other countries are considering similar bans.
Bans and Restrictions on Advertising, Marketing, Promotions and Sponsorships: For many years, the FCTC has called for, and countries have imposed, partial or total bans on tobacco advertising, marketing, promotions and sponsorships, including bans and restrictions on advertising on radio and television, in print and on the Internet. The FCTC's non-binding guidelines recommend that governments prohibit all forms of communication with adult smokers.
Restrictions on Product Design: Some members of the public health community are calling for the further standardization of tobacco products by requiring, for example, that cigarettes have a certain minimum diameter, which would amount to a ban on slim cigarettes, or requiring the use of standardized filter and cigarette paper designs. In addition, at its meeting in November 2016, the CoP adopted non-binding guidelines recommending that countries regulate product design features that increase the attractiveness of tobacco products, such as the diameter of cigarettes and the use of flavor capsules.
Restrictions on Public Smoking: The pace and scope of public smoking restrictions have increased significantly in most of our markets. Many countries around the world have adopted, or are likely to adopt, regulations that restrict or ban smoking in public and/or work places, restaurants, bars and nightclubs. Some public health groups have called for, and some countries, regional governments and municipalities have adopted or proposed, bans on smoking in outdoor places, as well as bans on smoking in cars (typically, when minors are present) and private homes.
Other Regulatory Issues: Some regulators are considering, or in some cases have adopted, regulatory measures designed to reduce the supply of tobacco products. These include regulations intended to reduce the number of retailers selling tobacco products by, for example, reducing the overall number of tobacco retail licenses available or banning the sale of tobacco products within specified distances of certain public facilities. In addition, South Africa banned the sale of tobacco products, e-cigarettes, and electronic devices that heat tobacco for several months during the COVID-19 pandemic. The ban, which was lifted on August 17, 2020, resulted in a significant increase of illicit trade of tobacco products.
In a limited number of markets, most notably Japan, we are dependent on governmental approvals that may limit our pricing flexibility.
The EU Single-Use Plastics Directive, which will require tobacco manufacturers and importers to cover the costs of public collection systems for tobacco product filters, entered into force on July 2, 2019, after which Member States will have two years to transpose it into national law. While we cannot predict the impact of this initiative on our business at this time, we are monitoring developments in this area.
Illicit Trade: Illicit tobacco trade creates a cheap and unregulated supply of tobacco products, undermines efforts to reduce smoking prevalence, especially among youth, damages legitimate businesses and intellectual property rights, stimulates organized crime, increases corruption and reduces government tax revenue. Without accounting for any potential COVID-19-related impact, we generally estimate that, excluding China and the U.S., illicit trade may account for as much as 10 to 12% of global cigarette consumption; this includes counterfeit, contraband and the persistent problem of “illicit whites,” which are
67
cigarettes legally produced in one jurisdiction for the sole purpose of being exported and illegally sold in another jurisdiction where they have no legitimate market. Currently, we estimate that illicit trade in the European Union accounted for approximately 8% of total cigarette consumption in 2019.
A number of jurisdictions are considering actions to prevent illicit trade. In November 2012, the FCTC adopted the Protocol to Eliminate Illicit Trade in Tobacco Products (the “Protocol”), which includes supply chain control measures, such as licensing of manufacturers and distributors, enforcement of these control measures in free trade zones, controls on duty free and Internet channels and the implementation of tracking and tracing technologies. To date, 62 Parties, including the European Union, have ratified it. The Protocol came into force in September 2018. Parties must start implementing its provisions in their national legislation. In October 2018, the first Meeting of the Parties to the Protocol decided to produce a comprehensive report on good practices for the implementation of tracking and tracing systems and to prepare a conceptual framework for global information sharing to combat illicit tobacco trade. We welcome this decision and expect that other Parties will ratify the Protocol.
We devote substantial resources to help prevent illicit trade in combustible tobacco products and RRPs. For example, we engage with governments, our business partners and other stakeholders to implement effective measures to combat illicit trade and, in some instances, pursue legal remedies to protect our intellectual property rights.
The tracking and tracing regulations for cigarettes and roll-your-own products manufactured or destined for the EU became effective on May 20, 2019. The effective date for other tobacco-containing products, including some of our RRPs such as heated tobacco units, is May 20, 2024. While we expect that this regulation will increase our operating expenses, we do not expect this increase to be significant.
In 2009, our Colombian subsidiaries entered into an Investment and Cooperation Agreement with the national and regional governments of Colombia to promote investment in, and cooperation on, anti-contraband and anti-counterfeit efforts. The agreement provides $200 million in funding over a 20-year period to address issues such as combating illegal cigarette trade and increasing the quality and quantity of locally-grown tobacco.
In May 2016, PMI launched PMI IMPACT, a global initiative that supports third-party projects dedicated to fighting illegal trade and related crimes such as corruption, organized criminal networks and money laundering. The centerpiece of PMI IMPACT is a council of external independent experts in the fields of law, anti-corruption and law enforcement. The experts are responsible for evaluating and approving funding proposals for PMI IMPACT grants. PMI has pledged $100 million to fund projects within PMI IMPACT over three funding rounds.
Reduced-Risk Products (RRPs)
Our Approach to RRPs: We recognize that smoking cigarettes causes serious diseases and that the best way to avoid the harms of smoking is never to start or to quit. Nevertheless, it is predicted that over the next decade the number of smokers will remain largely unchanged from the current estimate of 1.1 billion, despite the considerable efforts to discourage smoking.
Cigarettes burn tobacco, which produces smoke. As a result of the combustion process, the smoker inhales various toxic substances. In contrast, RRPs do not burn tobacco and produce an aerosol that contains significantly lower levels of harmful and potentially harmful constituents ("HPHCs") than found in cigarette smoke.
For adult smokers who would otherwise continue to smoke, we believe that RRPs, while not risk-free, offer a much better consumer choice. Accordingly, our key strategic priorities are: to develop and commercialize products that present less risk of harm to adult smokers who switch to those products versus continued smoking; and to convince current adult smokers who would otherwise continue to smoke to switch to those products.
We recognize that this transformation from cigarettes to RRPs will take time and that the speed of transformation will depend in part upon factors beyond our control, such as the willingness of governments, regulators and other policy groups to embrace RRPs as a desired alternative to continued cigarette smoking. We also recognize that our part in this transformation must be funded from our existing cigarette business. For as long as a significant number of adult smokers continues to smoke, it is critical that the industry be led by responsible and ethical manufacturers. Therefore, during the transformation, we intend to remain a leading international cigarette manufacturer.
We have a range of RRPs in various stages of development, scientific assessment and commercialization. We conduct rigorous scientific assessments of our RRP platforms to substantiate that they reduce exposure to HPHCs and, ultimately, that these products present, are likely to present, or have the potential to present less risk of harm to adult smokers who switch to them
68
versus continued smoking. We draw upon a team of expert scientists and engineers from a broad spectrum of scientific disciplines and our extensive learnings of adult consumer preferences to develop and assess our RRPs. Our efforts are guided by the following key objectives:
•to develop RRPs that adult smokers who would otherwise continue to smoke find to be satisfying alternatives to smoking;
•for those adult smokers, our goal is to offer RRPs with a scientifically substantiated risk-reduction profile that approaches as closely as possible that associated with smoking cessation;
•to substantiate the reduction of risk for the individual adult smoker and the reduction of harm to the population as a whole, based on scientific evidence of the highest standard that is made available for scrutiny and review by external independent scientists and relevant regulatory bodies; and
•to advocate for the development of science-based regulatory frameworks for the development and commercialization of RRPs, including the communication of scientifically substantiated information to enable adult smokers to make better consumer choices.
Our RRP Platforms: Our product development is based on the elimination of combustion via tobacco heating and other innovative systems for aerosol generation, which we believe is the most promising path to providing a better consumer choice for those who would otherwise continue to smoke. We recognize that no single product will appeal to all adult smokers. Therefore, we are developing a portfolio of products intended to appeal to a variety of distinct adult consumer preferences.
Four RRP platforms are in various stages of development and commercialization readiness:
Platform 1 uses a precisely controlled heating device incorporating our IQOS HeatControl technology, into which a specially designed and proprietary tobacco unit is inserted and heated to generate an aerosol. We have conducted a series of clinical studies for this platform, the results of which were included in our submission to the U.S. Food and Drug Administration (“FDA”) described below. We completed a 6+6 month exposure response study and shared the results with the FDA in April 2020. The study showed that for the group that switched to our Platform 1 product, the eight clinical risk endpoints that were tested as co-primary endpoints in the first six-month term moved in the same direction as observed for smoking cessation after 12 months of use of this product. In addition, we completed an 18-month combined chronic toxicity and carcinogenicity study in mice, which was on-going at the time of our FDA submission. We shared the results with the FDA in August 2018.
Platform 2 uses a pressed carbon heat source which, when ignited, generates a nicotine-containing aerosol by heating tobacco. The results of our pharmacokinetic study (that measured the nicotine pharmacokinetic profile as well as subjective effects) and of our five-day reduced exposure study indicate that this platform could be an acceptable substitute for adult smokers who seek an alternative to cigarettes. The reduced exposure study results showed a substantial reduction in relevant biomarkers of exposure to the measured HPHCs in those who switched to Platform 2 compared to those who continued to smoke cigarettes over a five-day period. The sustainability of this reduction as well as changes in clinical risk markers were assessed in a three-month reduced exposure study. The results of this study were received at the end of 2017, and the related report was finalized in the second quarter of 2018.
Platform 3 provides an aerosol of nicotine salt. We have explored two routes for this platform, one with electronics and one without, and conducted nicotine pharmacokinetic studies with both versions. The results of the pharmacokinetic study related to the version without electronics were received, and the related report was finalized in the fourth quarter of 2018. The results indicate this product's potential as an acceptable alternative to continued cigarette smoking in terms of product satisfaction. In February 2020, we completed a product use and adaptation study in adult smokers for the product variant without electronics. The analysis was completed and the related report finalized in the second quarter of 2020.
Platform 4 covers e-vapor products, which are battery-powered devices that produce an aerosol by vaporizing a nicotine-containing liquid solution. Our e-vapor products comprise devices using current generation technology and our new e-vapor mesh technology that addresses certain challenges presented by some e-vapor products currently on the market. Our IQOS MESH products are designed to ensure the consistency and quality of the generated aerosol. We conducted a nicotine pharmacokinetic study in 2017. The results of this study were received in the second quarter of 2018 for analysis, and the related report was finalized in the fourth quarter of 2018. The results of this study indicate that IQOS MESH products are an effective means of nicotine delivery while being a satisfying alternative for e-cigarette users. In March 2019, a six-month pre-clinical study in mice evaluating the impact of e-cigarette vapor on the risks of pulmonary and cardiovascular disease compared to cigarette smoke was completed; this study did not pertain to a specific product. The study demonstrated that e-cigarette
69
vapors induce significantly lower biological responses associated with cardiovascular and pulmonary diseases compared with cigarette smoke.
After we receive the results of our scientific studies mentioned above, in accordance with standard scientific practices, we intend to share the conclusions in scientific forums and to submit them for inclusion in peer-reviewed publications.
Commercialization of RRPs: We are building a new product category and tailor our commercialization strategy to the characteristics of each specific market. We focus our commercialization efforts on consumer retail experience, guided consumer trials and customer care, and increasingly, digital communication programs. In order to accelerate switching to our Platform 1 product, our initial market introductions typically entail one-on-one consumer engagement and introductory device discounts. These initial commercialization efforts require substantial investment, which we believe will moderate over time.
In 2014, we introduced our Platform 1 product in pilot city launches in Nagoya, Japan, and in Milan, Italy. Since then, we have continuously expanded our commercialization activities, and the product has been commercialized in 61 markets in key cities or nationwide. While our Platform 1 products are currently available for sale in Mexico, that country recently banned the importation of e-cigarettes and devices that heat tobacco.
We estimate that only a very small percentage of adult smokers who convert to our Platform 1 product switch back to cigarettes.
We have integrated the production of our heated tobacco units into a number of our existing manufacturing facilities, are progressing with our plans to build manufacturing capacity for our other RRP platforms, and continue to optimize our manufacturing infrastructure.
An adequate supply chain for our RRP portfolio, including the supply of electronic devices, is important to our business. We work with two electronics manufacturing service providers for the supply of our Platform 1 and IQOS MESH devices and a small number of other providers for other products in our RRP portfolio and related accessories. Due to the COVID-19 pandemic, the operations of our two electronic manufacturing service providers were temporarily suspended at different times. Even though these suspensions did not materially affect our operations, if both of these service providers were significantly constrained at the same time, the supply of the devices could be disrupted. Although we work closely with these service providers on monitoring their production capability and financial health, we cannot guarantee that they will remain capable of meeting their commitments, particularly during the COVID-19 pandemic; if they will not, the commercialization of our RRPs could be adversely affected. The production of our RRP portfolio requires various metals, and we believe that there is an adequate supply of such metals in the world markets to satisfy our current and anticipated production requirements. However, some components and materials necessary for the production of our RRPs, including those for the electronic devices, are obtained from single or limited sources, and can be subject to industry-wide shortages and price fluctuations. While we were successful in maintaining adequate supply of such components and materials so far, we may not be able to secure such supply going forward, particularly during the COVID-19 pandemic; this could negatively impact the commercialization of our RRPs. For details on the impact of COVID-19 on our production and supply chain, see the "Executive Summary" section of this MD&A.
Our Platform 1 and IQOS MESH devices are subject to standard product warranties generally for a period of 12 months from the date of purchase or such other periods as required by law. We discuss product warranties in more detail in Note 16. Product Warranty. The significance of warranty claims is dependent on a number of factors, including device version mix, product failure rates, logistics and service delivery costs, and warranty policies, and may increase with the number of devices sold.
Product quality may affect consumer acceptance of our RRPs.
Our commercialization efforts for the other RRP platforms are as follows:
•We started commercializing an improved version of our IQOS MESH product in New Zealand under the IQOS VEEV brand name and currently plan to launch these products in additional markets under the IQOS VEEV or VEEV brand names.
•With respect to TEEPS, our Platform 2 product, we are finalizing our improvements to this product and plan to conduct a consumer test in the beginning of 2021.
•Following a consumer test, we are improving our Platform 3 product.
70
Due to the COVID-19 pandemic, these plans may be delayed.
RRP Regulation and Taxation: RRPs contain nicotine and are not risk-free. As we describe in more detail above, we support science-based regulation and taxation of RRPs and believe that regulation and taxation should differentiate between cigarettes and products that present, are likely to present, or have the potential to present less risk of harm to adult smokers who switch to these products versus continued smoking and should recognize a continuum of risk for tobacco and other nicotine-containing products. Regulation, as well as industry practices, should reflect the fact that youth should not consume nicotine in any form.
Some governments have banned or are seeking to ban or severely restrict emerging tobacco and nicotine-containing products such as our RRPs and communication of truthful and non-misleading information about such products. For example, the commercialization of e-cigarettes and heat-not-burn products is prohibited in Australia, the commercialization of e-cigarettes is prohibited in Argentina, the importation of e-cigarettes and heat-not-burn products is prohibited in Turkey, and the importation of e-cigarettes and devices that heat tobacco is prohibited in Mexico.
These regulations might foreclose or unreasonably restrict adult consumer access even to products that might be shown to be a better consumer choice than continuing to smoke. During the COVID-19 pandemic, governments may temporarily be unable to focus on the development of science-based regulatory frameworks for the development and commercialization of RRPs or on the enforcement or implementation of regulations that are significant to our business.
We oppose blanket bans and unreasonable restrictions of products that have the potential to present less risk of harm compared to continued smoking. By contrast, we support regulation that sets clear standards for all RRP categories and propels innovation to benefit adult smokers who would otherwise continue to smoke.
In the United States, an established regulatory framework for assessing “Modified Risk Tobacco Products” and “New Tobacco Products” exists under the jurisdiction of the FDA. We submitted to the FDA a Modified Risk Tobacco Product Application (“MRTPA”) for our Platform 1 product in December 2016, and a Premarket Tobacco Product Application (“PMTA”) for our Platform 1 product in March 2017.
On April 30, 2019, the FDA determined that a version of our Platform 1 product is appropriate for the protection of public health and authorized it for sale in the United States. The FDA’s decision followed its comprehensive assessment of our PMTA. We filed a supplemental PMTA application for the IQOS 3 device in March 2020.
On July 7, 2020, the FDA determined that the available scientific evidence demonstrates that the issuance of an exposure modification order would be appropriate for the promotion of public health and authorized the marketing of a version of our Platform 1 product as a "modified risk tobacco product." The FDA authorized the marketing of this product in the U.S. with the following information:
"AVAILABLE EVIDENCE TO DATE:
•the IQOS system heats tobacco but does not burn it.
•this significantly reduces the production of harmful and potentially harmful chemicals.
•scientific studies have shown that switching completely from conventional cigarettes to the IQOS system significantly reduces your body’s exposure to harmful or potentially harmful chemicals."
We must request and receive authorization from the FDA in order to continue marketing this product with the same modified exposure information after the present order expires in four years.
There are two types of MRTP orders the FDA may issue: a “risk modification” order or an “exposure modification” order. We had requested both types of orders for our Platform 1 product. After review, the FDA determined that the evidence did not support issuing a "risk modification" order at this time but that it did support issuing an "exposure modification" order for the product. This determination included a finding that issuance of the exposure modification order is expected to benefit the health of the population as a whole.
We look forward to working with the FDA to provide any additional information they may require in order to market this product with reduced risk claims.
71
The FDA’s PMTA and MRTP orders do not mean that the agency “approved” our Platform 1 product. Both authorizations are subject to strict marketing, reporting and other requirements and are not a guarantee that the product will remain authorized, particularly if there is a significant uptake in youth or non-smoker initiation. The FDA will monitor the marketing of the product.
Some states and municipalities in the U.S. have introduced severe restrictions for the sale and marketing of e-cigarettes and products, including those authorized by the FDA. We believe that the restrictions on FDA-authorized products will not advance public health and will unreasonably limit adult consumer access to products that are shown to be a better alternative to continued smoking.
In March 2020, we requested a clarification from the FDA regarding the applicability of its new health warning requirements to our heated tobacco units sold in the United States.
In the U.S., tobacco and nicotine-containing products that were not commercially marketed as of February 15, 2007 are subject to review and authorization by the FDA. Manufacturers of all non-authorized products currently on the market were required to file a PMTA with the FDA by September 9, 2020.
The FDA announced on September 9, 2020 that it will prioritize enforcement against any tobacco and nicotine-containing product sold without a PMTA.
We continue to support regulation and industry practices that reflect the fact that youth should not consume nicotine in any form.
FDA actions may influence the regulatory approach of other governments.
Until recently, there were no countries with specific product standards for heat-not-burn products. Currently, national standards setting minimum quality and safety requirements for such products have been adopted in several countries with technical heat-not-burn specifications and/or methods for demonstrating the absence of combustion. They are mandatory in Egypt, Jordan, Saudi Arabia and the UAE, and voluntary in the U.K., Russia, Ukraine, Kazakhstan, and Kyrgyzstan. In Japan, a voluntary standard has minimum safety requirements for tobacco heating devices. We expect other governments to consider similar product standards and encourage making them mandatory.
In the EU, all EU Member States have transposed the EU Tobacco Products Directive, including the provisions on novel tobacco products, such as heated tobacco units, and e-cigarettes. Most of the EU Member States require a notification submitted six months before the intended placing on the market of a novel tobacco product, while some require pre-market authorizations for the introduction of such products. To date, we have filed a comprehensive dossier summarizing our scientific assessment of our Platform 1 product in over 20 Member States.
In addition, in Italy, in April 2018, we submitted an application for HEETS, used with the IQOS device, requesting regulatory recognition of the reduction of toxic substances and potential risk reduction resulting from switching to this product compared to continued cigarette smoking. In January 2019, our application was not granted primarily on the grounds of insufficient data and questions of methodology. Due to the constraints of the review process, we had been unable to supplement the application with all the data we subsequently filed with the FDA and to address methodological questions during the review. We plan to submit a new application where we will clarify the concerns raised by the decision and further strengthen our application by submitting additional evidence that became available since we submitted our first application, consistent with our FDA filing. We are confident that our evidence supports our application.
On October 31, 2019, our Australian subsidiary, Philip Morris Limited (“PML”), submitted an application to the Scheduling Committee of the Therapeutic Goods Administration of Australia (“TGA”) seeking to exempt heated tobacco products from being prohibited in Australia. In August 2020, the TGA issued its decision denying the application and stating that it does not present compelling evidence to establish a public health benefit from greater access to nicotine in heated tobacco products.
To date, several governmental agencies have published their scientific findings that analyze the harm-reduction potential of certain RRPs versus continuing smoking, including:
In December 2017, at the request of the U.K. Department of Health and Public Health England, the U.K. Committee on Toxicity published its assessment of the risk of heat-not-burn products relative to cigarette smoking. This assessment included analysis of scientific data for two heat-not-burn products, one of which was our Platform 1 product. The assessment concluded
72
that, while still harmful to health, compared with the known risks from cigarettes, heat-not-burn products are probably less harmful. Subsequently, in February 2018, Public Health England published a report stating that the available evidence suggests that heat-not-burn products may be considerably less harmful than cigarettes and more harmful than e-cigarettes.
In May 2018, the German Federal Institute for Risk Assessment (“BfR”) published a study on the Platform 1 aerosol relative to cigarette smoke using the Health Canada Intense Smoking Regimen. BfR found reductions in selected HPHCs in a range of 80-99%. This publication indicates that significant reductions in the levels of selected toxicants are likely to reduce toxicant exposure, which BfR stated might be regarded as a discrete benefit compared to combustible cigarettes.
In May 2018, the Dutch National Institute for Public Health and Environment (“RIVM”) published a factsheet on novel tobacco products that heat rather than burn tobacco, focusing on our Platform 1 product. RIVM analyzed the aerosol generated by our Platform 1 product and concluded that the use of this product, while still harmful to health, is probably less harmful than continued smoking.
In June 2018, the Korean Food and Drug Administration (“KFDA”) issued a statement on products that heat rather than burn tobacco. The KFDA tested three heat-not-burn products, one of which was our Platform 1 product. The KFDA confirmed that the levels of the nine HPHCs tested in the aerosol of these products were on average approximately 90% lower compared to those measured in the cigarette smoke of the top five cigarette brands in South Korea. However, the KFDA stated that it could not establish that the tested heat-not-burn products are less harmful than cigarettes. In October 2018, our Korean subsidiary filed a request with a local court seeking information underlying KFDA’s analysis, conclusions and public statements. In May 2020, the court ordered KFDA to produce certain records.
In August 2018, the Science & Technology Committee of the U.K. House of Commons published a report of its inquiry into e-cigarettes and heat-not-burn products. The report concluded that e-cigarettes are significantly less harmful to health than smoking tobacco. The report also observed that for those smokers who don’t accept e-cigarettes, heat-not-burn products may offer a public health benefit despite their relative risk. The report called for a risk-proportionate regulatory environment for both e-cigarettes and heat-not-burn products and noted that e-cigarettes should remain the least taxed, cigarettes the most taxed, with heat-not-burn products falling between the two. The U.K. Committee on Advertising Practice announced the removal of a prohibition of health claims in the advertising of e-cigarettes in the U.K. effective November 2018.
In November 2018, the Eurasian Economic Commission (regulatory body of the Eurasian Union consisting of Armenia, Belarus, Kazakhstan, Kyrgyzstan and Russia) published the results of its commissioned study on novel nicotine-containing products, including our Platform 1 product. The study confirms significantly lower levels of HPHCs in the aerosol generated by this product compared to cigarette smoke.
In January 2019, scientific media published the results of the study of the China National Tobacco Quality Supervision and Test Centre (“CNTQST”) comparing the aerosol generated by our Platform 1 product with cigarette smoke. The CNTQST found that the former contained fewer, and lower levels of, harmful constituents than the latter and concluded that the lower temperature of heating tobacco in our Platform 1 product contributed to the difference. The CNTQST stated that the reduction in emissions of harmful constituents cannot be interpreted as equivalent to a proportionate harm/risk reduction for smokers.
The foregoing scientific findings of government agencies may not be indicative of the measures that the relevant government authorities could take in regulating our products.
We make our scientific findings publicly available for scrutiny and peer review through several channels, including our websites. From time to time, adult consumers, competitors, members of the scientific community, and others inquire into our scientific methodologies, challenge our scientific conclusions or request further study of certain aspects of our RRPs and their health effects. We are committed to a robust and open scientific debate and believe that such debate should be based on accurate and reliable scientific information. We seek to provide accurate and reliable scientific information about our RRPs; nonetheless, we may not be able to prevent third-party dissemination of false, misleading or unsubstantiated information about these products. The dissemination of scientifically unsubstantiated information or studies with a strong confirmation bias by third parties may cause confusion among adult smokers and affect their decision to switch to better alternatives to continued smoking, such as our RRPs.
To date, we have been largely successful in demonstrating to regulators that our heated tobacco units are not cigarettes due to the absence of combustion, and as such they are generally taxed either as a separate category or as other tobacco products, which typically yields more favorable tax rates than cigarettes. Although we believe that this is sensible from the public health perspective, we cannot guarantee that regulators will continue this approach.
73
There can be no assurance that we will succeed in our efforts to replace cigarettes with RRPs or that regulation will allow us to commercialize RRPs in all markets, to communicate about our RRPs, including making scientifically substantiated risk-reduction claims, or to treat RRPs differently from cigarettes.
Legal Challenges to RRPs: We face various administrative and legal challenges related to certain RRP activities, including allegations concerning product classification, advertising restrictions, corporate communications, product coach activities, scientific substantiation, product liability, and unfair competition. While we design our programs to comply with relevant regulations, we expect these or similar challenges to continue as we expand our efforts to commercialize RRPs and to communicate publicly. The outcomes of these matters may affect our RRP commercialization and public communication activities and performance in one or more markets.
Our RRP Business Development Initiatives: In December 2013, we established a strategic framework with Altria Group, Inc. (“Altria”) setting out terms on how the parties would collaborate to develop and commercialize e-vapor products and commercialize two of our RRPs in the U.S. In late 2018, Altria announced that it will participate in the e-vapor category only through another e-vapor company in which Altria acquired a minority interest. In September 2019, Altria's subsidiary, Philip Morris USA Inc. (“PM USA”), began commercialization of a version of our Platform 1 product in the U.S. PM USA is responsible for the marketing of this product in the U.S. and implementation of the messages authorized by the FDA in its MRTP order described above.
In January 2020, we announced an agreement with KT&G, a leading tobacco and nicotine company in South Korea, for the commercialization of KT&G’s smoke-free products outside of South Korea on an exclusive basis. For more information, see Acquisitions and Other Business Arrangements below.
Other Developments: In September 2017, we announced our support of the Foundation for a Smoke-Free World. In September 2020, our pledge agreement with the Foundation was amended. We contributed $45 million in 2020 and expect to contribute $40 million in 2021 and $35 million annually from 2022 through 2029, as specified in the amended pledge agreement. To date, we contributed a total of $209.5 million. The Foundation is an independent body and is governed by its independent Board of Directors. The Foundation’s role, as set out in its corporate charter, includes funding research in the field of tobacco harm reduction, encouraging measures that reduce the harm caused by smoking, and assessing the effect of reduced cigarette consumption on the industry value chain.
Governmental Investigations
From time to time, we are subject to governmental investigations on a range of matters, including tax, customs, antitrust, advertising, and labor practices. We describe certain matters pending in Thailand, Russia and South Korea in Note 8. Contingencies.
In November 2010, a WTO panel issued its decision in a dispute relating to facts that arose from August 2006 between the Philippines and Thailand concerning a series of Thai customs and tax measures affecting cigarettes imported by PM Thailand into Thailand (see Note 8. Contingencies for additional information). The WTO panel decision, which was upheld by the WTO Appellate Body, concluded that Thailand had no basis to find that PM Thailand's declared customs values and taxes paid were too low, as alleged by the DSI in 2009. The decision also created obligations for Thailand to revise its laws, regulations, or practices affecting the customs valuation and tax treatment of future cigarette imports. Thailand agreed in September 2011 to fully comply with the decision by October 2012. The Philippines asserts that to date Thailand has not fully complied with the WTO panel decision and commenced challenges at the WTO Appellate Body. The WTO Appellate Body is not operational, and the appeals by Thailand are suspended indefinitely. It is not possible to predict any future developments in these proceedings.
The Public Prosecutor’s office of Rome, Italy, notified our Italian subsidiary, Philip Morris Italia S.r.l. (“PM Italia”), as well as three former or current employees and a former external consultant of PM Italia in July and March 2020, respectively, that it concluded a preliminary investigation against them for alleged contravention of anti-corruption laws and related disruption of trade freedom. The Public Prosecutor alleges that the individuals involved promised certain personal favors to government officials from January to July of 2018 in exchange for favorable treatment for PM Italia, and that PM Italia lacked appropriate organizational controls to prevent the alleged actions by the individuals. In September 2020, the Public Prosecutor referred the matter to trial. PM Italia believes the charges brought against it by the Public Prosecutor are without merit and will defend them vigorously.
74
Asset Impairment and Exit Costs
We discuss asset impairment and exit costs in Note 18. Asset Impairment and Exit Costs to our condensed consolidated financial statements.
Acquisitions and Other Business Arrangements
In January 2020, PMI announced a global collaboration agreement with the leading tobacco and nicotine company in South Korea, KT&G, to commercialize KT&G’s smoke-free products outside of the country. The agreement will run for an initial period of three years. The two companies plan for global collaboration with the intention to actively expand to cover many markets, based on commercial success. The agreement allows PMI to distribute current KT&G smoke-free products, and their evolutions, on an exclusive basis, and does not restrict PMI from distributing its own or third-party products. KT&G’s smoke-free product brand portfolio includes heat-not-burn tobacco products (e.g., LIL Mini and LIL Plus), hybrid technologies that combine heat-not-burn tobacco and e-vapor technologies (e.g., LIL HYBRID), and e-vapor products (e.g., LIL Vapor). PMI will be responsible for the commercialization of smoke-free products supplied under the agreement.
Products sold under the agreement are subject to careful assessment to ensure they meet the regulatory requirements in the markets where they are launched, as well as our standards of quality and scientific substantiation to confirm the absence of combustion and significant reductions of emissions of harmful chemicals compared to cigarettes. PMI and KT&G will seek any necessary regulatory approvals that may be required on a market-by-market basis. There are no current plans to commercialize KT&G products in the United States.
In the third quarter of 2020, we launched commercial initiatives for licensed KT&G products in select markets.
Investments in Unconsolidated Subsidiaries and Equity Securities
We discuss our investments in unconsolidated subsidiaries and equity securities in Note 11. Fair Value Measurements and Note 14. Related Parties - Investments in Unconsolidated Subsidiaries, Equity Securities and Other to our condensed consolidated financial statements.
Trade Policy
We are subject to various trade restrictions imposed by the United States of America and countries in which we do business (“Trade Sanctions”), including the trade and economic sanctions administered by the U.S. Department of the Treasury's Office of Foreign Assets Control and the U.S. Department of State. It is our policy to comply fully with these Trade Sanctions.
Tobacco products are agricultural products under U.S. law and are not technological or strategic in nature. From time to time we make sales in countries subject to Trade Sanctions, either where such sanctions do not apply to our business or pursuant to exemptions or licenses.
A subsidiary sells products to distributors that, in turn, sell those products to duty free customers that supply U.N. peacekeeping forces around the world, including those in the U.N. peacekeeping mission located in Abyei, a special administrative territory in Sudan. We do not believe that these sales, which are not subject to Trade Sanctions, and are de minimis in volume and value, present a material risk to our shareholders, our reputation or the value of our shares. We have no employees, operations or assets in Sudan.
We do not sell products in Iran, North Korea and Syria. From time to time, we explore opportunities to sell our products in one or more of these countries, as permitted by law.
Certain states within the U.S. have enacted legislation permitting or requiring state pension funds to divest or abstain from future investment in stocks of companies that do business with certain countries that are sanctioned by the U.S. We do not believe such legislation has had a material effect on the price of our shares.
75
Operating Results – Three Months and Nine Months Ended September 30, 2020
The following discussion compares operating results within each of our operating segments for the three months and nine months ended September 30, 2020, with the three months and nine months ended September 30, 2019.
Unless otherwise stated, references to total industry, total market, our shipment volume and our market share performance reflect cigarettes and heated tobacco units.
European Union:
| Financial Summary - Quarters Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 2,950 | $ | 2,645 | 11.5 | % | 10.0 | % | $ | 305 | $ | 40 | $ | 82 | $ | 183 | $ | — | ||||||||||||||||||||
| Operating Income | $ | 1,588 | $ | 1,255 | 26.5 | % | 24.7 | % | $ | 333 | $ | 23 | $ | 82 | $ | 158 | $ | 70 | ||||||||||||||||||||
For the three months ended September 30, 2020, net revenues, excluding favorable currency, increased by 10.0%, reflecting: favorable volume/mix, mainly driven by higher heated tobacco unit volume (notably in Germany, Italy and Poland), partly offset by lower cigarette volume (notably in Poland); and a favorable pricing variance (driven by higher combustible pricing, notably in Germany).
Operating income, excluding favorable currency, increased by 24.7%, reflecting: favorable volume/mix, driven by the same factors as for net revenues noted above; a favorable pricing variance; lower manufacturing costs across the Region; and lower marketing, administration and research costs.
| Financial Summary - Nine Months Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 7,960 | $ | 7,381 | 7.8 | % | 9.6 | % | $ | 579 | $ | (130) | $ | 142 | $ | 567 | $ | — | ||||||||||||||||||||
| Operating Income | $ | 3,924 | $ | 3,346 | 17.3 | % | 20.5 | % | $ | 578 | $ | (107) | $ | 142 | $ | 534 | $ | 9 | ||||||||||||||||||||
For the nine months ended September 30, 2020, net revenues, excluding unfavorable currency, increased by 9.6%, reflecting: favorable volume/mix, mainly driven by higher heated tobacco unit volume across the Region (notably in the Czech Republic, Germany, Hungary, Italy and Poland), partly offset by lower cigarette volume (notably in Italy, Poland and Spain, partly offset by Germany); and a favorable pricing variance (driven by higher combustible pricing across the Region, notably in France and Germany, partly offset by lower heated tobacco unit and IQOS device pricing).
Operating income, excluding unfavorable currency, increased by 20.5%, mainly reflecting: favorable volume/mix, driven by the same factors as for net revenues noted above; a favorable pricing variance; and lower manufacturing costs (notably in Germany and Italy); partly offset by higher marketing, administration and research costs (largely related to increased investments behind reduced-risk products, notably in Poland, as well as 2020 asset impairment and exit costs).
Excluding asset impairment and exit costs of $27 million in 2020 and unfavorable currency of $107 million, operating income increased by 21.3%.
European Union - Total Market, PMI Shipment Volume and Market Share Commentaries
Total market, PMI shipment volume and market share performance are shown in the table below:
76
| European Union Key Data | Third-Quarter | Nine Months Year-to-Date | ||||||||||||||||||||||||
| Change | Change | |||||||||||||||||||||||||
| 2020 | 2019 | % / pp | 2020 | 2019 | % / pp | |||||||||||||||||||||
| Total Market (billion units) | 131.5 | 132.3 | (0.6) | % | 356.3 | 363.9 | (2.1) | % | ||||||||||||||||||
| PMI Shipment Volume (million units) | ||||||||||||||||||||||||||
| Cigarettes | 45,179 | 47,238 | (4.4) | % | 126,142 | 133,093 | (5.2) | % | ||||||||||||||||||
| Heated Tobacco Units | 5,181 | 3,474 | 49.1 | % | 14,069 | 8,810 | 59.7 | % | ||||||||||||||||||
| Total European Union | 50,360 | 50,712 | (0.7) | % | 140,211 | 141,903 | (1.2) | % | ||||||||||||||||||
| PMI Market Share | ||||||||||||||||||||||||||
| Marlboro | 17.7 | % | 18.0 | % | (0.3) | 17.7 | % | 18.1 | % | (0.4) | ||||||||||||||||
| L&M | 6.0 | % | 6.7 | % | (0.7) | 6.3 | % | 6.7 | % | (0.4) | ||||||||||||||||
| Chesterfield | 5.5 | % | 5.7 | % | (0.2) | 5.6 | % | 5.8 | % | (0.2) | ||||||||||||||||
| Philip Morris | 2.5 | % | 2.7 | % | (0.2) | 2.5 | % | 2.7 | % | (0.2) | ||||||||||||||||
| HEETS | 3.9 | % | 2.4 | % | 1.5 | 3.9 | % | 2.3 | % | 1.6 | ||||||||||||||||
| Others | 3.1 | % | 3.1 | % | — | 3.1 | % | 3.2 | % | (0.1) | ||||||||||||||||
| Total European Union | 38.7 | % | 38.6 | % | 0.1 | 39.1 | % | 38.8 | % | 0.3 | ||||||||||||||||
In the third quarter, the estimated total market in the EU decreased by 0.6% to 131.5 billion units, notably driven by:
•Denmark, down by 36.1%, mainly reflecting the net unfavorable impact of estimated trade inventory movements related to a significant excise tax increase on April 1, 2020. Excluding these movements, the total estimated market decreased by 11.8%, mainly due to the impact of excise tax-driven price increases;
•Romania, down by 7.9%, primarily reflecting the impact of pandemic-related lockdown measures; and
•Spain, down by 7.3%, mainly reflecting lower in-bound tourism and border sales due to the pandemic;
partly offset by
•Poland, up by 8.6%, notably reflecting the pandemic-related impact of: lower cross-border (non-domestic) purchases, reduced out-bound tourism and a lower estimated prevalence of illicit trade due to border restrictions.
In the third quarter, our total shipment volume decreased by 0.7% to 50.4 billion units, reflecting:
•lower cigarette shipment volume, mainly due to lower market share (notably in Italy and Poland, partly reflecting out-switching to heated tobacco units);
partly offset by
•higher heated tobacco unit shipment volume across the Region, driven by higher market share (notably in Italy and Poland).
For the nine months year-to-date, the estimated total market in the EU decreased by 2.1% to 356.3 billion units, notably due to:
•Czech Republic, down by 7.5%, primarily reflecting lower border sales due to lockdown measures, particularly during the second quarter;
•France, down by 3.7%, mainly reflecting the impact of price increases in the fourth quarter of 2019 and the first quarter of 2020, partly offset by the pandemic-related impact of lower cross-border (non-domestic) purchases and a lower estimated prevalence of illicit trade due to border restrictions;
•Greece, down by 8.5%, primarily reflecting lower in-bound tourism due to the pandemic;
77
•Romania, down by 7.1%, mainly reflecting the same factor as in the quarter; and
•Spain, down by 7.8%, primarily reflecting the same factors as in the quarter;
partly offset by
•Germany, up by 2.4%, notably reflecting the pandemic-related impact of lower cross-border (non-domestic) purchases and reduced out-bound tourism, partly offset by the impact of retail price increases in the first quarter of 2020 and adult smoker out-switching to other combustible tobacco products.
For the nine months year-to-date, our total shipment volume decreased by 1.2% to 140.2 billion units, reflecting:
•lower cigarette shipment volume, mainly due to the lower total market and lower cigarette market share (notably in Italy and Poland, partly reflecting out-switching to heated tobacco units);
partly offset by
•higher heated tobacco unit shipment volume across the Region (notably in Germany, Italy and Poland), driven by higher market share.
Eastern Europe:
| Financial Summary - Quarters Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 899 | $ | 899 | — | % | 7.3 | % | $ | — | $ | (66) | $ | 64 | $ | 2 | $ | — | ||||||||||||||||||||
| Operating Income (Loss) | $ | 245 | $ | (101) | +100% | +100% | $ | 346 | $ | (105) | $ | 64 | $ | 17 | $ | 370 | ||||||||||||||||||||||
For the three months ended September 30, 2020, net revenues, excluding unfavorable currency, increased by 7.3%, mainly reflecting: a favorable pricing variance, driven by higher combustible pricing (predominantly in Russia); and favorable volume/mix, mainly driven by higher heated tobacco unit volume across the Region (primarily in Russia and Ukraine), largely offset by unfavorable cigarette volume/mix in Russia.
Operating income, excluding unfavorable currency, increased by +100%, notably reflecting a favorable comparison, shown in "Cost/Other," due to a charge recorded in the third quarter of 2019 of $374 million related to the Russia excise and VAT audit.
Excluding this charge and unfavorable currency of $105 million, operating income increased by 28.2%, reflecting: a favorable pricing variance; favorable volume/mix, reflecting the same drivers as for net revenues noted above; and lower manufacturing costs (mainly in Russia); partially offset by higher marketing, administration and research costs due to Russia.
78
| Financial Summary - Nine Months Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 2,470 | $ | 2,300 | 7.4 | % | 13.7 | % | $ | 170 | $ | (145) | $ | 105 | $ | 210 | $ | — | ||||||||||||||||||||
| Operating Income | $ | 610 | $ | 284 | +100% | +100% | $ | 326 | $ | (208) | $ | 105 | $ | 156 | $ | 273 | ||||||||||||||||||||||
For the nine months ended September 30, 2020, net revenues, excluding unfavorable currency, increased by 13.7%, reflecting: favorable volume/mix, predominantly driven by higher heated tobacco unit volume across the Region (notably in Russia and Ukraine) and higher heated tobacco unit mix (mainly in Russia), partly offset by unfavorable cigarette volume (primarily in Russia and Ukraine, partially offset by Israel) and unfavorable cigarette mix (mainly in Russia); and a favorable pricing variance, driven by higher combustible pricing (primarily in Russia and Ukraine), partly offset by lower IQOS device pricing (mainly in Russia).
Operating income, excluding unfavorable currency, increased by +100%, notably reflecting a favorable comparison, shown in "Cost/Other," of charges recorded September year-to-date 2020 of $7 million, related to asset impairment and exit costs associated with organizational design optimization, to a charge recorded in the same period in 2019 of $374 million, related to the Russia excise and VAT audit.
Excluding these 2019 and 2020 charges and unfavorable currency of $208 million, operating income increased by 25.4%, reflecting: favorable volume/mix, driven by the same factors as for net revenues noted above; and a favorable pricing variance; partly offset by higher marketing, administration and research costs (partly related to increased investments behind reduced-risk products, notably in Russia); and higher manufacturing costs due to Russia.
Eastern Europe - Total Market, PMI Shipment Volume and Market Share Commentaries
In the third quarter, the estimated total market in Eastern Europe decreased, mainly due to:
•Russia, down by 2.9%, primarily reflecting the impact of price increases; and
•Ukraine, down by 7.4%, mainly reflecting the impact of excise tax-driven price increases.
For the nine months year-to-date, the estimated total market in Eastern Europe decreased, notably due to:
•Russia, down by 2.7%, or by 4.3% excluding the net favorable impact of estimated trade inventory movements, primarily reflecting the same factor as in the quarter; and
•Ukraine, down by 9.7%, mainly reflecting the impact of excise tax-driven price increases, as well as reduced adult smoker average daily consumption, notably in the second quarter of 2020, due to lockdown measures.
| PMI Shipment Volume (million units) | Third-Quarter | Nine Months Year-to-Date | |||||||||||||||||||||
| 2020 | 2019 | Change | 2020 | 2019 | Change | ||||||||||||||||||
| Cigarettes | 25,661 | 27,379 | (6.3) | % | 70,737 | 74,779 | (5.4) | % | |||||||||||||||
| Heated Tobacco Units | 4,882 | 3,858 | 26.5 | % | 14,374 | 8,213 | 75.0 | % | |||||||||||||||
| Total Eastern Europe | 30,543 | 31,237 | (2.2) | % | 85,111 | 82,992 | 2.6 | % | |||||||||||||||
In the third quarter, our total shipment volume decreased by 2.2% to 30.5 billion units, notably due to:
•Russia, down by 1.4%. Excluding the net unfavorable impact of estimated distributor inventory movements, PMI's in-market sales increased by 0.4%, mainly reflecting a higher market share, driven by heated tobacco units, partly offset by the lower total market.
79
For the nine months year-to-date, our total shipment volume increased by 2.6% to 85.1 billion units, mainly due to:
•Russia, up by 5.8%, primarily reflecting a higher market share, driven by heated tobacco units, partly offset by the lower total market;
partly offset by
•Ukraine, down by 4.8%, mainly due to the lower total market, partly offset by a higher market share, driven by heated tobacco units.
Middle East & Africa:
| Financial Summary - Quarters Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 768 | $ | 1,127 | (31.9) | % | (29.4) | % | $ | (359) | $ | (28) | $ | 6 | $ | (325) | $ | (12) | ||||||||||||||||||||
| Operating Income | $ | 261 | $ | 519 | (49.7) | % | (46.8) | % | $ | (258) | $ | (15) | $ | 6 | $ | (271) | $ | 22 | ||||||||||||||||||||
For the three months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 29.4%, primarily reflecting: unfavorable volume/mix, mainly due to lower cigarette and heated tobacco unit volume in PMI Duty Free; partly offset by a favorable pricing variance, driven by PMI Duty Free and Saudi Arabia, largely offset by Turkey.
Operating income, excluding unfavorable currency, decreased by 46.8%, notably reflecting: unfavorable volume/mix, due to the same factor as for net revenues noted above; partly offset by lower marketing, administration and research costs; lower manufacturing costs; and a favorable pricing variance.
| Financial Summary - Nine Months Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 2,348 | $ | 3,058 | (23.2) | % | (21.7) | % | $ | (710) | $ | (46) | $ | 123 | $ | (736) | $ | (51) | ||||||||||||||||||||
| Operating Income | $ | 819 | $ | 1,304 | (37.2) | % | (35.0) | % | $ | (485) | $ | (29) | $ | 123 | $ | (565) | $ | (14) | ||||||||||||||||||||
For the nine months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 21.7%, reflecting: unfavorable volume/mix, mainly due to lower cigarette volume (predominantly in PMI Duty Free and Turkey) and lower heated tobacco unit volume in PMI Duty Free; and lower fees for certain distribution rights billed to customers in certain markets, shown in "Cost/Other" partially offset by a favorable pricing variance, driven by combustible pricing (mainly in the GCC, particularly Saudi Arabia, as well as Egypt and PMI Duty Free, partly offset by Turkey).
Operating income, excluding unfavorable currency, decreased by 35.0%, mainly reflecting: unfavorable volume/mix, predominantly due to lower cigarette and heated tobacco unit volume in PMI Duty Free; and unfavorable "Cost/Other," mainly due to lower fees for certain distribution rights, as noted above for net revenues; partially offset by a favorable pricing variance; and lower marketing, administration and research costs.
Excluding asset impairment and exit costs of $9 million in 2020 and unfavorable currency of $29 million, operating income decreased by 34.3%.
Middle East & Africa - Total Market, PMI Shipment Volume and Market Share Commentaries
In the third quarter, the estimated total market in the Middle East & Africa decreased, mainly due to:
•International Duty Free, down by 72.8%, reflecting the impact of government travel restrictions and reduced passenger traffic due to the pandemic; and
80
•South Africa, down by 25.8%, reflecting the impact of the pandemic-related ban on all tobacco sales through August 17, 2020.
For the nine months year-to-date, the estimated total market in the Middle East & Africa decreased, mainly due to:
•Egypt, down by 5.4%, notably reflecting pandemic-related supply-chain shortages involving competitors' products and reductions in adult smoker average daily consumption during the lockdown;
•International Duty Free, down by 63.1%, reflecting the same factors as in the quarter;
•South Africa, down by 42.7%, primarily reflecting the same factor as in the quarter; and
•Turkey, down by 7.3%, mainly reflecting the impact of lockdown measures on adult smoker average daily consumption, as well as a higher prevalence of illicit trade related to cut tobacco, particularly during the first-half of 2020, following significant industry-wide price increases in 2019.
| PMI Shipment Volume (million units) | Third-Quarter | Nine Months Year-to-Date | |||||||||||||||||||||
| 2020 | 2019 | Change | 2020 | 2019 | Change | ||||||||||||||||||
| Cigarettes | 30,903 | 36,994 | (16.5) | % | 88,087 | 101,957 | (13.6) | % | |||||||||||||||
| Heated Tobacco Units | 179 | 588 | (69.6) | % | 834 | 2,061 | (59.5) | % | |||||||||||||||
| Total Middle East & Africa | 31,082 | 37,582 | (17.3) | % | 88,921 | 104,018 | (14.5) | % | |||||||||||||||
In the third quarter, our total shipment volume decreased by 17.3% to 31.1 billion units, notably due to:
•PMI Duty Free, down by 93.7%, or by 68.7% excluding the net unfavorable impact of estimated distributor inventory movements (driven by cigarettes), mainly reflecting the lower total market; and
•Turkey, down by 4.9%, primarily reflecting a lower market share mainly due to adult smoker down-trading following 2019 retail price increases;
partly offset by
•Saudi Arabia, up by 23.4%. Excluding the net favorable impact of estimated distributor inventory movements of 0.7 billion cigarettes, PMI's in-market sales decreased by 9.4%, mainly reflecting a lower market share due to adult smoker down-trading following VAT-driven retail price increases and the temporary adverse impact of the transition to a new distributor.
For the nine months year-to-date, our total shipment volume decreased by 14.5% to 88.9 billion units, notably due to:
•PMI Duty Free, down by 68.9%, or by 59.2% excluding the net unfavorable impact of estimated distributor inventory movements (driven by cigarettes), mainly reflecting the lower total market; and
•Turkey, down by 13.0%, mainly reflecting the lower total market and a lower market share, notably due to adult smoker down-trading following the 2019 price increases.
81
South & Southeast Asia:
| Financial Summary - Quarters Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 1,071 | $ | 1,246 | (14.0) | % | (14.3) | % | $ | (175) | $ | 3 | $ | (90) | $ | (88) | $ | — | ||||||||||||||||||||
| Operating Income | $ | 402 | $ | 539 | (25.4) | % | (24.9) | % | $ | (137) | $ | (3) | $ | (90) | $ | (60) | $ | 16 | ||||||||||||||||||||
For the three months ended September 30, 2020, net revenues, excluding favorable currency, decreased by 14.3%, reflecting: an unfavorable pricing variance, due to Indonesia; and unfavorable volume/mix, principally due to lower cigarette volume in Indonesia, partly offset by favorable cigarette mix in Indonesia.
Operating income, excluding unfavorable currency, decreased by 24.9%, primarily reflecting: an unfavorable pricing variance; and unfavorable volume/mix, due to the same factors as for net revenues noted above; partly offset by lower marketing, administration and research costs (notably in Indonesia).
| Financial Summary - Nine Months Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 3,211 | $ | 3,607 | (11.0) | % | (10.3) | % | $ | (396) | $ | (24) | $ | 54 | $ | (426) | $ | — | ||||||||||||||||||||
| Operating Income | $ | 1,290 | $ | 1,471 | (12.3) | % | (12.3) | % | $ | (181) | $ | — | $ | 54 | $ | (296) | $ | 61 | ||||||||||||||||||||
For the nine months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 10.3%, reflecting: unfavorable volume/mix, primarily due to lower cigarette volume in Indonesia and the Philippines, partly offset by favorable cigarette mix in Indonesia; partially offset by a favorable pricing variance, principally driven by combustible pricing in the Philippines, partly offset by Indonesia.
Operating income, excluding currency, decreased by 12.3%, mainly reflecting: unfavorable volume/mix, due to the same factors as for net revenues noted above; partially offset by a favorable pricing variance; and lower marketing, administration and research costs (notably in Indonesia).
Excluding lower asset impairment and exit costs of $9 million and currency, operating income decreased by 12.7%.
South & Southeast Asia - Total Market, PMI Shipment Volume and Market Share Commentaries
In the third quarter, the estimated total market in South & Southeast Asia decreased, notably due to:
•India, down by 14.0%, primarily reflecting the impact of lockdown restrictions on the movement of certain products, including tobacco; and
•Indonesia, down by 9.3%, or by 6.1% excluding the net unfavorable impact of estimated trade inventory movements, mainly reflecting the impact of excise tax-driven price increases and pandemic-related measures on adult smoker average daily consumption;
partly offset by
•Pakistan, up by 32.3%, or by 1.0% excluding the net favorable impact of estimated trade inventory movements related to the timing of price increases in 2019.
82
For the nine months year-to-date, the estimated total market in South & Southeast Asia decreased, notably due to:
•India, down by 20.4%, mainly reflecting the same factor as in the quarter;
•Indonesia, down by 9.4%, or by 11.8% excluding the net favorable impact of estimated trade inventory movements, primarily reflecting the same factors as in the quarter;
•Pakistan, down by 14.8%, mainly reflecting the impact of excise tax-driven price increases in June 2019 and value brand price increases in February 2020, coupled with the impact of trade supply disruption on tobacco product availability due to lockdown measures; and
•the Philippines, down by 10.7%, mainly reflecting the impact of pandemic-related quarantines, as well as industry-wide price increases in the third quarter of 2019.
| PMI Shipment Volume (million units) | Third-Quarter | Nine Months Year-to-Date | |||||||||||||||||||||
| 2020 | 2019 | Change | 2020 | 2019 | Change | ||||||||||||||||||
| Cigarettes | 37,238 | 42,362 | (12.1) | % | 108,179 | 130,230 | (16.9) | % | |||||||||||||||
| Heated Tobacco Units | 10 | — | — | % | 10 | — | — | % | |||||||||||||||
| Total South & Southeast Asia | 37,248 | 42,362 | (12.1) | % | 108,189 | 130,230 | (16.9) | % | |||||||||||||||
In the third quarter, our total shipment volume decreased by 12.1% to 37.2 billion units, notably due to:
•Indonesia, down by 20.8%, reflecting the lower total market, as well as a lower market share, mainly due to: adult smoker down-trading to the tax-advantaged 'below tier one' segment, the impact of elevated price gaps in the tier one segment (partly due to the delay in minimum price enforcement), and the disproportionate impact of stricter public mobility restrictions in urban areas, where PMI’s share is higher; and
•the Philippines, down by 5.5%, mainly reflecting a lower market share for mid-price Fortune due to the impact of price increases in the third quarter of 2019;
partly offset by
•Pakistan, up by 36.0%, primarily reflecting the higher total market.
For the nine months year-to-date, our total shipment volume decreased by 16.9% to 108.2 billion units, notably due to:
•Indonesia, down by 19.1%, reflecting the lower total market, as well as a lower market share, mainly due to the same factors as in the quarter;
•Pakistan, down by 26.2%, mainly reflecting the lower total market and a lower market share, mainly due to low-price Morven; and
•the Philippines, down by 13.7%, mainly reflecting the lower total market and a lower market share, primarily due to the same factor as in the quarter.
83
East Asia & Australia:
| Financial Summary - Quarters Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ 1,358 | $ 1,252 | 8.5 | % | 7.0 | % | $ | 106 | $ | 18 | $ | 92 | $ | (4) | $ | — | ||||||||||||||||||||||
| Operating Income | $ 637 | $ 451 | 41.2 | % | 37.5 | % | $ | 186 | $ | 17 | $ | 92 | $ | (38) | $ | 115 | ||||||||||||||||||||||
For the three months ended September 30, 2020, net revenues, excluding favorable currency, increased by 7.0%, reflecting: a favorable pricing variance, primarily driven by higher heated tobacco and combustible pricing in Japan (due to the impact of distributor inventory revaluation associated with the October 1, 2020, excise tax-driven price increases); partially offset by unfavorable volume/mix, mainly due to: lower cigarette volume in Japan and unfavorable cigarette mix in Australia, largely offset by higher IQOS device and heated tobacco unit volume (principally in Japan).
Operating income, excluding favorable currency, increased by 37.5%, mainly reflecting: a favorable pricing variance; lower manufacturing costs (primarily related to Japan); and lower marketing, administration and research costs (mainly related to reduced-risk products, notably in Japan); partly offset by unfavorable volume/mix, due to the same factors as for net revenues noted above.
| Financial Summary - Nine Months Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 4,045 | $ | 4,094 | (1.2) | % | (1.1) | % | $ | (49) | $ | (3) | $ | 135 | $ | (181) | $ | — | ||||||||||||||||||||
| Operating Income | $ | 1,792 | $ | 1,520 | 17.9 | % | 17.8 | % | $ | 272 | $ | 2 | $ | 135 | $ | (134) | $ | 269 | ||||||||||||||||||||
For the nine months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 1.1%, reflecting: unfavorable volume/mix, mainly due to lower cigarette volume (primarily in Australia and Japan) and unfavorable cigarette mix in Australia, partly offset by higher heated tobacco unit volume in Japan; partially offset by a favorable pricing variance, mainly driven by higher heated tobacco and combustible pricing in Japan (partly reflecting the same factor as in the third quarter) and higher combustible pricing in Australia, partly offset by lower IQOS device pricing in Japan.
Operating income, excluding favorable currency, increased by 17.8%, mainly reflecting: lower marketing, administration and research costs (notably in Japan); lower manufacturing costs (mainly related to Japan and Korea); and a favorable pricing variance; partly offset by unfavorable volume/mix, primarily due to the same factors as for net revenues noted above.
Excluding asset impairment and exit costs of $13 million in 2020 and favorable currency of $2 million, operating income increased by 18.6%.
East Asia & Australia - Total Market, PMI Shipment Volume and Market Share Commentaries
In the third quarter, the estimated total market in East Asia & Australia, excluding China, increased, primarily due to:
•Korea, up by 10.2%, or by 4.7% excluding the net favorable impact of estimated trade inventory movements, mainly driven by the shift of adult smokers from duty-free to domestic purchases due to the pandemic-related decline in international travel; and
•Taiwan, up by 8.0%, mainly driven by the same factor as for Korea;
partly offset by
84
•Japan, down by 2.8%, or by 9.0% excluding the net favorable impact of estimated trade inventory movements (primarily related to the October 1, 2020, excise tax-driven retail price increases), notably due to adult smoker out-switching from cigarettes to the cigarillo category.
For the nine months year-to-date, the estimated total market in East Asia & Australia, excluding China, decreased, notably due to:
•Japan, down by 7.3%, or by 10.2% excluding the net favorable impact of estimated trade inventory movements. The lower total market mainly reflected the impact of reduced adult smoker consumption occasions due to pandemic-related measures, as well as adult smoker out-switching from cigarettes to the cigarillo category; and
•Australia, down by 9.9%, or by 3.9% excluding the net unfavorable impact of estimated trade inventory movements, primarily reflecting the impact of excise tax-driven price increases;
partly offset by
•Korea, up by 6.0%, notably driven by the same factor as in the quarter.
| PMI Shipment Volume (million units) | Third-Quarter | Nine Months Year-to-Date | |||||||||||||||||||||
| 2020 | 2019 | Change | 2020 | 2019 | Change | ||||||||||||||||||
| Cigarettes | 10,784 | 12,692 | (15.0) | % | 35,154 | 38,650 | (9.0) | % | |||||||||||||||
| Heated Tobacco Units | 8,601 | 7,976 | 7.8 | % | 24,799 | 23,253 | 6.6 | % | |||||||||||||||
| Total East Asia & Australia | 19,385 | 20,668 | (6.2) | % | 59,953 | 61,903 | (3.2) | % | |||||||||||||||
In the third quarter, our total shipment volume decreased by 6.2% to 19.4 billion units, notably in:
•Japan, down by 10.6%. Excluding the net unfavorable impact of estimated distributor inventory movements of 1.3 billion cigarettes and 0.7 billion heated tobacco units, PMI's in-market sales increased by 4.2%, mainly driven by a higher market share for heated tobacco units, partly offset by the lower total market and a lower market share for cigarettes.
For the nine months year-to-date, our total shipment volume decreased by 3.2% to 60.0 billion units, notably in:
•Japan, down by 4.2%, or by 0.9% excluding the net unfavorable impact of estimated distributor inventory movements, mainly due to the lower total market and a lower market share for cigarettes, partly offset by a higher market share for heated tobacco units; and
•Korea, down by 4.2%, primarily due to a lower market share, mainly reflecting the unfavorable impact of the growth of the cigarette new taste dimension segment, in which PMI has a relatively low share, partly offset by the higher total market.
85
Latin America & Canada:
| Financial Summary - Quarters Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 400 | $ | 473 | (15.4) | % | (5.9) | % | $ | (73) | $ | (45) | $ | 17 | $ | (43) | $ | (2) | ||||||||||||||||||||
| Operating Income | $ | 110 | $ | 125 | (12.0) | % | 23.2 | % | $ | (15) | $ | (44) | $ | 17 | $ | (31) | $ | 43 | ||||||||||||||||||||
Note: Net Revenues include revenues from shipments of Platform 1 devices, heated tobacco units and accessories to Altria Group, Inc., commencing in the third quarter of 2019, for sale under license in the United States.
For the three months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 5.9%, mainly reflecting: unfavorable volume/mix, notably due to lower cigarette volume in Argentina; partly offset by a favorable pricing variance, predominantly driven by higher combustible pricing in most markets across the Region.
Operating income, excluding unfavorable currency, increased by 23.2%, notably reflecting a favorable comparison, shown in "Cost/Other," due to asset impairment and exit costs of $22 million recorded in the third quarter of 2019 associated with a plant closure in Colombia.
Excluding 2019 asset impairment and exit costs of $22 million and unfavorable currency of $44 million, operating income increased by 4.8%, primarily reflecting: a favorable pricing variance; and lower marketing, administration and research costs (notably in Argentina); partly offset by unfavorable volume/mix (due to the same factor as for net revenues noted above).
| Financial Summary - Nine Months Ended September 30, | Change Fav./(Unfav.) | Variance Fav./(Unfav.) | ||||||||||||||||||||||||||||||||||||
| 2020 | 2019 | Total | Excl. Curr. | Total | Cur- rency | Price | Vol/ Mix | Cost/ Other(1) | ||||||||||||||||||||||||||||||
| (in millions) | ||||||||||||||||||||||||||||||||||||||
| Net Revenues | $ | 1,216 | $ | 1,652 | (26.4) | % | (19.1) | % | $ | (436) | $ | (121) | $ | 107 | $ | (232) | $ | (190) | ||||||||||||||||||||
| Operating Income | $ | 328 | $ | 100 | +100% | +100% | $ | 228 | $ | (85) | $ | 107 | $ | (175) | $ | 381 | ||||||||||||||||||||||
(1) Cost/Other variance includes the impact of the RBH deconsolidation.
Note: Net Revenues include revenues from shipments of Platform 1 devices, heated tobacco units and accessories to Altria Group, Inc., commencing in the third quarter of 2019, for sale under license in the United States.
For the nine months ended September 30, 2020, net revenues, excluding unfavorable currency, decreased by 19.1%, reflecting: unfavorable volume/mix, due to lower cigarette volume, mainly in Argentina and Mexico; and the unfavorable impact of the deconsolidation of RBH shown in "Cost/Other" partly offset by a favorable pricing variance, driven by higher combustible pricing across the Region (notably in Mexico).
Operating income, excluding unfavorable currency, increased by +100%, notably reflecting a favorable comparison, shown in "Cost/Other," of charges recorded September year-to-date 2020 of $4 million, related to asset impairment and exit costs associated with organizational design optimization, to charges recorded in the same period in 2019 of $478 million, related to: asset impairment and exit costs ($45 million) associated with a plant closure in Colombia, the loss on the deconsolidation of RBH ($239 million), and the Canadian tobacco litigation-related expense ($194 million).
Excluding these 2020 and 2019 charges and unfavorable currency of $85 million, operating income decreased by 27.9%, mainly reflecting: unfavorable volume/mix, due to the same factor as for net revenues noted above; and the unfavorable impact of the deconsolidation of RBH, included in "Cost/Other" partly offset by a favorable pricing variance; and lower marketing, administration and research costs (notably in Argentina).
86
Latin America & Canada - Total Market, PMI Shipment Volume and Market Share Commentaries
In the third quarter, the estimated total market in Latin America & Canada was essentially stable, notably reflecting:
•Brazil, up by 17.3%, mainly reflecting a lower estimated prevalence of illicit trade due to: reduced price gaps with legal products and the impact of border restrictions imposed as a result of the pandemic;
partly offset by
•Colombia, down by 19.0%, primarily reflecting reduced adult smoker average daily consumption due to the impact of pandemic-related mobility restrictions.
For the nine months year-to-date, the estimated total market in Latin America & Canada decreased, notably due to:
•Colombia, down by 16.5%, primarily reflecting reduced product availability (mainly in the second quarter of 2020) and adult smoker average daily consumption due to the impact of pandemic-related mobility restrictions; and
•Mexico, down by 12.7%, mainly due to the impact of excise tax-driven price increases in January 2020, as well as the impact of pandemic-related measures on adult smoker average daily consumption;
partly offset by
•Brazil, up by 12.2%, mainly reflecting the same factors as in the quarter.
| PMI Shipment Volume (million units) | Third-Quarter | Nine Months Year-to-Date | |||||||||||||||||||||
| 2020 | 2019 | Change | 2020 | 2019 | Change | ||||||||||||||||||
| Cigarettes | 15,699 | 16,854 | (6.9) | % | 45,542 | 52,906 | (13.9) | % | |||||||||||||||
| Heated Tobacco Units | 114 | 89 | 28.1 | % | 316 | 202 | 56.4 | % | |||||||||||||||
| Total Latin America & Canada | 15,813 | 16,943 | (6.7) | % | 45,858 | 53,108 | (13.7) | % | |||||||||||||||
In the third quarter, our total shipment volume decreased by 6.7% to 15.8 billion units, mainly due to:
•Argentina, down by 16.9%, primarily reflecting a lower market share, mainly due to adult smoker down-trading to ultra-low-price brands produced by local manufacturers; and
•Colombia, down by 19.6%, mainly due to the lower total market;
partly offset by
•Brazil, up by 13.9%, mainly reflecting the higher total market.
For the nine months year-to-date, our total shipment volume decreased by 13.7% to 45.9 billion units, or by 12.0% excluding the impact of the RBH deconsolidation, notably due to
•Argentina, down by 15.1%, primarily reflecting a lower market share, mainly due to the impact of retail out-of-stock of PMI brands, during the second quarter, as well as adult smoker down-trading to ultra-low-price brands produced by local manufacturers;
•Canada, down by 26.0%, due to the unfavorable impact of the deconsolidation of RBH; and
•Colombia, down by 17.8%, primarily reflecting the same factor as in the quarter; and
•Mexico, down by 17.9%, mainly due to a lower total market and a lower market share, primarily reflecting: adult smoker down-trading following the January 2020 price increases and the impact of the pandemic on adult smoker consumption patterns;
87
partly offset by
•Brazil, up by 10.2%, mainly reflecting the higher total market.
Financial Review
Cash Flow Highlights



| For the Nine Months Ended September 30, | ||||||||
| (in millions) | 2020 | 2019 | ||||||
| Net cash provided by operating activities | $ | 6,650 | $ | 6,766 | ||||
| Net cash used in investing activities | (568) | (1,627) | ||||||
| Net cash used in financing activities | (8,031) | (5,102) | ||||||
Net Cash Provided by Operating Activities
During the first nine months of 2020, net cash provided by operating activities decreased by $0.1 billion compared with the first nine months of 2019. Excluding unfavorable currency movements of $0.3 billion, net cash provided by operating activities increased by $0.2 billion, due primarily to higher net earnings (excluding 2019 non-cash charges related to the Canadian tobacco litigation-related expense and the loss on deconsolidation of RBH), partially offset by higher working capital requirements of $0.2 billion and higher cash payments in 2020 for asset impairment and exit costs.
Higher net earnings for the first nine months of 2020, excluding the impact of the above 2019 non-cash charges, were partly attributable to the Russia excise and VAT audit charge of $374 million which was paid in the third quarter of 2019. For further details, see Note 8. Contingencies.
The higher working capital requirements were primarily due to the timing of shipments and excise tax payments partially offset by less cash used for accounts receivable, due to the varying levels of usage of our factoring arrangements to sell trade receivables and timing of sales and cash collections.
88
Net Cash Used in Investing Activities
During the first nine months of 2020, net cash used in investing activities decreased by $1.1 billion compared with the first nine months of 2019. This decrease in net cash used in investing activities was primarily due to the reduction of cash in 2019 resulting from the deconsolidation of RBH and lower capital expenditures, partially offset by higher cash collateral posted to secure derivatives designated as net investment hedges of Euro assets principally related to changes in exchange rates between the Euro and the U.S. dollar. For further details on the deconsolidation of RBH, see Note 19. Deconsolidation of RBH. For further details on our derivatives designated as net investment hedges, see Note 5. Financial Instruments.
During the first nine months of 2020, capital expenditures decreased by $234 million compared with the first nine months of 2019. The 2020 and 2019 capital expenditures were primarily related to our ongoing investments in RRPs. We expect total capital expenditures in 2020 to be approximately $0.6 billion, compared to approximately $0.7 billion disclosed previously.
Net Cash Used in Financing Activities
During the first nine months of 2020, net cash used in financing activities increased by $2.9 billion compared with the first nine months of 2019. The change was due primarily to lower proceeds from long-term debt issuances ($2.2 billion proceeds from our U.S. dollar debt issuances in 2020 compared with $3.8 billion proceeds from our U.S. dollar and Euro debt issuances in 2019) and higher long-term debt repayments ($4.0 billion in 2020 compared with $3.0 billion in 2019).
Debt and Liquidity
We define cash and cash equivalents as short-term, highly liquid investments, readily convertible to known amounts of cash that mature within a maximum of three months and have an insignificant risk of change in value due to interest rate or credit risk changes. As a policy, we do not hold any investments in structured or equity-linked products. Our cash and cash equivalents are predominantly held in demand deposits with institutions that have investment-grade long-term credit rating. As part of our cash management strategy and in order to manage counterparty exposure, we also enter into reverse repurchase agreements. Such agreements are collateralized with government or corporate securities held by a custodial bank and, at maturity, cash is paid back to PMI, and the collateral is returned to the bank. For the nine months ended September 30, 2020 and the full-year 2019, the activities for such reverse repurchase agreements were not material.
We utilize long-term and short-term debt financing, including a commercial paper program that is regularly used to finance ongoing liquidity requirements, as part of our overall cash management strategy. Our ability to access the capital and credit markets as well as overall dynamics of these markets may impact borrowing costs. We expect that the combination of our long-term and short-term debt financing, the commercial paper program and the committed credit facilities, coupled with our operating cash flows, will enable us to meet our liquidity requirements.
Credit Ratings – The cost and terms of our financing arrangements as well as our access to commercial paper markets may be affected by applicable credit ratings. At September 30, 2020, our credit ratings and outlook by major credit rating agencies were as follows:
| Short-term | Long-term | Outlook | ||||||||||||||||||
| Moody’s | P-1 | A2 | Stable | |||||||||||||||||
| Standard & Poor’s | A-1 | A | Stable | |||||||||||||||||
| Fitch | F1 | A | Stable | |||||||||||||||||
Credit Facilities – On January 31, 2020, we entered into an agreement to amend and extend the term of our $2.0 billion 364-day revolving credit facility from February 4, 2020, to February 2, 2021.
On February 10, 2020, we entered into a new $2.0 billion multi-year revolving credit facility, expiring on February 10, 2025. The new credit facility replaced the $2.5 billion multi-year revolving credit facility, which was terminated effective February 10, 2020. We had no borrowings outstanding under the terminated facility, which was due to expire on February 28, 2021.
89
At September 30, 2020, our committed credit facilities were as follows:
| (in billions) | ||||||||
| Type | Committed Credit Facilities | |||||||
| 364-day revolving credit, expiring February 2, 2021 | $ | 2.0 | ||||||
| Multi-year revolving credit, expiring October 1, 2022 | 3.5 | |||||||
| Multi-year revolving credit, expiring February 10, 2025 | 2.0 | |||||||
Total facilities | $ | 7.5 | ||||||
At September 30, 2020, there were no borrowings under the committed credit facilities, and the entire committed amounts were available for borrowing.
All banks participating in our committed credit facilities have an investment-grade long-term credit rating from the credit rating agencies. We continuously monitor the credit quality of our banking group, and at this time we are not aware of any potential non-performing credit provider.
These facilities do not include any credit rating triggers, material adverse change clauses or any provisions that could require us to post collateral. The $3.5 billion multi-year revolving credit facility in the table above requires us to maintain a ratio of consolidated earnings before interest, taxes, depreciation and amortization (“consolidated EBITDA”) to consolidated interest expense of not less than 3.5 to 1.0 on a rolling four-quarter basis. At September 30, 2020, our ratio calculated in accordance with the agreements was 12.3 to 1.0. We expect to continue to meet our covenants. The terms “consolidated EBITDA” and “consolidated interest expense,” both of which include certain adjustments, are defined in the facility agreement previously filed with the U.S. Securities and Exchange Commission.
In addition to the committed credit facilities discussed above, certain of our subsidiaries maintain short-term credit arrangements to meet their respective working capital needs. These credit arrangements, which amounted to approximately $2.6 billion at September 30, 2020 and $2.7 billion at December 31, 2019, are for the sole use of our subsidiaries. Borrowings under these arrangements and other bank loans amounted to $152 million at September 30, 2020, and $338 million at December 31, 2019.
Commercial Paper Program – We continue to have access to liquidity in the commercial paper market through programs in place in the U.S. and in Europe having an aggregate issuance capacity of $8.0 billion. At September 30, 2020 and December 31, 2019, we had no commercial paper outstanding. The average commercial paper balance outstanding during the first nine months of 2020 was $1.5 billion. The average commercial paper balance outstanding during 2019 was $2.3 billion.
Sale of Accounts Receivable – To mitigate credit risk and enhance cash and liquidity management we sell trade receivables to unaffiliated financial institutions. These arrangements allow us to sell, on an ongoing basis, certain trade receivables without recourse. The trade receivables sold are generally short-term in nature and are removed from the condensed consolidated balance sheets. We sell trade receivables under two types of arrangements, servicing and nonservicing.
Our operating cash flows were positively impacted by the amount of the trade receivables sold and derecognized from the condensed consolidated balance sheets, which remained outstanding with the unaffiliated financial institutions. The trade receivables sold that remained outstanding under these arrangements as of September 30, 2020, and September 30, 2019 were $0.6 billion and $0.6 billion, respectively. The net proceeds received are included in cash provided by operating activities in the condensed consolidated statements of cash flows.
For further details, see Note 15. Sale of Accounts Receivable to our condensed consolidated financial statements.
Debt – Our total debt was $29.5 billion at September 30, 2020 and $31.0 billion at December 31, 2019.
On February 11, 2020, we filed a shelf registration statement with the U.S. Securities and Exchange Commission, under which we may from time to time sell debt securities and/or warrants to purchase debt securities over a three-year period.
90
Our debt issuances in the first nine months of 2020 were as follows:
| (in millions) | ||||||||||||||||||||||||||
| Type | Face Value | Interest Rate | Issuance | Maturity | ||||||||||||||||||||||
| U.S. dollar notes | (a) | $750 | 1.125% | May 2020 | May 2023 | |||||||||||||||||||||
| U.S. dollar notes | (a) | $750 | 1.500% | May 2020 | May 2025 | |||||||||||||||||||||
| U.S. dollar notes | (a) | $750 | 2.100% | May 2020 | May 2030 | |||||||||||||||||||||
(a) Interest on these notes is payable semi-annually in arrears beginning in November 2020.
Guarantees – At September 30, 2020, we were contingently liable for guarantees of our own performance, of which $0.3 billion were related to our obligations under indemnity agreements to enable appeals of customs assessments against our distributors. In October 2020, we guaranteed an obligation for an equity method investee. For further details, see Note 8. Contingencies to our condensed consolidated financial statements. Additionally, we have other guarantees of our own performance, which are primarily related to excise taxes on the shipment of our products. There is no liability in the condensed consolidated financial statements associated with these guarantees. These guarantees have not had, and are not expected to have, a significant impact on PMI’s liquidity.
Equity and Dividends
We discuss our stock awards as of September 30, 2020 in Note 2. Stock Plans to our condensed consolidated financial statements.
During 2019, we did not repurchase any shares under a share repurchase program, and we do not presently intend to repurchase shares of our common stock in 2020.
Dividends paid in the first nine months of 2020 were $5.5 billion. During the third quarter of 2020, our Board of Directors approved a 2.6% increase in the quarterly dividend to $1.20 per common share. As a result, the present annualized dividend rate is $4.80 per common share.
Market Risk
Counterparty Risk - We predominantly work with financial institutions with strong short- and long-term credit ratings as assigned by Standard & Poor’s and Moody’s. These banks are also part of a defined group of relationship banks. Non-investment grade institutions are only used in certain emerging markets to the extent required by local business needs. We have a conservative approach when it comes to choosing financial counterparties and financial instruments. As such, we do not invest or hold investments in any structured or equity-linked products. The majority of our cash and cash equivalents is currently invested in demand deposits maturing within less than 30 days.
We continuously monitor and assess the credit worthiness of all our counterparties.
Derivative Financial Instruments - We operate in markets outside of the United States of America, with manufacturing and sales facilities in various locations throughout the world. Consequently, we use certain financial instruments to manage our foreign currency and interest rate exposure. We use derivative financial instruments principally to reduce our exposure to market risks resulting from fluctuations in foreign exchange and interest rates by creating offsetting exposures. We are not a party to leveraged derivatives and, by policy, do not use derivative financial instruments for speculative purposes.
See Note 5. Financial Instruments, Note 11. Fair Value Measurements, and Note 13. Balance Sheet Offsetting to our condensed consolidated financial statements for further details on our derivative financial instruments and the related collateral arrangements.
Contingencies
See Note 8. Contingencies to our condensed consolidated financial statements for a discussion of contingencies.
91
Cautionary Factors That May Affect Future Results
Forward-Looking and Cautionary Statements
We may from time to time make written or oral forward-looking statements, including statements contained in filings with the SEC, in reports to stockholders and in press releases and investor webcasts. You can identify these forward-looking statements by use of words such as "strategy," "expects," "continues," "plans," "anticipates," "believes," "will," "estimates," "intends," "projects," "goals," "targets," "forecasts" and other words of similar meaning. You can also identify them by the fact that they do not relate strictly to historical or current facts.
We cannot guarantee that any forward-looking statement will be realized, although we believe we have been prudent in our plans and assumptions. Our RRPs constitute a new product category in its early stages that is less predictable than our mature cigarette business. Achievement of future results is subject to risks, uncertainties and inaccurate assumptions. Should known or unknown risks or uncertainties materialize, or should underlying assumptions prove inaccurate, actual results could vary materially from those anticipated, estimated or projected. Investors should bear this in mind as they consider forward-looking statements and whether to invest in or remain invested in our securities. In connection with the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, we are identifying important factors that, individually or in the aggregate, could cause actual results and outcomes to differ materially from those contained in any forward-looking statements made by us; any such statement is qualified by reference to the following cautionary statements. We elaborate on these and other risks we face throughout this document, particularly in the “Business Environment” section. You should understand that it is not possible to predict or identify all risk factors. Consequently, you should not consider the following to be a complete discussion of all potential risks or uncertainties. We do not undertake to update any forward-looking statement that we may make from time to time, except in the normal course of our public disclosure obligations.
Risks Related to Our Business and Industry
Consumption of tax-paid cigarettes continues to decline in many of our markets.
This decline is due to multiple factors, including increased taxes and pricing, governmental actions, the diminishing social acceptance of smoking, continuing economic and geopolitical uncertainty, and the continuing prevalence of illicit products. These factors and their potential consequences are discussed more fully below and in the "Business Environment" section.
Cigarettes are subject to substantial taxes. Significant increases in cigarette-related taxes have been proposed or enacted and are likely to continue to be proposed or enacted in numerous jurisdictions. These tax increases may disproportionately affect our profitability and make us less competitive versus certain of our competitors.
Tax regimes, including excise taxes, sales taxes and import duties, can disproportionately affect the retail price of cigarettes versus other combustible tobacco products, or disproportionately affect the relative retail price of our cigarette brands versus cigarette brands manufactured by certain of our competitors. Because our portfolio is weighted toward the premium-price cigarette category, tax regimes based on sales price can place us at a competitive disadvantage in certain markets. As a result, our volume and profitability may be adversely affected in these markets.
Increases in cigarette taxes are expected to continue to have an adverse impact on our sales of cigarettes, due to resulting lower consumption levels, a shift in sales from manufactured cigarettes to other combustible tobacco products and from the premium-price to the mid-price or low-price cigarette categories, where we may be under-represented, from local sales to legal cross-border purchases of lower price products, or to illicit products such as contraband, counterfeit and "illicit whites."
Our business faces significant governmental action aimed at increasing regulatory requirements with the goal of reducing or preventing the use of tobacco products.
Governmental actions, combined with the diminishing social acceptance of smoking and private actions to restrict smoking, have resulted in reduced industry volume in many of our markets, and we expect that such factors will continue to reduce consumption levels and will increase down-trading and the risk of counterfeiting, contraband, "illicit whites" and legal cross-border purchases. Significant regulatory developments will continue to take place over the next few years in most of our markets, driven principally by the World Health Organization's Framework Convention on Tobacco Control ("FCTC"). Since it came into force in 2005, the FCTC has led to increased efforts by tobacco control advocates and public health organizations to promote increasingly restrictive regulatory measures on the marketing and sale of tobacco products to adult smokers. Regulatory initiatives that have been proposed, introduced or enacted include:
92
•restrictions on or licensing of outlets permitted to sell cigarettes;
•the levying of substantial and increasing tax and duty charges;
•restrictions or bans on advertising, marketing and sponsorship;
•the display of larger health warnings, graphic health warnings and other labeling requirements;
•restrictions on packaging design, including the use of colors, and plain packaging;
•restrictions on packaging and cigarette formats and dimensions;
•restrictions or bans on the display of tobacco product packaging at the point of sale and restrictions or bans on cigarette vending machines;
•requirements regarding testing, disclosure and performance standards for tar, nicotine, carbon monoxide and other smoke constituents;
•disclosure, restrictions, or bans of tobacco product ingredients;
•increased restrictions on smoking in public and work places and, in some instances, in private places and outdoors;
•regulation, restrictions or prohibitions of novel tobacco or nicotine-containing products;
•elimination of duty free sales and duty free allowances for travelers;
•encouraging litigation against tobacco companies; and
•excluding tobacco companies from transparent public dialogue regarding public health and other policy matters.
Our financial results could be significantly affected by regulatory initiatives resulting in a significant decrease in demand for our brands, in particular requirements that lead to a commoditization of tobacco products or impede adult consumers' ability to convert to our RRPs, as well as any significant increase in the cost of complying with new regulatory requirements.
Litigation related to tobacco use and exposure to environmental tobacco smoke could substantially reduce our profitability and could severely impair our liquidity.
There is litigation related to tobacco products pending in certain jurisdictions. Damages claimed in some tobacco-related litigation are significant and, in certain cases in Brazil, Canada, and Nigeria, range into the billions of U.S. dollars. We anticipate that new cases will continue to be filed. The FCTC encourages litigation against tobacco product manufacturers. It is possible that our consolidated results of operations, cash flows or financial position could be materially affected in a particular fiscal quarter or fiscal year by an unfavorable outcome or settlement of certain pending litigation. See Note 8. Contingencies to our condensed consolidated financial statements for a discussion of pending litigation and "Business Environment—Reduced-Risk Products (RRPs)—Legal Challenges to RRPs."
We face intense competition, and our failure to compete effectively could have a material adverse effect on our profitability and results of operations.
We compete primarily on the basis of product quality, brand recognition, brand loyalty, taste, R&D, innovation, packaging, customer service, marketing, advertising and retail price and, increasingly, adult smoker willingness to convert to our RRPs. We are subject to highly competitive conditions in all aspects of our business. The competitive environment and our competitive position can be significantly influenced by weak economic conditions, erosion of consumer confidence, competitors' introduction of lower-price products or innovative products, higher tobacco product taxes, higher absolute prices and larger gaps between retail price categories, and product regulation that diminishes the ability to differentiate tobacco products and restricts adult consumer access to truthful and non-misleading information about our RRPs. Competitors include three large international tobacco companies, new market entrants, particularly with respect to innovative products, several regional and local tobacco companies and, in some instances, state-owned tobacco enterprises, principally in Algeria, Egypt, the PRC, Taiwan, Thailand and Vietnam. Industry consolidation and privatizations of state-owned enterprises have led to an overall increase in competitive pressures. Some competitors have different profit and volume objectives, and some international competitors are susceptible to changes in different currency exchange rates. Certain new market entrants may alienate consumers from innovative products through inappropriate marketing campaigns and messaging and inferior product satisfaction, while not relying on scientific substantiation based on appropriate R&D protocols and standards. The growing use of digital media could increase the speed and extent of the dissemination of inaccurate and misleading information about our RRPs.
93
Because we have operations in numerous countries, our results may be influenced by economic, regulatory and political developments, natural disasters, pandemics or conflicts.
Some of the countries in which we operate face the threat of civil unrest and can be subject to regime changes. In others, nationalization, terrorism, conflict and the threat of war may have a significant impact on the business environment. Natural disasters, pandemics, economic, political, regulatory or other developments could disrupt our supply chain, manufacturing capabilities or distribution capabilities. In addition, such developments could increase costs of our materials and operations and lead to loss of property or equipment that are critical to our business in certain markets and difficulty in staffing and managing our operations, all of which could reduce our volumes, revenues and net earnings. We discuss risks associated with the COVID-19 pandemic below.
In certain markets, we are dependent on governmental approvals of various actions such as price changes, and failure to obtain such approvals could impair growth of our profitability.
In addition, despite our high ethical standards and rigorous control and compliance procedures aimed at preventing and detecting unlawful conduct, given the breadth and scope of our international operations, we may not be able to detect all potential improper or unlawful conduct by our employees and partners.
Our business, results of operations, cash flows and financial position will be adversely impacted during the continuation of the COVID-19 pandemic.
The COVID-19 pandemic has created significant societal and economic disruption, and resulted in closures of stores, factories and offices, and restrictions on manufacturing, distribution and travel, all of which will adversely impact our business, results of operations, cash flows and financial position during the continuation of the pandemic. Our business continuity plans and other safeguards may not be effective to mitigate the results of the pandemic.
Currently, significant risks include our diminished ability to convert adult smokers to our RRPs, significant volume declines in our duty-free business and certain other key markets, disruptions or delays in our manufacturing and supply chain, increased currency volatility, and delays in certain cost saving, transformation and restructuring initiatives. Our business could also be adversely impacted if key personnel or a significant number of employees or business partners become unavailable due to the COVID-19 outbreak. The significant adverse impact of COVID-19 on the economic or political conditions in markets in which we operate could result in changes to the preferences of our adult consumers and lower demand for our products, particularly for our mid-price or premium-price brands. Continuation of the pandemic could disrupt our access to the credit markets or increase our borrowing costs. Governments may temporarily be unable to focus on the development of science-based regulatory frameworks for the development and commercialization of RRPs or on the enforcement or implementation of regulations that are significant to our business. In addition, messaging about the potential negative impacts of the use of our products on COVID-19 risks may lead to increasingly restrictive regulatory measures on the sale and use of our products, negatively impact demand for our products, the willingness of adult consumers to switch to our RRPs and our efforts to advocate for the development of science-based regulatory frameworks for the development and commercialization of RRPs.
The impact of these risks also depends on factors beyond our knowledge or control, including the duration and severity of the outbreak, its recurrence in our key markets, actions taken to contain its spread and to mitigate its public health effects, and the ultimate economic consequences thereof.
We may be unable to anticipate changes in adult consumer preferences.
Our business is subject to changes in adult consumer preferences, which may be influenced by local economic conditions. To be successful, we must:
•promote brand equity successfully;
•anticipate and respond to new adult consumer trends;
•develop new products and markets and broaden brand portfolios;
•improve productivity;
•convince adult smokers to convert to our RRPs;
•ensure adequate production capacity to meet demand for our products; and
•be able to protect or enhance margins through price increases.
94
In periods of economic uncertainty, adult consumers may tend to purchase lower-price brands, and the volume of our premium-price and mid-price brands and our profitability could suffer accordingly. Such down-trading trends may be reinforced by regulation that limits branding, communication and product differentiation.
The financial and business performance of our reduced-risk products is less predictable than our cigarette business.
Our RRPs are novel products in a new category, and the pace at which adult smokers adopt them may vary, depending on the competitive, regulatory, fiscal and cultural environment, and other factors in a specific market. There may be periods of accelerated growth and periods of slower growth for these products, the timing and drivers of which may be more difficult for us to predict versus our mature cigarette business. The impact of this lower predictability on our projected results for a specific period may be significant, particularly during the early stages of this new product category and during the COVID-19 pandemic.
We lose revenues as a result of counterfeiting, contraband, cross-border purchases, "illicit whites," non-tax-paid volume produced by local manufacturers, and counterfeiting of our Platform 1 device and heated tobacco units.
Large quantities of counterfeit cigarettes are sold in the international market. We believe that Marlboro is the most heavily counterfeited international cigarette brand, although we cannot quantify the revenues we lose as a result of this activity. In addition, our revenues are reduced by contraband, legal cross-border purchases, "illicit whites" and non-tax-paid volume produced by local manufacturers. Our revenues and consumer satisfaction with our Platform 1 device and heated tobacco units may be adversely affected by counterfeit products that do not meet our product quality standards and scientific validation procedures.
From time to time, we are subject to governmental investigations on a range of matters.
Investigations include allegations of contraband shipments of cigarettes, allegations of unlawful pricing activities within certain markets, allegations of underpayment of income taxes, customs duties and/or excise taxes, allegations of false and misleading usage of descriptors, allegations of unlawful advertising, and allegations of unlawful labor practices. We cannot predict the outcome of those investigations or whether additional investigations may be commenced, and it is possible that our business could be materially affected by an unfavorable outcome of pending or future investigations. See Note 8. Contingencies—Other Litigation and “Management's Discussion and Analysis of Financial Condition and Results of Operations—Operating Results by Business Segment—Business Environment—Governmental Investigations” for a description of certain governmental investigations to which we are subject.
We may be unsuccessful in our attempts to introduce reduced-risk products, and regulators may not permit the commercialization of these products or the communication of scientifically substantiated risk-reduction claims.
Our key strategic priorities are: to develop and commercialize products that present less risk of harm to adult smokers who switch to those products versus continued smoking; and to convince current adult smokers who would otherwise continue to smoke to switch to those RRPs. For our efforts to be successful, we must: develop RRPs that such adult smokers find acceptable alternatives to smoking; conduct rigorous scientific studies to substantiate that they reduce exposure to harmful and potentially harmful constituents in smoke and, ultimately, that these products present, are likely to present, or have the potential to present less risk of harm to adult smokers who switch to them versus continued smoking; and effectively advocate for the development of science-based regulatory frameworks for the development and commercialization of RRPs, including communication of scientifically substantiated information to enable adult smokers to make better consumer choices. We might not succeed in our efforts. If we do not succeed, but others do, or if heat-not-burn products are inequitably regulated compared to other RRP categories without regard to the totality of the scientific evidence available for such products, we may be at a competitive disadvantage. In addition, actions of some market entrants, such as the inappropriate marketing of e-vapor products to youth, as well as alleged health consequences associated with the use of certain e-vapor products, may unfavorably impact public opinion and/or mischaracterize all e-vapor products or other RRPs to consumers, regulators and policy makers without regard to the totality of scientific evidence for specific products. This may impede our efforts to advocate for the development of science-based regulatory frameworks for the development and commercialization of RRPs. We cannot predict whether regulators will permit the sale and/or marketing of RRPs with scientifically substantiated risk-reduction claims. Such restrictions could limit the success of our RRPs. Moreover, the FDA’s premarket tobacco product and modified risk tobacco product authorizations of a version of our Platform 1 product are subject to strict marketing, reporting and other requirements and is not a guarantee that the product will remain authorized, particularly if there is a significant uptake in youth or non-smoker initiation.
95
We may be unsuccessful in our efforts to differentiate reduced-risk products and cigarettes with respect to taxation.
To date, we have been largely successful in demonstrating to regulators that our RRPs are not cigarettes, and as such they are generally taxed either as a separate category or as other tobacco products, which typically yields more favorable tax rates than cigarettes. If we cease to be successful in these efforts, RRP unit margins may be adversely affected.
Our reported results could be adversely affected by unfavorable currency exchange rates, and currency devaluations could impair our competitiveness.
We conduct our business primarily in local currency and, for purposes of financial reporting, the local currency results are translated into U.S. dollars based on average exchange rates prevailing during a reporting period. During times of a strengthening U.S. dollar, our reported net revenues, operating income and EPS will be reduced because the local currency translates into fewer U.S. dollars. During periods of economic crises, such as during the ongoing COVID-19 pandemic, foreign currencies may be devalued significantly against the U.S. dollar, reducing our margins. Actions to recover margins may result in lower volume and a weaker competitive position.
Changes in the earnings mix and changes in tax laws may result in significant variability in our effective tax rates. Our ability to receive payments from foreign subsidiaries or to repatriate royalties and dividends could be restricted by local country currency exchange controls and other regulations.
The Tax Cuts and Jobs Act that was signed into law in December 2017 constitutes a major change to the U.S. tax system. Our estimated impact of the Tax Cuts and Jobs Act is based on management’s current interpretations, and our analysis is ongoing. Our final tax liability may be materially different from current estimates due to developments such as implementing regulations and clarifications. In future periods, our effective tax rate and our ability to recover deferred tax assets could be subject to additional uncertainty as a result of such developments. Furthermore, changes in the earnings mix or applicable foreign tax laws may result in significant variability in our effective tax rates. Because we are a U.S. holding company, our most significant source of funds is distributions from our non-U.S. subsidiaries. Certain countries in which we operate have adopted or could institute currency exchange controls and other regulations that limit or prohibit our local subsidiaries' ability to convert local currency into U.S. dollars or to make payments outside the country. This could subject us to the risks of local currency devaluation and business disruption.
Our ability to grow profitability may be limited by our inability to introduce new products, enter new markets or improve our margins through higher pricing and improvements in our brand and geographic mix.
Our profit growth may suffer if we are unable to introduce new products or enter new markets successfully, to raise prices or to improve the proportion of our sales of higher margin products and in higher margin geographies.
We may be unable to expand our brand portfolio through successful acquisitions or the development of strategic business relationships.
One element of our growth strategy is to strengthen our brand portfolio and market positions through selective acquisitions and the development of strategic business relationships. Acquisition and strategic business development opportunities are limited and present risks of failing to achieve efficient and effective integration, strategic objectives and anticipated revenue improvements and cost savings. There is no assurance that we will be able to acquire attractive businesses on favorable terms, or that future acquisitions or strategic business developments will be accretive to earnings.
Government mandated prices, production control programs, shifts in crops driven by economic conditions and the impact of climate change may increase the cost or reduce the quality of the tobacco and other agricultural products used to manufacture our products.
As with other agricultural commodities, the price of tobacco leaf and cloves can be influenced by imbalances in supply and demand and the impacts of natural disasters and pandemics such as COVID-19, and crop quality can be influenced by variations in weather patterns, including those caused by climate change. Tobacco production in certain countries is subject to a variety of controls, including government mandated prices and production control programs. Changes in the patterns of demand for agricultural products could cause farmers to produce less tobacco or cloves. Any significant change in tobacco leaf and clove prices, quality and quantity could affect our profitability and our business.
Our ability to achieve our strategic goals may be impaired if we fail to attract and retain the best global talent.
To be successful, we must continue transforming our culture and ways of working, align our talent with our business needs, innovate and transform to a consumer-centric business. We compete for talent, including in areas that are new to us, such as
96
digital and technical solutions, with companies in the consumer products, technology and other sectors that enjoy greater societal acceptance. As a result, we may be unable to attract and retain the best global talent with the right degree of diversity, experience and skills to achieve our strategic goals.
The failure of our information systems to function as intended or their penetration by outside parties with the intent to corrupt them or our failure to comply with privacy laws and regulations could result in business disruption, litigation and regulatory action, and loss of revenue, assets or personal or other confidential data.
We use information systems to help manage business processes, collect and interpret data and communicate internally and externally with employees, suppliers, consumers, customers and others. Some of these information systems are managed by third-party service providers. We have backup systems and business continuity plans in place, and we take care to protect our systems and data from unauthorized access. Nevertheless, failure of our systems to function as intended, or penetration of our systems by outside parties intent on extracting or corrupting information or otherwise disrupting business processes, could place us at a competitive disadvantage, result in a loss of revenue, assets or personal or other sensitive data, litigation and regulatory action, cause damage to our reputation and that of our brands and result in significant remediation and other costs. Failure to protect personal data, respect the rights of data subjects, and adhere to strict cybersecurity protocols could subject us to substantial fines and other legal challenges under regulations such as the EU General Data Protection Regulation. As we are increasingly relying on digital platforms in our business, the magnitude of these risks is likely to increase.
We may be unable to adequately protect our intellectual property rights, and disputes relating to intellectual property rights could harm our business.
Our intellectual property rights are valuable assets, and their protection is important to our business. If the steps we take to protect our intellectual property rights globally, including through a combination of trademark, design, patent and other intellectual property rights, are inadequate, or if others infringe or misappropriate our intellectual property rights, notwithstanding legal protection, our business could suffer. Intellectual property rights of third parties may limit our ability to commercialize our products or improve product quality in one or more markets. Competitors or other third parties may claim that we infringe their intellectual property rights. Any such claims, regardless of merit, could divert management’s attention, be costly, disruptive, time-consuming and unpredictable and expose us to litigation costs and damages, and impede our ability to manufacture, commercialize and improve our products. If, as a result, we are unable to manufacture or sell our RRPs or improve their quality in one or more markets, our ability to convert adult smokers to our RRPs in such markets would be adversely affected. See Note 8. Contingencies— Other Litigation to our condensed consolidated financial statements for a description of certain intellectual property proceedings.
We may be required to replace third-party contract manufacturers or service providers with our own resources.
In certain instances, we contract with third parties to manufacture some of our products or product parts or to provide other services. We may be unable to renew these agreements on satisfactory terms for numerous reasons, including government regulations. Accordingly, our costs may increase significantly if we must replace such third parties with our own resources.
97
Item 4. Controls and Procedures.
PMI carried out an evaluation, with the participation of PMI’s management, including PMI’s Chief Executive Officer and Chief Financial Officer, of the effectiveness of PMI’s disclosure controls and procedures (as defined in Rule 13a-15(e) under the Securities Exchange Act of 1934, as amended) as of the end of the period covered by this report. Based upon that evaluation, PMI’s Chief Executive Officer and Chief Financial Officer concluded that PMI’s disclosure controls and procedures are effective. There have been no changes in PMI’s internal control over financial reporting during the most recent fiscal quarter that have materially affected, or are reasonably likely to materially affect, PMI’s internal control over financial reporting.
98
Part II - OTHER INFORMATION
Item 1.Legal Proceedings.
See Note 8. Contingencies of the Notes to the Condensed Consolidated Financial Statements included in Part I – Item 1 of this report for a discussion of legal proceedings pending against Philip Morris International Inc. and its subsidiaries.
Item 1A. Risk Factors.
Information regarding Risk Factors appears in “MD&A – Cautionary Factors That May Affect Future Results,” in Part I – Item 2 of this Form 10-Q and in Part I – Item 1A. Risk Factors of our Annual Report on Form 10-K for the year ended December 31, 2019.
99
Item 2.Unregistered Sales of Equity Securities and Use of Proceeds.
Our share repurchase activity for each of the three months in the quarter ended September 30, 2020 was as follows:
| Period | Total Number of Shares Repurchased | Average Price Paid Per Share | Total Number of Shares Purchased as Part of Publicly Announced Plans or Programs | Approximate Dollar Value of Shares that May Yet be Purchased Under the Plans or Programs | ||||||||||||||||||||||
| July 1, 2020 – July 31, 2020 (1) | — | $ | — | — | $ | — | ||||||||||||||||||||
| August 1, 2020 – August 31, 2020 (1) | — | $ | — | — | $ | — | ||||||||||||||||||||
| September 1, 2020 – September 30, 2020 (1) | — | $ | — | — | $ | — | ||||||||||||||||||||
| Pursuant to Publicly Announced Plans or Programs | — | $ | — | |||||||||||||||||||||||
| July 1, 2020 – July 31, 2020 (2) | 3,603 | $ | 70.38 | |||||||||||||||||||||||
| August 1, 2020 – August 31, 2020 (2) | 280 | $ | 76.76 | |||||||||||||||||||||||
| September 1, 2020 – September 30, 2020 (2) | 2,074 | 2,342 | $ | 79.77 | ||||||||||||||||||||||
| For the Quarter Ended September 30, 2020 | 5,957 | $ | 73.95 | |||||||||||||||||||||||
(1)During this reporting period, we did not have an authorized share repurchase program.
(2)Shares repurchased represent shares tendered to us by employees who vested in restricted and performance share unit awards and used shares to pay all, or a portion of, the related taxes.
100
Item 6.Exhibits.
| 3.1 | ||||||||
| 31.1 | ||||||||
| 31.2 | ||||||||
| 32.1 | ||||||||
| 32.2 | ||||||||
| 101.INS | XBRL Instance Document - the instance document does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document. | |||||||
| 101.SCH | XBRL Taxonomy Extension Schema. | |||||||
| 101.CAL | XBRL Taxonomy Extension Calculation Linkbase. | |||||||
| 101.DEF | XBRL Taxonomy Extension Definition Linkbase. | |||||||
| 101.LAB | XBRL Taxonomy Extension Label Linkbase. | |||||||
| 101.PRE | XBRL Taxonomy Extension Presentation Linkbase. | |||||||
| 104 | Cover Page Interactive Data File (formatted as Inline XBRL and contained in Exhibit 101) | |||||||
101
Signature
Pursuant to the requirements of the Securities Exchange Act of 1934, the Registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
| PHILIP MORRIS INTERNATIONAL INC. | ||
| /s/ EMMANUEL BABEAU | ||
| Emmanuel Babeau | ||
| Chief Financial Officer | ||
| October 27, 2020 | ||
102
Similar companies
See also British American Tobacco p.l.c.See also ALTRIA GROUP, INC. - Annual report 2022 (10-K 2022-12-31) Annual report 2023 (10-Q 2023-09-30)
See also SWEDISH MATCH CORP
See also RLX Technology Inc.
See also VECTOR GROUP LTD - Annual report 2022 (10-K 2022-12-31) Annual report 2023 (10-Q 2023-09-30)
